Uranium, Cesium, and Mercury Leaching and Recovery from Cemented Radioactive Wastes in Sulfuric Acid and Iodide Media
Abstract
:1. Introduction
2. Experimental Section
2.1. SRCW Preparation
| Element | Concentration | Added as |
|---|---|---|
| Aluminum | 26,371 ppm | Al(NO3)3·9H2O |
| Uranium | 4760 ppm | UO2(NO3)3·6H2O |
| Mercury | 6622 ppm | Hg(NO3)2·H2O |
| Rubidium | 6.44 ppm | RbNO3 |
| Cesium | 29.1 ppm | CsNO3 |
| Strontium | 25.9 ppm | Sr(NO3)2 |
| Barium | 31.4 ppm | Ba(NO3)2 |
| Ruthenium | 30.5 ppm | 1.5% RuNO(NO3)3 solution |
| Lanthanum | 20.0 ppm | La(NO3)2·6H2O |
| Cerium | 54.5 ppm | Ce(NO3)3·6H2O |
| Praseodymium | 14.3 ppm | Pr(NO3)3·6H2O |
| Neodymium | 41.0 ppm | Nd(NO3)3·6H2O |
| Samarium | 5.11 ppm | Sm(NO3)3·6H2O |
| Europium | 0.75 ppm | Eu(NO3)3·6H2O |
