1. Introduction
The formation of new capillaries or angiogenesis, is considered a vital part of the natural recovery after an ischemic injury and is, therefore, a key factor for post-ischemic repair of the infarcted myocardium [
1]. Since angiogenesis is associated with post-infarct remodeling of the left ventricle, and thus prognosis following myocardial infarction, it is of particular interest to be able to noninvasively monitor angiogenesis [
2,
3]. This might not only permit for a risk stratification of patients following myocardial infarction, but also facilitate development and improvement of new therapies directed towards stimulation of the natural angiogenic response.
Myocardial angiogenesis following myocardial infarction might be focal or non-transmural making it difficult to detect noninvasively, and existing noninvasive imaging methods directed towards the evaluation of angiogenesis have been somewhat limited.
During angiogenesis, endothelial cells must adhere to one another and to the extracellular matrix in order to form new microvessels and to extend existing ones [
4,
5]. The cross-talk with the extracellular matrix is mediated by integrins, which are a family of heterodimeric cell surface receptors involved in many cellular processes, including adhesion, migration, proliferation, and survival [
6]. Specifically, the α
vβ
3 integrin has been identified as a critical modulator of angiogenesis. It is highly expressed in activated endothelial cells, and therefore serves as a potential target for directly imaging angiogenesis [
5,
7,
8,
9].
The aim of this study was to present data for the uptake of a newly developed angiogenesis positron-emission-tomography (PET) tracer targeted towards α
vβ
3 integrin (
68Ga-NODAGA-E[c(RGDyK)]
2 (RGD)) [
7] and correlate it with histology and further to illustrate the differences in angiogenesis in Göttingen minipigs treated with saline or porcine adipose-derived mesenchymal stromal cell (pASC) suspended in alginate hydrogel after induction of acute myocardial infarction.
2. Materials and Methods
2.1. Study Design
Five Göttingen minipigs were used in this study. The study was approved by The National Committee for the Protection of Animals used for Scientific Purposes in Denmark (1 July 2014, Authorization number 2014-15-0201-00191). The infarction model was an acute model of myocardial infarction with permanent ligation and pASC or saline treatment immediately after the infarct induction. The minipigs underwent PET-imaging examining myocardial perfusion (
82Rubidium(
82Rb)-PET) and neo-angiogenesis (
68Ga-NODAGA-RGD) at baseline and one week and four weeks after infarct induction and treatment (
Figure 1).
2.2. Preparation for Scans
The minipigs were sedated with an intramuscular injection of 0.08 or 0.1 mL/kg (before and after an induced myocardial infarction) of a mixture of zolezapam 10.9 mg/mL and tiletamine 10.9 mg/mL (Zoletil 50/50® vet; Virbac, Carros, France), xylazine 10.9 mg/mL (Xysol vet; ScanVet Animal Health A/S, Fredensborg, Denmark), ketamine 10.9 mg/mL (Ketaminol vet, Intervet International B.V., Boxmeer, the Netherlands), methadone 1.7 mg/mL (Comfortan vet, Dechra Veterinary Products A/S, Uldum, Denmark) and butorphanol 1.7 mg/mL (Torbugesic vet, ScanVet Animal Health A/S, Fredensborg, Denmark). When in lateral recumbency minipigs were moved to the preparation room for instrumentation. For each scan minipigs were instrumented with 3 intravenous accesses (one in each ear (lateral auricular vene) and one in the hind leg (branch of v. saphena)) for anesthesia and PET tracer administration, a urinary bladder catheter, and a tracheal tube for ventilation. During anesthesia saturation (SpO2), ECG, non-invasive blood pressure, temperature and end-tidal CO2 were monitored using a Datex-Ohmeda S/5 compact monitor (GE Healthcare, Brøndbyvester, Denmark). When fully instrumented the minipigs were transported to the scanner and put on respirator. Respiration was set to obtain normo ventilation with an end-tidal CO2 of approximately 55 cmH2O. Minipigs were kept anesthetized with a maintenance dose of intravenous propofol 10 mg/kg/h during the 82Rb-PET computed tomography (CT) (Siemens mCT, Siemens, 128-slice CT, Knoxville, TN, USA).
