Study on Selective Removal of Impurity Iron from Leached Copper-Bearing Solution Using a Chelating Resin
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Adsorption Experiment
2.3. Measurements
3. Results and Discussions
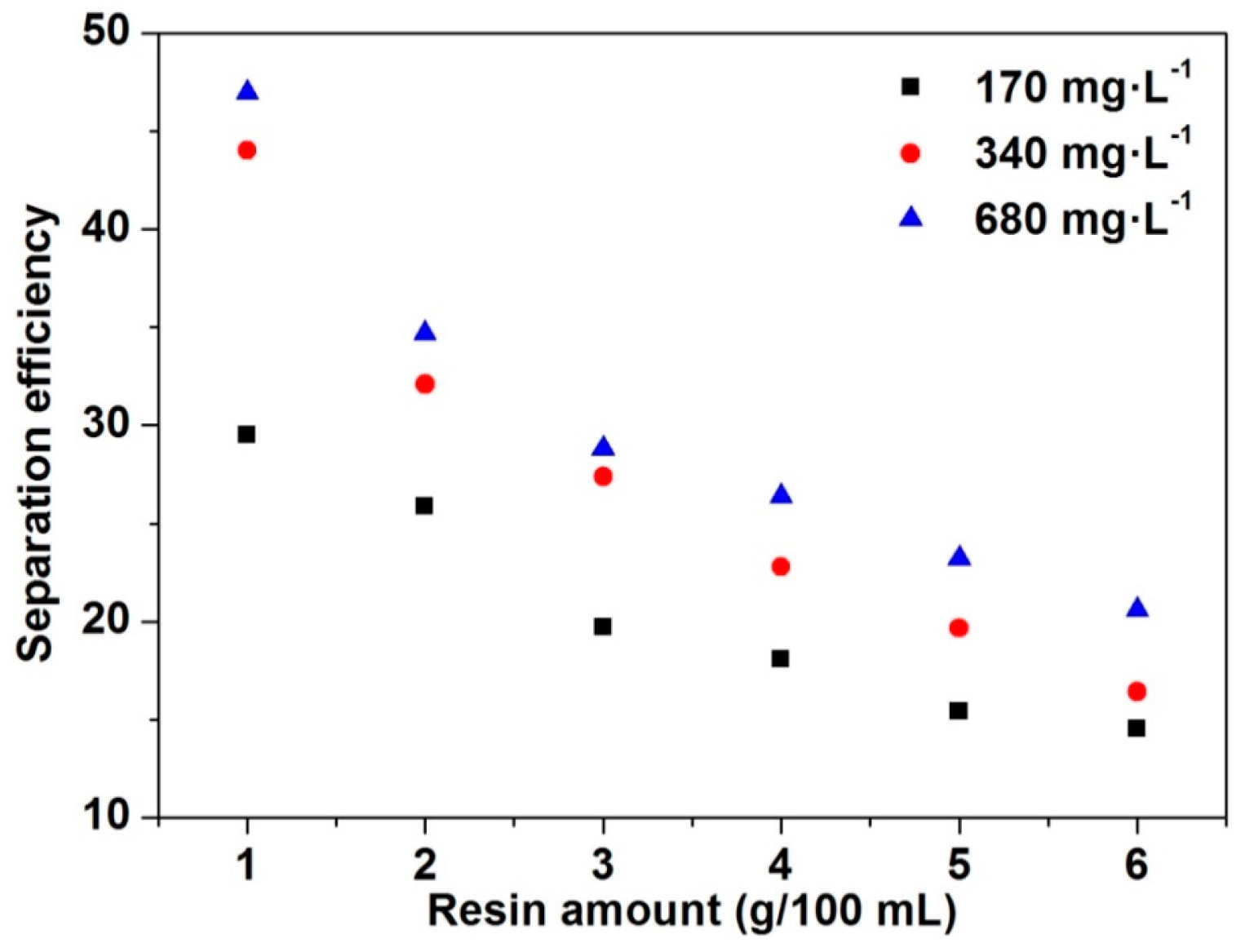
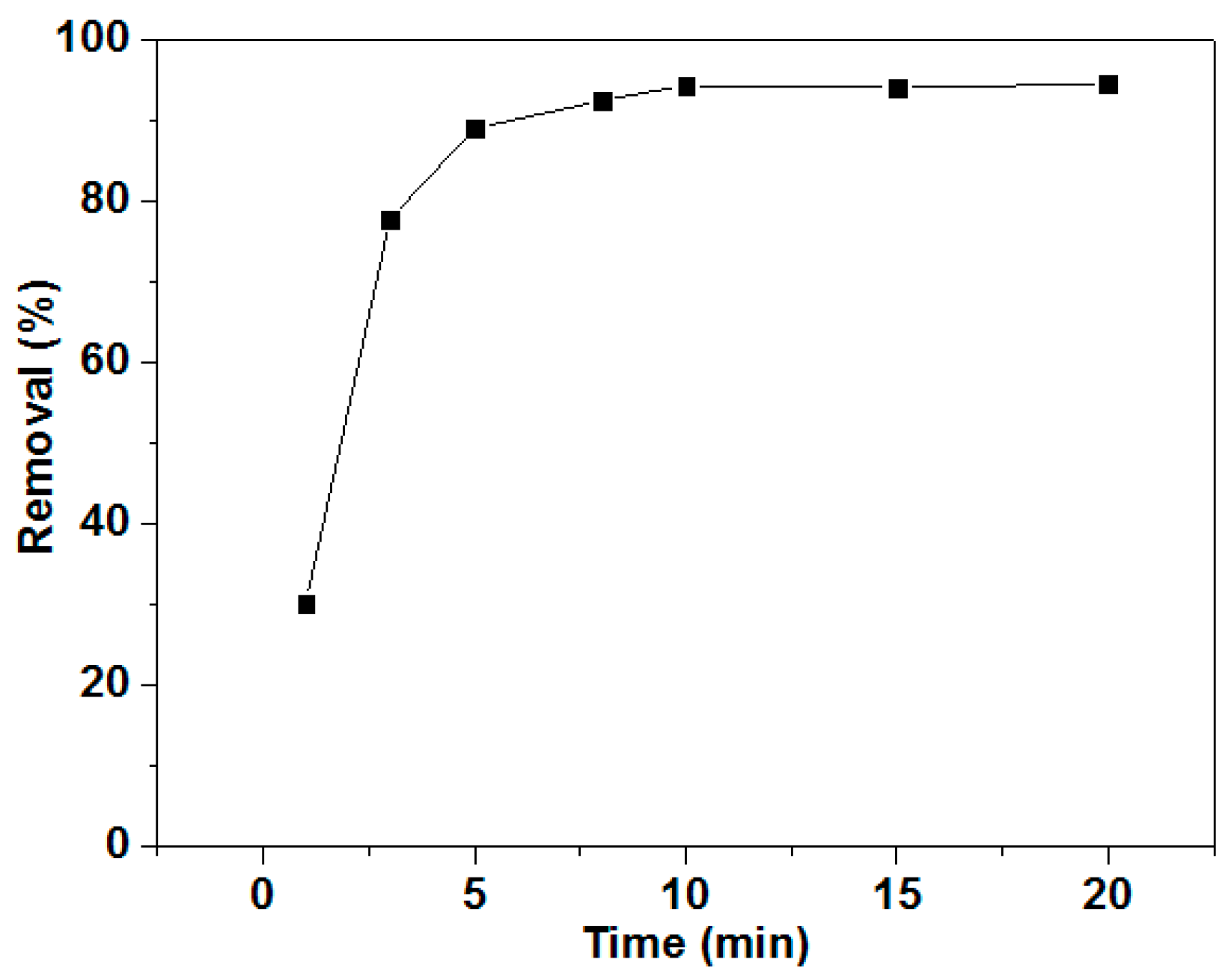
3.1. Separation Efficiency
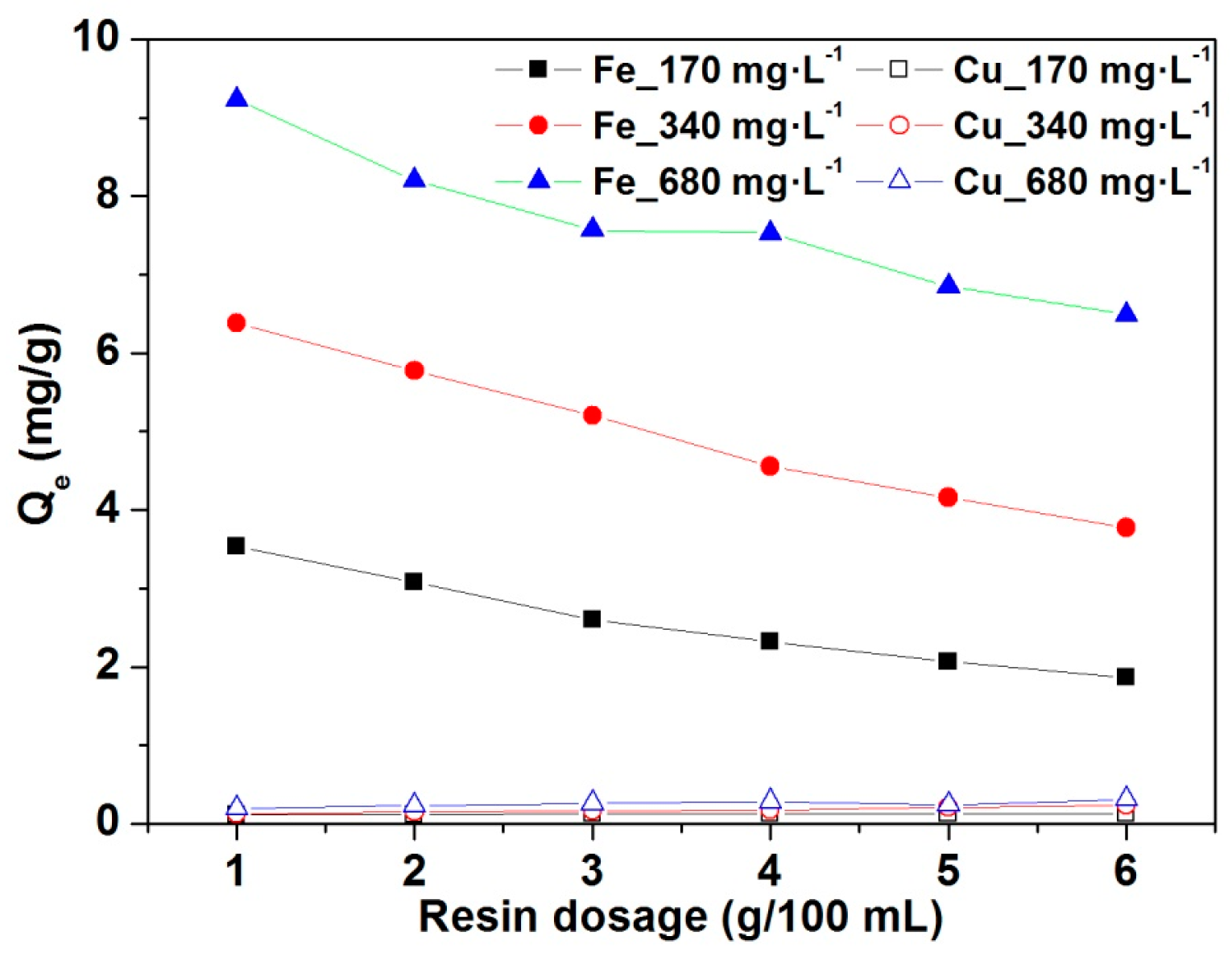
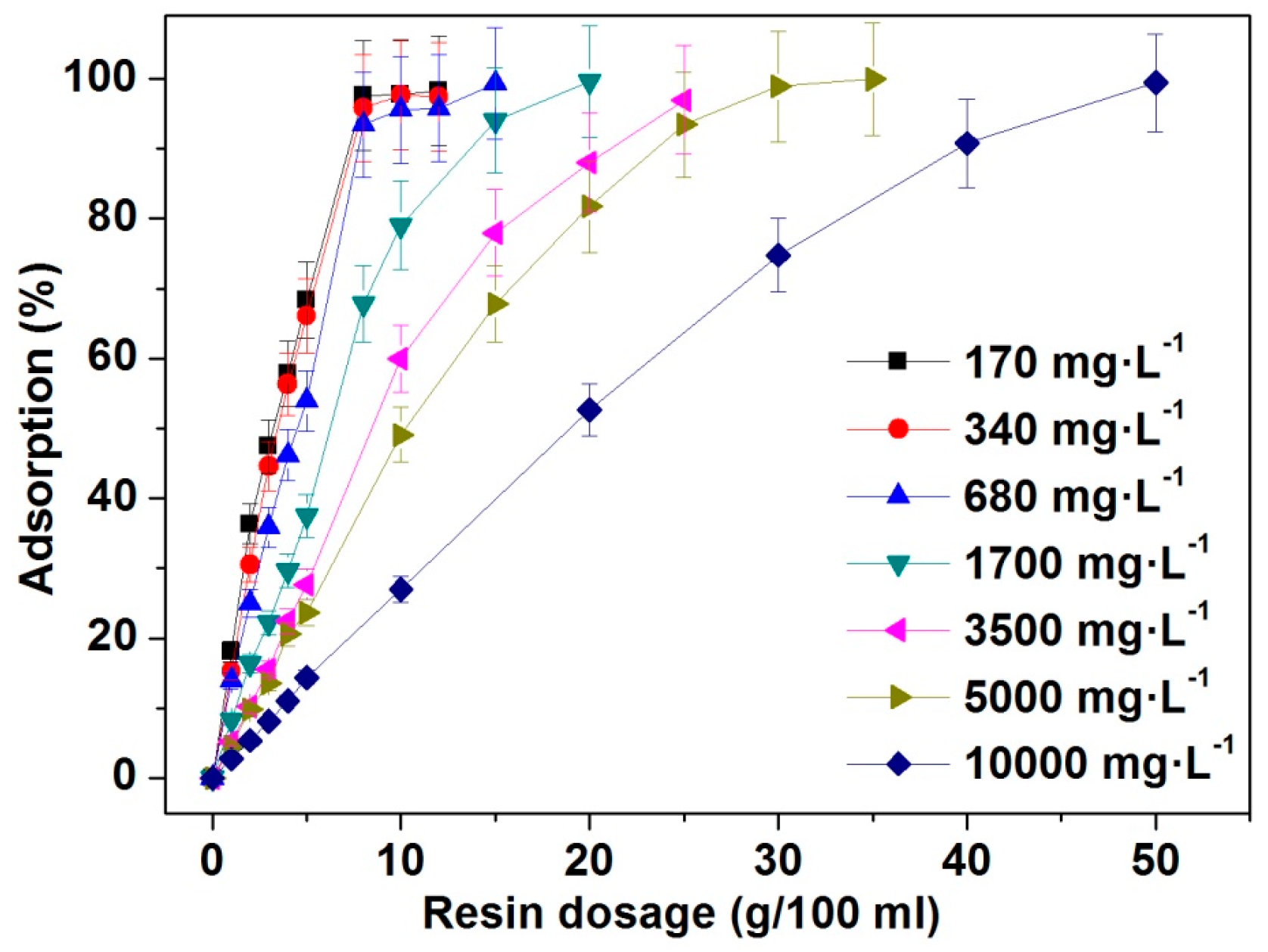
3.2. Effects of Resin Amount
3.3. Adsorption Isotherms
3.4. Thermodynamic Analysis
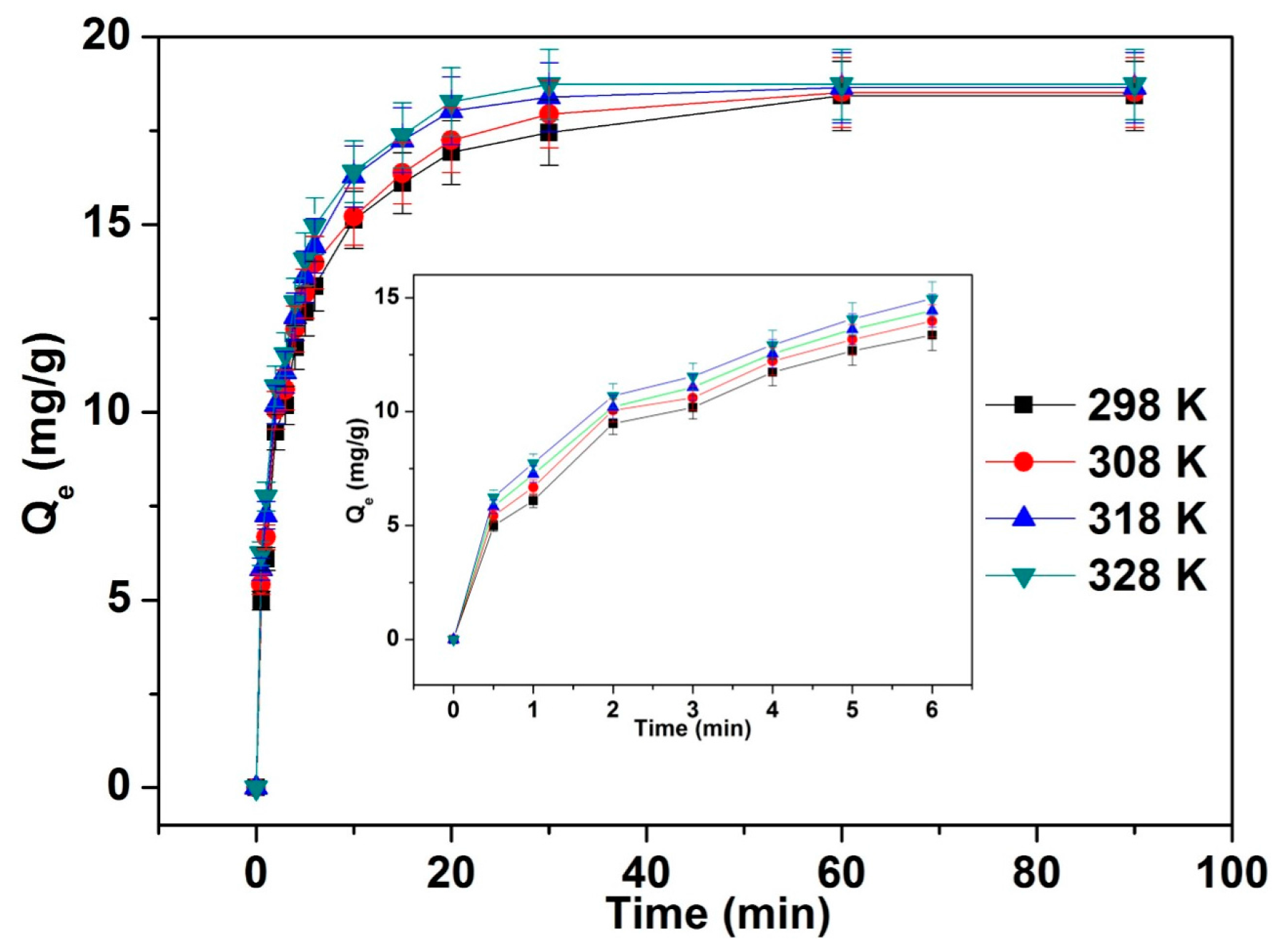
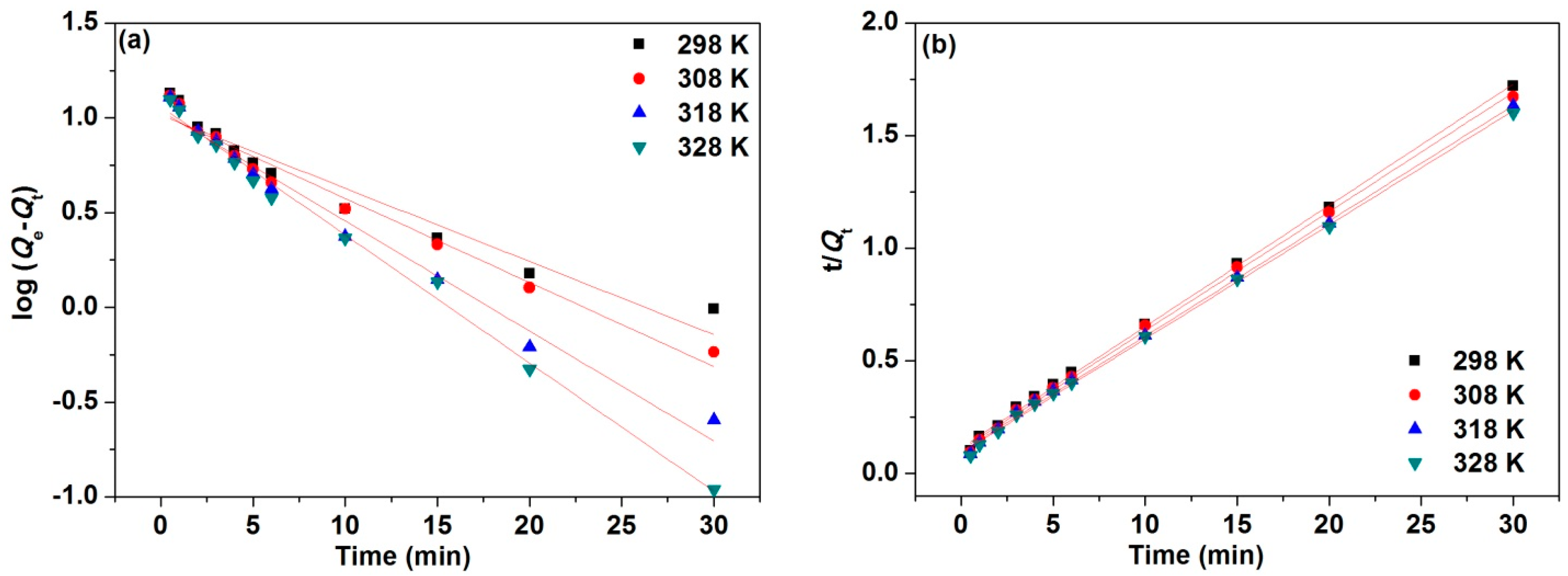
3.5. Adsorption Kinetics
3.6. Regeneration and Reuse
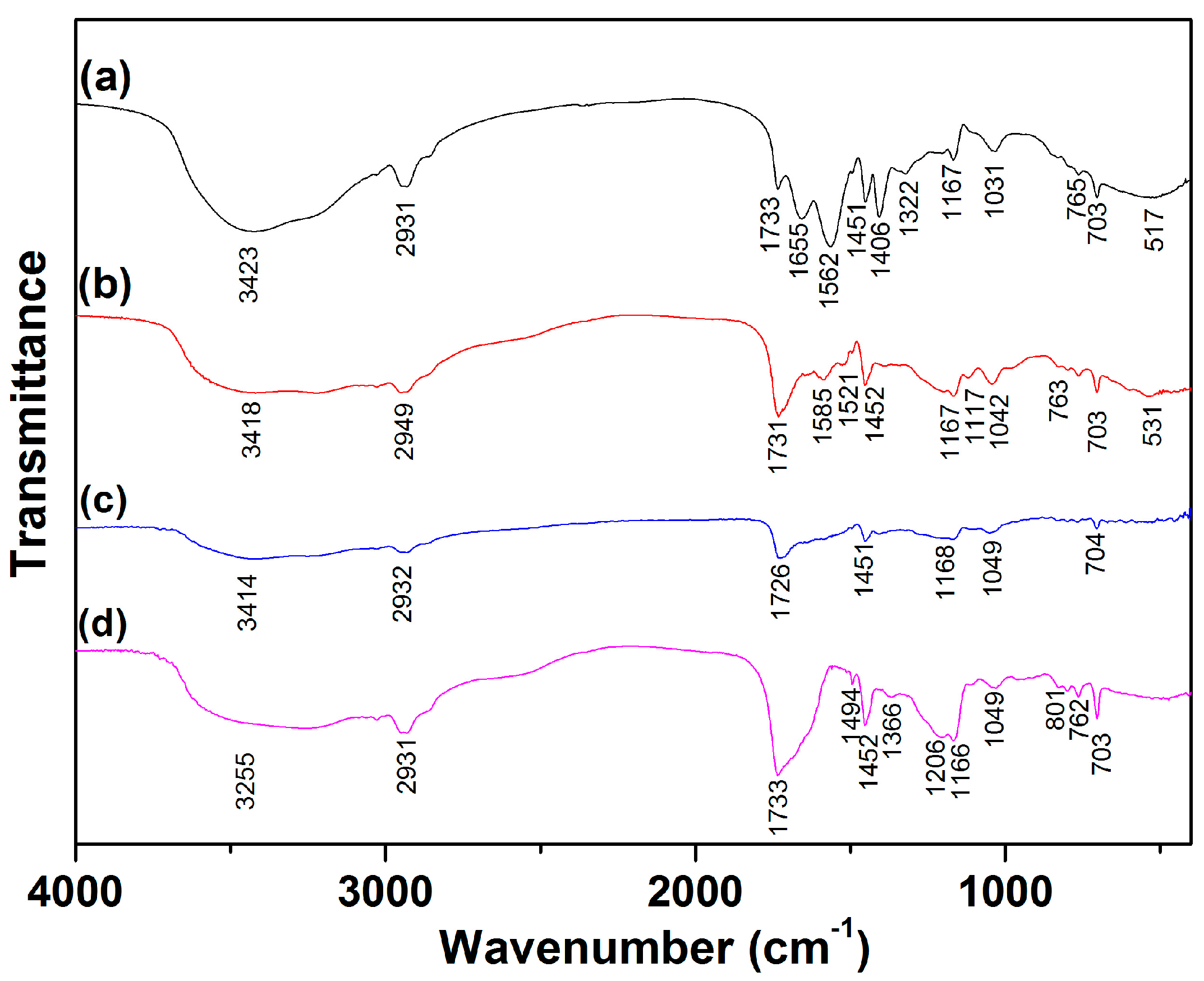
3.7. Adsorption Mechanisms
