1. Introduction
Titanosilicates constitute an important group of minerals that have found many applications as materials, including ion-exchange, sorption, catalysis, optics, biocide technologies, etc. [
1,
2,
3,
4,
5]. Of particular interest are layered titanosilicates of the seidozerite supergroup that currently contains more than forty-five mineral species [
6] with several new minerals described very recently [
7,
8,
9,
10,
11,
12,
13]. These species occur mainly in alkaline massifs such as those in Kola Peninsula, Russia, above the Polar circle. Belov and Organova [
14] were the first who considered the crystal chemistry of several minerals of the current murmanite group of seidozerite supergroup [
6]. Belov [
15] and Pyatenko et al. [
16] made further generalizations and called minerals with a “seidozerite block” (=TS block, [
17]) and astrophyllite-group minerals titanosilicate analogues of micas. The modular approach to these minerals has been developed by Egorov-Tismenko and Sokolova [
18,
19], who described a homologous series of Ti-analogues of micas, and Ferraris [
20,
21,
22], who named those minerals heterophyllosilicates and described them as members of a single polysomatic series. Sokolova [
17] quantitatively divided TS-block minerals into four groups based on the content of Ti, structural topology and stereochemistry of the TS block.
Shkatulkalite, Na
10MnTi
3Nb
3(Si
2O
7)
6(OH)
2F·12H
2O, was described by Menshikov et al. [
23] from the pegmatite “Shkatulka” of the Lovozero alkaline massif, Kola Peninsula, Russia. The mineral was considered to be a Ti–Nb–sorosilicate of the “epistolite group” (now considered a part of the lamprophyllite group [
24]). In their review on the seidozerite-supergroup minerals, Sokolova and Cámara [
6] pointed out that shkatulkalite is a potential member of the supergroup, but the final assignment of the mineral to a particular group remained unclear, due to the fact that its crystal structure was unknown until now. Menshikov et al. [
23] established that the mineral is monoclinic,
a = 5.468(9),
b = 7.18(1),
c = 31.1(1) Å,
β = 94.0(2)°,
V = 1218(8) Å
3, Z = 1, and commented on the proximity of the
a and
b parameters of shkatulkalite to those typical for other known Ti and Nb sorosilicates. On the basis of systematic absences, the space groups
Pm,
P2,
P2/
m were proposed as possible for the mineral. However, due to the poor quality of single-crystal X-ray diffraction data, the structure of the mineral could not be solved at the time. Németh et al. [
25] investigated syntactic intergrowths of epistolite, murmanite and shkatulkalite using transmission electron microscopy (TEM) and selected-area electron diffraction (SAED), and reported on the absence of the
l = 2
n + 1 reflections for the latter mineral, pointing out that its
c parameter is halved with respect to the value of 31.1 Å reported by Menshikov et al. [
23]. Later, shkatulkalite was described in nepheline syenites of the alkaline sill of St. Amable, Quebec, Canada [
26], as forming prismatic crystals and radial intergrowth of crystals in small miarolic voids as well as in a hydrothermal cavity in the southeastern part of the Demix quarry. The authors [
26] noted that the shkatulkalite is visually indistinguishable from vuonnemite and epistolite found in the same voids. At the same time, Menshikov et al. [
23] pointed out that shkatulkalite sometimes forms pseudomorphs after vuonnemite and thus can be considered as a transformation mineral species [
27,
28], that is, mineral species that forms as a result of a secondary transformation of a primary proto-phase. However, this hypothesis could not be confirmed until the crystal structure of the mineral is solved.
The aim of the present paper is to report the results of crystal-structure determination of shkatulkaite and to re-consider its status as both seidozerite-supergroup mineral and transformation mineral species.
