The Effect of Polystyrene on the Carrier Flotation of Fine Smithsonite
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials and Reagents
2.2. SEM Analysis
2.3. Microflotation Tests
2.4. Particle Size Analysis
2.5. Adsorption Tests
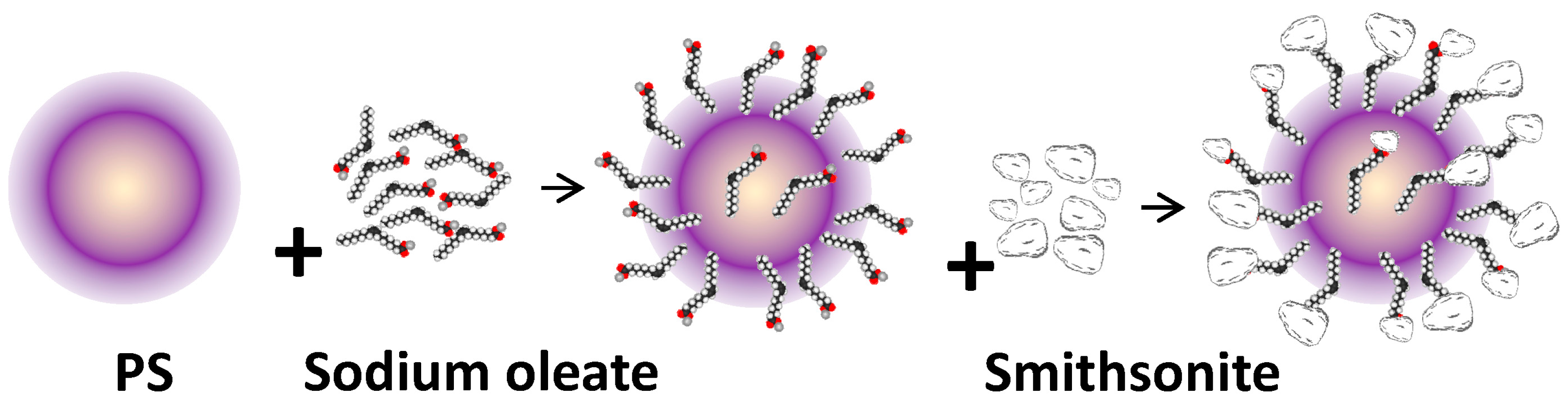
3. Results and Discussion
3.1. SEM Results
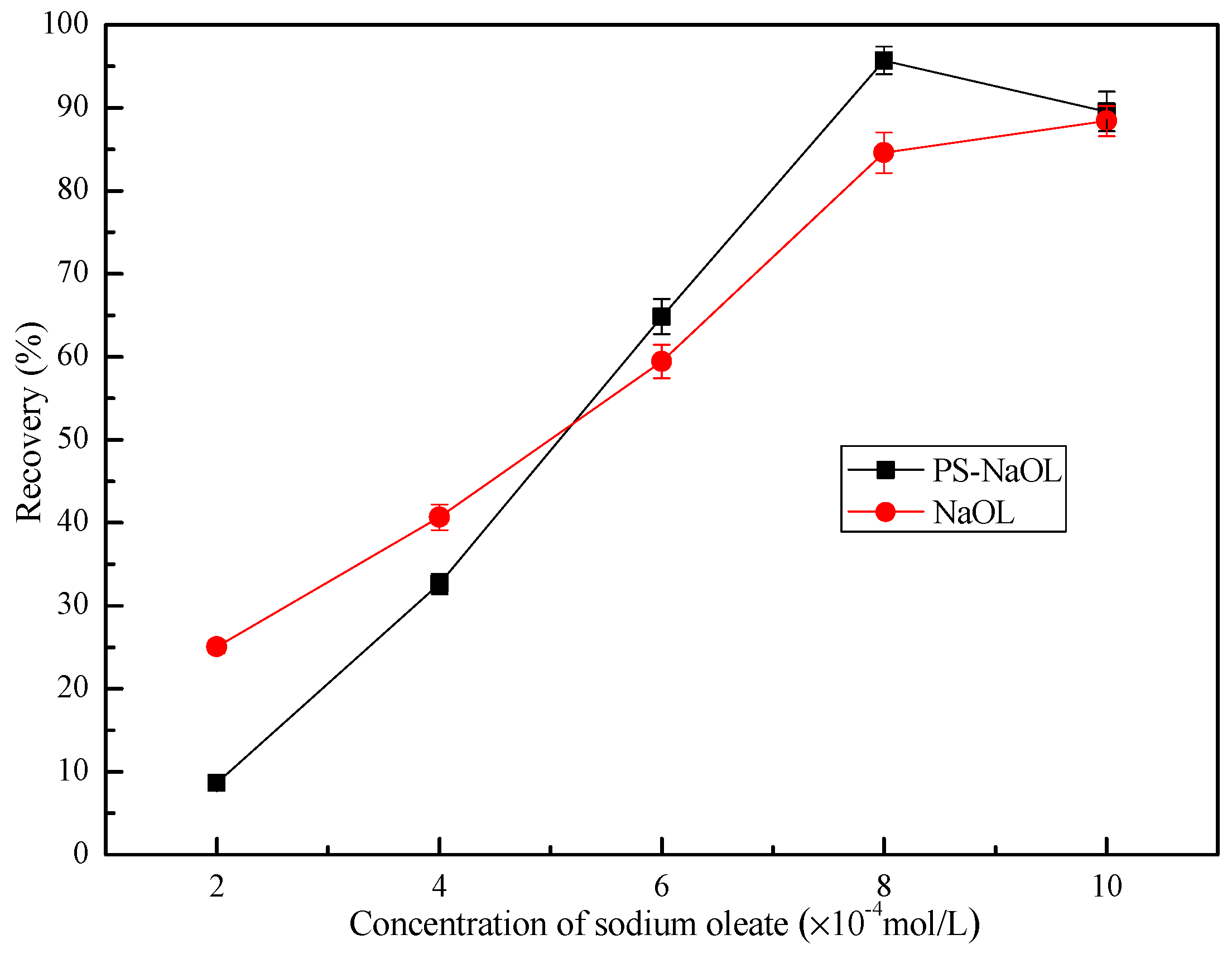
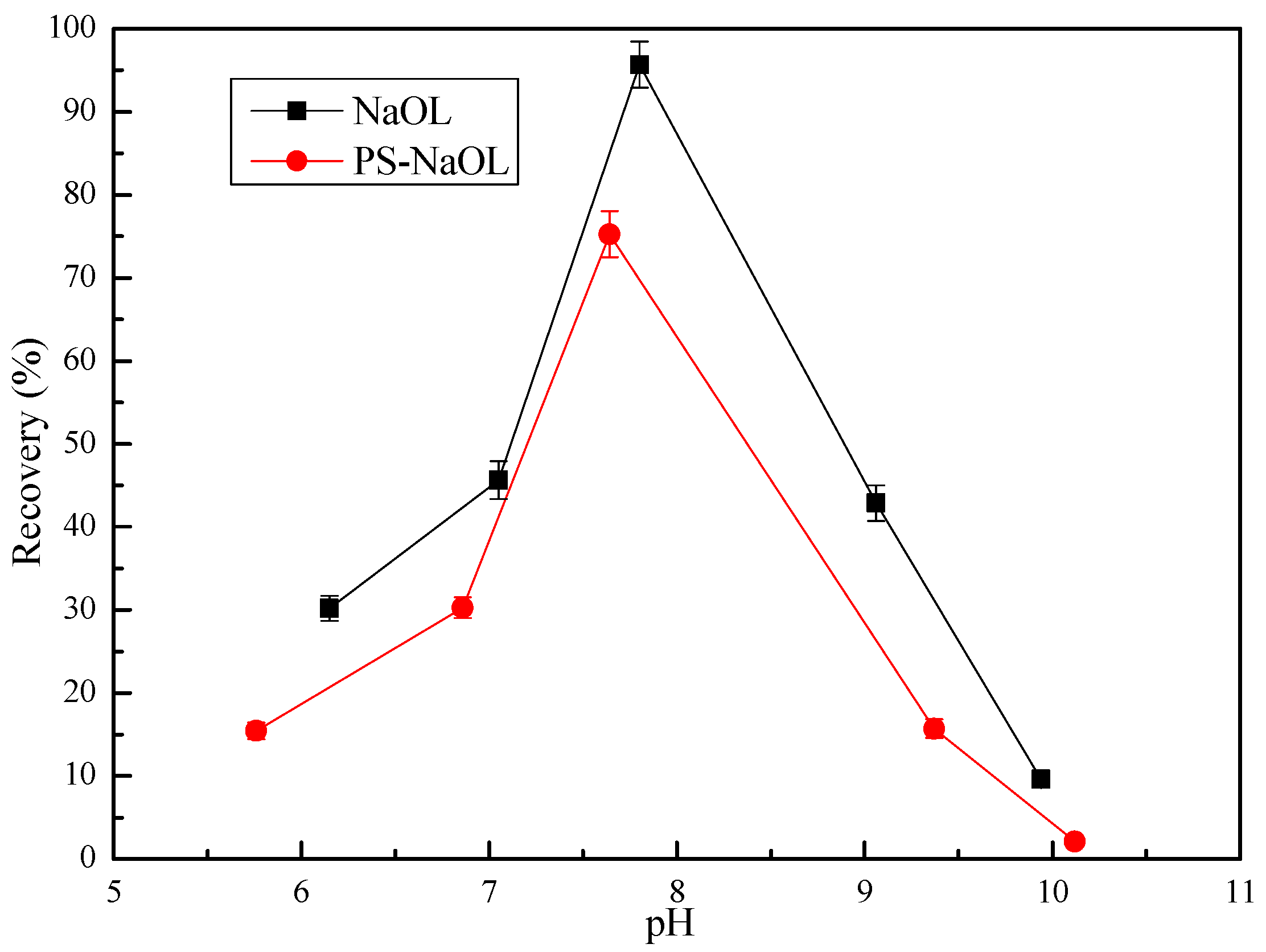
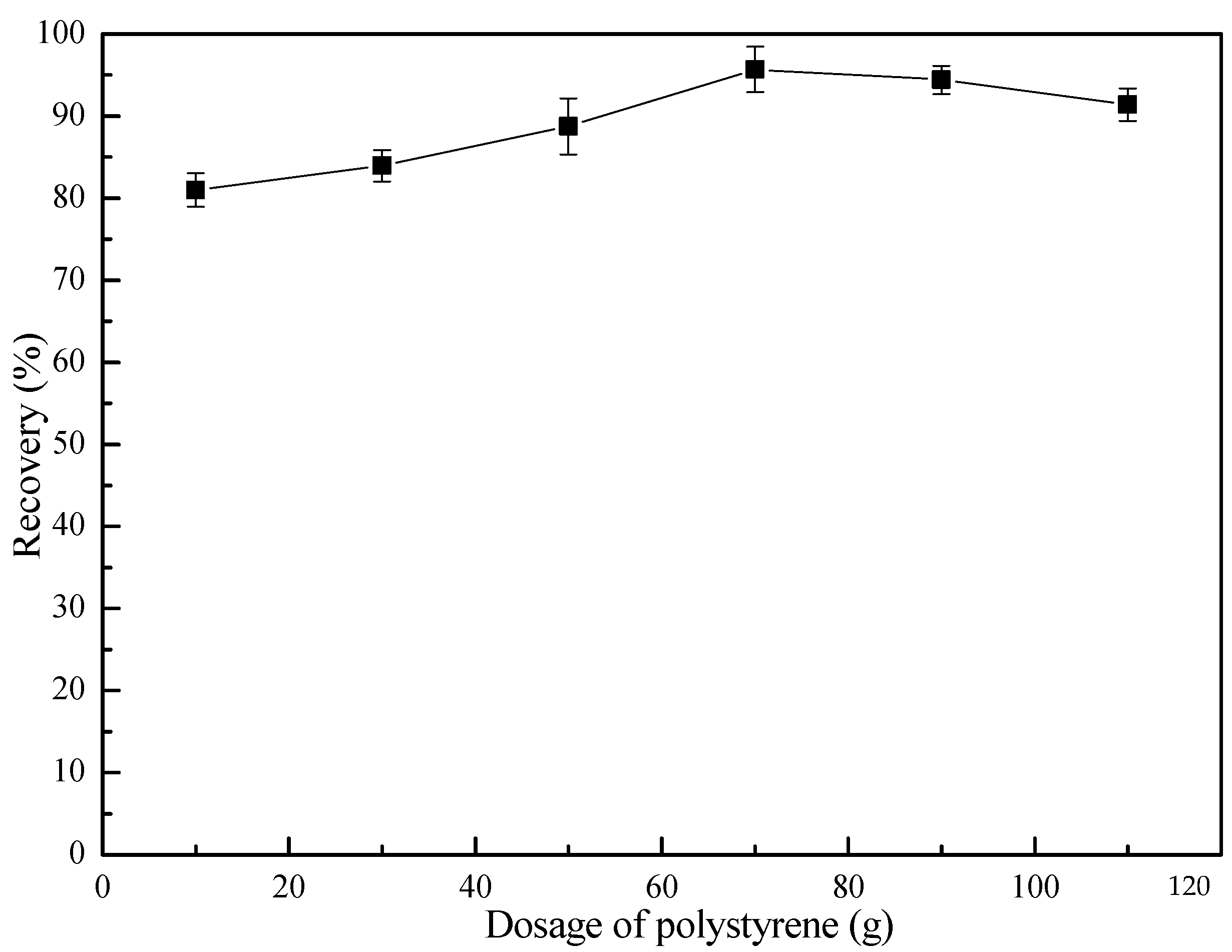
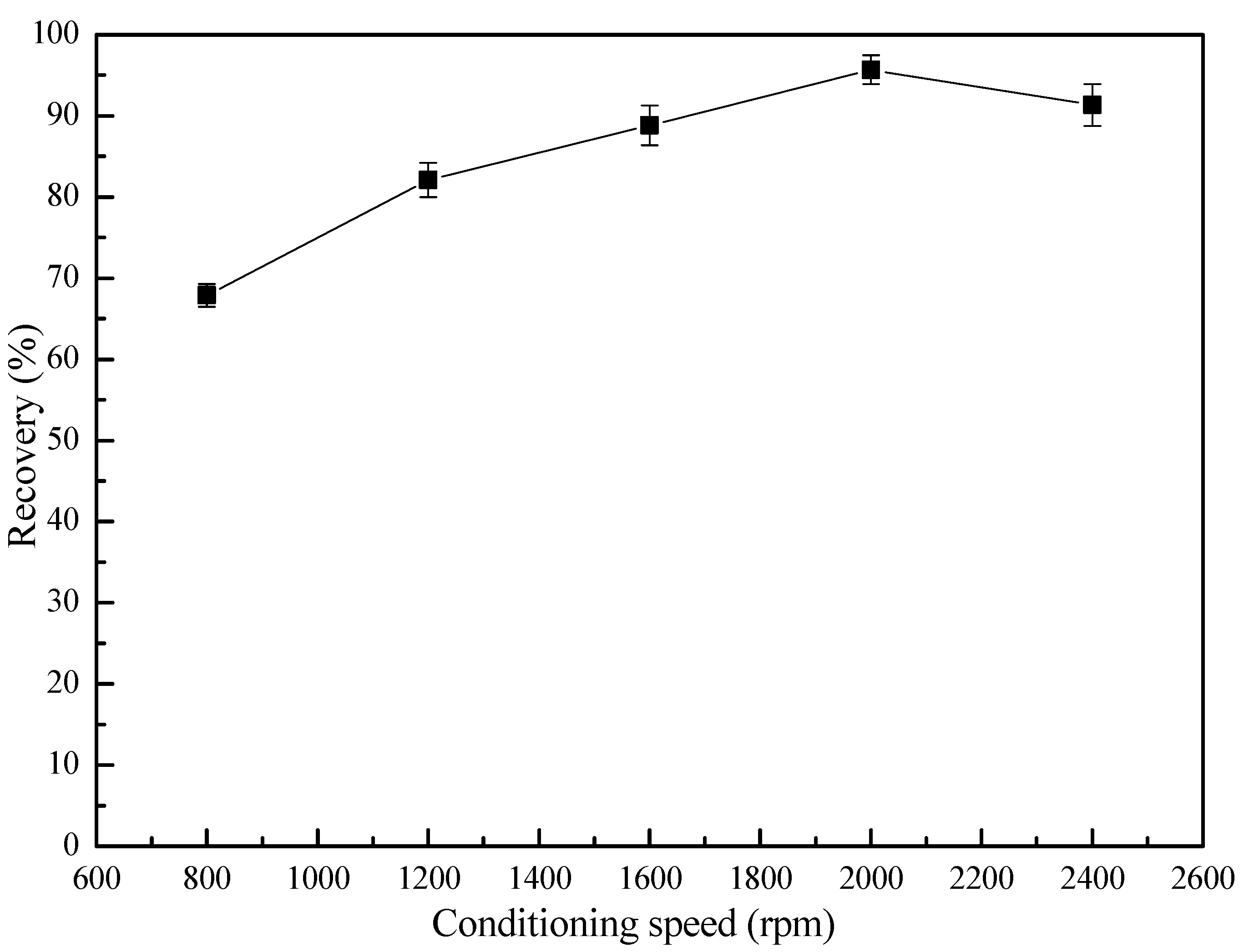
3.2. Flotation Behavior
3.3. Particle Size Analysis
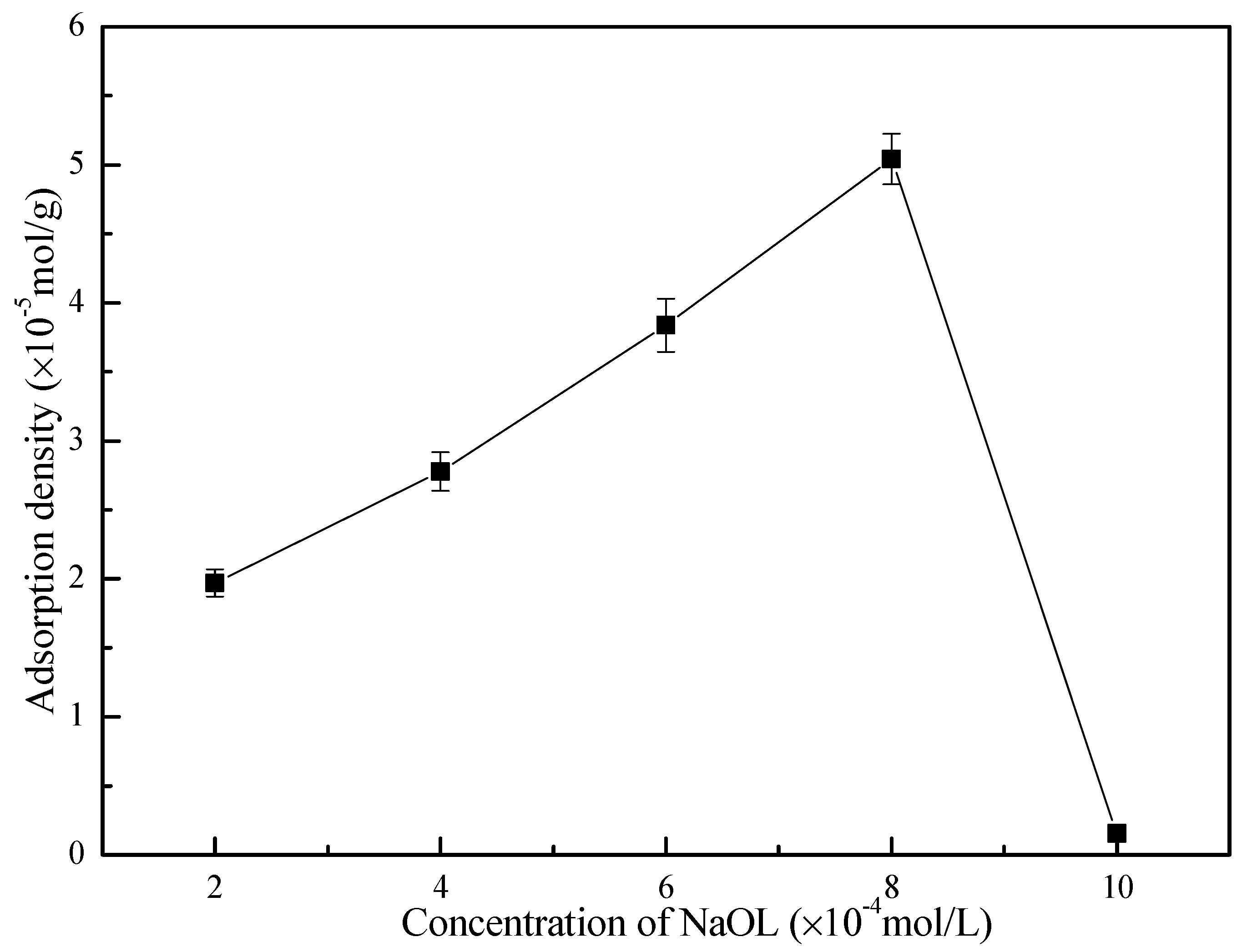
3.4. Adsorption Results
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Miettinen, T.; Ralston, J.; Fornasiero, D. The limits of fine particle flotation. Miner. Eng. 2010, 23, 420–437. [Google Scholar] [CrossRef]
- Xu, L.; Hu, Y.; Dong, F.; Gao, Z.; Wu, H.; Wang, Z. Anisotropic adsorption of oleate on diaspore and kaolinite crystals: Implications for their flotation separation. Appl. Surf. Sci. 2014, 321, 331–338. [Google Scholar] [CrossRef]