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Li, Y.; Qian, G.; Li, J.; Gerson, A.R. Kinetics and roles of solution and surface species of chalcopyrite dissolution at 650 mV. Geochim. Cosmochim. Acta 2015, 161, 188–202. [Google Scholar] [CrossRef]
- Li, Y.; Wei, Z.; Qian, G.; Li, J.; Gerson, A. Kinetics and mechanisms of chalcopyrite dissolution at controlled redox potential of 750 mV in sulfuric acid solution. Minerals 2016, 6, 83. [Google Scholar] [CrossRef]
- Dong, C.L.; Mattesini, M.; Augustsson, A.; Wen, X.G.; Zhang, W.X.; Yang, S.H.; Persson, C.; Ahuja, R.; Lüning, J.; Chang, C.L.; et al. Electronic structure and surface structure of Cu2S nanorods from polarization dependent X-ray absorption spectroscopy. J. Electron Spectrosc. Relat. Phenom. 2006, 151, 64–70. [Google Scholar] [CrossRef]
- Majuste, D.; Ciminelli, V.S.T.; Osseo-Asare, K.; Dantas, M.S.S.; Magalhães-Paniago, R. Electrochemical dissolution of chalcopyrite: Detection of bornite by synchrotron small angle X-ray diffraction and its correlation with the hindered dissolution process. Hydrometallurgy 2012, 111–112, 114–123. [Google Scholar] [CrossRef]
- Wei, Z.; Li, Y.; Xiao, Q.; Song, S. The influence of impurity monovalent cations adsorption on reconstructed chalcopyrite (001)-s surface in leaching process. Minerals 2016, 6, 89. [Google Scholar] [CrossRef]
- Li, Y.; Kawashima, N.; Li, J.; Chandra, A.P.; Gerson, A.R. A review of the structure, and fundamental mechanisms and kinetics of the leaching of chalcopyrite. Adv. Colloid Interface Sci. 2013, 197–198, 1–32. [Google Scholar] [CrossRef] [PubMed]