| Gadolinium | 0.23 ppm | Gd(NO3)3·6H2O |
| Yttrium | 11.3 ppm | Y(NO3)3·6H2O |
| Iron | 55.9 ppm | Fe(NO3)3·9H2O |
| Nickel | 10.3 ppm | Ni(NO3)2·6H2O |
| Chromium | 17.0 ppm | Cr(NO3)3·9H2O |
| Nitric acid | 0.36 M | HNO3 (16M) |
| Batch Code | Cement Type | S/C Ratio* | Aging Time | Aging Temperature | Cs (ppm) | Hg (ppm) | U (ppm) |
|---|---|---|---|---|---|---|---|
| U5 | GU | 0.29 | 7 days | Ambient | 7.9 | 1598 | 1182 |
| U8 | GU | 0.39 | 18 months | Ambient | 8.6 | 1785 | 1177 |
| U10 | GU | 0.29 | 18 months | Ambient | 8.2 | 1462 | 959 |
| U13 | GU | 0.29 | 18 months | 60 °C | 3.5 | 1116 | 893 |
| U18 | GU | 0.39 | 18 months | Ambient | 5.8 | 1093 | 759 |
| U20 | HE | 0.39 | 18 months | Ambient | 8.1 | 1644 | 1243 |
| U22 | HE | 0.29 | 18 months | Ambient | 7.3 | 1415 | 1,018 |