3. Results
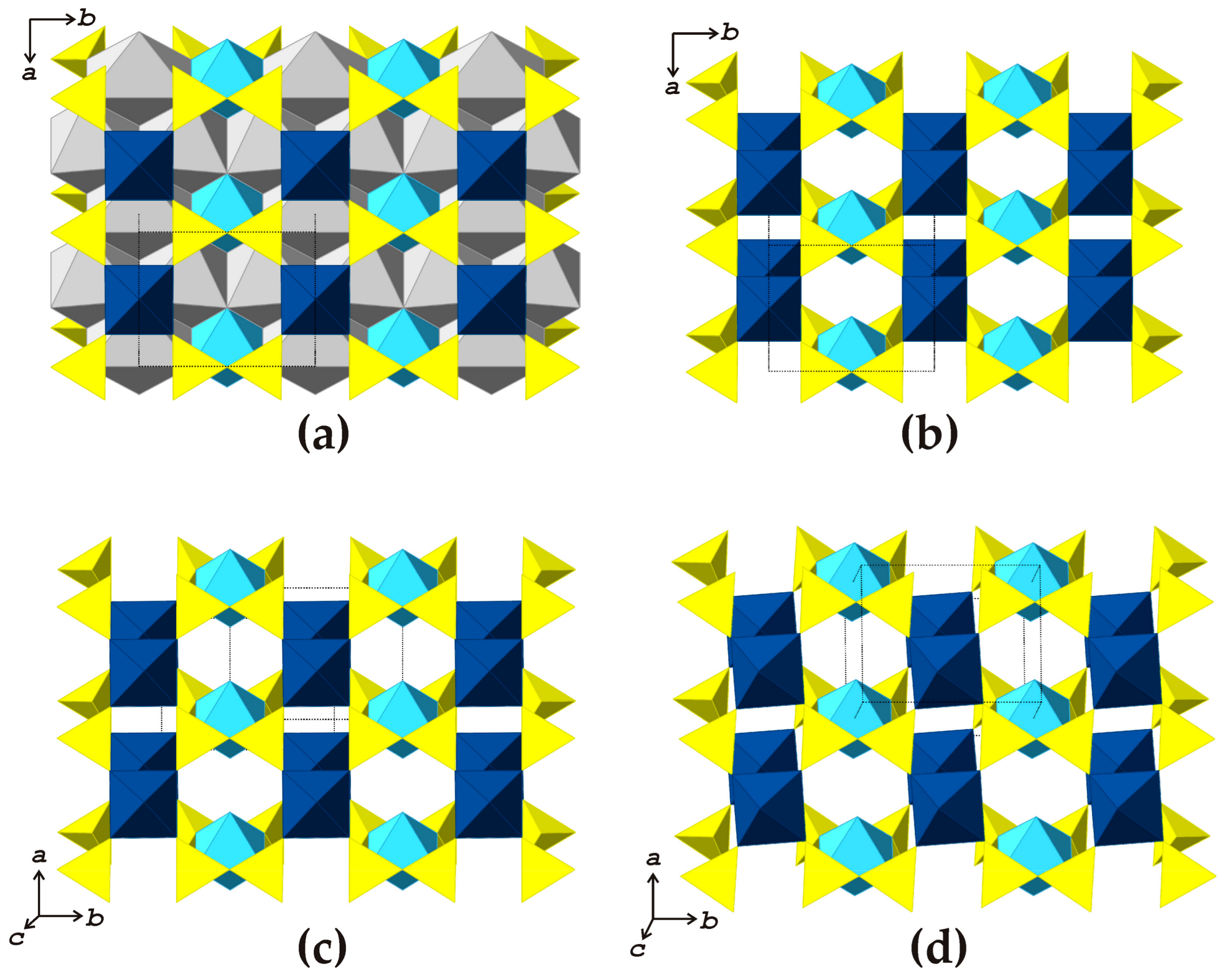
The crystal structure of shkatulkalite is based upon the
HOH blocks consisting of one octahedral (
O) sheet sandwiched between two heteropolyhedral (
H) sheets (
Figure 2a). According to Sokolova [
17], the
HOH blocks are of the type III with one Ti
apfu present in the
O sheet (
Figure 2b). The blocks are parallel to (001) and are separated from each other (
Figure 3a) with interlayer space occupied by
Na1 atoms and H
2O groups. The
Na2,
Na3, and
Ti sites are located within the
O sheet. The
H sheets are formed by
NbO
6 octahedra and Si
2O
7 groups sharing common O atoms.
