A High-Efficiency Approach for the Synthesis of N235-Impregnated Resins and the Application in Enhanced Adsorption and Separation of Vanadium(V)
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of SIRs
2.3. Static Adsorption Experiments
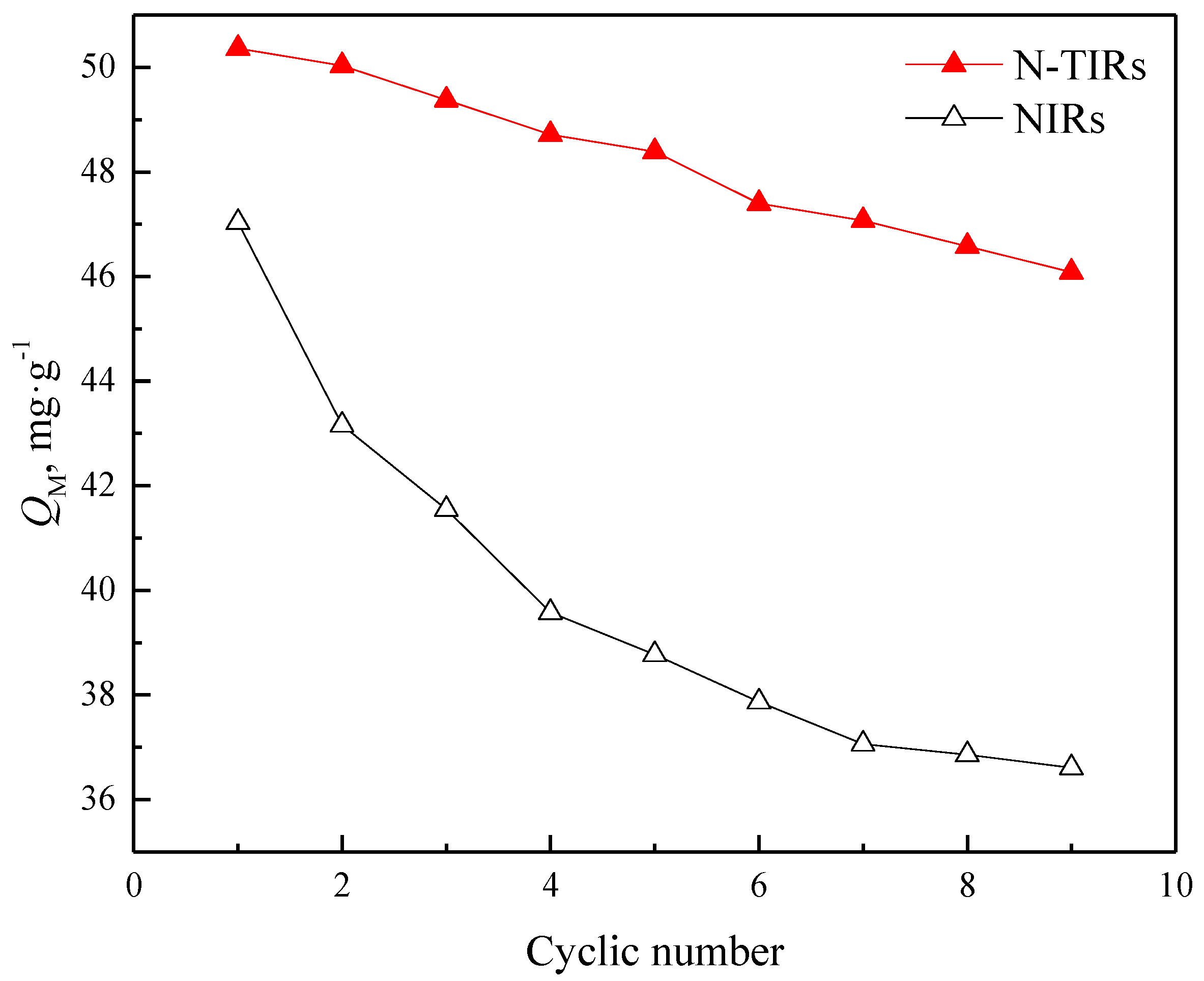
