1. Introduction
The Niquelândia igneous complex, exposed in Central Goias (
Figure 1A), was recognized as a layered intrusion [
1]. According to geochemical and geochronological data, the complex was emplaced in the Middle Proterozoic (1560–1600 Ma) as a result of continental rifting and underwent amphibolite to granulite facies metamorphism at about 770–795 Ma [
2,
3]. Final convergence and continental collision caused deformation and disruption at about 630 Ma [
4]. During Lower-Tertiary erosion, corresponding to the
Sul-Americano cycle, ultramafic units in the lower part of the intrusion underwent supergene alteration and weathering originating in a thick laterite cover with silicate-Ni deposits [
5].
The Niquelândia complex is similar to many other layered intrusions (
i.e., Bushveld, Stillwater, Great Dyke, Campo Formoso), and it contains chromite deposits (
Figure 1B) with associated platinum-group element (PGE) geochemical anomalies [
6]. A study of polished sections and heavy-mineral concentrates revealed that the majority of the PGE occur in specific platinum-group minerals (PGM) occurring as microscopic grains (<20 µm) unevenly disseminated in chromitites and their host rock [
7,
8,
9]. As pointed out by [
7], the chromitites contain two genetically distinct populations of PGM: The “primary” PGM, crystallized at high temperatures, and the “secondary” PGM, formed at relatively low temperatures, during post-magmatic evolution of the complex.
In this study, we summarize data from previous works, and present new results of a detailed investigation of the chromite-PGM paragenesis showing that the primary PGM were deposited at high temperature under relatively high sulfur fugacity, and that they underwent mineralogical reworking at low-temperature under variable redox conditions. This was a result of serpentinization (hydrothermal?) and weathering alteration (lateritization) of the ultramafic rocks under favorable morphologic and climatic conditions.