2.3. Infarct Induction and Stromal Cell Treatment
2.3.1. Göttingen Minipig Adipose-Derived Mesenchymal Stromal Cells
Abdominal subcutaneous adipose tissue was harvested from recently euthanized Göttingen minipigs. The tissue was manually cut from the abdominal region and stored in phosphate-buffered saline (PBS) pH 7.4 (Gibco, Life Technologies, Carlsbad, CA, USA) with 5% penicillin/streptomycin for transportation. The tissue was manually cut into small pieces before being processed as with human samples. The pieces were treated with 0.6 PZ U/mL collagenase NB4 (SERVA Electrophoresis) dissolved in Hank’s buffered saline solution (Gibco, diluted to a concentration of 2 mM Ca2+) for one hour at 37 °C. The solution was filtered through a 100 µm filter (Cell Strainer, BD Falcon) and washed trice with PBS. The resulting cells were seeded in T75 flasks in a density of 4.5 × 106 cells/flask in complete medium (alpha-MEM (Gibco, Life Technologies), 10% fetal bovine serum (FBS) (Gibco, Life Technologies), 1% penicillin/streptomycin. The resulting stromal vascular fraction was incubated in an incubator with 37 °C and 5% CO2 for 2–3 days after which suspension cells were removed by washing with PBS, before continuing culture. This resulted in a homogeneous population of pASCs, which were stored in liquid nitrogen at a density of 5 × 106 per mL in 5% DMSO (WAK-Chemie Medical, Steinbach, Germany) in FBS. Two weeks before the operation the cells were thawed and cultured in complete medium again. The pASCs used for treatment were in passage 4 or 5.
The cells were tested for mesenchymal stromal cell abilities by performing differentiation towards adipogenic, osteogenic, and chondrogenic lineage using StemPro differentiation kit (Gibco, Life Technologies) according to manufacturer’s protocol. Differentiation was verified by lipid droplets stained with Oil Red O (Sigma-Aldrich, St. Louis, MO, USA) for adipogenic lineage, calcium deposition by Alizarin Red S for osteogenic lineage, and glycosaminoglycans by Alcian Blue 8GX (Sigma-Aldrich, St. Louis, MO, USA) for chondrogenic lineage.
Three minipigs were treated with pASC suspended in alginate hydrogel and two minipigs with saline.
2.3.2. Alginate Hydrogel
The injectable alginate hydrogel was cast using very low viscosity (5cP) high guluronic acid content (G > 65%) alginate (VLVG, NovaMatrix, FMC Biopolymers, Drammen, Norway) dissolved in sterile water. The alginate solution was crosslinked using
d-gluconic acid hemi calcium (Sigma-Aldrich, St. Louis, MO, USA) yielding a free-flow injectable alginate hydrogel consisting of 1.5% (
w/
v) alginate and 0.9% (
w/
v) calcium ions, which was filtered through a 20 µm filter [
10]. Centrifuged pASCs were re-suspended in the alginate hydrogel and transferred to a sterile syringe prior to the operation.