2.4. Cyclic Adsorption Experiments
2.5. Analytical and Testing Methods
3. Results and Discussion
3.1. Effects of Impregnation Conditions on the N-TIRs
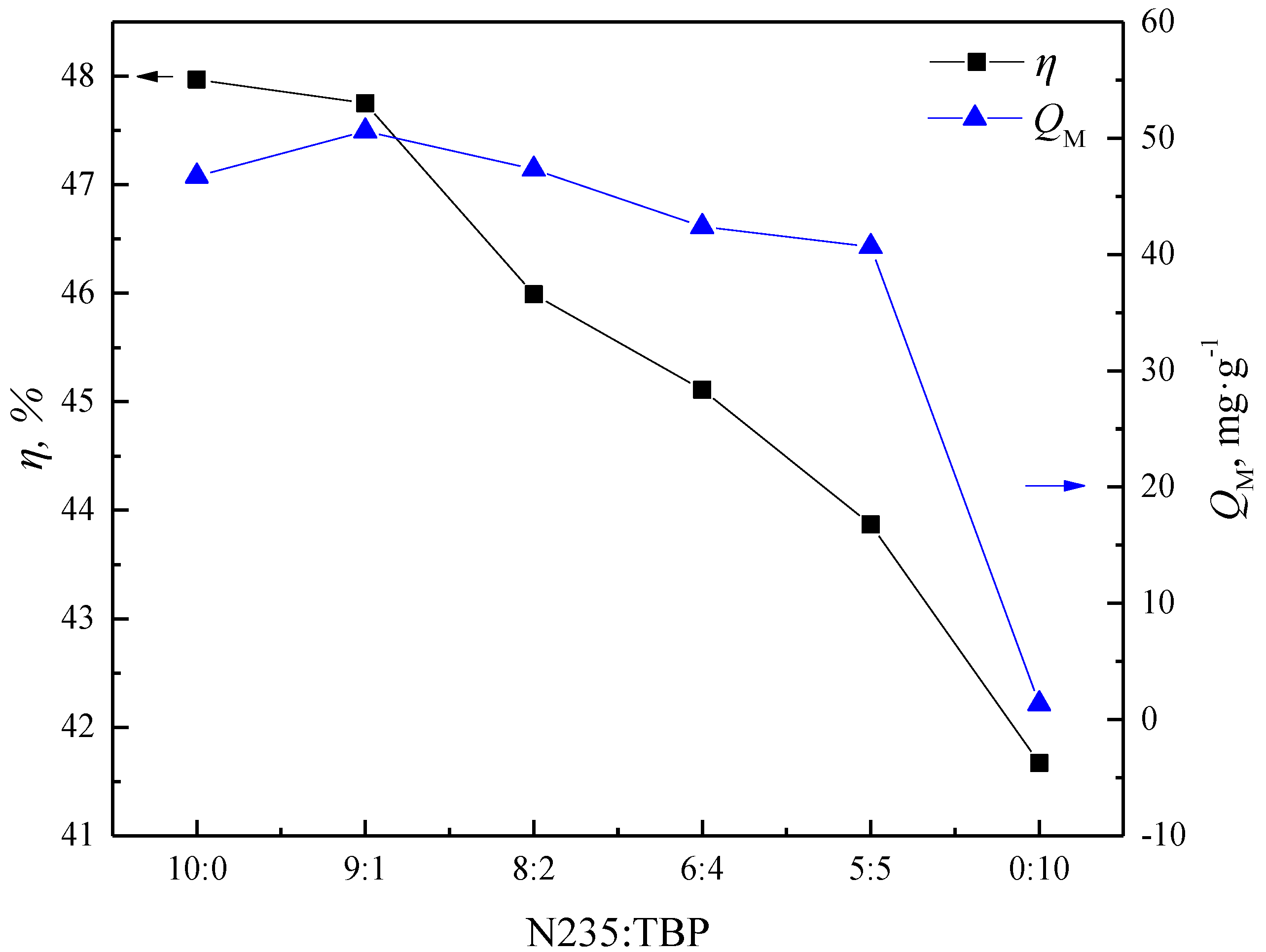
3.1.1. Effects of the Used Mixtures
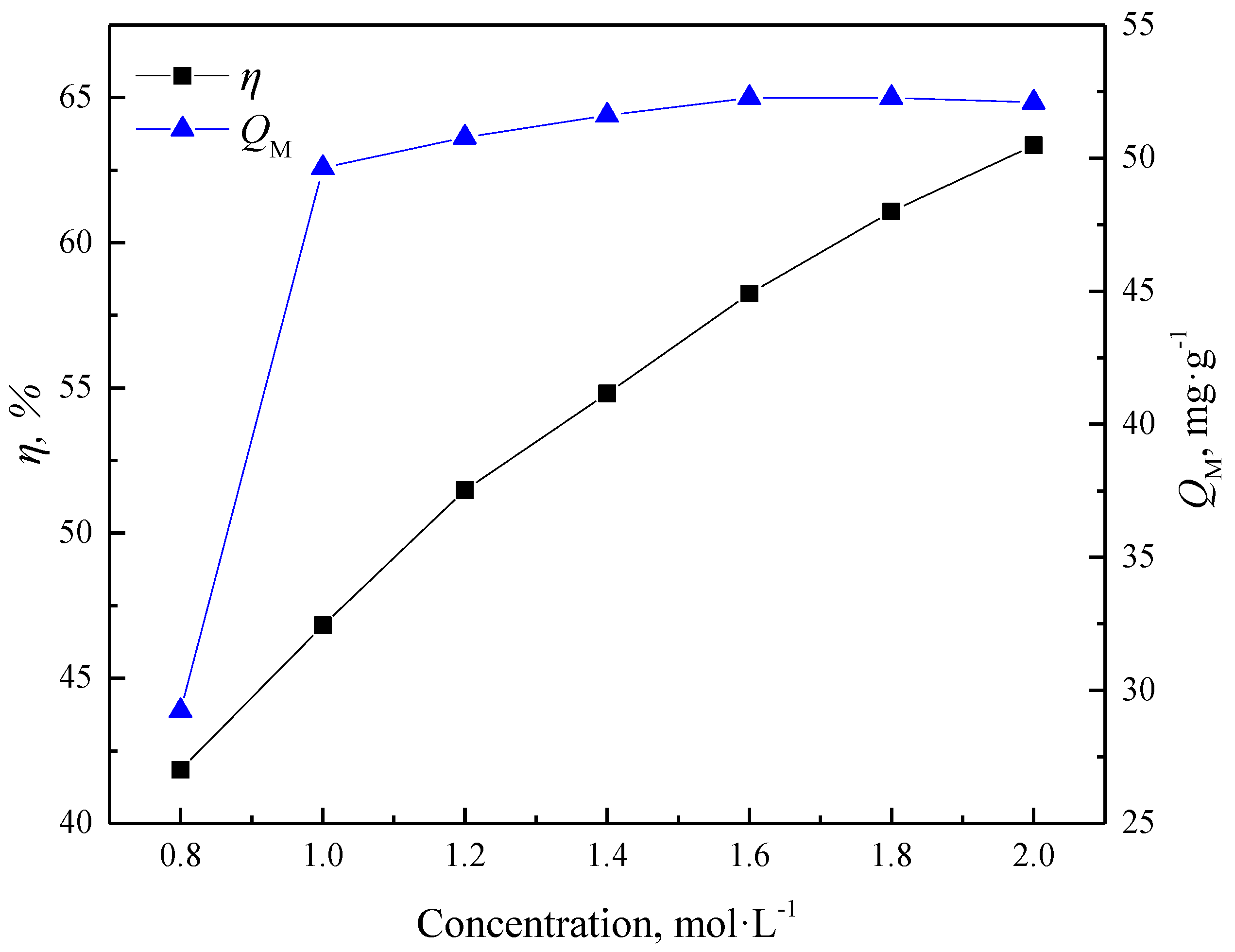
3.1.2. Effects of Extractants Concentration