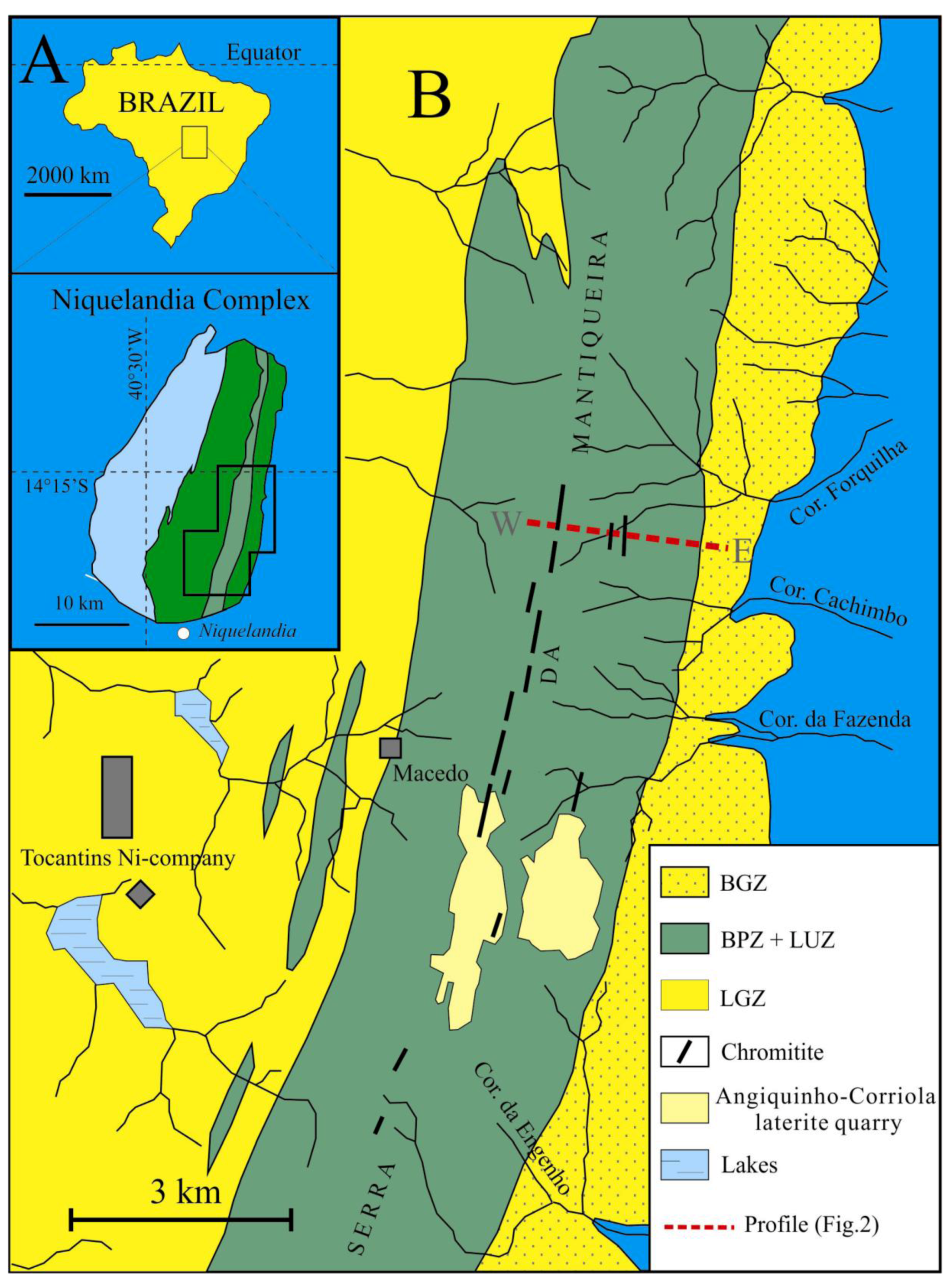
Figure 1.
(
A) Geographic location of the Niquelândia igneous complex; (
B) Geological setting of the chromitite layers; BGZ = basal gabbro zone, BPZ + LUZ = basal peridotite zone and layered ultramafic zone, LGZ = lower gabbro zone, W−E = geologic profile in
Figure 2 (Modified after [
6]).
Figure 1.
(
A) Geographic location of the Niquelândia igneous complex; (
B) Geological setting of the chromitite layers; BGZ = basal gabbro zone, BPZ + LUZ = basal peridotite zone and layered ultramafic zone, LGZ = lower gabbro zone, W−E = geologic profile in
Figure 2 (Modified after [
6]).
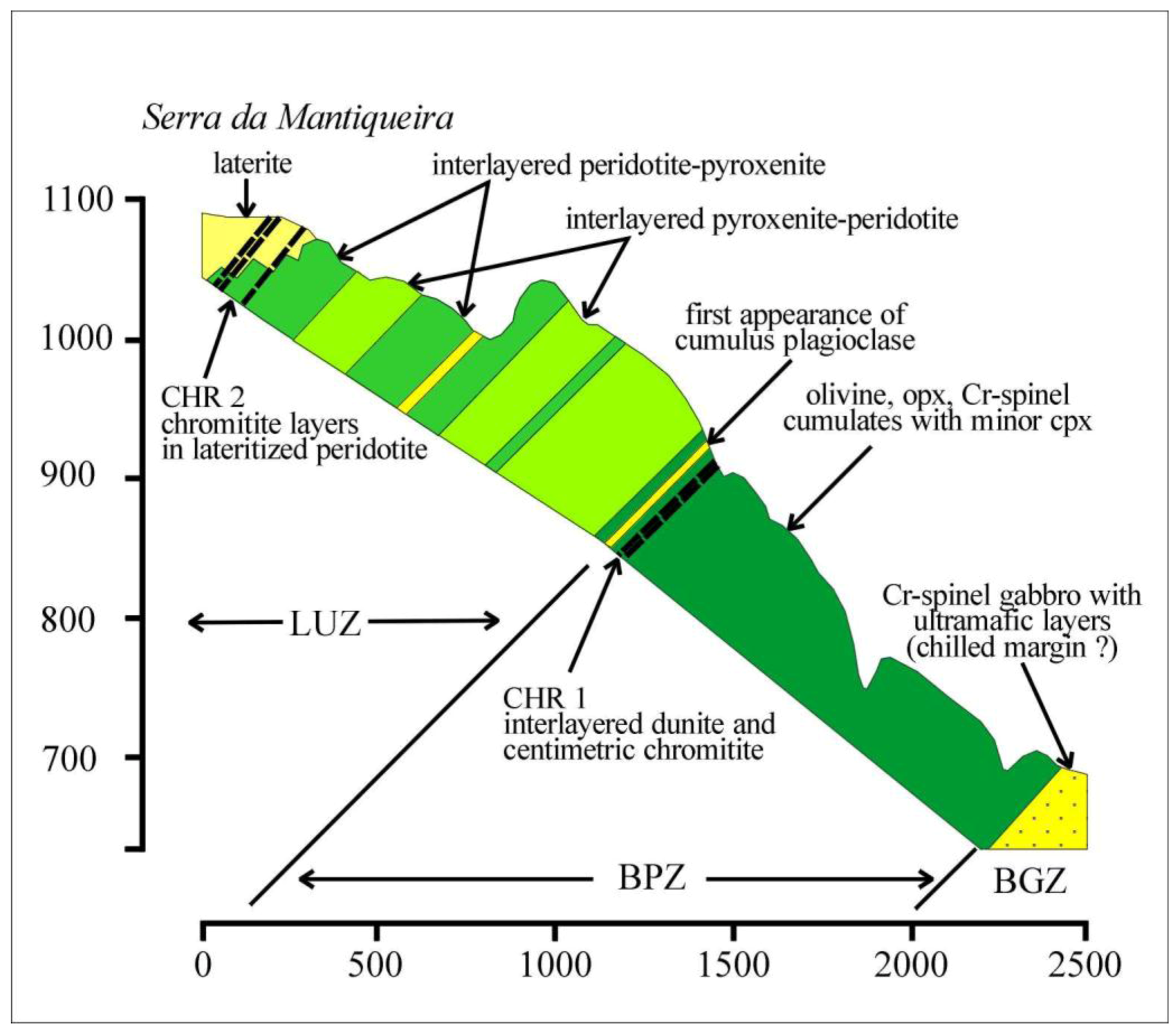
Figure 2.
Geologic profile W–E showing stratigraphic location of the two chromitite horizons CHR-1 and CHR-2; Note that the upper chromitite CHR-2 occurs within the laterite cover in the highland of Serra da Mantiqueira. (Vertical and horizontal scale are in meters).
Figure 2.
Geologic profile W–E showing stratigraphic location of the two chromitite horizons CHR-1 and CHR-2; Note that the upper chromitite CHR-2 occurs within the laterite cover in the highland of Serra da Mantiqueira. (Vertical and horizontal scale are in meters).
2. Analytical Techniques
This study is based on the investigation of 50 polished sections obtained from 16 chromitite samples and 6 host rocks, collected along a geologic profile cutting across the two major chromitite horizons in the ultramafic zone of the Niquelândia layered intrusion (
Figure 2).