| U24 | HE | 0.21 | 18 months | Ambient | 6.0 | 1121 | 785 |
| U28 | GU | 0.39 | 30 months | 60 °C | 8.5 | 1973 | 1150 |
| U29 | GU | 0.29 | 30 months | Ambient | 7.2 | 1977 | 1104 |
| U32 | GU | 0.29 | 30 months | 60 °C | 6.0 | 1277 | 850 |
| U34 | HE | 0.39 | 30 months | Ambient | 8.0 | 2651 | 1355 |
2.2. Leaching Experiments
2.3. Key Elements Recovery
2.4. Analytical
3. Results and Discussion
3.1. Mercury Oxidation in Old Wastes

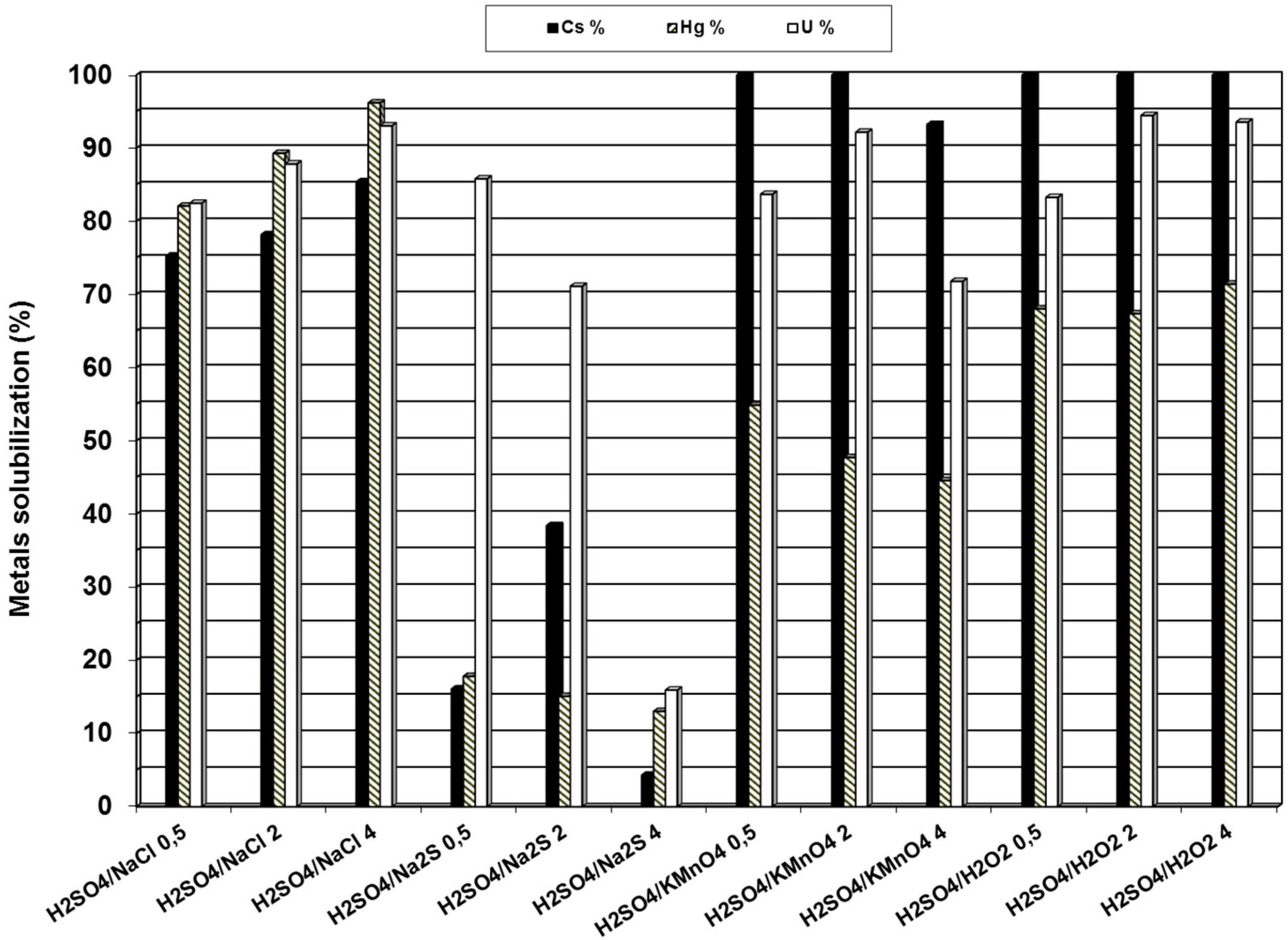
3.2. Use of Potassium Iodide as Mercury Oxidant
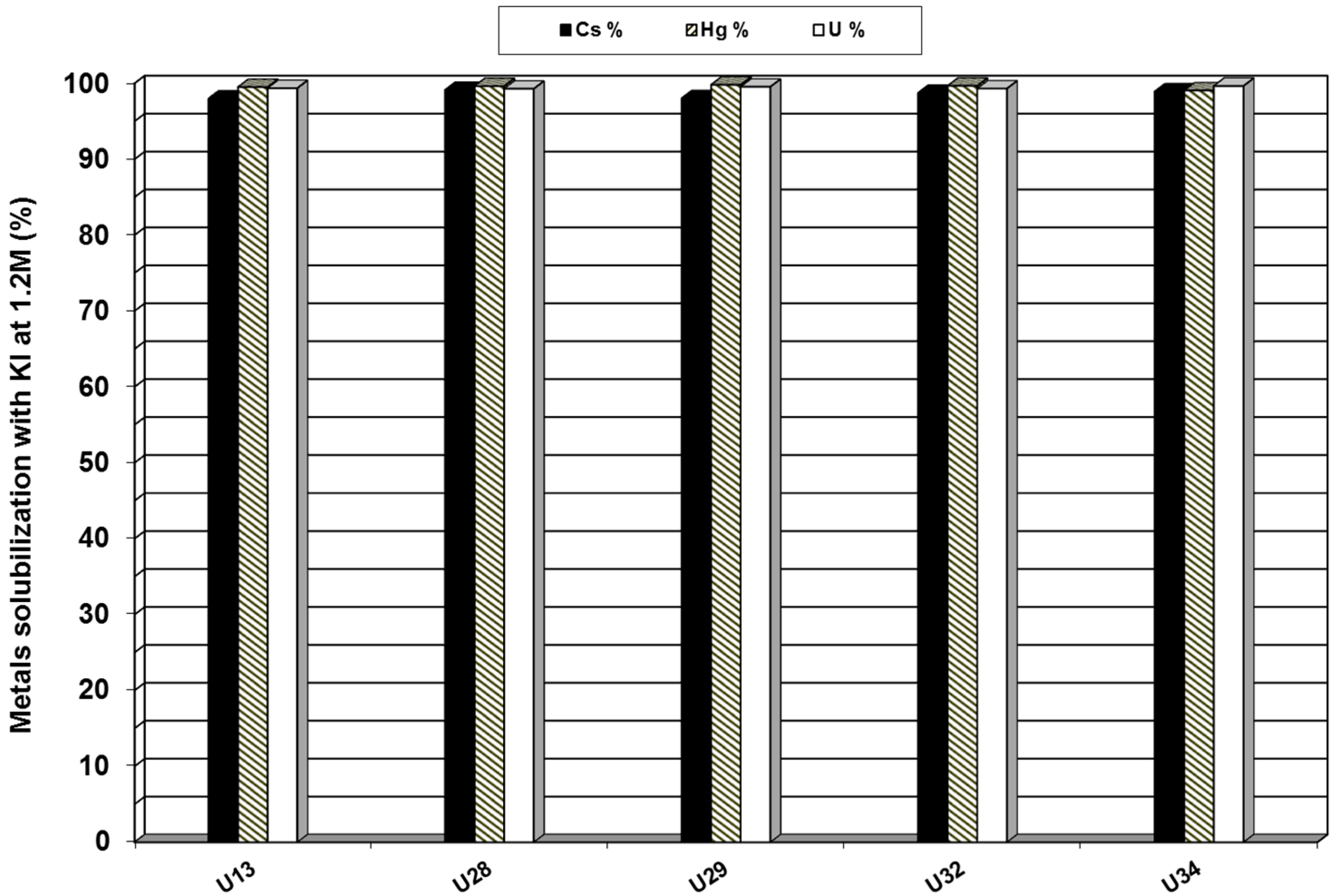
3.3. Leaching Optimization in Iodide Media
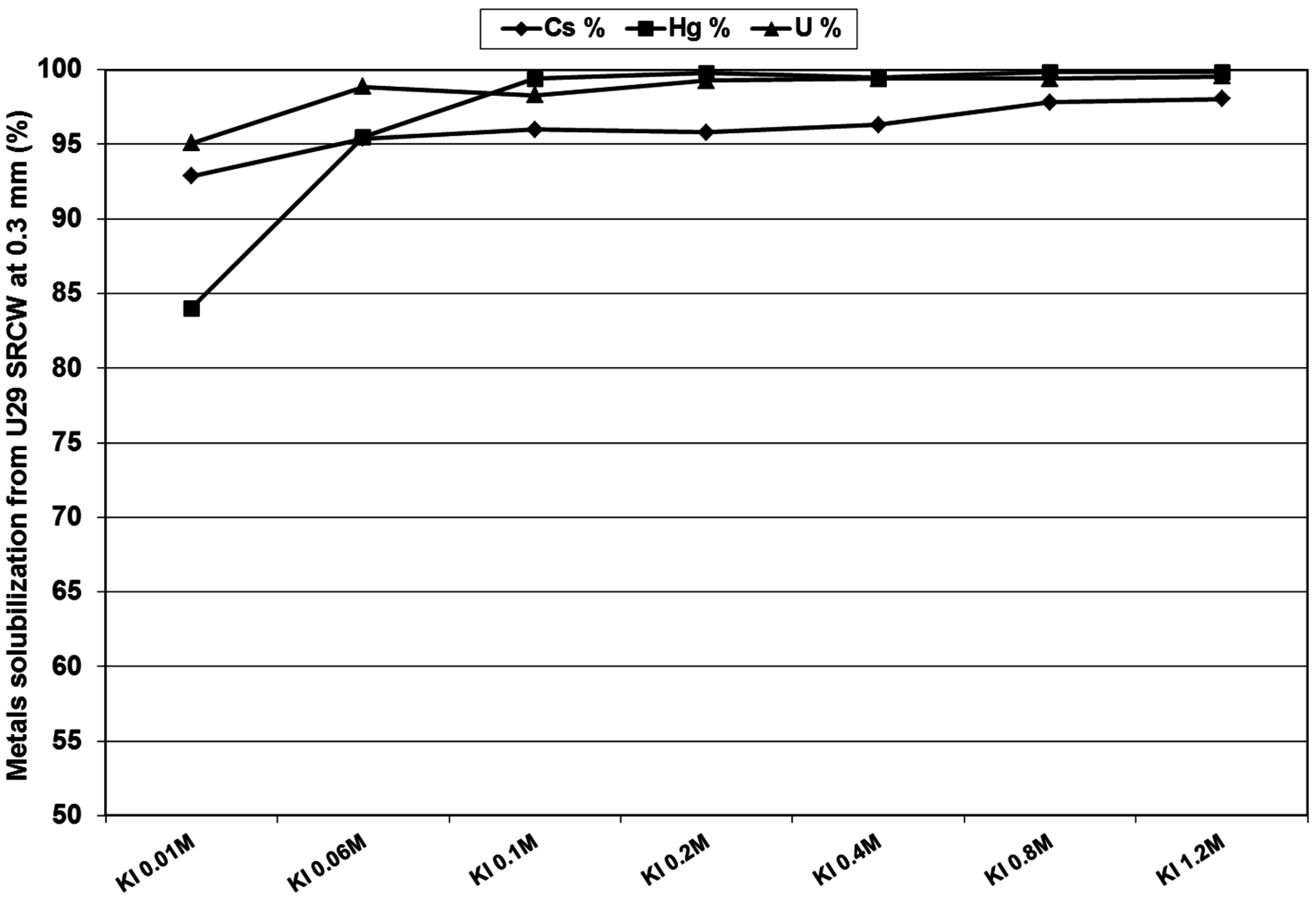
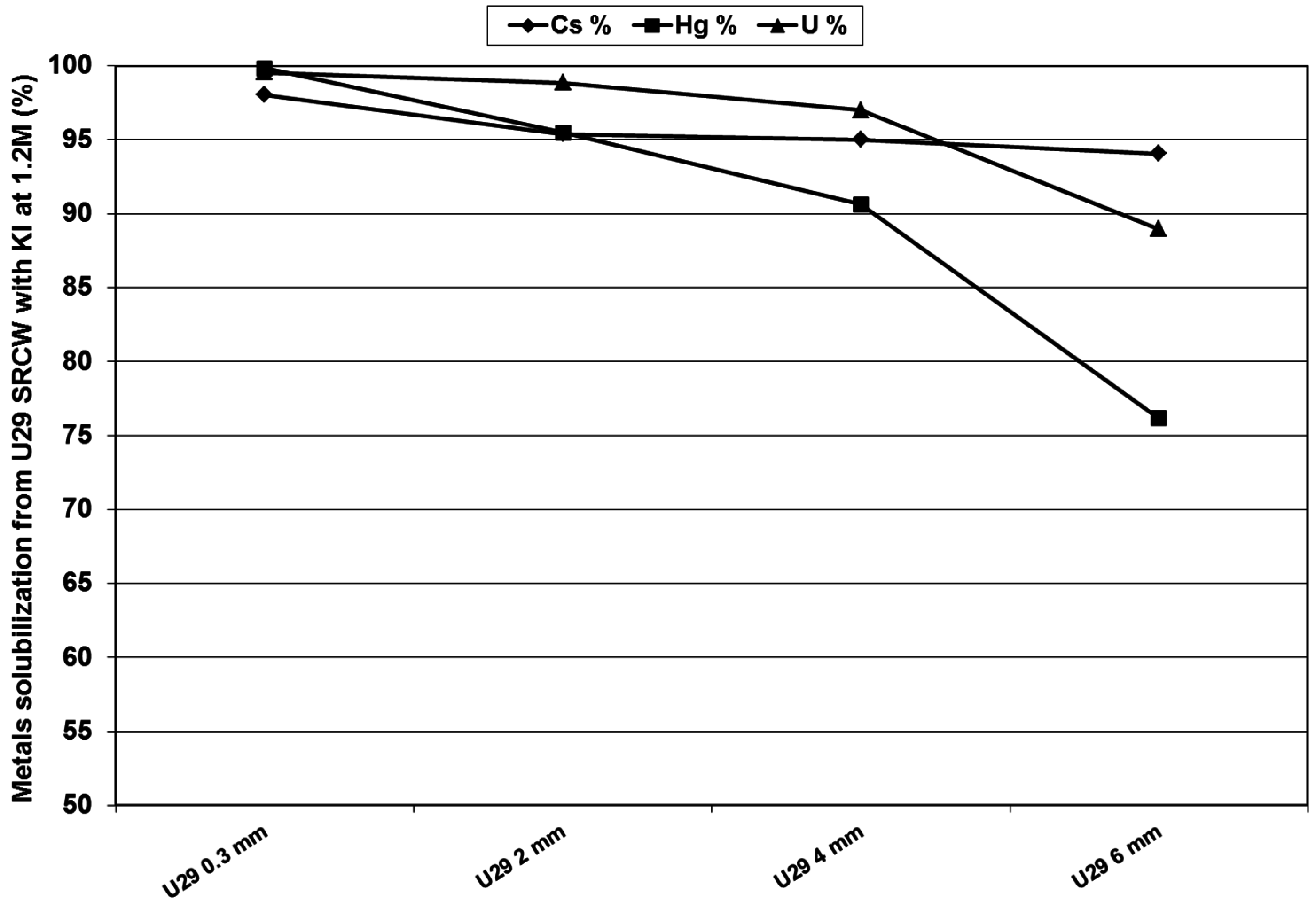

3.4. Separation and Recovery of Uranium