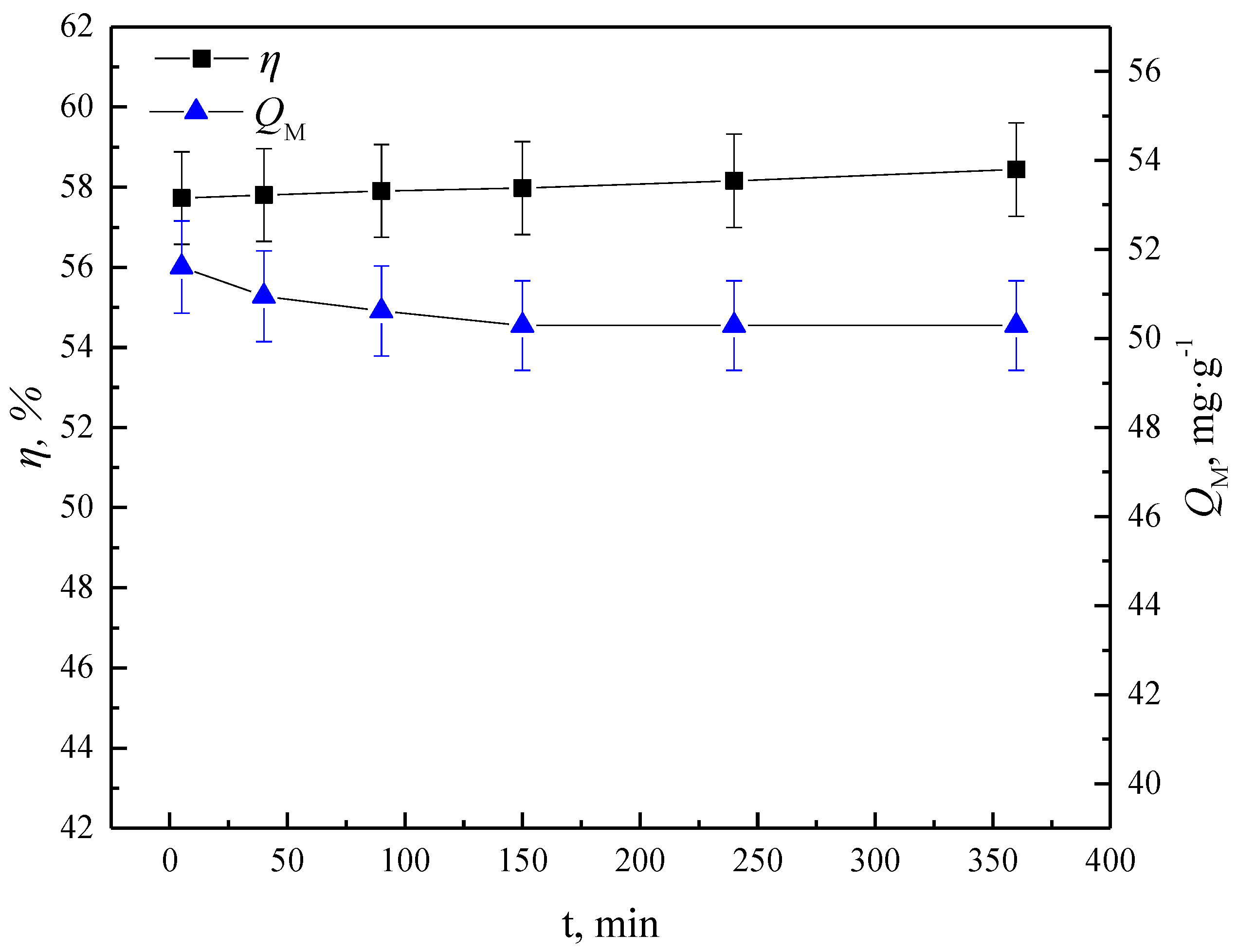
3.1.3. Effects of Impregnation Time
3.2. Characterizations of N-TIRs and NIRs
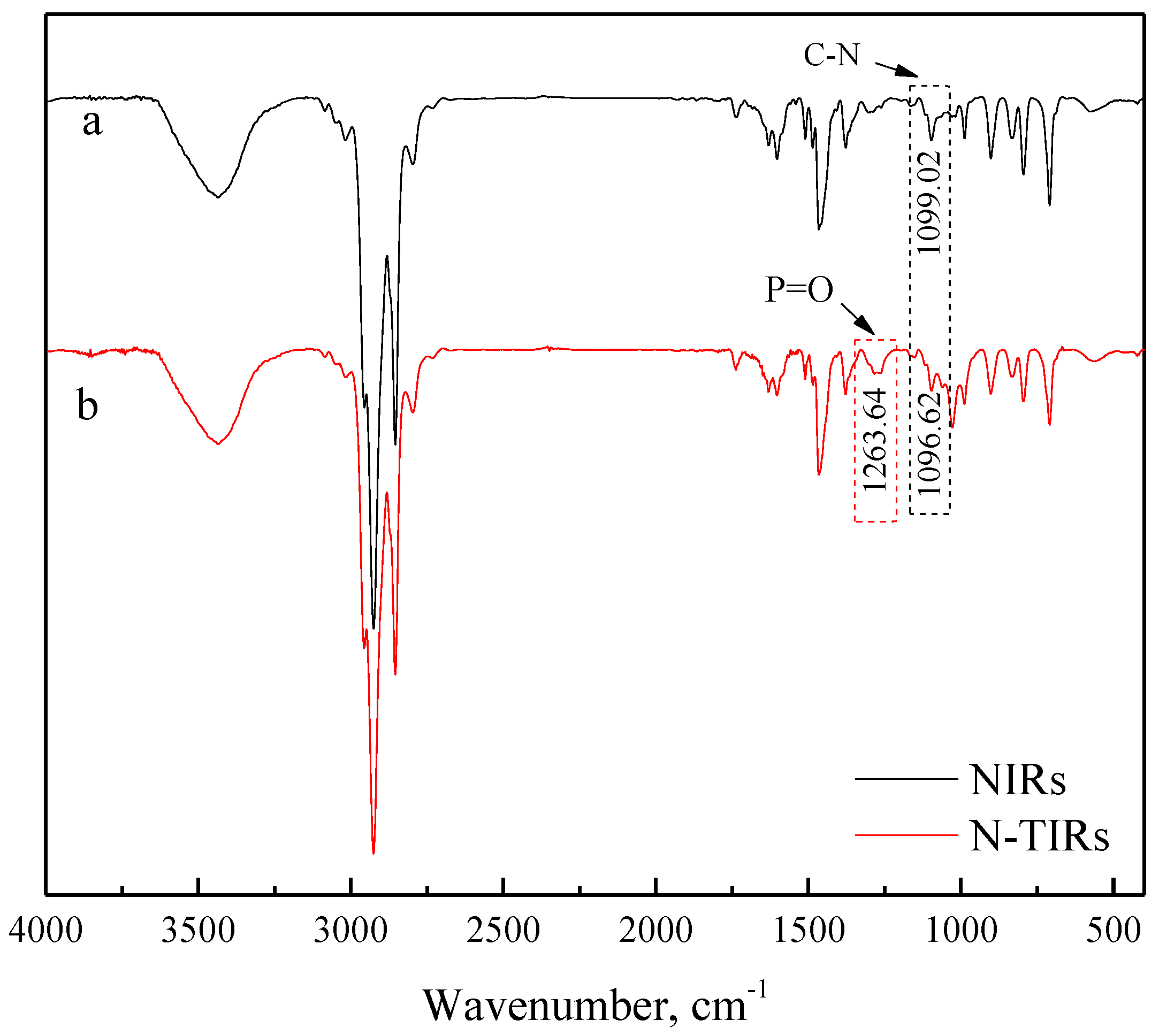
3.2.1. FTIR Analyses
3.2.2. SEM-EDS Analyses
3.3. Adsorption of V(V) onto the N-TIRs and NIRs