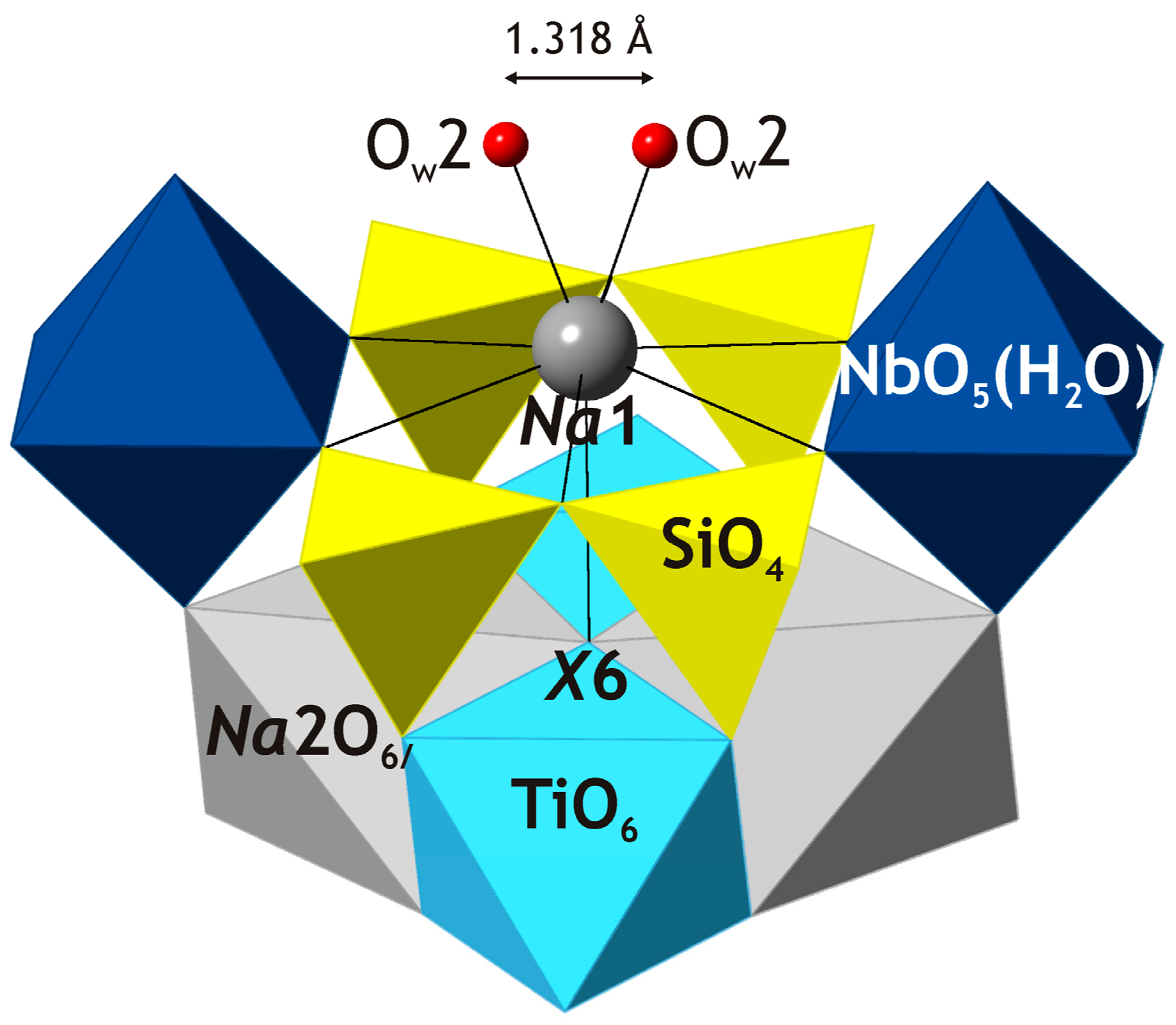
The
Na1 site is coordinated by eight anions (taking into account the disorder observed for the O
w2 site with adjacent O
w2 sites located at 1.318 Å) and is located approximately in the center of a six-membered ring in the
H layer (
Figure 4). The relatively high coordination number (8) and the long average <
Na1–O> bond length of 2.534 Å indicate the capability of this site to accumulate large cations such as K
+, Sr
2+ and Ba
2+ present in shkatulkalite. The structure refinement indicated the presence of rather short
Na1–O
w2 contact of 2.01 Å, which we explain by the static disorder observed for both
Na1 and O
w2 sites (note also the high values of their atomic displacement parameters). The
Na2 and
Na3 sites are octahedrally coordinated by six anions each. The
Na2O
6 octahedron is more compact, with the mean <
Na2–O> bond length of 2.342 Å, the bond-valence sum (BVS) of 1.40 valence units (v.u.) and the site-scattering factor (SSF) of 12.54
e− all pointing out that, in addition to Na
+, this site also incorporates Ca
2+ and Mn
2+ cations (
Table 3). The refinement of the SSF of the
Na3 site is consistent with its full occupancy by Na
+ cations, which is also confirmed by its BVS (1.06 v.u.) and the <
Na3–O> bond length of 2.458 Å.