3.5. Reuse of Washing Solutions for Subsequent Leaching
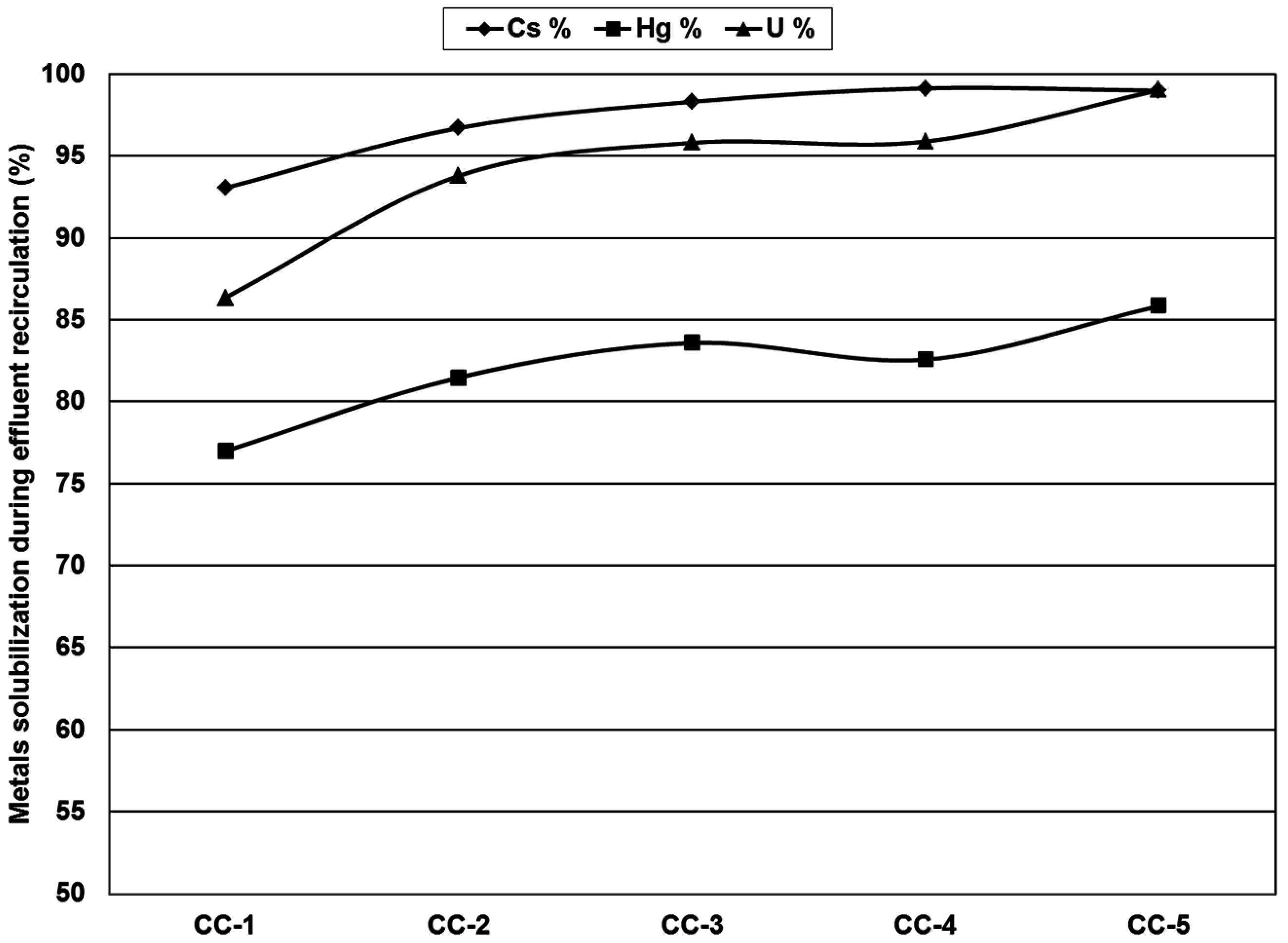
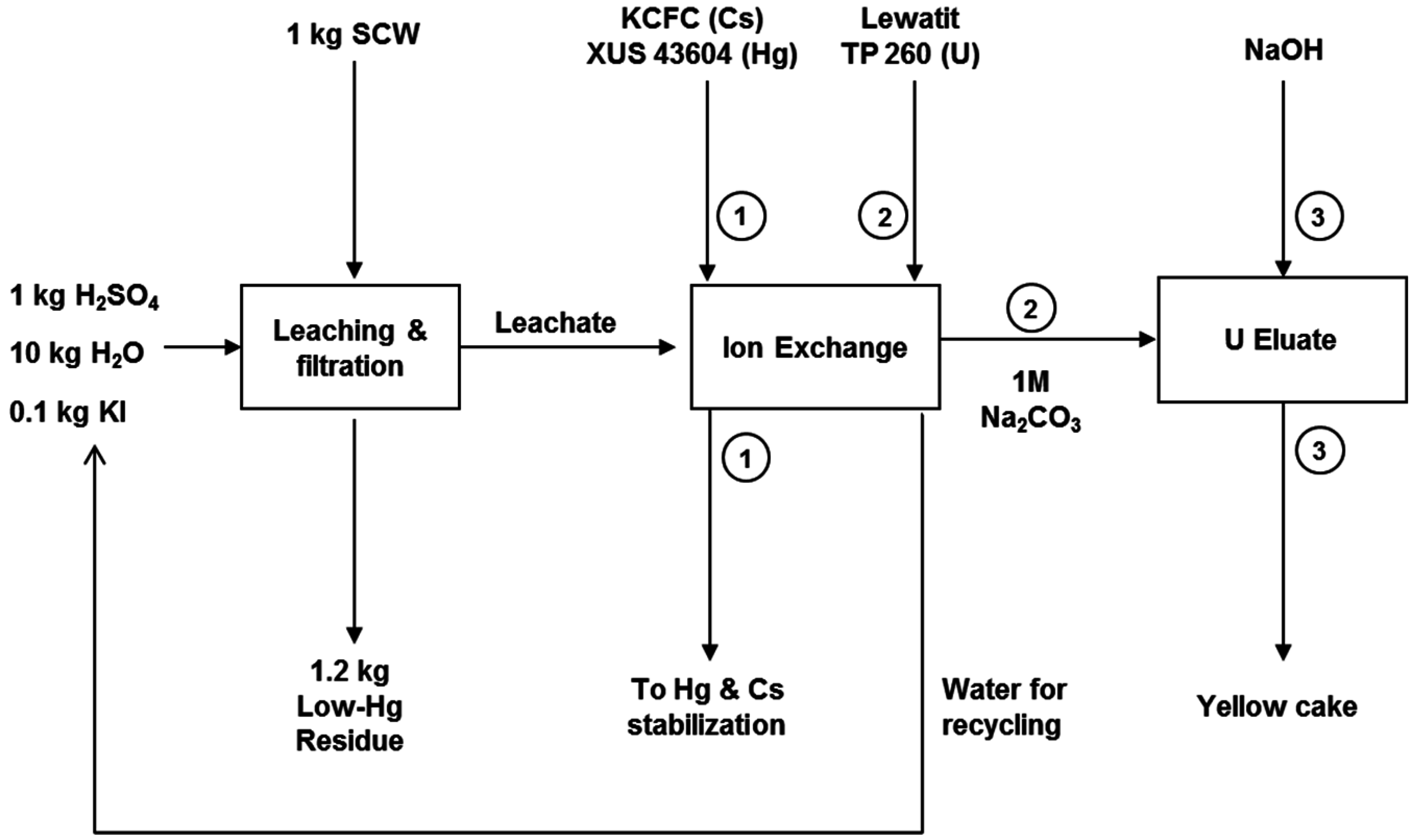
3.6. Process Overview, Flow Sheet and Economic Analysis
4. Conclusions
Author Contributions
Conflicts of Interest
References
- Lemire, R.J.; Larouche, M.M.A.; Tosello, N.B. Scoping Studies on the Treatment of Cemented Waste from 99Mo Production; AECL Report RC-1245; AECL: Chalk River, ON, Canada, 1994. [Google Scholar]
- Bilodeau, A.; Bouzoubaâ, N.; Chevrier, R.; Kwong, Y.T.J.; Nkinamubanzi, P.C.; Riveros, P. Literature Review Related to Cemented Nuclear Wastes; Report 07–061(CR); CANMET-MMSL: Ottawa, ON, Canada, 2008. [Google Scholar]
- Riveros, P.A. A Literature Survey of the Cement-Based Solidification/Stabilization of Radioactive Wastes Containing Cesium, Mercury and Uranium; Report 10–016(CR); CANMET-MMSL: Ottawa, ON, Canada, 2010. [Google Scholar]