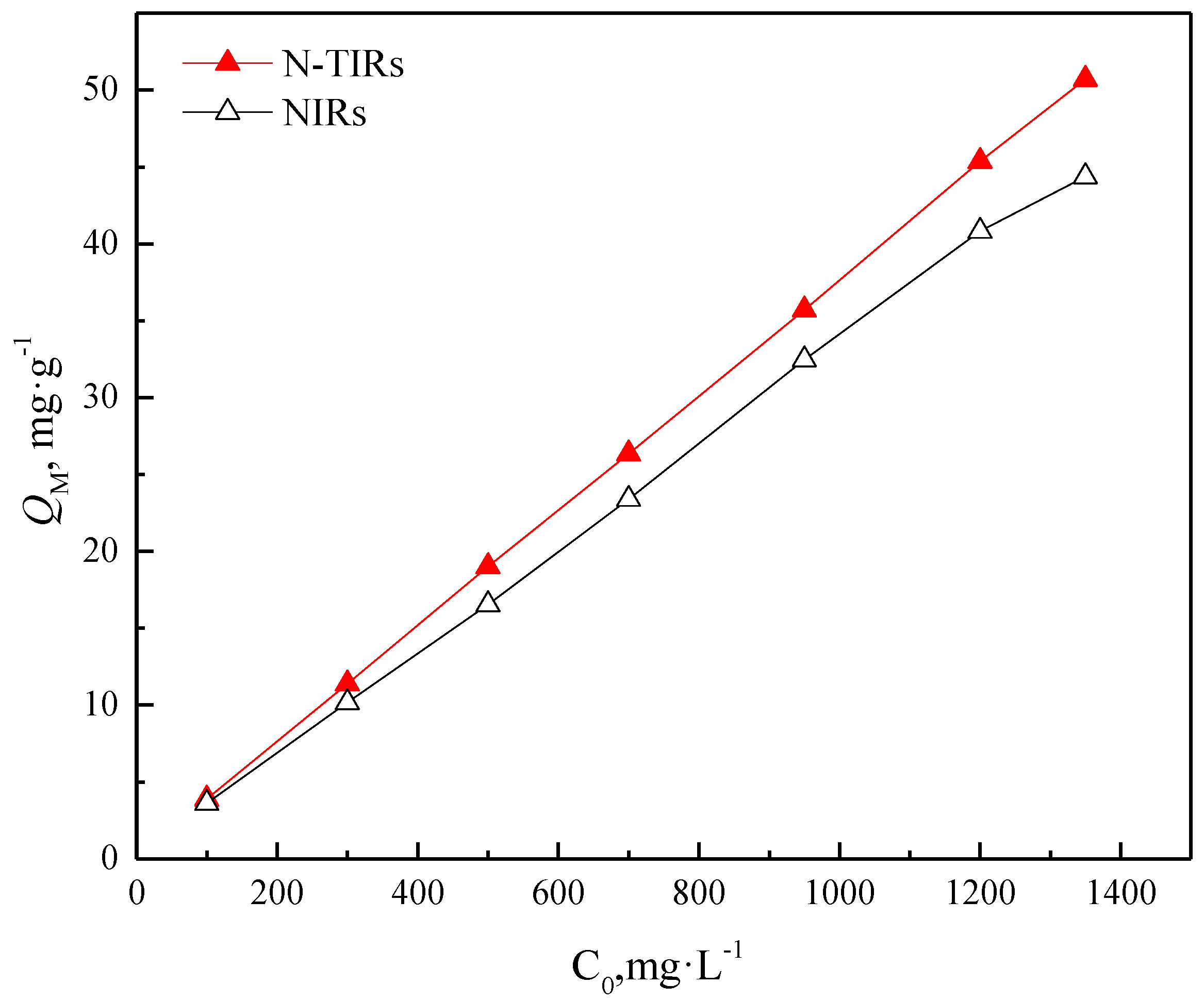
3.3.1. Effects of Initial V(V) Concentration
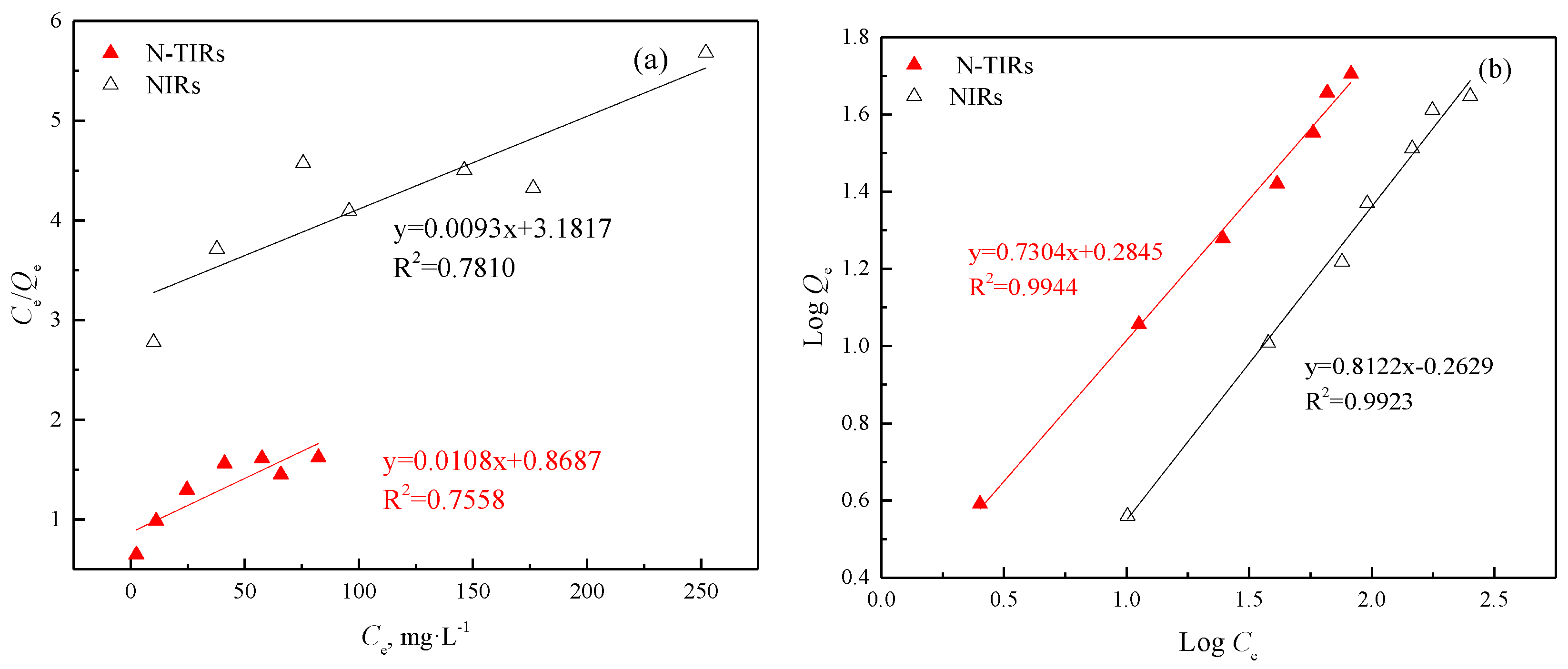
3.3.2. Adsorption Isotherm
3.3.3. Effects of Adsorption Time
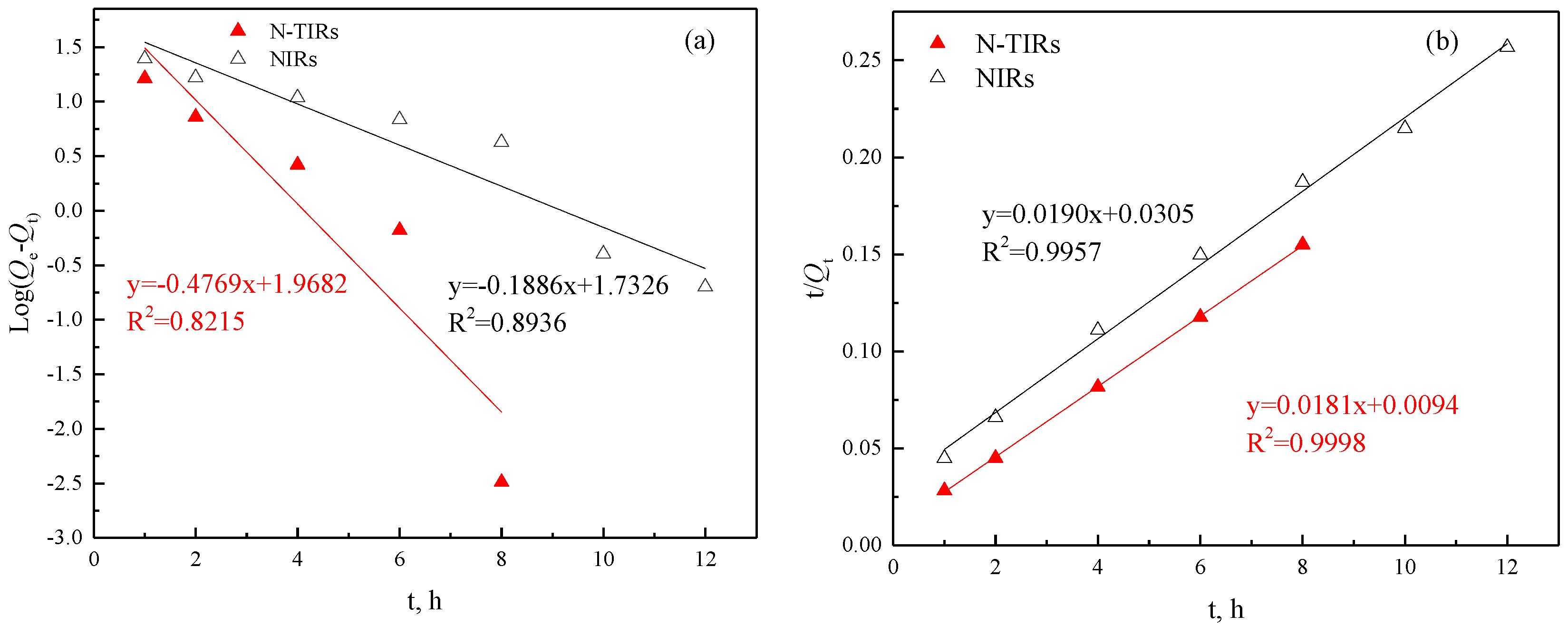
3.3.4. Adsorption Kinetics
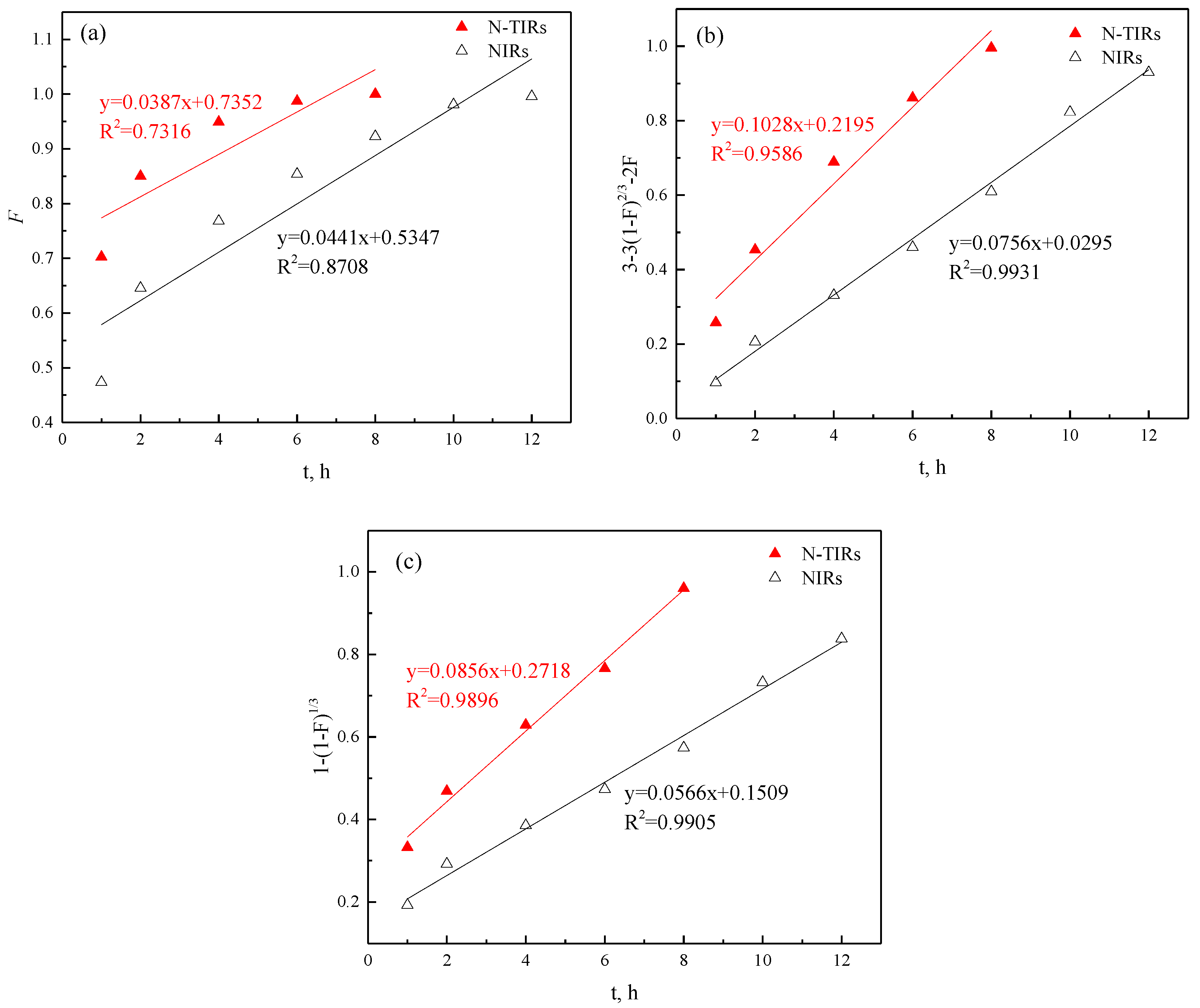
- (a)
- When the fluid film diffusion controls:
- (b)
- when the intraparticle diffusion controls:
- (c)
- when the chemical reaction controls:
3.4. Separation of V(V) from Complex Solution
3.5. Stability of the SIRs
4. Conclusions
- (1)
- The addition of TBP can accelerate the impregnation rate and improve the impregnation ratio of N235 onto the macroporous resins. The impregnation equilibrium time of N-TIRs can be significantly shortened to 5 min in comparison to that of NIRs (240 min) and the impregnation ratio of N-TIRs (57.73%) is higher than that of the NIRs (36.95%).