- Xu, L.; Hu, Y.; Dong, F.; Jiang, H.; Wu, H.; Wang, Z.; Liu, R. Effects of particle size and chain length on flotation of quaternary ammonium salts onto kaolinite. Mineral. Petrol. 2015, 109, 309–316. [Google Scholar] [CrossRef]
- Subrahmanyam, T.V.; Forssberg, K.E. Fine particles processing: Shear-flocculation and carrier flotation—A review. Int. J. Miner. Process. 1990, 30, 265–286. [Google Scholar] [CrossRef]
- Yoon, R.H.; Luttrell, G.H. The effect of bubble size on fine particle flotation. Miner. Process. Extr. Metall. Rev. 1989, 5, 101–122. [Google Scholar] [CrossRef]
- Shibata, J.; Fuerstenau, D.W. Flocculation and flotation characteristics of fine hematite with sodium oleate. Int. J. Miner. Process. 2003, 72, 25–32. [Google Scholar] [CrossRef]
- Gao, Z.; Hu, Y.; Sun, W.; Drelich, J.W. Surface-charge anisotropy of scheelite crystals. Langmuir 2016, 32, 6282–6288. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Hu, Y.; Wu, H.; Tian, J.; Liu, J.; Gao, Z.; Wang, L. Surface crystal chemistry of spodumene with different size fractions and implications for flotation. Sep. Purif. Technol. 2016, 169, 33–42. [Google Scholar] [CrossRef]