- Córdoba, E.M.; Muñoz, J.A.; Blázquez, M.L.; González, F.; Ballester, A. Leaching of chalcopyrite with ferric ion. Part I: General aspects. Hydrometallurgy 2008, 93, 81–87. [Google Scholar] [CrossRef]
- Sinha, M.K.; Sahu, S.K.; Pramanik, S.; Prasad, L.B.; Pandey, B.D. Recovery of high value copper and zinc oxide powder from waste brass pickle liquor by solvent extraction. Hydrometallurgy 2016, 165, 182–190. [Google Scholar] [CrossRef]
- Cheng, C.Y.; Barnard, K.R.; Zhang, W.; Zhu, Z.; Pranolo, Y. Recovery of nickel, cobalt, copper and zinc in sulphate and chloride solutions using synergistic solvent extraction. Chin. J. Chem. Eng. 2016, 24, 237–248. [Google Scholar] [CrossRef]
- Shahcheraghi, S.H.; Khayati, G.R.; Ranjbar, M. Improved dynamic modeling and simulation of an industrial copper solvent extraction process. Hydrometallurgy 2016. [Google Scholar] [CrossRef]
- Devi, N. Solvent extraction and separation of copper from base metals using bifunctional ionic liquid from sulfate medium. Trans. Nonferrous Met. Soc. China 2016, 26, 874–881. [Google Scholar] [CrossRef]
- Karnib, M.; Kabbani, A.; Holail, H.; Olama, Z. Heavy metals removal using activated carbon, silica and silica activated carbon composite. Energy Procedia 2014, 50, 113–120. [Google Scholar] [CrossRef]
- Sheet, I.; Kabbani, A.; Holail, H. Removal of heavy metals using nanostructured graphite oxide, silica nanoparticles and silica/graphite oxide composite. Energy Procedia 2014, 50, 130–138. [Google Scholar] [CrossRef]
- El-Bahy, S.M.; El-Bahy, Z.M. Synthesis and characterization of polyamidoxime chelating resin for adsorption of Cu(II), Mn(II) and Ni(II) by batch and column study. J. Environ. Chem. Eng. 2016, 4, 276–286. [Google Scholar] [CrossRef]
- Mukherjee, R.; Bhunia, P.; De, S. Impact of graphene oxide on removal of heavy metals using mixed matrix membrane. Chem. Eng. J. 2016, 292, 284–297. [Google Scholar] [CrossRef]
- Bowden, L.I.; Johnson, K.L.; Jarvis, A.P.; Robinson, H.; Ghazireh, N.; Younger, P.L. The use of Basic Oxygen Steel Furnace Slag (BOS) as a High Surface Area Media for the Removal of Iron from Circumneutral Mine Waters. In Proceedings of the 7th International Conference on Acid Rock Drainage (ICARD), St. Louis, MO, USA, 26–30 March 2006; pp. 25–30.