- (2)
- FTIR analyses demonstrate that N235 can interact with TBP during the impregnation process, resulting in the improvement of the extractability of N235, which enhances the adsorption capacity and the separation capability of the N-TIRs for V(V) when compared with that of the NIRs (50.95 mg·g−1 versus 46.73 mg·g−1). It is confirmed by SEM-EDS analyses that more extractants can enter deeper into the pores of the resins with the addition of TBP, thus the stability of the N-TIRs is higher than that of the NIRs during the cyclic use.
- (3)
- The kinetics results show that the pseudo-second order model fits better for the adsorption process of V(V) onto the N-TIRs and NIRs, and the chemical reaction is the rate-limiting step of the adsorption process of V(V). The reaction constant (Ks) implies that the reaction rate of V(V) with extractants impregnated onto N-TIRs is faster than that onto NIRs, which results in the shorter adsorption equilibrium time of the NTIRs (6 h) when compared to NIRs (10 h).
Author Contributions
Funding
Conflicts of Interest
References
- Saitoh, T.; Suzuki, S.; Hiraide, M. Solid phase extraction of some precious metals from hydrochloric acid to polystyrene-divinylbenzene porous resin impregnated with polyoxyethylene-type nonionic surfactant. J. Chromatogr. A 2005, 1097, 179–182. [Google Scholar] [CrossRef] [PubMed]
- Lee, G.S.; Uchikoshi, M.; Mimura, K.; Isshiki, M. Separation of major impurities Ce, Pr, Nd, Sm, Al, Ca, Fe, and Zn from La using bis(2-ethylhexyl) phosphoric acid (D2EHPA)-impregnated resin in a hydrochloric acid medium. Sep. Purif. Technol. 2010, 71, 186–191. [Google Scholar] [CrossRef]
- Metwally, S.S.; Hassan, M.A.; Aglan, R.F. Extraction of copper from ammoniacal solution using impregnated amberlite XAD-7 resin loaded with LIX-54. J. Environ. Chem. Eng. 2013, 1, 252–259. [Google Scholar] [CrossRef]
- Babić, K.; Ham, L.V.D.; Haan, A.D. Recovery of benzaldehyde from aqueous streams using extractant impregnated resins. React. Funct. Polym. 2006, 66, 1494–1505. [Google Scholar] [CrossRef]
- Burghoff, B.; Schiferli, J.; Marques, J.S.; Haan, A.B.D. Extractant screening and selection for methyl tert-butyl ether removal from aqueous streams. Chem. Eng. Sci. 2009, 64, 2887–2892. [Google Scholar] [CrossRef]
- Liang, L.; Bao, S.; Zhang, Y.; Tang, Y. Separation and recovery of V(IV) from sulfuric acid solutions containing Fe(III) and Al(III) using bis(2-ethylhexyl)phosphoric acid impregnated resin. Chem. Eng. Res. Des. 2016, 111, 109–116. [Google Scholar] [CrossRef]
- Tang, Y.; Bao, S.; Zhang, Y.; Liang, L. Effect of support properties on preparation process and adsorption performances of solvent impregnated resins. React. Funct. Polym. 2017, 113, 50–57. [Google Scholar] [CrossRef]
- Warshawsky, A. South African Patent 71/5637, 1971.
- Grinstead, R. On Contract NO. 14-12-808 to the Water Quality Office of US Environmental Protection Administration; Reported by the Dow Chemical Co.: Midland, Michigan, USA, January 1971. [Google Scholar]
- Kroebel, R.; Meyer, A. West German Patent 2,162,951, 1971.