Electron microprobe analyses of chromite, silicates and PGM were performed using a Superprobe JEOL JXA 8200 instrument operated at the Eugen F. Stumpfl Laboratory of Leoben University (Austria). Back scattered electron (BSE) images of the PGM were obtained at the Leoben laboratory as well as at the Serveis Cientificotècnicos of Barcelona University.
Chromite and silicates were analyzed in WDS mode with an accelerating voltage of 15 kV and a beam current of 10 nA, using the Kα lines for Mg, Si, Ca, K, Al, F, Cl, Ti, V, Cr, Mn, Fe, Ni and Zn. Natural K-feldspar, albite, atacamite, diopside, chromite, rhodonite, ilmenite and metallic V, Ni and Zn were used as reference materials. The counting times for peak and background were 20 s and 10 s respectively. The amount of Fe3+ in the spinel was calculated assuming the ideal spinel stoichiometry, R2+O and R3+2O3. X-ray Diffraction was used in the study of the laterite matrix of the CHR-2 chromitite.
The PGM and associated sulfides had been previously located by scanning polished sections using both reflected light (in air) and electron microscopes. They were quantitatively analyzed in the WDS mode, with an accelerating voltage of 20 kV and 10 nA beam current, allowing a beam diameter of about 1 µm or less. The counting times on peak and backgrounds were 15 s and 5 s respectively. The Kα lines were used for S, As, Fe, Cu and Ni; Lα for Rh, Pd, Pt, Sb and Te, and Mα for Hg. The reference materials were pure metals for the PGE, synthetic NiS, SbBi and Pd3HgTe, natural chalcopyrite and niccolite for Ni, Hg, Fe, Cu, S, Te, Sb and As. The following diffracting crystals were selected: PETJ for S, Te; PETH for Ru, Os, Rh, Hg, Sb; LIF for Cu; LIFH for Ni, Ir, Pt; and TAP for As. Automatic corrections were performed for interferences involving Rh-Pd overlap. The detection limits (wt %) of the analyzed elements are: As = 0.40, S = 0.01, Sb = 0.05, Ni = 0.04, Fe = 0.02, Cu = 0.05, Os = 0.08, Ir = 0.1, Ru = 0.02, Rh = 0.01, Pt = 0.1, Pd = 0.02.