- Name, T.; Sheridan, C. Remediation of acid mine drainage using metallurgical slags. Miner. Eng. 2014, 64, 15–22. [Google Scholar] [CrossRef]
- Kruse, N.; Mackey, A.; Bowman, J.; Brewster, K.; Riefler, R.G. Alkalinity production as an indicator of failure in steel slag leach beds treating acid mine drainage. Environ. Earth Sci. 2012, 67, 1389–1395. [Google Scholar] [CrossRef]
- Deepatana, A.; Valix, M. Recovery of nickel and cobalt from organic acid complexes: Adsorption mechanisms of metal-organic complexes onto aminophosphonate chelating resin. J. Hazard. Mater. 2006, 137, 925–933. [Google Scholar] [CrossRef] [PubMed]
- Shukla, S.; Pai, R.S.; Shendarkar, A.D. Adsorption of Ni(II), Zn(II) and Fe(II) on modified coir fibres. Separat. Purif. Technol. 2006, 47, 141–147. [Google Scholar] [CrossRef]
- Pramanik, S.; Dhara, P.K.; Chattopadhyay, P. A chelating resin containing bis(2-benzimidazolylmethyl) amine: Synthesis and metal-ion uptake properties suitable for analytical application. Talanta 2004, 63, 485–490. [Google Scholar] [CrossRef] [PubMed]
- Jing, X.; Liu, F.; Yang, X.; Ling, P.; Li, L.; Long, C.; Li, A. Adsorption performances and mechanisms of the newly synthesized N,N′-di(carboxymethyl) dithiocarbamate chelating resin toward divalent heavy metal ions from aqueous media. J. Hazard. Mater. 2009, 167, 589–596. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Zhao, H.; Teasdale, P.; John, R.; Zhang, S. Synthesis and characterisation of a polyacrylamide-polyacrylic acid copolymer hydrogel for environmental analysis of Cu and Cd. React. Funct. Polym. 2002, 52, 31–41. [Google Scholar] [CrossRef]
- Dakova, I.; Karadjova, I.; Ivanov, I.; Georgieva, V.; Evtimova, B.; Georgiev, G. Solid phase selective separation and preconcentration of Cu(II) by Cu(II)-imprinted polymethacrylic microbeads. Anal. Chim. Acta 2007, 584, 196–203. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Chang, X.; Yang, D.; Guo, Y.; Meng, S. Highly selective determination of inorganic mercury (II) after preconcentration with Hg(II)-imprinted diazoaminobenzene-vinylpyridine copolymers. Anal. Chim. Acta 2005, 538, 85–91. [Google Scholar] [CrossRef]
- Bao, Y.; Yuan, F.; Zhao, X.; Liu, Q.; Gao, Y. Equilibrium and kinetic studies on the adsorption debittering process of ponkan (citrus reticulata blanco) juice using macroporous resins. Food Bioprod. Process. 2015, 94, 199–207. [Google Scholar] [CrossRef]
- Liu, Y.; Di, D.; Bai, Q.; Li, J.; Chen, Z.; Lou, S.; Ye, H. Preparative separation and purification of rebaudioside a from steviol glycosides using mixed-mode macroporous adsorption resins. J. Agric. Food Chem. 2011, 59, 9629–9636. [Google Scholar] [CrossRef] [PubMed]
- Namasivayam, C.; Kadirvelu, K. Uptake of mercury(II) from wastewater by activated carbon from an unwanted agricultural solid by-product: Coirpith. Carbon 1999, 37, 79–84. [Google Scholar] [CrossRef]
- Ho, Y.S.; McKay, G. Kinetic models for the sorption of dye from aqueous solution by wood. Process Saf. Environ. Prot. 1998, 76, 183–191. [Google Scholar] [CrossRef]
- Yeddou, N.; Bensmaili, A. Kinetic models for the sorption of dye from aqueous solution by clay-wood sawdust mixture. Desalination 2005, 185, 499–508. [Google Scholar] [CrossRef]
- Zhang, Y.L.; Yu, X.J.; Wang, Q.L.; Jiang, Z.J.; Fang, T. Adsorption of zinc onto anionic ion-exchange resin from cyanide barren solution. Chin. J. Chem. Eng. 2015, 23, 646–651. [Google Scholar] [CrossRef]