2.3.3. Myocardial Infarct Induction and Treatment
The minipigs were sedated, instrumented, ventilated and monitored like described above and kept anaesthetized on propofol (Propofol “B. Braun”; 10 mg/mL; B. Braun Medical A/S, Frederiksberg, Denmark) with a dose range of 10–15 mg/kg/h depending on the individual response to anesthesia. Fentanyl 0.5 µg/kg/h (Fentanyl “Hameln”; 50 µg/mL; Hameln Pharmaceuticals Gmbh, Hameln, Germany) was given as infusion as intraoperative analgesic. Amoxicillin 15 mg/kg (Curamox Prolongatum Vet.; 150 mg/mL; Boehringer Ingelheim DK A/S, Kalundborg, Denmark) and meloxicam 0.4 mg/kg (Boehringer Ingelheim DK A/S, Denmark) were given prophylactic before operation. Before surgical incision 50 mg of amiodaronhydrochlorid was administered intravenously as a bolus. The heart was accessed through partial lower median sternotomy. A sternal retractor was inserted, and the pericardium was opened with a scissor. The left anterior descending artery and a large diagonal branch were identified. Myocardial infarction was induced by ligation with a 5-0 prolene suture of the apical part of the LAD artery or a large diagonal artery (D2). After ligation of the artery, a sharp demarcation line between ischemic and non-ischemic myocardium was observed. Depending on the treatment arm, either isotonic saline or pASCs in alginate hydrogel was injected in 4–6 injections with a 25 G needle in the peri-infarct area on one side of the infarction. The minipigs in the treatment group received approximately 40 × 106 pASCs in 2.5 mL alginate hydrogel. Afterward, the sternum was closed with PDS 0 suture, the fascia and subcutis with 0 vicryl, and the cutis with a 4-0 monocryl intradermal suture. The minipigs were treated post-operative with butorphanole 0.2 mg/kg (Torbugesic vet 10 mg/mL, ScanVet Animal Health A/S, Denmark) and methadone 0.2 mg/kg (Comfortan vet 10 mg/mL, Dechra Veterinary Products A/S, Denmark) every 3–4 h for the first 24 h. Subsequently, they were treated with buprenorfin 0.01–0.02 mg/kg (Vetergesic 0.3 mg/mL, Orion Pharma Animal Health, Copenhagen, Denmark) every 8th hour in 2–3 days. The opioid regime was supported by oral administration of meloxicam 0.4 mg/kg (metacam oral suspension 15 mg/mL, Boehringer Ingelheim DK A/S) for 5 days post-operative. Antibiotic treatment with amoxicillin 20 mg/kg (Clamoxyl VET. 51%, Orion Pharma Animal Health, Denmark) was continued orally in 6 days post-operative.
2.4. Radiochemistry: 68Ga-NODAGA-E[c(RGDyK)]2 Synthesis
NODAGA-E[c(RGDyK)]2 acetate was obtained from ABX GmbH (Radeberg, Germany). Gallium-68 (t1/2 = 68 min; Emax,β+ = 1.90 MeV (89%)) labelling of NODAGA-E[c(RGDyK)]2 acetate was performed using a Modular-Lab eazy module (Eckert & Ziegler, Berlin, Germany). The 68Ge/68Ga generator (IGG100, Eckert & Ziegler) was eluted with 6 mL 0.1 M HCl. The eluate was concentrated on a Bond Elut SCX cartridge and eluted with 600 µL 5 M NaCl/5.5 M HCl (41:1). NODAGA-E[c(RGDyK)]2 (30 nmol) was labelled in 1000 µL 0.7 M NaOAc buffer pH 4.5 and 400 µL 50% EtOH at 60 °C for 400 s. The resulting 68Ga-NODAGA-E[c(RGDyK)]2 was formulated with saline or phosphate buffer.
The radiochemical purity was more than 96% on HPLC, and the amount of unlabeled 68Ga in the product was less than 1%, as demonstrated by radio–thin layer chromatography.
All reagents and cassettes were purchased from Eckert & Ziegler. For analysis, a high-performance liquid chromatograph (Ultimate 3000; Dionex, Sunnyvale, CA, USA) was used with a 2.6-μm, 100-Å, 50 × 4.6 mm C18 Kinetex column (Phenomenex, Torrance, CA, USA). The mobile phases were: eluent A: 10% MeCN in H2O with 0.1% trifluoroacetic acid; eluent B: 10% H2O in MeCN with 0.1% trifluoroacetic acid.
2.5. 82Rubidium and 68GA-NODAGA-RGD PET Imaging
82Rb rest and stress myocardial perfusion PET-CT were performed before induction of myocardial infarction, one week and one month after infarction (
Figure 1). The
68GA-NODAGA-RGD (RGD) PET-CT was performed as a 10 min ECG-gated scan 45 min after administration of 100 MBq RGD. The
82Rb rest and stress myocardial perfusion PET-CT has previously been described in detail [
11,
12]. In brief, a 7 min dynamic PET myocardial perfusion rest scan under administration of 1000–1200 MBq
82Rb was performed. Subsequently, a 7 min dynamic dipyridamole stress PET scan was performed. Dipyridamole (140 µg/kg/min) was given as a continuous intravenous infusion over 4 min prior to
82Rb-tracer injection which followed 3–5 min after the completion of dipyridamole infusion. PET images were analyzed semi-automatically using Cedars-Sinai Cardiac Suite QPS/QGS
® (Cedars-Sinai Medical Center, Los Angeles, CA, USA) for Syngo.Via (Siemens, Knoxville, TN, USA). The accuracy of slice-alignments in the ventricle was assessed by planes and intervened if necessary. Perfusion defects were subsequently quantified in the total left ventricle myocardium. The magnitude of the rest perfusion defects was determined automatically by comparing the polar plot of a minipig to that of the human normal database on a pixel-by-pixel basis. A 2.5 standard deviation cut-off was used to define whether a pixel count fell below a normal value. Mean Standardized Uptake Values were measured in the infarcted area (SUV
mean,infarct) as well as the non-infarcted myocardium including blood pool (SUV
mean,background) and the ratio between the two (SUV
index) was calculated.