- Kurama, H.; Karagüzel, C.; Mergan, T.; Çelik, M.S. Ammonium removal from aqueous solutions by dissolved air flotation in the presence of zeolite carrier. Desalination 2010, 253, 147–152. [Google Scholar] [CrossRef]
- Feng, D.; Aldrich, C.; Tan, H. Removal of heavy metal ions by carrier magnetic separation of adsorptive particulates. Hydrometallurgy 2000, 56, 359–368. [Google Scholar]
- Ateşok, G.; Boylu, F.; Çelĭk, M.S. Carrier flotation for desulfurization and deashing of difficult-to-float coals. Miner. Eng. 2001, 14, 661–670. [Google Scholar] [CrossRef]
- Chia, Y.H.; Somasundaran, P. A theoretical approach to flocculation in carrier flotation for beneficiation of clay. Colloids Surf. 1983, 8, 187–202. [Google Scholar] [CrossRef]
- Li, H.; Chow, R.; Roberge, K. Role of carrier flotation in accelerating bitumen extraction recovery from mineable Athabasca oil sands. Can. J. Chem. Eng. 2013, 91, 1340–1348. [Google Scholar]
- Basařová, P.; Bartovská, L.; Kořínek, K.; Horn, D. The influence of flotation agent concentration on the wettability and flotability of polystyrene. J. Colloid Interface Sci. 2005, 286, 333–338. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Pelton, R.; Raegen, A.; Montgomery, M.; Dalnoki-Veress, K. Nanoparticle flotation collectors: Mechanisms behind a new technology. Langmuir 2011, 27, 10438–10446. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Wang, J.K.; Wei, C.; Liu, C.X.; Jiang, J.B.; Wang, F. Sulfidation roasting of low grade lead–zinc oxide ore with elemental sulfur. Miner. Eng. 2010, 23, 563–566. [Google Scholar] [CrossRef]
- Ejtemaei, M.; Irannajad, M.; Gharabaghi, M. Role of dissolved mineral species in selective flotation of smithsonite from quartz using oleate as collector. Int. J. Miner. Process. 2012, 114, 40–47. [Google Scholar] [CrossRef]
- Ejtemaei, M.; Gharabaghi, M.; Irannajad, M. A review of zinc oxide mineral beneficiation using flotation method. Adv. Colloid Interface Sci. 2014, 206, 68–78. [Google Scholar] [CrossRef] [PubMed]