- Doğan, M.; Abak, H.; Alkan, M. Adsorption of methylene blue onto hazelnut shell: Kinetics, mechanism and activation parameters. J. Hazard. Mater. 2009, 164, 172–181. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.-Y.; Chiang, C.-L.; Chen, C.-R. Removal of heavy metal ions by a chelating resin containing glycine as chelating groups. Separat. Purif. Technol. 2007, 54, 396–403. [Google Scholar] [CrossRef]
- Al-Muallem, H.A.; Wazeer, M.I.M.; Ali, S.A. Synthesis and solution properties of a new pH-responsive polymer containing amino acid residues. Polymer 2002, 43, 4285–4295. [Google Scholar] [CrossRef]
- Lee, W.; Lee, S.-E.; Lee, C.-H.; Kim, Y.-S.; Lee, Y.-I. A chelating resin containing 1-(2-thiazolylazo)-2-naphthol as the functional group; synthesis and sorption behavior for trace metal ions. Microchem. J. 2001, 70, 195–203. [Google Scholar] [CrossRef]
- Gupta, A.P.; Varshney, P.K. Studies on tetracycline hydrochloride sorbed zirconium tungstophosphate; La(III)-selective chelating ion exchanger. React. Funct. Polym. 1996, 31, 111–116. [Google Scholar] [CrossRef]

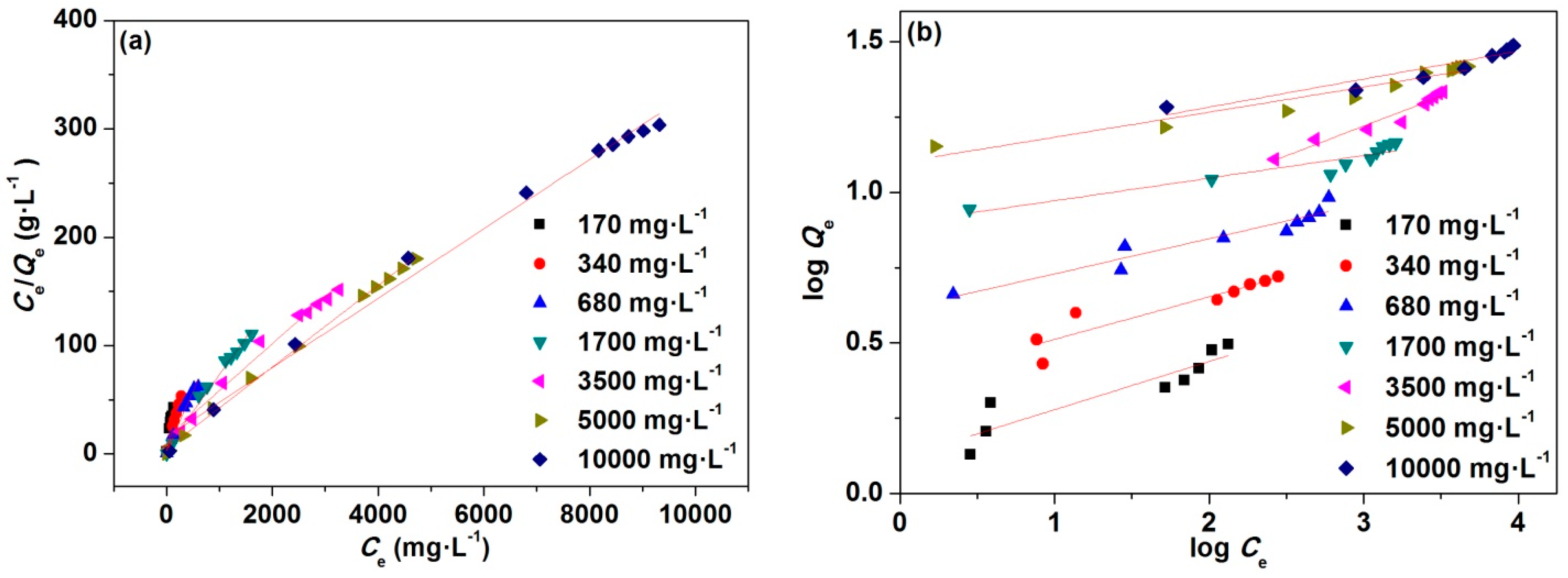


| Concentration (mg·L−1) | Langmuir Isotherm | Freundlich Isotherms | |||||
|---|---|---|---|---|---|---|---|
| Qm (exp) (mg·g−1) | Qm (cal) (mg·g−1) | b (L·mg−1) | R2 | Kf | 1/n | R2 | |
| 170 | 3.1273 | 3.0562 | 0.1288 | 0.9655 | 1.3152 | 0.1598 | 0.8169 |
| 340 | 5.2458 | 5.2604 | 0.1020 | 0.9946 | 2.3496 | 0.1409 | 0.8369 |
| 680 | 9.5836 | 9.0580 | 0.0401 | 0.9806 | 4.0986 | 0.1167 | 0.9102 |
| 1700 | 14.5470 | 14.6628 | 0.0132 | 0.9861 | 7.8864 | 0.0749 | 0.8714 |
| 3500 | 21.5379 | 23.1481 | 0.0098 | 0.9838 | 4.2565 | 0.1969 | 0.9447 |
| 5000 | 26.1574 | 26.5957 | 0.0077 | 0.9977 | 12.5707 | 0.0831 | 0.9316 |
| 10,000 | 30.6689 | 31.2500 | 0.0021 | 0.9919 | 12.4825 | 0.0930 | 0.8924 |
| T (K) | First Order | Second Order | ||||||
|---|---|---|---|---|---|---|---|---|
| Qe (exp, mg·g−1) | k1 (min−1) | Qe (mg·g−1) | R2 | k2 (g·min−1·mg−1) | Qe (mg·g−1) | t1/2 (min) | R2 | |
| 298 | 18.43 | 0.0888 | 10.3412 | 0.9342 | 0.0253 | 18.5908 | 2.1257 | 0.9987 |
| 308 | 18.52 | 0.1020 | 10.4323 | 0.9683 | 0.0263 | 18.9322 | 2.0112 | 0.9983 |
| 318 | 18.65 | 0.1336 | 10.8665 | 0.9815 | 0.0267 | 19.5389 | 1.9134 | 0.9987 |
| 328 | 18.74 | 0.1557 | 11.4583 | 0.9926 | 0.0281 | 19.7472 | 1.8021 | 0.9987 |
| T (K) | (exp) (J·mol−1) | (cal) (J·mol−1) | |
|---|---|---|---|
| 298 | −6613.66 | −6588.58 | 14.4121 |
| 308 | −7036.82 | −7081.67 | 15.5904 |
| 318 | −7582.72 | −7574.75 | 17.5790 |
| 328 | −8078.50 | −8067.84 | 19.3186 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, Y.; Wang, X.; Xiao, Q.; Zhang, X. Study on Selective Removal of Impurity Iron from Leached Copper-Bearing Solution Using a Chelating Resin. Minerals 2016, 6, 106. https://doi.org/10.3390/min6040106
Li Y, Wang X, Xiao Q, Zhang X. Study on Selective Removal of Impurity Iron from Leached Copper-Bearing Solution Using a Chelating Resin. Minerals. 2016; 6(4):106. https://doi.org/10.3390/min6040106
Chicago/Turabian StyleLi, Yubiao, Xinyu Wang, Qing Xiao, and Xu Zhang. 2016. "Study on Selective Removal of Impurity Iron from Leached Copper-Bearing Solution Using a Chelating Resin" Minerals 6, no. 4: 106. https://doi.org/10.3390/min6040106