During the examination heart rate was measured continuously and non-invasive blood pressure was measured every minute. The hemodynamic response to dipyridamole was evaluated by calculating Rate-Pressure-Products (RPP) at rest and stress as the product of non-invasive systolic blood pressure and heart rate at rest and stress, respectively.
2.6. Histology
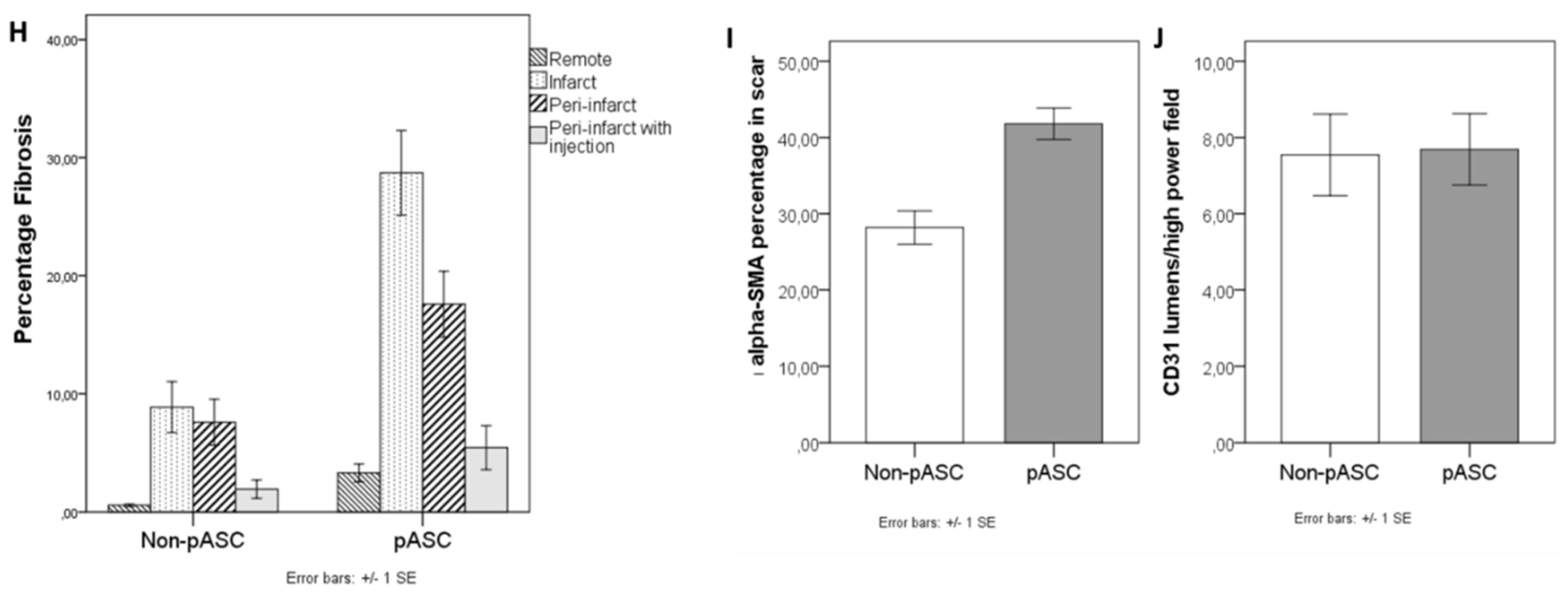
After euthanasia, the heart of the minipig was removed and the infarct area was located together with the suture. The heart was cut into 1.5 cm thick short axis slices, and 4 areas from the slice below the ligation were selected for histology. The four areas were: (1) Infarct area; (2) Peri-infarct area; (3) Peri-infarct area at the side of injections; and (4) Remote myocardium, beyond the peri-infarct area. The tissue was fixed in 4% paraformaldehyde and embedded in paraffin. Slices of 5 µm thickness were cut for staining at four different levels in each tissue sample. Antigen retrieval was performed before all stainings. The tissue was stained for collagen by Masson’s Trichrome (Hospital Pharmacy, Rigshospitalet), tissue organization and inflammation by hematoxylin and eosin (H&E) (Hospital Pharmacy, Rigshospitalet), macrophages by CD68 (Agilent Technologies, Glostrup, Denmark, 1:100), myofibroblasts by α-smooth muscle actin (α-SMA) (Agilent Technologies), CD31 (Bio-Rad Laboratories, Copenhagen, Denmark, 1:50), and integrin αvβ3 (Merck Millipore, Darmstadt, Germany, 1:75). Visualization was performed used Carl Zeiss Axio Imager Z.1 microscope and Axiovision 4.6.3 software. Mason’s Trichrome was analyzed by three random areas at ×1.25 magnification for each slice, resulting in a total of 12 random fields for each heart area from each minipig. The percentage of collagen staining compared to the whole image was analyzed using Image J software (Fiji). A similar number of fields were acquired for each staining. The level of inflammation was assessed by scoring each ×5 magnification field from 0 to 3, ranging from 0 = no visible inflammation, 1 = <10% inflammatory cells, 2 = 10–50% inflammatory cells, and 3 = >50% inflammatory cells of total cells. High power fields of ×20 magnification were used for total manual CD68 count. Similarly, the same magnification was used for α-SMA fibroblast percentage measurements in the scar area, and CD31 and integrin αvβ3. All manual assessment was performed by two independent observers.