- Schubert, H. On the optimization of hydrodynamics in fine particle flotation. Miner. Eng. 2008, 21, 930–936. [Google Scholar] [CrossRef]
- Mehdilo, A.; Zarei, H.; Irannajad, M.; Arjmandfar, H. Flotation of zinc oxide ores by cationic and mixed collectors. Miner. Eng. 2012, 36, 331–334. [Google Scholar] [CrossRef]
- Ejtemaei, M.; Irannajad, M.; Gharabaghi, M. Influence of important factors on flotation of zinc oxide mineral using cationic, anionic and mixed (cationic/anionic) collectors. Miner. Eng. 2011, 24, 1402–1408. [Google Scholar] [CrossRef]
- Irannajad, M.; Ejtemaei, M.; Gharabaghi, M. The effect of reagents on selective flotation of smithsonite–calcite–quartz. Miner. Eng. 2009, 22, 766–771. [Google Scholar] [CrossRef]
- Shi, Q.; Feng, Q.; Zhang, G.; Deng, H. Electrokinetic properties of smithsonite and its floatability with anionic collector. Colloids Surf. A 2012, 410, 178–183. [Google Scholar] [CrossRef]
- Hosseini, S.H.; Forssberg, E. Adsorption studies of smithsonite flotation using dodecylamine and oleic acid. Miner. Metall. Process. 2006, 23, 87–96. [Google Scholar]
- Shi, Q.; Zhang, G.; Feng, Q.; Deng, H. Effect of solution chemistry on the flotation system of smithsonite and calcite. Int. J. Miner. Process. 2013, 119, 34–39. [Google Scholar] [CrossRef]
- Gao, Y.; Gao, Z.; Sun, W.; Hu, Y. Selective flotation of scheelite from calcite: A novel reagent scheme. Int. J. Miner. Process. 2016, 154, 10–15. [Google Scholar] [CrossRef]
- Deng, W.; Xu, L.; Tian, J.; Hu, Y.; Han, Y. Flotation and adsorption of a new polysaccharide depressant on pyrite and talc in the presence of a pre-adsorbed xanthate collector. Minerals 2017, 7, 40. [Google Scholar] [CrossRef]
- Xu, L.; Wu, H.; Dong, F.; Wang, L.; Wang, Z.; Xiao, J. Flotation and adsorption of mixed cationic/anionic collectors on muscovite mica. Miner. Eng. 2013, 41, 41–45. [Google Scholar] [CrossRef]
- Tian, J.; Xu, L.; Deng, W.; Jiang, H.; Gao, Z.; Hu, Y. Adsorption mechanism of new mixed anionic/cationic collectors in a spodumene-feldspar flotation system. Chem. Eng. Sci. 2017, 164, 99–107. [Google Scholar] [CrossRef]
