The Activation Mechanism of Bi3+ Ions to Rutile Flotation in a Strong Acidic Environment
Abstract
:1. Introduction
2. Materials and Methods
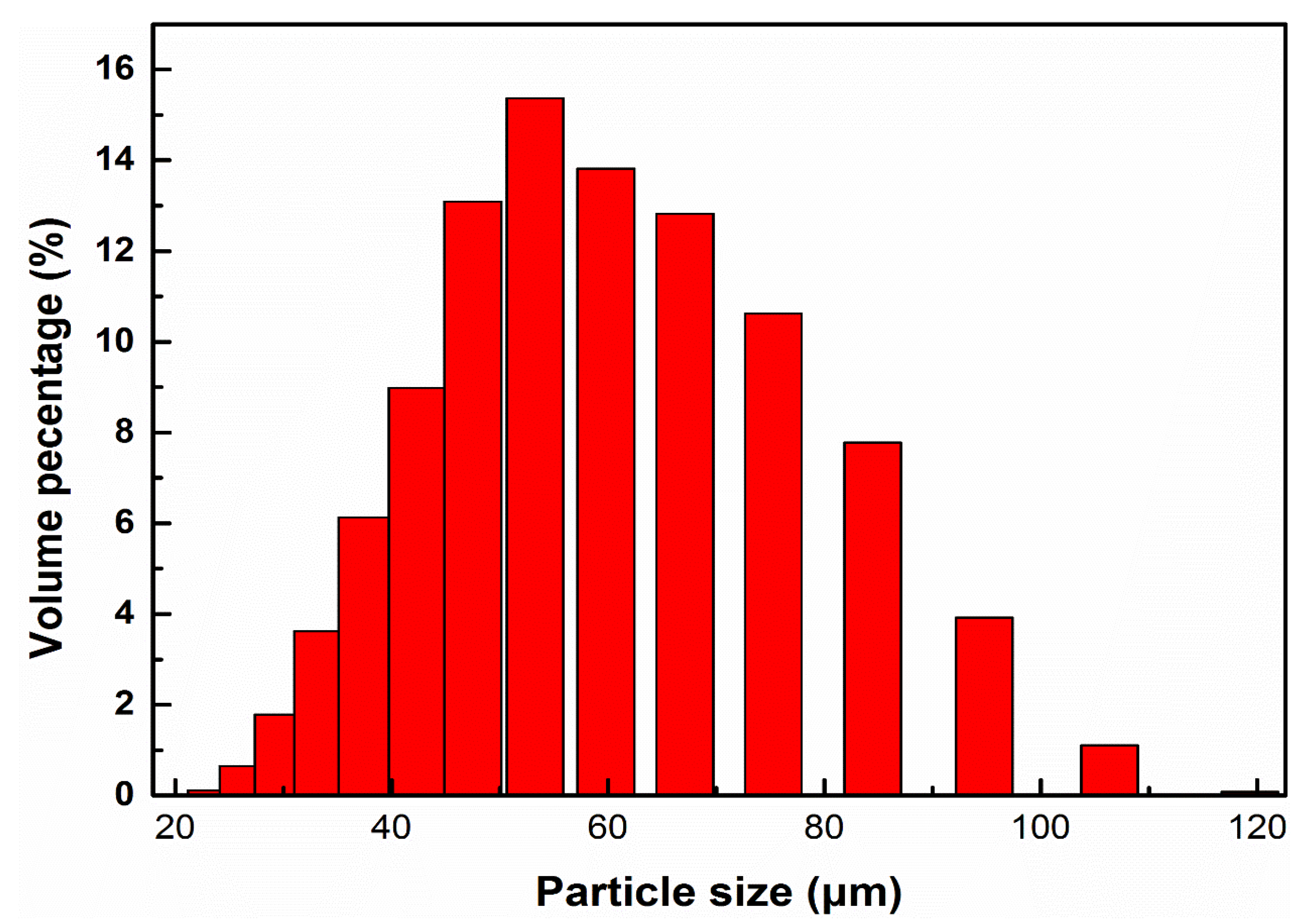
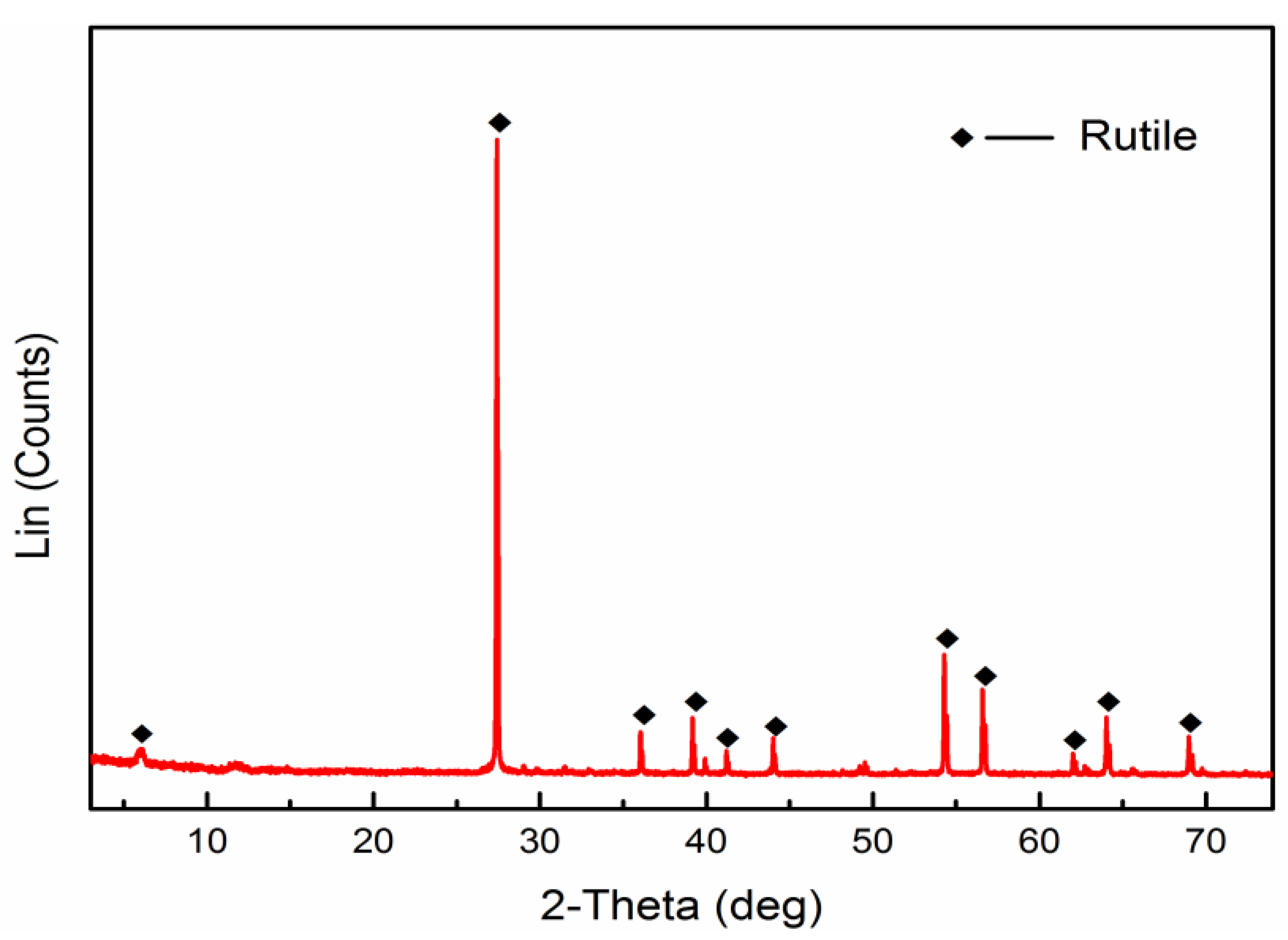
2.1. Materials