- Bao, S.; Tang, Y.; Zhang, Y.; Liang, L. Recovery and Separation of Metal Ions from Aqueous Solutions by Solvent–Impregnated Resins. Chem. Eng. Technol. 2016, 39, 1377–1392. [Google Scholar] [CrossRef]
- Navarro, R.; Saucedo, I.; Núñez, A.; Ávila, M.; Guibal, E. Cadmium extraction from hydrochloric acid solutions using Amberlite XAD-7 impregnated with Cyanex 921 (tri-octyl phosphine oxide). React. Funct. Polym. 2008, 68, 557–571. [Google Scholar] [CrossRef]
- Ma, Y.; Wang, X.; Wang, M.; Jiang, C.; Xiang, X.; Zhang, X. Separation of V(IV) and Fe(III) from the acid leach solution of stone coal by D2EHPA/TBP. Hydrometallurgy 2015, 153, 38–45. [Google Scholar] [CrossRef]
- Cheraghi, A.; Ardakani, M.S.; Alamdari, E.K.; Fatmesari, D.H.; Darvishi, D.; Sadrnezhaad, S.K. Thermodynamics of vanadium (V) solvent extraction by mixture of D2EHPA and TBP. Int. J. Miner. Process. 2015, 138, 49–54. [Google Scholar] [CrossRef]
- Li, X.; Deng, Z.; Wei, C.; Li, C.; Li, M.; Fan, G.; Huang, H. Solvent extraction of vanadium from a stone coal acidic leach solution using D2EHPA/TBP: Continuous testing. Hydrometallurgy 2015, 154, 40–46. [Google Scholar] [CrossRef]
- Alamdari, E.K.; Darvishi, D.; Sadrnezhaad, S.K.; Haghshenas, D.F. Synergistic effect of TBP on separation of zinc and cadmium with D2EHPA. In Proceedings of the International Solvent Extraction Conference (ISEC), Beijing, China, 19–23 September 2005. [Google Scholar]
- Zheng, R.; Bao, S.; Zhang, Y.; Chen, B. Synthesis of Di-(2-ethylhexyl) phosphoric acid (D2EHPA)-Tributyl phosphate) (TBP) Impregnated Resin and Application in Adsorption of Vanadium(IV). Minerals 2018, 8, 206. [Google Scholar] [CrossRef]
- Chen, B.; Bao, S.; Zhang, Y. Preparation of N235-impregnated resin and its adsorption for vanadium. Chin. J. Rare Met. 2017, 42, 891–896. [Google Scholar]
- Shen, C.; Zhang, Y.; Bao, S.; Huang, J.; Yang, X. Separation and enrichment of vanadium from stone coal acidic leach solution using tertiary amine N235. Chin. J. Rare. Met. 2017, 41, 422–428. [Google Scholar]
- Chen, B.; Bao, S.; Zhang, Y.; Zheng, R. Ultrasound-assisted synthesis of N235-impregnated resins for vanadium (V) adsorption. R. Soc. Open Sci. 2018, 5, 171746. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Wei, C.; Deng, Z.; Li, M.; Li, C.; Fan, G. Selective solvent extraction of vanadium over iron from a stone coal/black shale acid leach solution by D2EHPA/TBP. Hydrometallurgy 2011, 105, 359–363. [Google Scholar] [CrossRef]
- Baaden, M.; Burgard, M.; Wipff, A.G. TBP at the water-oil interface: The effect of TBP concentration and water acidity investigated by molecular dynamics simulations. J. Phys. Chem. B 2001, 105, 11131–11141. [Google Scholar] [CrossRef]
- Fatmehsari, D.H.; Darvishi, D.; Etemadi, S.; Hollagh, A.R.E.; Alamdari, E.K.; Salardini, A.A. Interaction between TBP and D2EHPA during Zn, Cd, Mn, Cu, Co and Ni solvent extraction: A thermodynamic and empirical approach. Hydrometallurgy 2009, 98, 143–147. [Google Scholar] [CrossRef]
- Nguyen, N.V.; Lee, J.C.; Jeong, J.; Pandey, B.D. Enhancing the adsorption of chromium(VI) from the acidic chloride media using solvent impregnated resin (SIR). Chem. Eng. J. 2013, 219, 174–182. [Google Scholar] [CrossRef]
- Nguyen, N.V.; Lee, J.; Jha, M.K.; Yoo, K.; Jeong, J. Copper recovery from low concentration waste solution using Dowex G-26 resin. Hydrometallurgy. 2009, 97, 237–242. [Google Scholar] [CrossRef]
- Ho, Y.S.; Mckay, G. The kinetics of sorption of divalent metal ions onto sphagnum moss peat. Water. Res. 2000, 34, 735–742. [Google Scholar] [CrossRef]
- Cortina, J.L.; Miralles, N. Kinetic studies on heavy metal ions removal by impregnated resins containing di-(2,4,4-trymethylpentyl) phosphinic acid. Solvent. Extr. Ion Exc. 1997, 15, 1067–1083. [Google Scholar] [CrossRef]




| Parameters | Value |
|---|---|
| Pore volume | 1.82 mL·g−1 |
| Specific surface area | ≥800 m2·g−1 |
| Average pore diameter | 15 nm |
| Particle size | 0.425–0.850 mm |
| Matrix | Phenyl ethylene-divinyl benzene |
| SIRs | Langmuir | Freundlich | ||||
|---|---|---|---|---|---|---|
| Q0 | KL | R2 | n | KF | R2 | |
| N-TIRs | 92.59 | 0.0124 | 0.7558 | 1.37 | 1.9253 | 0.9944 |
| NIRs | 107.53 | 0.0029 | 0.7810 | 1.23 | 0.5459 | 0.9923 |
| SIRs | Pseudo-First Order | Pseudo-Second Order | ||||
|---|---|---|---|---|---|---|
| Qe | K1 | R2 | Qe | K2 | R2 | |
| N-TIRs | 7.16 | 0.4769 | 0.8251 | 55.25 | 0.0349 | 0.9998 |
| NIRs | 5.66 | 0.1886 | 0.8936 | 52.63 | 0.0118 | 0.9957 |
| SIRs | R2 | KS | ||
|---|---|---|---|---|
| Fluid Film Diffusion | Intraparticle Diffusion | Chemical Reaction | ||
| N-TIRs | 0.7316 | 0.9586 | 0.9896 | 2.06 × 10−3 |
| NIRs | 0.8708 | 0.9931 | 0.9905 | 1.36 × 10−3 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, B.; Bao, S.; Zhang, Y.; Zheng, R. A High-Efficiency Approach for the Synthesis of N235-Impregnated Resins and the Application in Enhanced Adsorption and Separation of Vanadium(V). Minerals 2018, 8, 358. https://doi.org/10.3390/min8080358
Chen B, Bao S, Zhang Y, Zheng R. A High-Efficiency Approach for the Synthesis of N235-Impregnated Resins and the Application in Enhanced Adsorption and Separation of Vanadium(V). Minerals. 2018; 8(8):358. https://doi.org/10.3390/min8080358
Chicago/Turabian StyleChen, Bo, Shenxu Bao, Yimin Zhang, and Ruwei Zheng. 2018. "A High-Efficiency Approach for the Synthesis of N235-Impregnated Resins and the Application in Enhanced Adsorption and Separation of Vanadium(V)" Minerals 8, no. 8: 358. https://doi.org/10.3390/min8080358