- Merritt, R.C. The extractive metallurgy of uranium. In Colorado School of Mines Research Institute; Johnson Publishing Company, Boulder, Co.: Washington, D.C., USA, 1971. [Google Scholar]
- Wilkinson, W.D. Uranium Metallurgy, Volume I (Uranium Process Metallurgy); John Wiley and Sons: Hoboken, NJ, USA, 1962. [Google Scholar]
- Queneau, P.B.; Berthold, C.E. Silica in hydrometallurgy: An overview. Can. Metall. Q. 1986, 25, 201–209. [Google Scholar] [CrossRef]
- Riveros, P.; Lastra, R.; Khandelwal, A.; Laviolette, C.; Demers, A. Task C: Determination of the Chemical Characteristics of Surrogate Cemented Wastes; Report 13–013(CR); CANMET-MMSL: Ottawa, ON, Canada, 2013. [Google Scholar]
- Reynier, N.; Lastra, R.; Laviolette, C.; Bouzoubaa, N.; Chapman, M. Optimization and Validation of a Chemical Process for Uranium, Mercury and Cesium Leaching from Cemented Radioactive Wastes; AECL Nuclear Review, ANR-D-15–00033; AECL Nuclear review (ANR): Chalk River, ON, Canada, 2015. [Google Scholar]
- Gavrilescu, M.; Pavel, L.V.; Cretescu, I. Characterization and remediation of soils contaminated with uranium. J. Hazard. Mater. 2009, 163, 475–510. [Google Scholar] [CrossRef] [PubMed]
- Sors, M.; Bouzanne, M. The analysis of cements by atomic absorption spectrometry. Bull. Liaison Lab. Ponts Chaussees 1977, 88, 69–76. [Google Scholar]
- Marrero, T.W.; Morris, J.S.; Manahan, S.E. Radioactive waste forms stabilized by ChemChar gasification: Characterization and leaching behavior of cerium, thorium, protactinium, uranium, and neptunium. Chemosphere 2004, 54, 873–885. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Wang, T.; Mallhi, H.; Liu, Y.; Ban, H.; Ladwig, K. The role of ammonia on mercury leaching from coal fly ash. Chemosphere 2007, 69, 1586–1592. [Google Scholar] [CrossRef] [PubMed]
- Sellier, A.; Buffo-Lacarriere, L.; El Gonnouni, M.; Bourbon, X. Behavior of HPC nuclear waste disposal structures in leaching environment. Nucl. Eng. Des. 2011, 241, 40–414. [Google Scholar] [CrossRef]
- Wong, S.F.; Chui, P.C.; Cahyadi, J.H.; Goh, C.P. An accelerated leaching test for cement mortar. In Proceedings of the 28th Conference on Our World in Concrete & Structures, Singapore, Singapore, 28–29 August 2003.