2.2. Methods
2.2.1. Micro-Flotation Tests
2.2.2. Adsorption Experiments
2.2.3. Zeta Potential Measurements
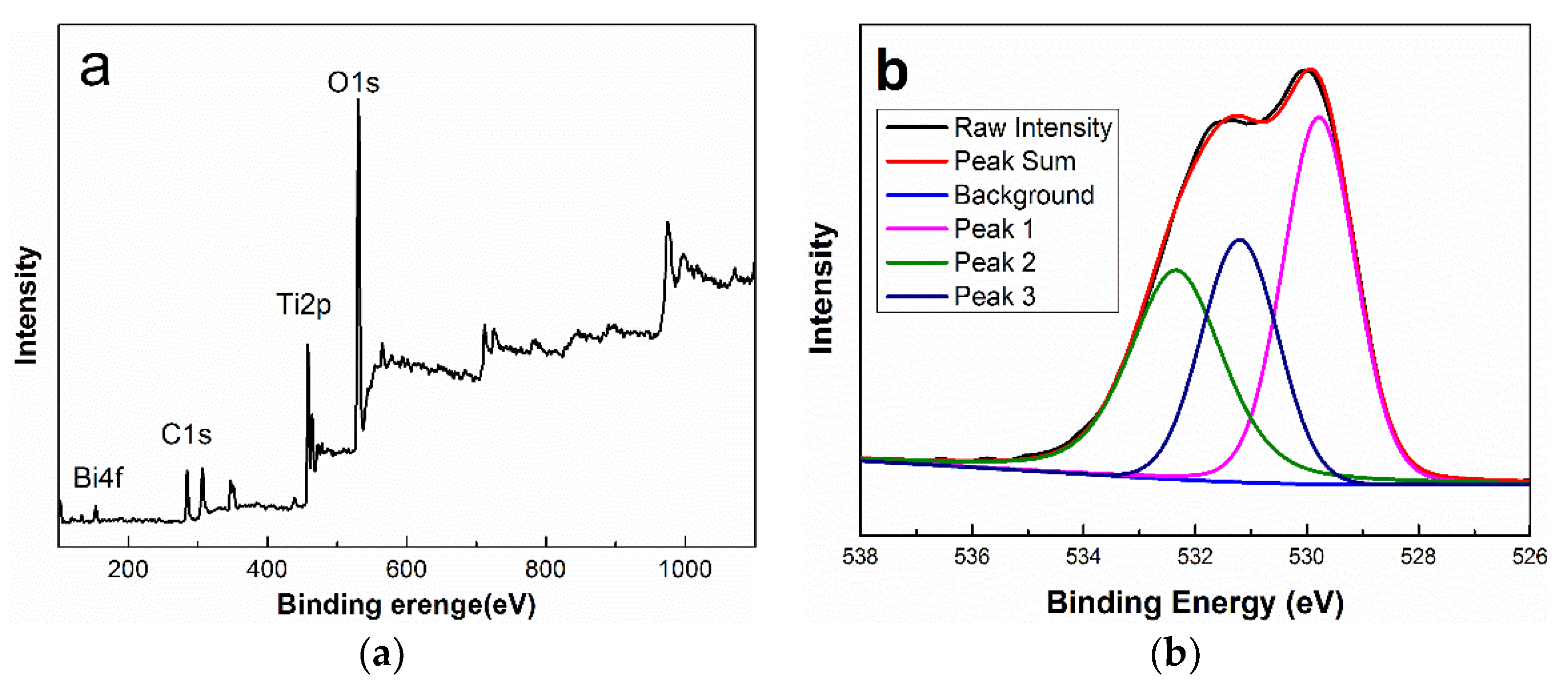
2.2.4. X-ray Photoelectron Spectroscopy
3. Results and Discussion