The
Nb and
Ti sites are both octahedrally coordinated by O atoms. The
Nb site contains significant amount of Ti (its refined SSF corresponds to the composition Nb
0.75Ti
0.25). The
NbO
6 octahedron is essentially distorted with one short (1.770 Å)
Nb–O5 bond opposite to one long
Nb–O
w1 (2.29 Å) bond, and four intermediate (~1.98 Å)
Nb–O bonds. This kind of octahedral distortion is typical for NbO
6 octahedra in
H sheets and was observed, for instance, in vuonnemite, Na
11TiNb
2(Si
2O
7)
2(PO
4)
2O
3F [
35,
36], and epistolite, Na
4TiNb
2(Si
2O
7)
2O
2(OH)
2(H
2O)
4 [
37], two minerals most closely related to shkatulkalite (see below). The refined SSF of the
Ti site is close to 22
e−, which would account for the full occupancy of this site by Ti. However, this would contradict the predominance of Nb over Ti observed in the chemical analyses (
Table 1), which prompted us to suggest that the
Ti site also accommodates Mn
2+ cations present in shkatulkalite. It is rather common for divalent cations and, in particular, Mn
2+ to substitute for Ti in the octahedral sites of the
O sheet in heterophyllosilicates. Such a substitution was reported, for instance, for sobolevite, Na
13Ca
2Mn
2Ti
3(Si
2O
7)
2(PO
4)
4O
3F
3 [
38,
39].
The BVS for the
X6 site in shkatulkalite is 1.22 v.u., which is compatible with its occupancy by (OH)
− or F
− anions. The (OH)
0.82F
0.18 assigned to this site in
Table 3 was calculated to conform with the results of the chemical analyses that demonstrate the presence in shkatulkalite of 0.36 F
apfu.
The interlayer between the adjacent HOH blocks in shkatulkalite is occupied by a number of partially occupied Ow sites that belong to H2O molecules. The Ow1 atom is bonded to the Nb site, forming a long apical Nb-H2O bond in the NbO6 octahedra. The Ow2 site is linked to the Na1 site and is split into two sites, with the total occupancy of 68%. The Ow3–Ow8 sites are purely interlayer with the occupancies in the range from 19% to 92%. However, there are several H2O positions that are mutually excluding (Ow4–Ow5, Ow6–Ow7, etc.). The maximum amount of H2O in the formula considering all incompatible sites is 10 H2O per formula unit. Therefore, the amount of H2O molecules in the ideal formula can be written as n, where n ≤ 10. It seems that the cohesion among different TS-blocks is ensured through hydrogen bonding between the H2O molecule of the Ow1 site (apical anion of NbO6 octahedra) and the H2O molecule of the Ow8 site. However, no precise picture of the hydrogen bonding in shkatulkalite can be derived, owing to the high degree of disorder observed for the interlayer sites.
According to the crystal-structure refinement, the crystal-chemical formula of shkatulkalite can be written as [Na
3.94☐
0.50Sr
0.14Mn
0.14Ba
0.12Ca
0.12K
0.04][Nb
1.50Ti
1.27Mn
2+0.20☐
0.03][Si
2O
7]
2O
2 [(OH)
0.82F
0.18]
2·7.43H
2O, which is in very general agreement with the empirical chemical formula given above, taking into account the difficulties associated with the study of the mineral by the electron-microprobe analysis. It is noteworthy that, according to the chemical analyses, the H
2O content in shkatulkalite ranges from 4.32 to 6.67 apfu. Thus, by analogy to selivanovaite and murmanite [
11,
25] the interlayer H
2O content in shkatulkalite is variable and may be defined as
nH
2O. Taking into account the cations prevalent at different atomic sites in shkatulkalite, its ideal chemical formula should be written as Na
5Nb
2Ti[Si
2O
7]
2O
2(OH)
2]·
nH
2O, where
n ≤ 10, which is not electroneutral and contains one extra positive charge. Therefore, we assume that the incorporations of Ti into
Nb and Mn into
Ti sites are important for charge-balance considerations, and suggest the general formula should be written as Na
5(Nb
1−xTi
x)
2(Ti
1−yMn
2+y)[Si
2O
7]
2O
2(OH)
2·
nH
2O, where
x +
y = 0.5,
n ≤ 10. For the sample of shkatulkalite under investigation x ≈ y ≈ 0.25. For the two critical cases of
x = 0.5 or
y = 0.5, the end-member formulae would be Na
5(NbTi)Ti[Si
2O
7]
2O
2(OH)
2·
nH
2O and Na
5Nb
2(Ti
0.5Mn
2+0.5)[Si
2O
7]
2O
2(OH)
2·
nH
2O (or Na
10Nb
4TiMn
2+[Si
2O
7]
4O
4(OH)
4·
nH
2O),
n ≤ 10, respectively.