3. Geological Setting and Petrography of the Chromitites
Two major chromitite horizons have been documented so far in the ultramafic sequence of the Niquelândia layered intrusion [
7]. They are located at about 1 km and 2 km from the base, respectively, being concordant with the N-S strike and the 30°–60° westwards dipping of the igneous stratigraphy (
Figure 2). The two chromitite horizons display different styles of low-temperature alteration, apparently controlled by their topographic location in the landscape.
The lower horizon (CHR-1) is exposed along the eastern, steep slope of Serra da Mantiqueira, along the Corrego Forquilha. Here, lateritic weathering is absent, and the chromitite occurs as centimeter thick seams and schlieren within partly serpentinized peridotite composed of olivine and pyroxene relics in a matrix of lizardite, chrysotile, talc, chlorite and carbonates. The chromitite contains rare accessory sulfides accounting for 50–350 ppm total sulfur. The sulfides tend to increase in the adjacent dunite (400–1200 ppm sulfur) suggesting that sulfur saturation was achieved during this stage of fractionation [
10]. The drop-like morphology of the interstitial sulfides is consistent with segregation of an intercumulus sulfide liquid, although their mineral assemblage, mainly composed of pentlandite veined with magnetite, appears to be a result of late alteration [
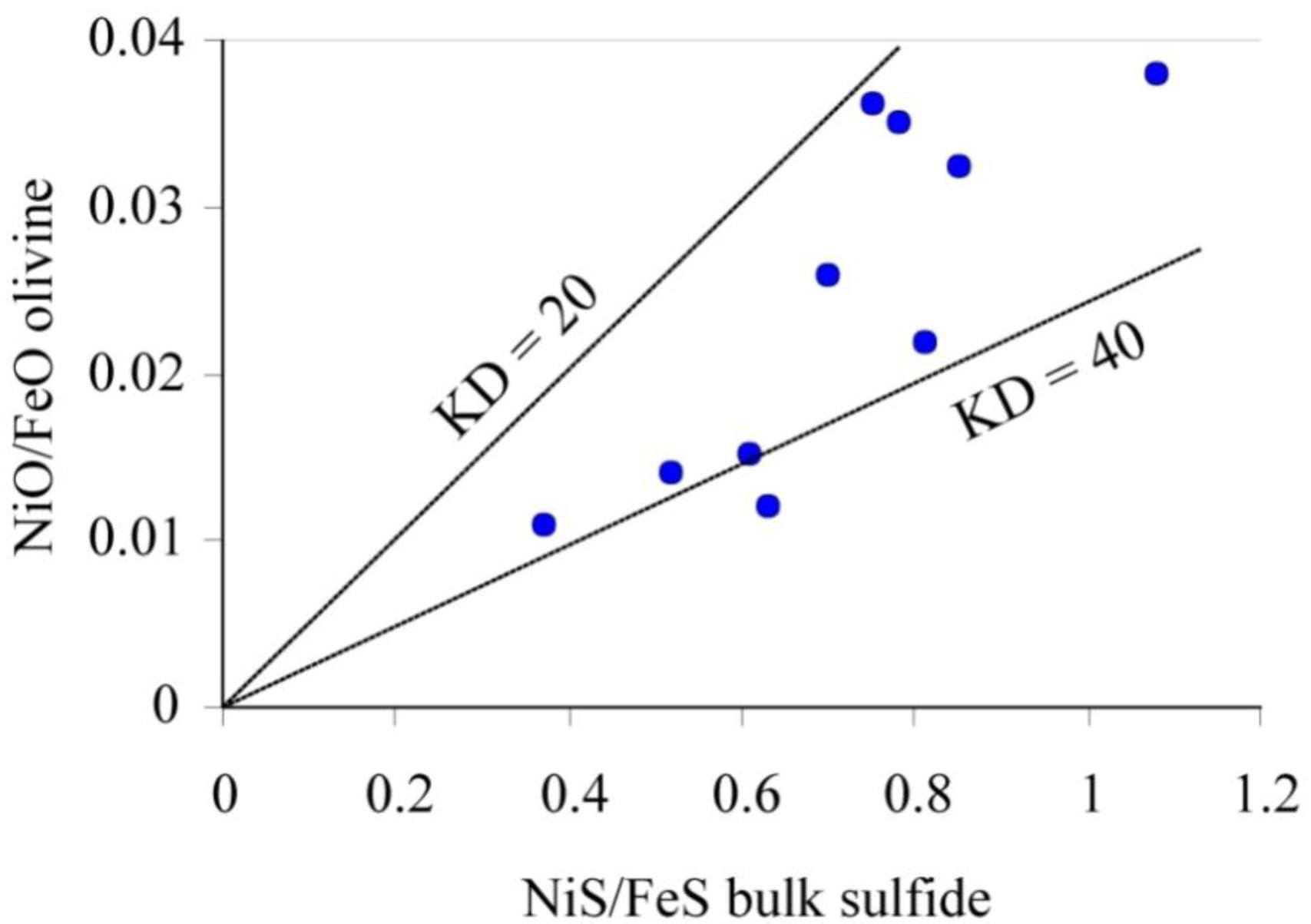
10]. The calculated Ni/Fe distribution-coefficient between olivine and sulfide is in excess of 20 (
Figure 3), indicating that the sulfide lost part of the Fe
2+ by oxidation, probably during serpentinization of the ultramafic rocks [
11].