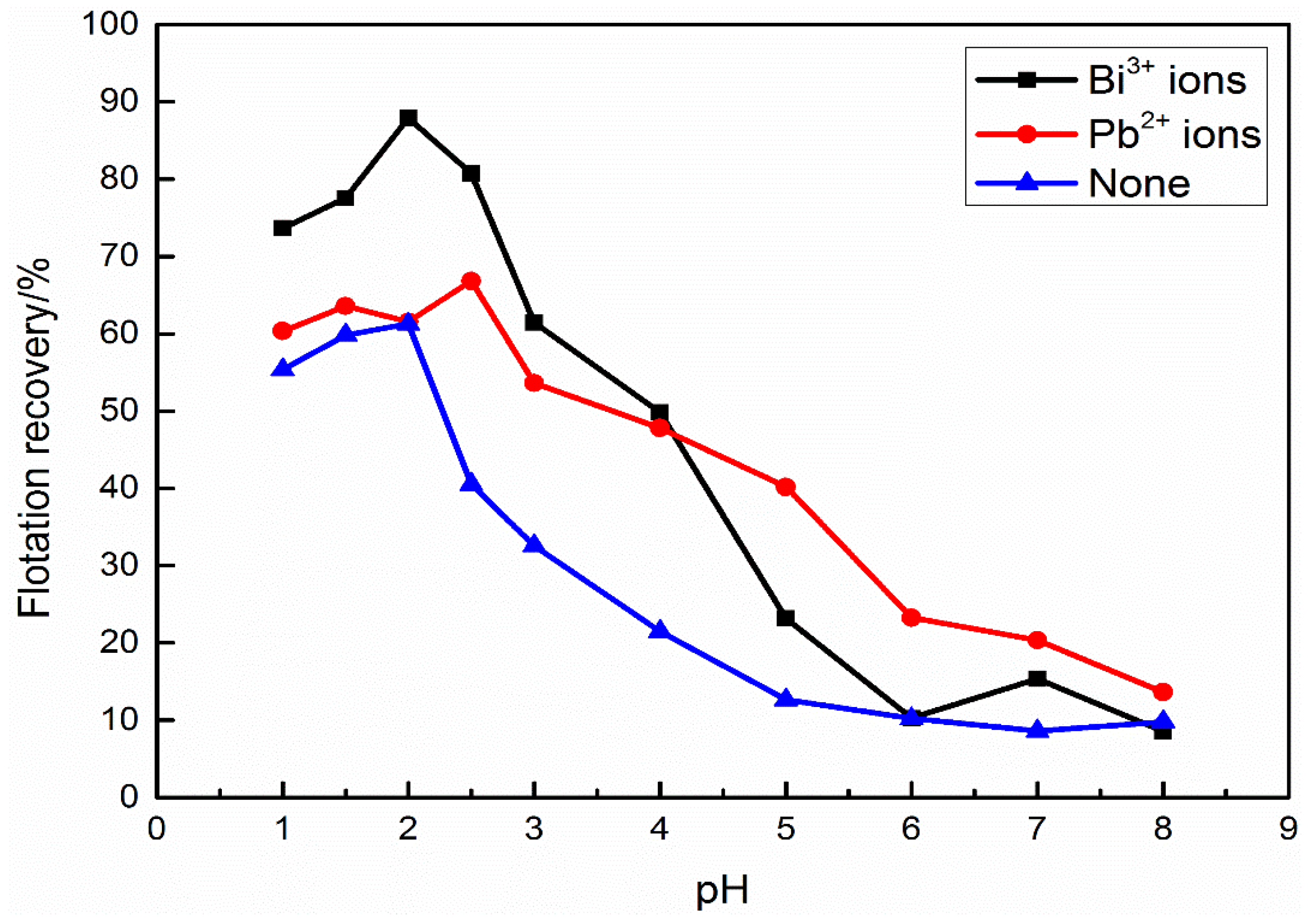
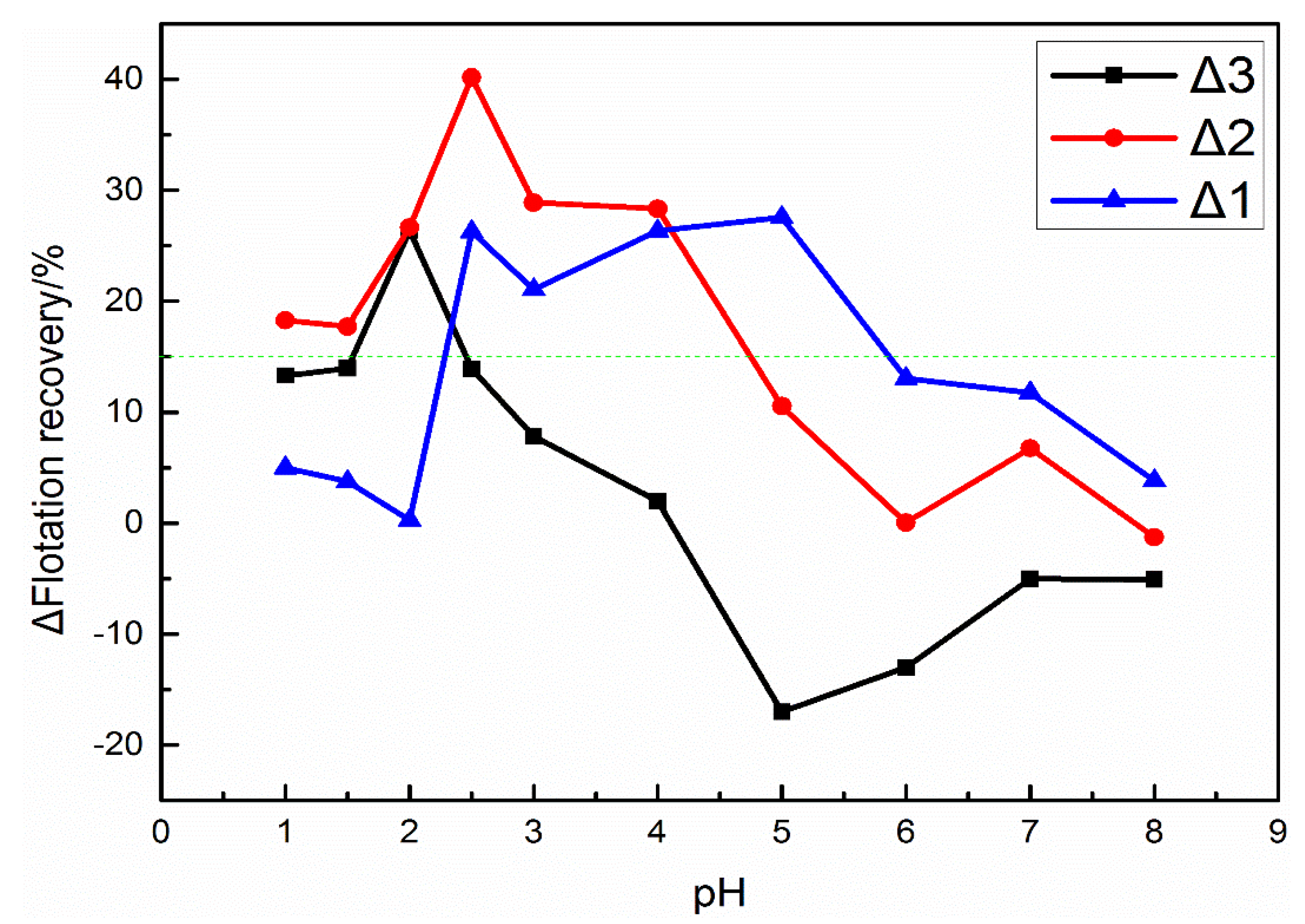
3.1. Micro-Flotation Tests
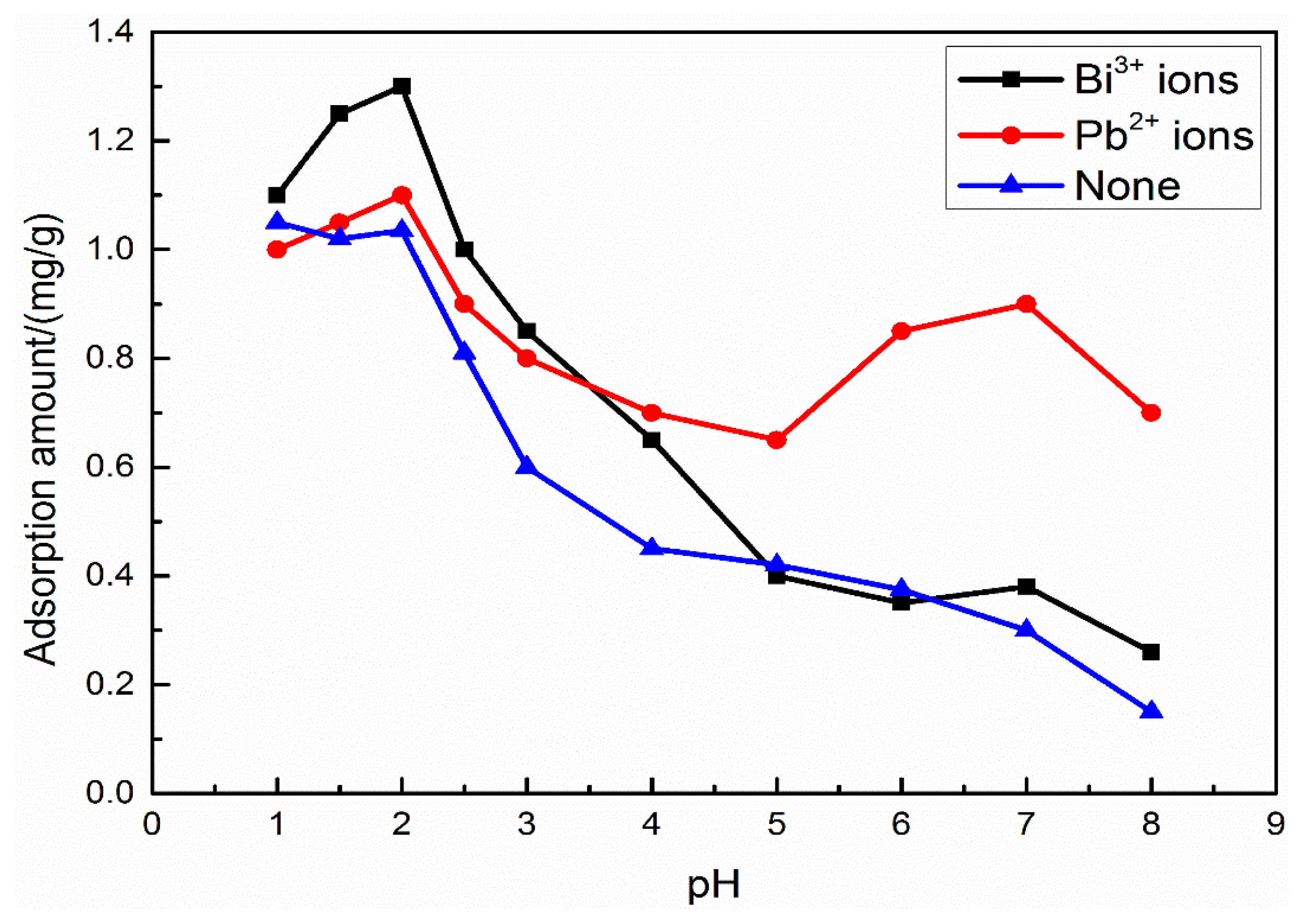
3.2. Adsorption Amount of SPA
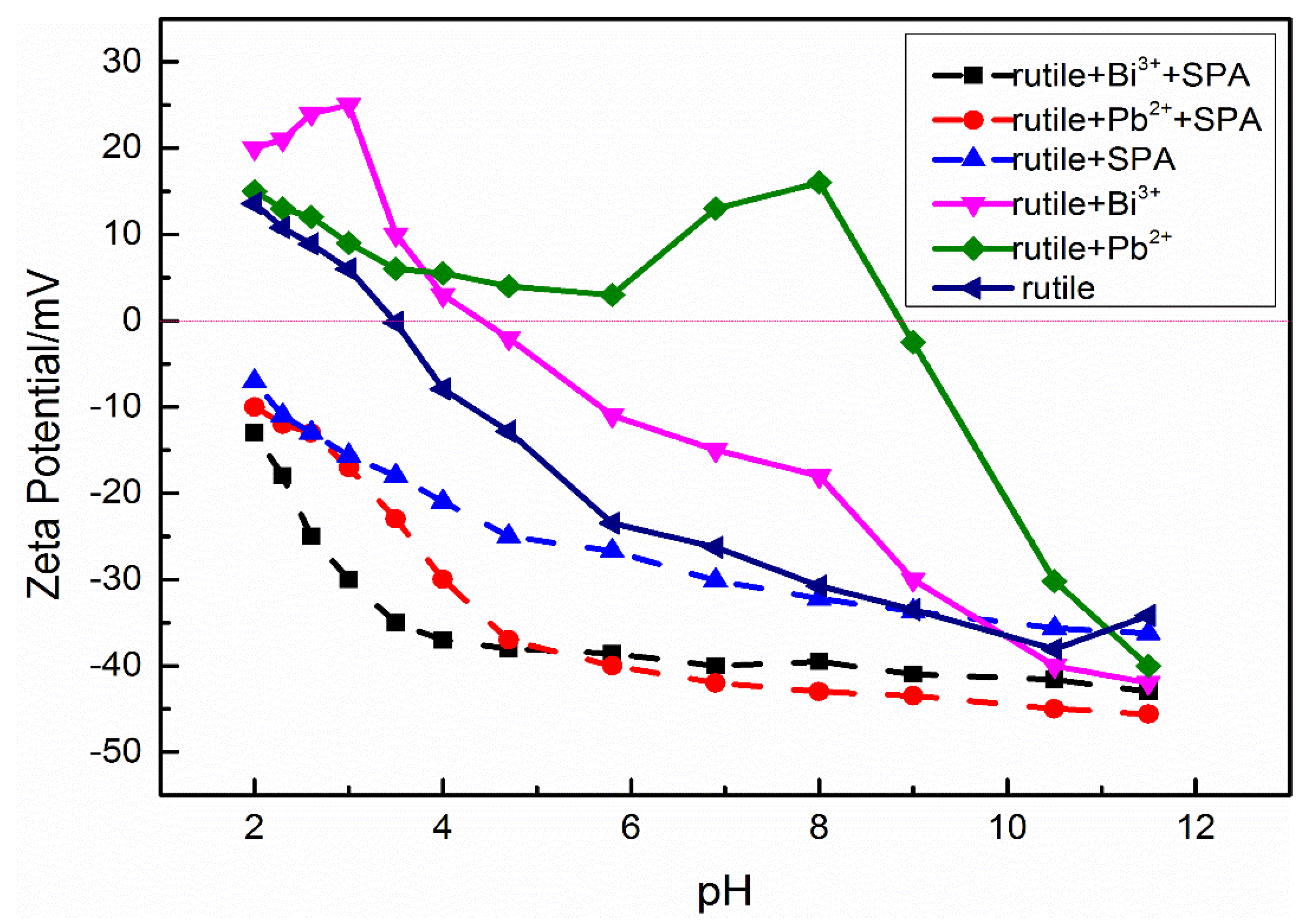
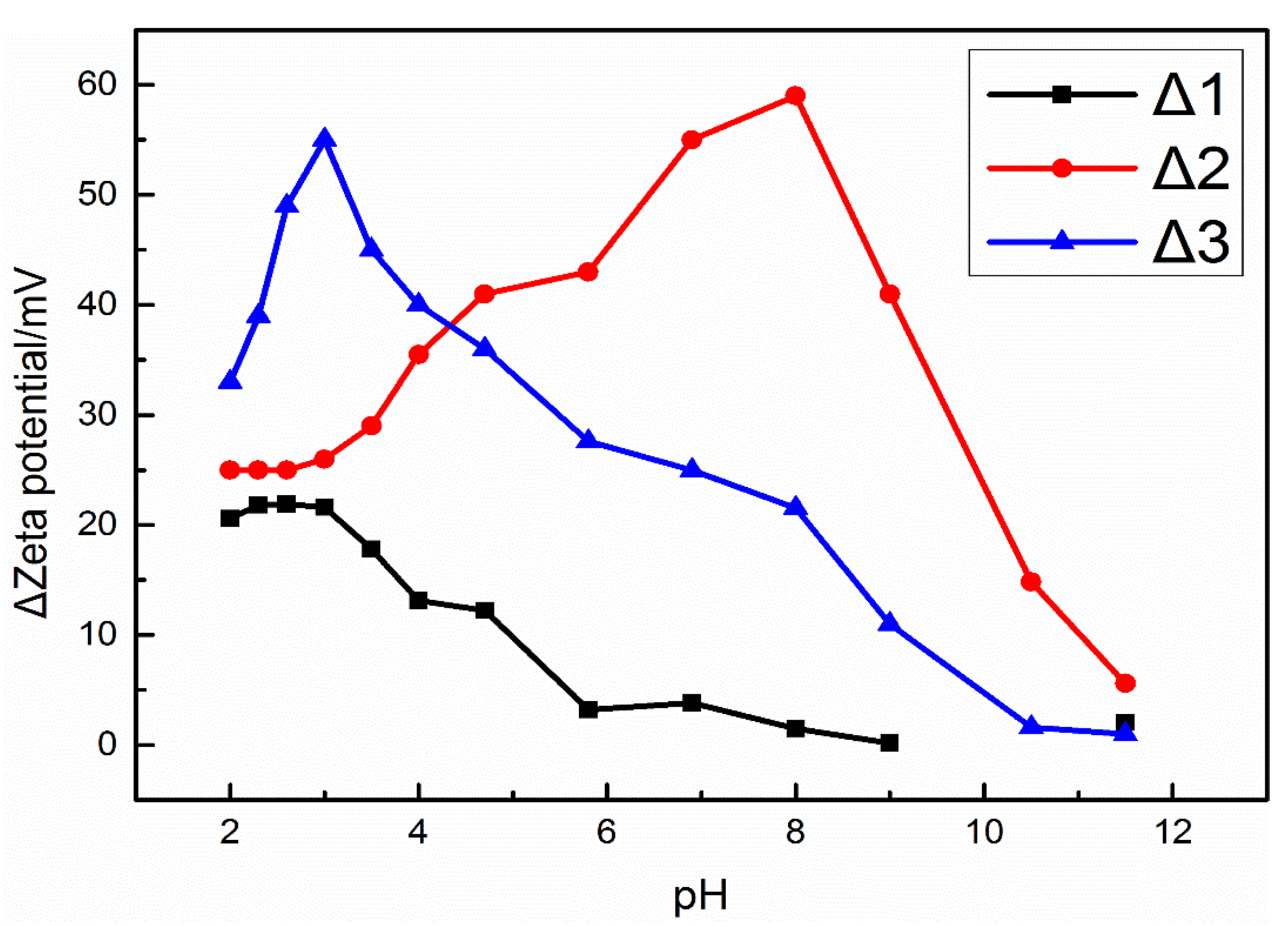