4. Discussion
The structure of shkatulkalite represents the basic structure type B5(GIII), according to Sokolova and Cámara [
40] that was inferred by inverse prediction. In fact, Sokolova and Cámara [
40] introduced the concept of basic and derivative structures for TS-block minerals and stated that a derivative structure is related to two or more basic structures of the same group. Hence a derivative structure can be built by adding basic structures
via sharing the central O sheet of the TS blocks of adjacent structural fragments. The inverse prediction of the structure of shkatulkalite to B5(GIII) is now completely confirmed with the present results. Incidentally, the ideal formula of B5(GII) predicted by Sokolova and Cámara [
40] is □
2Nb
2Na
2M
2+Ti (Si
2O
7)
2O
2(OH)
2(H
2O)
8 (with M
2+ = Mn, Ca), while the proposed ideal formula by us for shkatulkalite is Na
5(Nb
1−xTi
x)
2(Ti
1−yMn
2+y)[Si
2O
7]
2O
2(OH)
2·nH
2O, where x + y = 0.5 and n ≤ 10, which is a remarkable agreement. It is worth noting that Cámara et al. [
41] have already confirmed the right prediction of B7(GIV) structure type with the crystal structure of kolskyite.
In agreement with the suggestions by Németh et al. [
25] and Sokolova and Cámara [
6], shkatulkalite belongs to the seidozerite supergroup, since its structure is based upon the
HOH [
22] or TS [
17] blocks. More precisely, shatulkalite is a member of the lamprophyllite group, having Ti + (Nb + Mn) = 3
apfu, from which the
H and
O sheets has 2 and 1 (Ti + Nb + Mn)
apfu, respectively. The most closely related species to shkatulkalite are vuonnemite, Na
11TiNb
2(Si
2O
7)
2(PO
4)
2O
3F [
35,
36], and epistolite, Na
4TiNb
2(Si
2O
7)
2O
2(OH)
2(H
2O)
4 [
37,
38], crystal structures of which are shown in
Figure 3b,c, respectively. The three minerals are based upon the same topological type of the
HOH blocks (
Figure 2b–d) with different layer symmetries (
p2/
m in shkatulkalite, and
p − 1 in vuonnemite and epistolite). In all three minerals, the
HOH blocks are parallel to (001), but the
c parameters are different and equal to 15.573, 14.450 and 12.041 Å for shkatulkalite, vuonnemite and epistolite, respectively. In fact, both shkatulkalite and epistolite can be considered derivatives of vuonnemite and can be obtained from the latter at least through the
gedanken experiment by removing the some Na
+ and all (PO
4)
3− ions and subsequent hydration of the interlayer space. The hypothesis that shkatulkalite is a transformation mineral species that forms at the expense of vuonnemite (or some vuonnemite-related proto-mineral) seems quite reasonable, taking into account its high hydration state, which results in the very open packing of adjacent
HOH blocks that is manifested in the large value of the
c parameter and can be clearly seen in
Figure 3a. In contrast, in the crystal structure of epistolite (
Figure 3b), the packing of the
HOH blocks is quite dense, resulting in the shrinkage of the
c parameter. The syntactic intergrowths of shkatulkalite and epistolite reported by Németh et al. [
25] may point out to the following growth scenarios: (1) both minerals are primary phases that crystallize simultaneously; (2) both minerals are transformation species that form according to the sequence “vuonnemite (or vuonnemite-related proto-mineral) → shkatulkalite → epistolite”; or (3) both minerals are transformation species that form along two different pathways, “vuonnemite → shkatulkalite” and “vuonnemite → epistolite”. The reported observations of pseudomorphs of shkatulkalite after vuonnemite suggest that, at least in some environments, shkatulkalite is indeed a transformation mineral species that inherits basic structural features from vuonnemite in accordance with Khomyakov’s structural inheritance principle [
42]. The secondary nature and the status of shkatulkalite as a transformation mineral species may also account for the difficulties encountered when investigating the mineral by means of electron microprobe and crystal-structure analysis. The absence of precise agreement between the chemical and structural studies (the obvious cation deficiency observed in three series of independent chemical analyses and in the original report [
23]) remains an issue that we cannot resolve at the present time.