- Hu, Y.; Gao, Z.; Sun, W.; Liu, X. Anisotropic surface energies and adsorption behaviors of scheelite crystal. Colloids Surf. A 2012, 415, 439–448. [Google Scholar]
- Gao, Z.; Li, C.; Sun, W.; Hu, Y. Anisotropic surface properties of calcite: A consideration of surface broken bonds. Colloids Surf. A 2017, 520, 53–61. [Google Scholar] [CrossRef]
- Collins, G.L.; Jameson, G.J. Experiments on the flotation of fine particles: The influence of particle size and charge. Chem. Eng. Sci. 1976, 31, 985–991. [Google Scholar] [CrossRef]
- Ng, W.S.; Sonsie, R.; Forbes, E.; Franks, G.V. Flocculation/flotation of hematite fines with anionic temperature-responsive polymer acting as a selective flocculant and collector. Miner. Eng. 2015, 77, 64–71. [Google Scholar] [CrossRef]
- Hildebrand, A.; Garidel, P.; Neubert, R.; Blume, A. Thermodynamics of demicellization of mixed micelles composed of sodium oleate and bile salts. Langmuir 2004, 20, 320–328. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Hu, Y.; Tian, J.; Wu, H.; Wang, L.; Yang, Y.; Wang, Z. Synergistic effect of mixed cationic/anionic collectors on flotation and adsorption of muscovite. Colloids Surf. A 2016, 492, 181–189. [Google Scholar]



© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, X.; Hu, Y.; Sun, W.; Xu, L. The Effect of Polystyrene on the Carrier Flotation of Fine Smithsonite. Minerals 2017, 7, 52. https://doi.org/10.3390/min7040052
Zhang X, Hu Y, Sun W, Xu L. The Effect of Polystyrene on the Carrier Flotation of Fine Smithsonite. Minerals. 2017; 7(4):52. https://doi.org/10.3390/min7040052
Chicago/Turabian StyleZhang, Xiangfeng, Yuehua Hu, Wei Sun, and Longhua Xu. 2017. "The Effect of Polystyrene on the Carrier Flotation of Fine Smithsonite" Minerals 7, no. 4: 52. https://doi.org/10.3390/min7040052
APA StyleZhang, X., Hu, Y., Sun, W., & Xu, L. (2017). The Effect of Polystyrene on the Carrier Flotation of Fine Smithsonite. Minerals, 7(4), 52. https://doi.org/10.3390/min7040052