3.3. Zeta Potentials
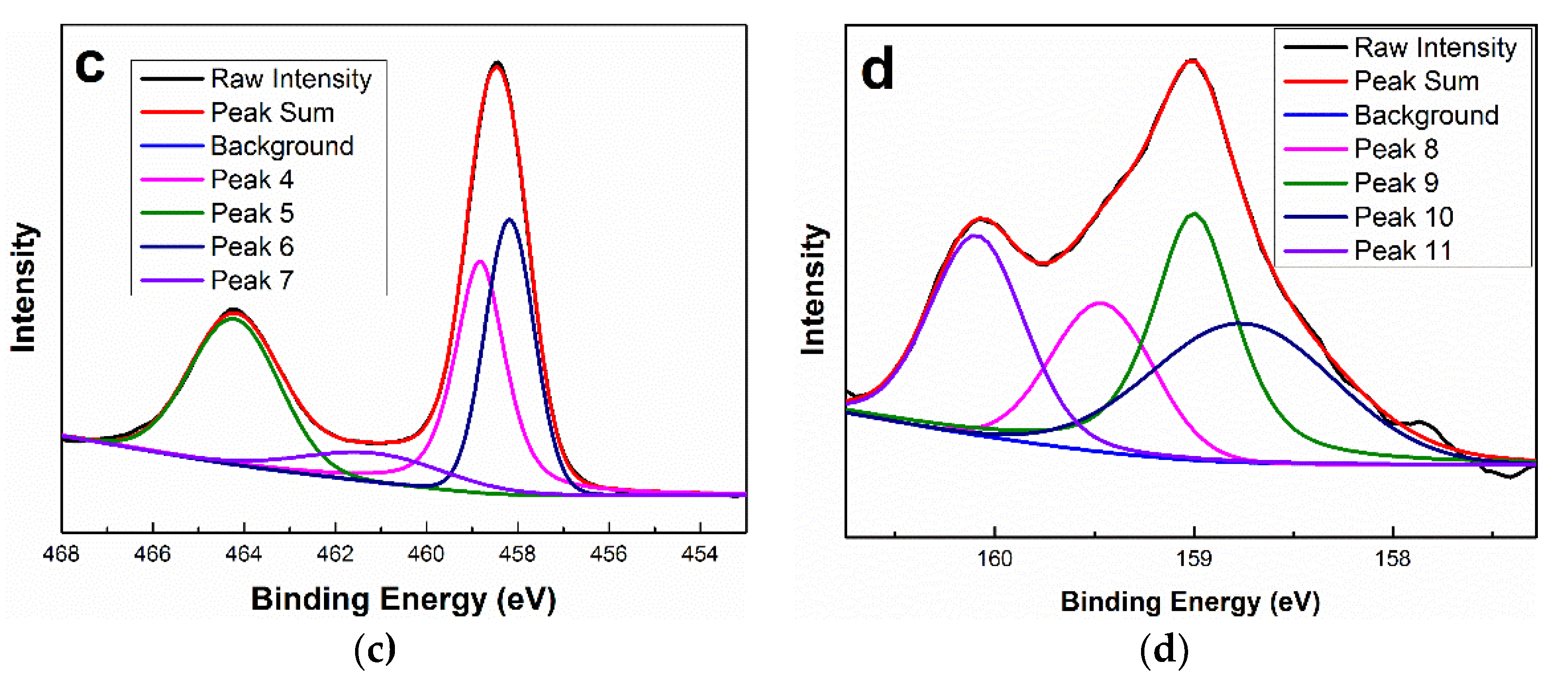
3.4. XPS Analysis
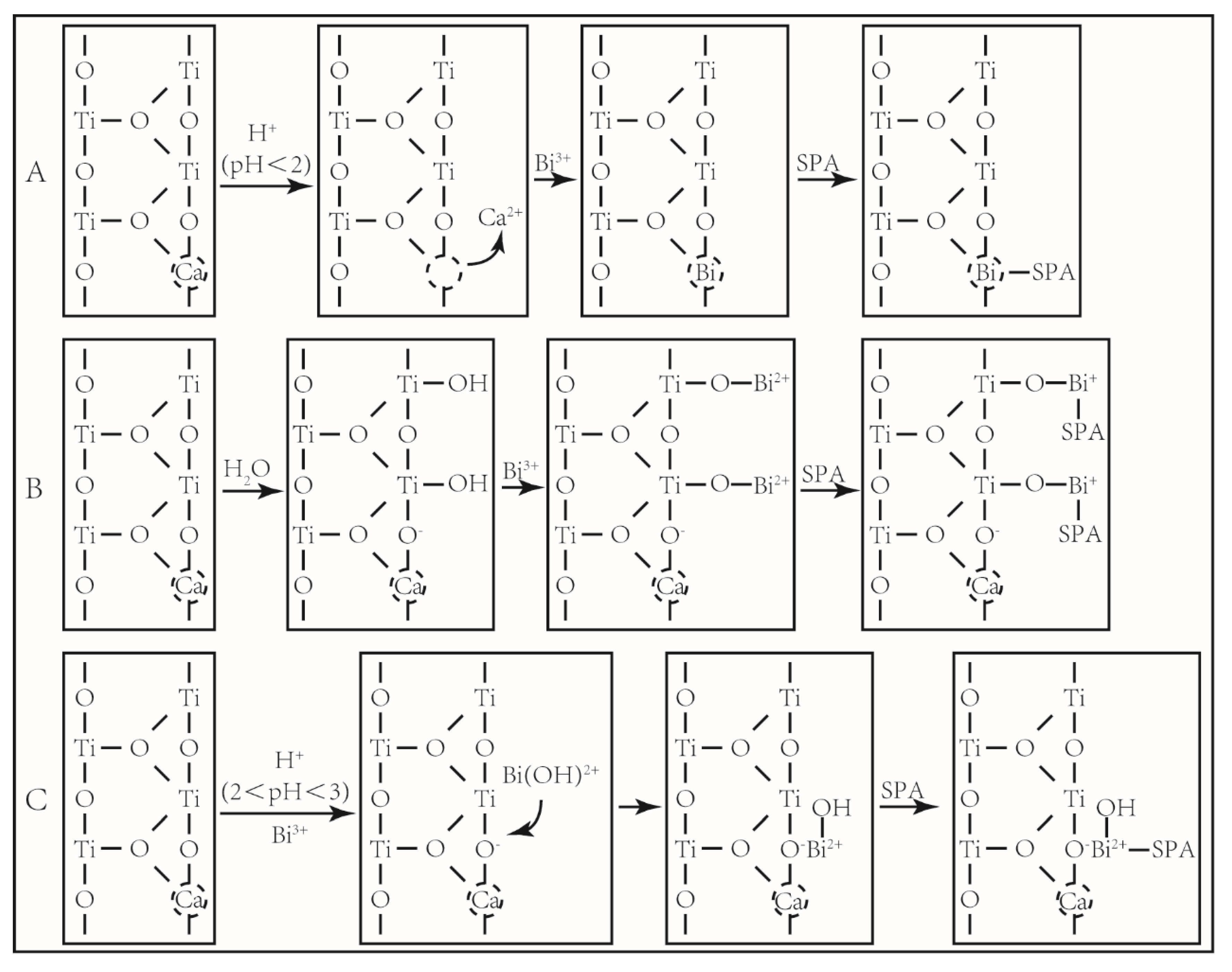
4. Conclusions
- (1)
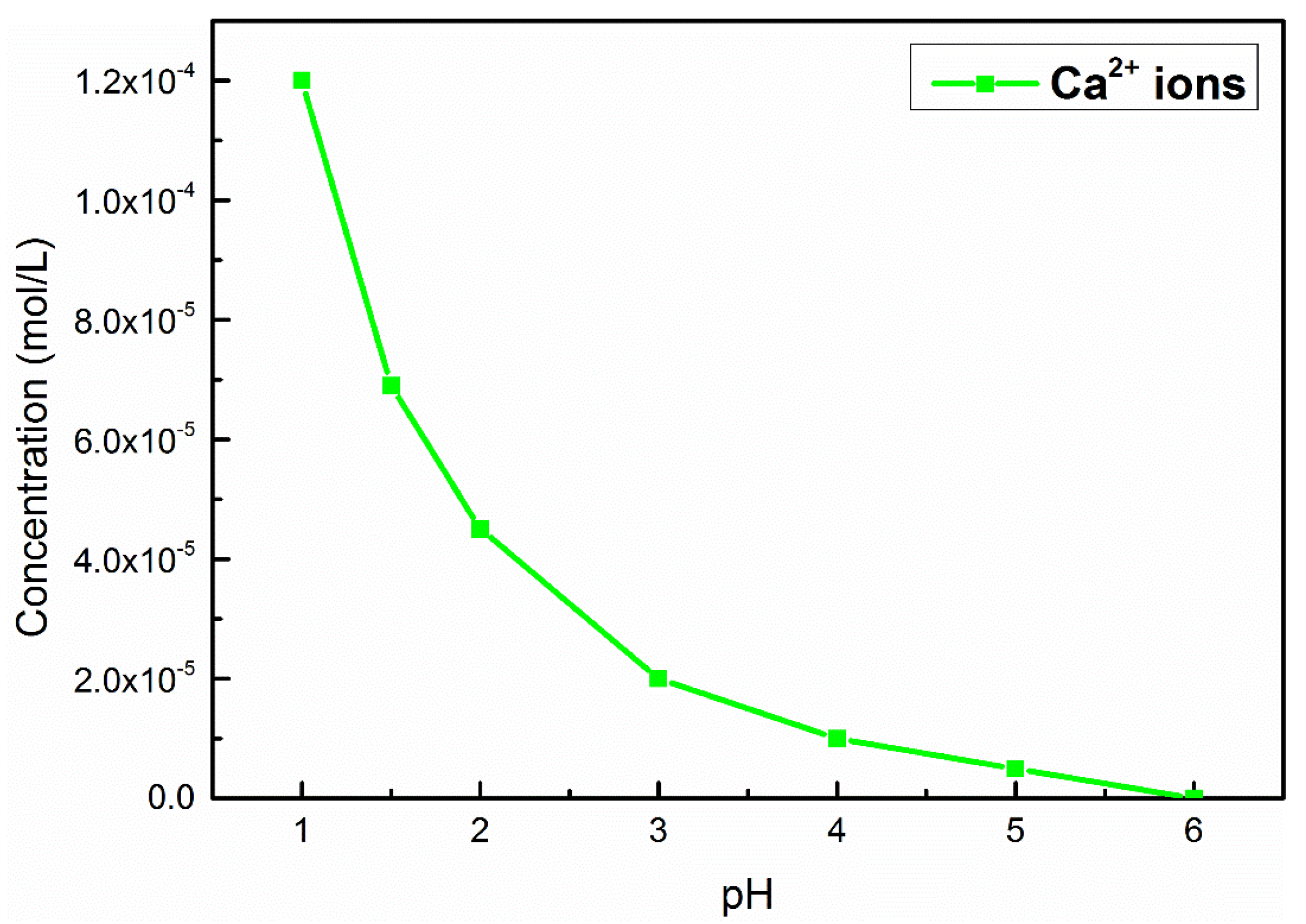
- The calcium impurity on the rutile surface is dissolved in strongly acidic conditions, and Bi3+ ions occupy the steric position of the original Ca2+ ions (in Figure 11A).
- (2)
- The proton substitution reaction occurs between the hydroxyl species of Bi3+ ions and hydroxylated rutile surface, producing the compounds of Ti-O-Bi2+ (in Figure 11B).
- (3)
- Bi3+ ions can adsorb on the rutile surface in the form of hydroxyl species, and increased the activation sites on the rutile surface (in Figure 11C).