The upper horizon (CHR-2) crops out in an almost flat area of the Serra da Mantiqueira highland. It embraces several disrupted layers of 5–30 cm, occasionally up to ~1 m thick, distributed over a stratigraphic interval of some hundred meters [
6].
The layers are completely embedded in a laterite soil derived from weathering of the original ultramafic country rock, presumably dunite or harzburgite. The CHR-2 chromitite consists of a hard, completely massive aggregate of closely interlocked chromite grains, up to 3 cm in size. Silicates are extremely scarce, completely altered to smectite, kaolinite, rare serpentine and amorphous silica, usually intergrown with amorphous Fe-hydroxides, goethite, limonite, hematite, ferrian-chromite and magnetite. Abundant Mg-ilmenite and rutile occurs as rounded or ameboid grains aligned along grain-boundaries and cracks in altered chromite (
Figure 4), suggesting splitting of Ti out of chromite during chemical weathering.
Figure 3.
Relationships between the NiO/FeO ratio in olivine and the NiS/FeS ratio in the coexisting sulfides from the partially serpentinized dunite hosting CHR-1 chromitite layers. Lines of constant distribution-coefficient KD = 20 and KD = 40 are indicated. The NiS/FeS ratio in sulfide was calculated combining the modal proportion and microprobe composition of pentlandite, chalcopyrite, and pyrrhotite constituting the interstitial droplets in the dunite.
Figure 3.
Relationships between the NiO/FeO ratio in olivine and the NiS/FeS ratio in the coexisting sulfides from the partially serpentinized dunite hosting CHR-1 chromitite layers. Lines of constant distribution-coefficient KD = 20 and KD = 40 are indicated. The NiS/FeS ratio in sulfide was calculated combining the modal proportion and microprobe composition of pentlandite, chalcopyrite, and pyrrhotite constituting the interstitial droplets in the dunite.
Figure 4.
Vermicular aggregates of Mg-ilmenite and rutile, and kaolinite filled pores in CHR-2 massive chromitite. Parallel Nichols reflected light picture, long side = 1.0 mm.
Figure 4.
Vermicular aggregates of Mg-ilmenite and rutile, and kaolinite filled pores in CHR-2 massive chromitite. Parallel Nichols reflected light picture, long side = 1.0 mm.