2.7. Statistical Analysis
Categorical variables were expressed as percentages and continuous variables were reported as means and standard deviations. Differences in continuous variables between groups were assessed with students t-test while differences in groups over time were assessed with paired t-test. A general linear model was used to analyse data with more than one time-point and the resulting regression lines were compared for differences between groups. A two tailed p-value < 0.05 was considered statistically significant. Statistical analyses were performed using SAS® for Windows, version 9.1 (SAS institute, Cary, NC, USA).
4. Discussion
The major finding of this study was that infarcted myocardium in minipigs had significantly higher RGD uptake after one week and one month, whether pASC-treated or non-pASC-treated. Overall, SUVindex tended to be lower one month after infarction compared to one week after infarction (p = 0.10) (not shown). Furthermore, we found no difference in SUVindex between pASC-treated vs. non-pASC-treated minipigs, which also was reflected in the histological findings of similar CD31 vessel density in the groups, with co-localization of integrin αvβ3 and CD31.
Our finding of integrin α
vβ
3 only in the infarcted area is in line with a recent study (Grönman et al.), using the same RGD peptide, but not the same tracer, to detect angiogenesis in pigs after induction of coronary stenosis [
13]. They found integrin α
vβ
3 and their tracer almost exclusively located in the injured myocardium.
In addition to this, we found no differences in ejection fractions or coronary flow reserves between pASC-treated vs. non-pASC-treated minipigs. This relates to the similar CD31 vessel density observed by histology. There was a tendency for a greater degree of fibrosis and a higher density of α-SMA expression in the scar of the pASC-treated pigs. The difference was apparently too small to be functionally relevant. It has been shown, that the effect of stromal cell therapy is dependent on the extent of the injury, with more injury resulting in more pronounced treatment effect [
14]. Hence, the relatively small infarcts could be the reason for the observed outcome.