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Li, T.; Wu, Y.; Wang, Q.; Zhang, D.; Zhang, A.; Miao, M. TiO2 crystalline structure and electrochemical performance in two-ply yarn CNT/TiO2 asymmetric supercapacitors. J. Mater. Sci. 2017, 52, 7733–7743. [Google Scholar] [CrossRef]
- Nam, I.; Park, J.; Park, S.; Bae, S.; Yoo, Y.G.; Han, J.W.; Yi, J. Observation of crystalline changes of titanium dioxide during lithium insertion by visible spectrum analysis. Phys. Chem. Chem. Phys. 2017, 19, 13140–13146. [Google Scholar] [CrossRef] [PubMed]
- Chachula, F.; Liu, Q. Upgrading a rutile concentrate produced from Athabasca oil sands tailings. Fuel 2003, 82, 929–942. [Google Scholar] [CrossRef]
- Wang, J.; Cheng, H.W.; Zhao, H.B.; Qin, W.Q.; Qiu, G.Z. Flotation behavior and mechanism of rutile with nonyl hydroxamic acid. Rare Met. 2016, 35, 419–424. [Google Scholar] [CrossRef]
- Liu, Q.; Peng, Y. The development of a composite collector for the flotation of rutile. Miner. Eng. 1999, 12, 1419–1430. [Google Scholar] [CrossRef]
- Madeley, J.D.; Graham, K. Flotation of rutile with anionic and cationic collectors. J. Appl. Chem. 2010, 16, 169–170. [Google Scholar] [CrossRef]
- Peng, Y. Effect of interactions between styryl phosphoric acid and aliphatic alcohol on rutile flotation. Chin. J. Nonferrous Metals 1999, 9, 358–361. [Google Scholar]
- Li, H.; Mu, S.; Weng, X.; Zhao, Y.; Song, S. Rutile Flotation with Pb2+ Ions as Activator: Adsorption of Pb2+ at Rutile/Water Interface. Colloids Surf. A Physicochem. Eng. Asp. 2016, 506, 431–437. [Google Scholar] [CrossRef]
- Fan, X.; Rowson, N.A. The effect of Pb(NO3)2 on ilmenite flotation. Miner. Eng. 2000, 13, 205–215. [Google Scholar] [CrossRef]
- Albrecht, T.W.J.; Addai-Mensah, J.; Fornasiero, D. Critical copper concentration in sphalerite flotation: Effect of temperature and collector. Int. J. Miner. Process. 2016, 146, 15–22. [Google Scholar] [CrossRef]
- Chandra, A.P.; Puskar, L.; Simpson, D.J.; Gerson, A.R. Copper and xanthate adsorption onto pyrite surfaces: Implications for mineral separation through flotation. Int. J. Miner. Process. 2012, 114–117, 16–26. [Google Scholar] [CrossRef]
- Han, J.; Liu, W.; Qin, W.; Zhang, T.; Chang, Z.; Xue, K. Effects of sodium salts on the sulfidation of lead smelting slag. Miner. Eng. 2017, 108, 1–11. [Google Scholar] [CrossRef]
- Luckay, R.; Cukrowski, I.; Mashishi, J.; Reibenspies, J.H.; Bond, A.H.; Rogers, R.D.; Hancock, R.D. Synthesis, stability and structure of the complex ofbismuth(III) with the nitrogen-donor macrocycle1,4,7,10-tetraazacyclododecane. The role of the lone pair onbismuth(III) and lead(II) in determiningco-ordination geometry. J. Chem. Soc. Dalton Trans. 1997, 5, 901–908. [Google Scholar] [CrossRef]
- Gillespie, R.J.; Nyholm, R.S.Q. Inorganic stereochemistry. Q. Rev. Chem. Soc. 1957, 11, 339–380. [Google Scholar] [CrossRef]
- Yang, N.; Sun, H. Biocoordination chemistry of bismuth: Recent advances. Coord. Chem. Rev. 2007, 251, 2354–2366. [Google Scholar] [CrossRef]
- Han, J.; Jiao, F.; Liu, W.; Qin, W.; Xu, T.; Xue, K.; Zhang, T. Innovative Methodology for Comprehensive Utilization of Spent MgO-Cr2O3 Bricks: Copper Flotation. ACS Sustain. Chem. Eng. 2016, 4, 5503–5510. [Google Scholar] [CrossRef]
- Zhao, H.; Wang, J.; Gan, X.; Hu, M.; Tao, L.; Qiu, G. Role of pyrite in sulfuric acid leaching of chalcopyrite: An elimination of polysulfide by controlling redox potential. Hydrometallurgy 2016, 164, 159–165. [Google Scholar] [CrossRef]
- Wang, J.; Gan, X.; Zhao, H.; Hu, M.; Li, K.; Qin, W.; Qiu, G. Dissolution and passivation mechanisms of chalcopyrite during bioleaching: DFT calculation, XPS and electrochemistry analysis. Miner. Eng. 2016, 98, 264–278. [Google Scholar] [CrossRef]
- Liu, W.; Zhang, S.; Wang, W.; Zhang, J.; Yan, W.; Deng, J.; Feng, Q.; Huang, Y. The effects of Ca(II) and Mg(II) ions on the flotation of spodumene using NaOL. Miner. Eng. 2015, 79, 40–46. [Google Scholar] [CrossRef]
- Fornasiero, D.; Ralston, J. Cu(II) and Ni(II) activation in the flotation of quartz, lizardite and chlorite. Int. J. Miner. Process. 2005, 76, 75–81. [Google Scholar] [CrossRef]
- Bagwasi, S.; Niu, Y.; Nasir, M.; Tian, B.; Zhang, J. The study of visible light active bismuth modified nitrogen doped titanium dioxide photocatlysts: Role of bismuth. Appl. Surf. Sci. 2013, 264, 139–147. [Google Scholar] [CrossRef]