- Carde, C.; Escadeillas, G.; François, R. Use of ammonium nitrate solution to simulate and accelerate the leaching of cemented pastes due to deionized water. Mag. Concrete Res. 1997, 49, 295–301. [Google Scholar] [CrossRef]
- Callaway, W.S.; Huber, H.J. Results of Physicochemical Characterization and Caustic Dissolution Tests on Tank 241-C-108 Heel Solids. Washington River Protection Solutions; LAB-RPT-10–00001, Prepared for the U.S. Department of Energy; Office of River Protection: Richland, WA, USA, 2010. [Google Scholar]
- Miller, R.L.; Welch, J.M.; Flinn, J.E. Uranium recovery from a nuclear fuel waste form. Trans. Am. Inst. Min. Metall. Petroleum Eng. Soc. 1985, 278, 51–52. [Google Scholar]
- Kitts, F.G.; Perona, J.J. A Preliminary Study, of Pre-Solvent Extraction Treatment of Stainless Steel-Uranium Fuels with Dilute Aqua Regia; Oak Ridge National Laboratory: Oak Ridge, TN, USA, 1957. [Google Scholar]
- Utton, C.A.; Hand, R.J.; Bingham, P.A.; Hyatt, N.C.; Swanton, S.W.; Williams, S.J. Dissolution of vitrified wastes in a high-pH calcium-rich solution. J. Nuclear Mater. 2013, 435, 112–122. [Google Scholar] [CrossRef]
- Francis, C.W.; Timpson, M.E.; Wilson, J.H. Bench- and pilot-scale studies relating to the removal of uranium from uranium-contaminated soils using carbonate and citrate lixiviants. J. Hazard. Mater. 1999, 66, 67–87. [Google Scholar] [CrossRef]
- Reynier, N.; Blais, J.F.; Mercier, G.; Besner, S. Optimization of arsenic and pentachlorophenol removal from soil using an experimental design methodology. J. Soils Sediments 2013, 13, 1189–1200. [Google Scholar] [CrossRef]
- Lafond, S.; Blais, J.F.; Martel, R.; Mercier, G. Chemical Leaching of Antimony and Other Metals from Small Arms Shooting Range Soil. Water Air Soil Pollut. 2013, 224, 1371. [Google Scholar] [CrossRef]
- Vosburgh, W.C.; Lackey, O.N. A mercury-basic mercury sulfate voltaic cell. J. Am. Chem. Soc. 1930, 52, 1407–1410. [Google Scholar] [CrossRef]
- Fiset, J.F.; Lastra, R.; Bilodeau, A.; Bouzoubaâ, N. Characterization of Surrogate Radioactive Cemented Waste: A Laboratory Study. In Proceedings of the Waste Management, Decommissioning and Environmental Restauration for Canada’s Nuclear Activities, Toronto, ON, Canada, 11–14 September 2011.
- Foust, D.F. Extraction of Mercury and Mercury Compounds from Contaminated Material and Solutions. U.S. Patent 5,226,545, 1993. [Google Scholar]
- Klasson, T.K.; Koran, L.J. Removal of Mercury from Solids Using the Potassium Iodide/Iodine Leaching Process; Oak Ridge National Laboratory: Oak Ridge, TN, USA, 1997. [Google Scholar]
- Zhang, H.; Jeffery, C.A.; Jeffrey, M.I. Ion exchange recovery of gold from iodine-iodide solutions. Hydrometallurgy 2012, 125, 69–75. [Google Scholar] [CrossRef]
- Yu, Z.; Feng, Q.; Ou, L.; Lu, Y.; Zhang, G. Selective leaching of a high-iron cobalt matte at atmospheric pressures. Sep. Purif. Technol. 2007, 53, 1–7. [Google Scholar] [CrossRef]
- Zakharov, M.K. Minimal extractant flow rate in counter-current leaching. Theor. Found. Chem. Eng. 2005, 39, 325–328. [Google Scholar] [CrossRef]
- Janin, A.; Riche, P.; Blais, J.-F.; Mercier, G.; Cooper, P.; Morris, P. Counter-current acid leaching process for copper azole treated wood waste. Environ. Technol. 2012, 33, 2111–2118. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors and Her Majesty the Queen in Right of Canada; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Reynier, N.; Lastra, R.; Laviolette, C.; Fiset, J.-F.; Bouzoubaâ, N.; Chapman, M. Uranium, Cesium, and Mercury Leaching and Recovery from Cemented Radioactive Wastes in Sulfuric Acid and Iodide Media. Minerals 2015, 5, 744-757. https://doi.org/10.3390/min5040522
Reynier N, Lastra R, Laviolette C, Fiset J-F, Bouzoubaâ N, Chapman M. Uranium, Cesium, and Mercury Leaching and Recovery from Cemented Radioactive Wastes in Sulfuric Acid and Iodide Media. Minerals. 2015; 5(4):744-757. https://doi.org/10.3390/min5040522
Chicago/Turabian StyleReynier, Nicolas, Rolando Lastra, Cheryl Laviolette, Jean-François Fiset, Nabil Bouzoubaâ, and Mark Chapman. 2015. "Uranium, Cesium, and Mercury Leaching and Recovery from Cemented Radioactive Wastes in Sulfuric Acid and Iodide Media" Minerals 5, no. 4: 744-757. https://doi.org/10.3390/min5040522