In a study by Cai et al., the angiogenic effect of vascular endothelial growth factor (VEGF) gene and/or bone marrow mesenchymal stem cells (BMSCs) following myocardial infarction was compared to a saline control, using Spraque–Dawley rats (
n = 4 in each group) [
15]. The angiogenic response was evaluated using
18F-alfatide II (
18F-AlF-NOTA-E[PEG
4-c(RGDfk)]
2), which, as RGD, targets α
vβ
3 integrin. Corresponding to our findings, RGD uptake was documented in all four groups one week after myocardial infarction. The infarct-to-normal myocardium ratio was considerably higher in all groups (3.94 ± 0.20 for VEGF group, 3.77 ± 0.16 for BMSCs group and 4.86 ± 0.08 for the combination group vs. 3.01 ± 0.03 for the control group) compared to our study. Contrary to our findings, the study documented an angiogenic effect of treatment in all treatment groups compared to the control group (
p < 0.005 for VEGF group,
p < 0.005 for BMSCs group and
p < 0.0001 for the combination group). Whether the differences in infarct to normal heart ratios rely on the different tracers used, differences in species or infarct size and whether the different conclusions rely on our relatively small sample size remains unsettled.
In another study angiogenesis after myocardial infarction by ligation of LAD in Spraque–Dawley rats using [
68Ga]DOTA-E-[c(RGDfK)]
2 PET was examined [
16]. The infarcted myocardium showed higher tracer uptake than the remote myocardium 7 days and 4 weeks post-infarct (infarct/remote myocardium ratio 2.25 (±0.14) and 2.13 (±0.37)). Compared to sham-operated-rats both infarcted and remote myocardium showed higher tracer uptake 7 days and 4 weeks postinfarct where it coincided with increased interstitial fibrosis. Again, whether the higher infarct/remote myocardium ratio compared to the ones measured in our study relies on tracer difference, species or infarct size remains unknown.
The primary outcomes for most clinical trials investigating the effect of cellular therapy are LVEF, EDV or end-systolic volume (ESV) measured by MRI, echocardiography, and/or CT. These are fine as surrogate endpoints for phase I/II clinical trials but do not relate directly to the mechanisms by which the cells exert their regenerative effect. The addition of other endpoints might clarify the effect of cell therapy [
17,
18].
Angiogenesis is a vital part of the reparative process following myocardial infarction. It is also thought to be one of the main mechanisms behind the regenerative effect of cardiac cellular therapy and is in pre-clinical studies traditionally assessed in histology by counting of vessel density in affected areas of the heart [
19,
20,
21]. Since this is not feasible in clinical studies, clinical trials with thorough functional design do not usually investigate angiogenesis due to lack of methods [
22]. While angiogenesis is associated with post-infarct remodeling of the left ventricle, stratification of patients with RGD-PET following myocardial infarction may prove effective [
2]. Although strictly hypothetical, one would assume a patient with RGD uptake to have a better prognosis than a patient not taking up RGD in a myocardial infarction of a similar size. Furthermore, the “clearance” of RGD at follow-up PET might be of importance. Indicative of the latter, although limited to two myocardial infarction patients, was a study by Luo et al. [
23]. Both patients showed retention of
68Ga-BNOTA-PRGD
2 in the infarcted region of the myocardium 1 week after the cardiac event. At follow-up, retention of
68Ga-BNOTA-PRGD
2 remained in the infarcted myocardium of the still symptomatic patient whereas no uptake could be visualized in the asymptomatic patient. Further studies are required to clarify the association between RGD uptake and post-infarct prognosis. Moreover, the most favorable time point for investigation of RGD uptake post infarct remains unsettled.
Interpretation of RGD uptake is not limited to ischemic heart disease; post-stroke repair processes after ischemic injury are subject to neurovascular research. Tumor angiogenesis has been well recognized as an essential hall mark of tumor growth, invasion and metastasis and, therefore, RGD PET can be used for tumor detection and staging in different types of cancer to provide additional information to existing imaging modalities [
24,
25]. A number of other RGD peptide PET tracers have been clinically investigated including
18F-Galacto-RGD,
18F-Fluciclatide,
18F-RGD-K5,
18F-FPPRGD
2,
18F-Alfatide,
68Ga-NOTA-RGD and
68Ga-NOTA-PRGD
2 [
25]. The detection efficiency of lesions in different regions is highly dependent on the difference in in vivo biodistribution patterns of each single RGD peptide.