- Qin, W.; Jiao, F.; Sun, W.; Wang, X.; Liu, B.; Wang, J.; Zeng, K.; Wei, Q.; Liu, K. Effects of sodium salt of N,N-dimethyldi-thiocarbamate on floatability of chalcopyrite, sphalerite, marmatite and its adsorption properties. Colloids Surf. A Physicochem. Eng. Asp. 2013, 421, 181–192. [Google Scholar] [CrossRef]
- Leppinen, J.O. FTIR and flotation investigation of the adsorption of ethyl xanthate on activated and non-activated sulfide minerals. Int. J. Miner. Process. 1990, 30, 245–263. [Google Scholar] [CrossRef]
- Jiang, H.; Sun, Z.; Xu, L.; Hu, Y.; Huang, K.; Zhu, S. A comparison study of the flotation and adsorption behaviors of diaspore and kaolinite with quaternary ammonium collectors. Miner. Eng. 2014, 65, 124–129. [Google Scholar] [CrossRef]
- Parks, G.A. The Isoelectric Points of Solid Oxides, Solid Hydroxides, and Aqueous Hydroxo Complex Systems. Chem. Rev. 1965, 65, 177–198. [Google Scholar] [CrossRef]
- Yu, X.; Ruan, D.; Wu, C.; Jing, W.; Shi, Z. Spiro-(1,1′)-bipyrrolidinium tetrafluoroborate salt as high voltage electrolyte for electric double layer capacitors. J. Power Sources 2014, 265, 309–316. [Google Scholar] [CrossRef]
- Han, T.; Park, M.S.; Kim, J.; Kim, J.H.; Kim, K. The smallest quaternary ammonium salts with ether groups for high-performance electrochemical double layer capacitors. Chem. Sci. 2016, 7, 1791–1796. [Google Scholar] [CrossRef]
- De Lara, L.S.; Rigo, V.A.; Michelon, M.F.; Metin, C.O.; Nguyen, Q.P.; Miranda, C.R. Molecular dynamics studies of aqueous silica nanoparticle dispersions: Salt effects on the double layer formation. J. Phys. Condens. Matter 2015, 27. [Google Scholar] [CrossRef] [PubMed]
- Anitha, B.; Khadar, M.A. Dopant concentration dependent magnetism of Cu-doped TiO2 nanocrystals. J. Nanoparticle Res. 2016, 18, 149–164. [Google Scholar] [CrossRef]
- Mikhailova, D.; Karakulina, O.M.; Batuk, D.; Hadermann, J.; Abakumov, A.M.; Herklotz, M.; Tsirlin, A.A.; Oswald, S.; Giebeler, L.; Schmidt, M.; et al. Layered-to-Tunnel Structure Transformation and Oxygen Redox Chemistry in LiRhO2 upon Li Extraction and Insertion. Inorg. Chem. 2016, 55, 7079–7089. [Google Scholar] [CrossRef] [PubMed]
- Bakhshayesh, A.M.; Bakhshayesh, N. Facile one-pot synthesis of uniform niobium-doped titanium dioxide microparticles for nanostructured dye-sensitized solar cells. J. Electroceramics 2016, 36, 112–121. [Google Scholar] [CrossRef]
- Halpegamage, S.; Wen, Z.H.; Gong, X.Q.; Batzill, M. Monolayer Intermixed Oxide Surfaces: Fe, Ni, Cr, and V Oxides on Rutile TiO2(011). J. Phys. Chem. C 2016, 120, 14782–14794. [Google Scholar] [CrossRef]
- Casella, I.G.; Contursi, M. Characterization of bismuth adatom-modified palladium electrodes: The electrocatalytic oxidation of aliphatic aldehydes in alkaline solutions. Electrochim. Acta 2006, 52, 649–657. [Google Scholar] [CrossRef]
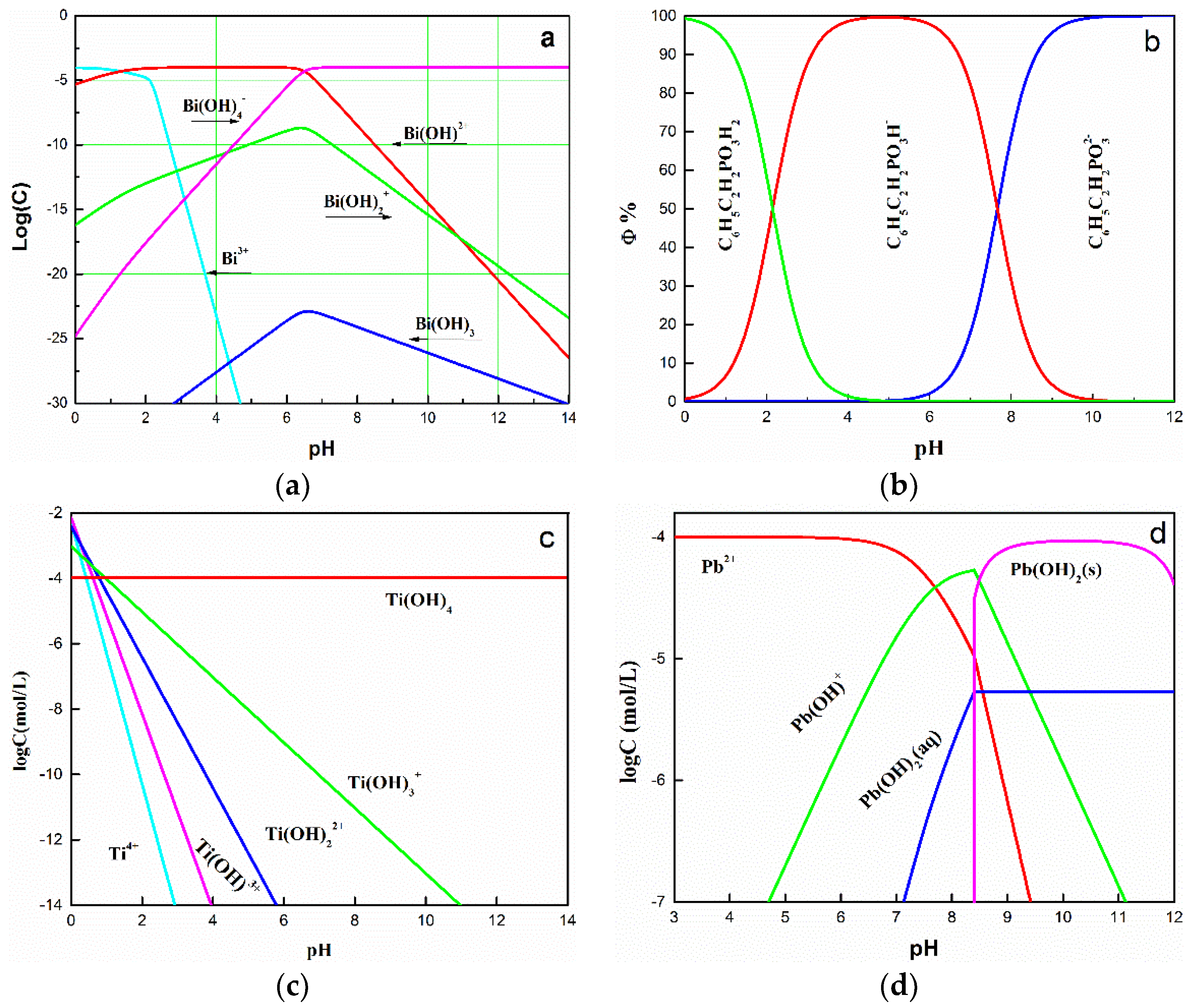
| Element | TiO2 | FeO | Fe2O3 | SiO2 | CaO | MgO | Al2O3 |
|---|---|---|---|---|---|---|---|
| Content | 93.80 | 1.53 | 1.33 | 2.17 | 0.17 | 0.31 | 0.99 |
| Ions type | Ca2+ | Fe2+ | Fe3+ | Mg2+ | Al3+ | Pb2+ | Bi3+ |
|---|---|---|---|---|---|---|---|
| Radius (Å) | 1.14 | 0.92 | 0.785 | 0.86 | 0.675 | 1.33 | 1.17 |
| Atom Orbit | Peak | Binding Energy (eV) | Chemical States |
|---|---|---|---|
| O 1s | Peak 1 | 531.19 | Ti-O-Bi (rutile surface) |
| Peak 2 | 532.34 | hydroxyl species (rutile surface) | |
| Peak 3 | 529.79 | Ti-O-Ti (rutile bulk) | |
| Ti 2p | Peak 4 | 458.84 | O-Ti-O (Ti 2p3/2) (rutile bulk) |
| Peak 5 | 464.24 | O-Ti-O (Ti 2p1/2) (rutile bulk) | |
| Peak 6 | 458.19 | Ti-OH (rutile surface) | |
| Peak 7 | 461.19 | Ti-O-Bi2+ (rutile surface) | |
| Bi 4f7/2 | Peak 8 | 159.47 | hydroxyl compounds (rutile surface) |
| Peak 9 | 158.99 | hydroxyl compounds (rutile surface) | |
| Peak 10 | 158.74 | hydroxyl compounds (rutile surface) | |
| Peak 11 | 160.10 | Ti-O-Bi2+ (rutile surface) |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xiao, W.; Cao, P.; Liang, Q.; Peng, H.; Zhao, H.; Qin, W.; Qiu, G.; Wang, J. The Activation Mechanism of Bi3+ Ions to Rutile Flotation in a Strong Acidic Environment. Minerals 2017, 7, 113. https://doi.org/10.3390/min7070113
Xiao W, Cao P, Liang Q, Peng H, Zhao H, Qin W, Qiu G, Wang J. The Activation Mechanism of Bi3+ Ions to Rutile Flotation in a Strong Acidic Environment. Minerals. 2017; 7(7):113. https://doi.org/10.3390/min7070113
Chicago/Turabian StyleXiao, Wei, Pan Cao, Qiannan Liang, Hong Peng, Hongbo Zhao, Wenqing Qin, Guanzhou Qiu, and Jun Wang. 2017. "The Activation Mechanism of Bi3+ Ions to Rutile Flotation in a Strong Acidic Environment" Minerals 7, no. 7: 113. https://doi.org/10.3390/min7070113