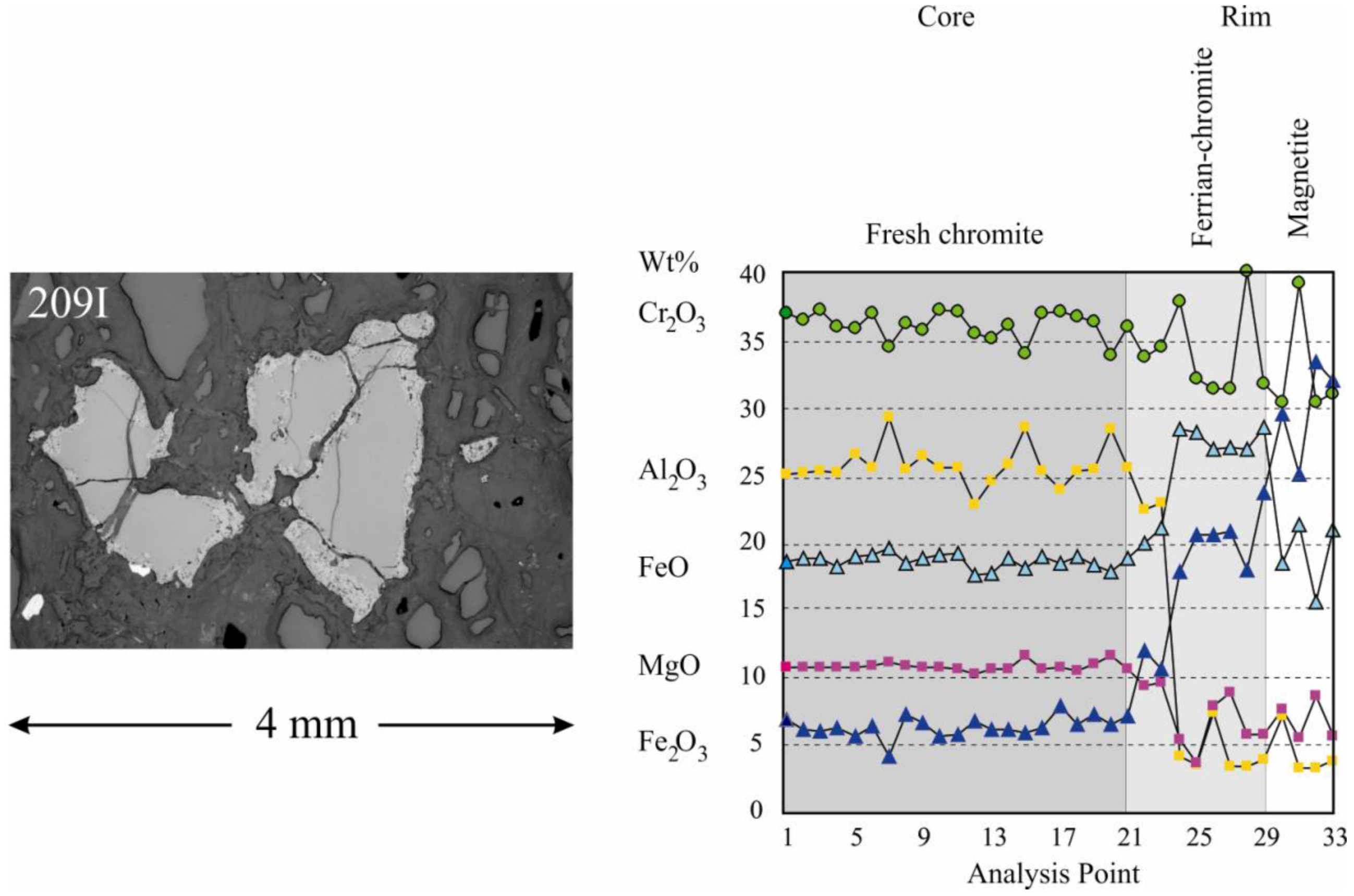
4. Chromite Alteration
Post-magmatic serpentinization and lateritic weathering have produced different types of alteration of the chromite.
Chromitites from the CHR-1 horizon are only slightly affected by serpentinization, showing limited alteration into ferrian-chromite along grain boundaries in contact with the serpentinized matrix. Alteration rims are more extensively developed in the accessory chromite in the dunite. The rims consist of spongy ferrian-chromite and chromian-magnetite locally intermixed with a fine grained chlorite (
Figure 5). Chemical changes involve sharp decrease of Al
2O
3 and MgO in the alteration rims, accompanied by an increase in FeO and Fe
2O
3, while Cr
2O
3 is floating but showing a general decrease. Notably, the Fe
2O
3/FeO increases remarkably in the outermost alteration zone, indicating progressive oxidation.
Figure 5.
Chemical variation across the chromitite core (fresh chromite) to rim (ferrian chromite) and boundary (chromian magnetite) zoning in accessory chromite from the serpentinized dunite host of the CHR-1 horizon.
Figure 5.
Chemical variation across the chromitite core (fresh chromite) to rim (ferrian chromite) and boundary (chromian magnetite) zoning in accessory chromite from the serpentinized dunite host of the CHR-1 horizon.
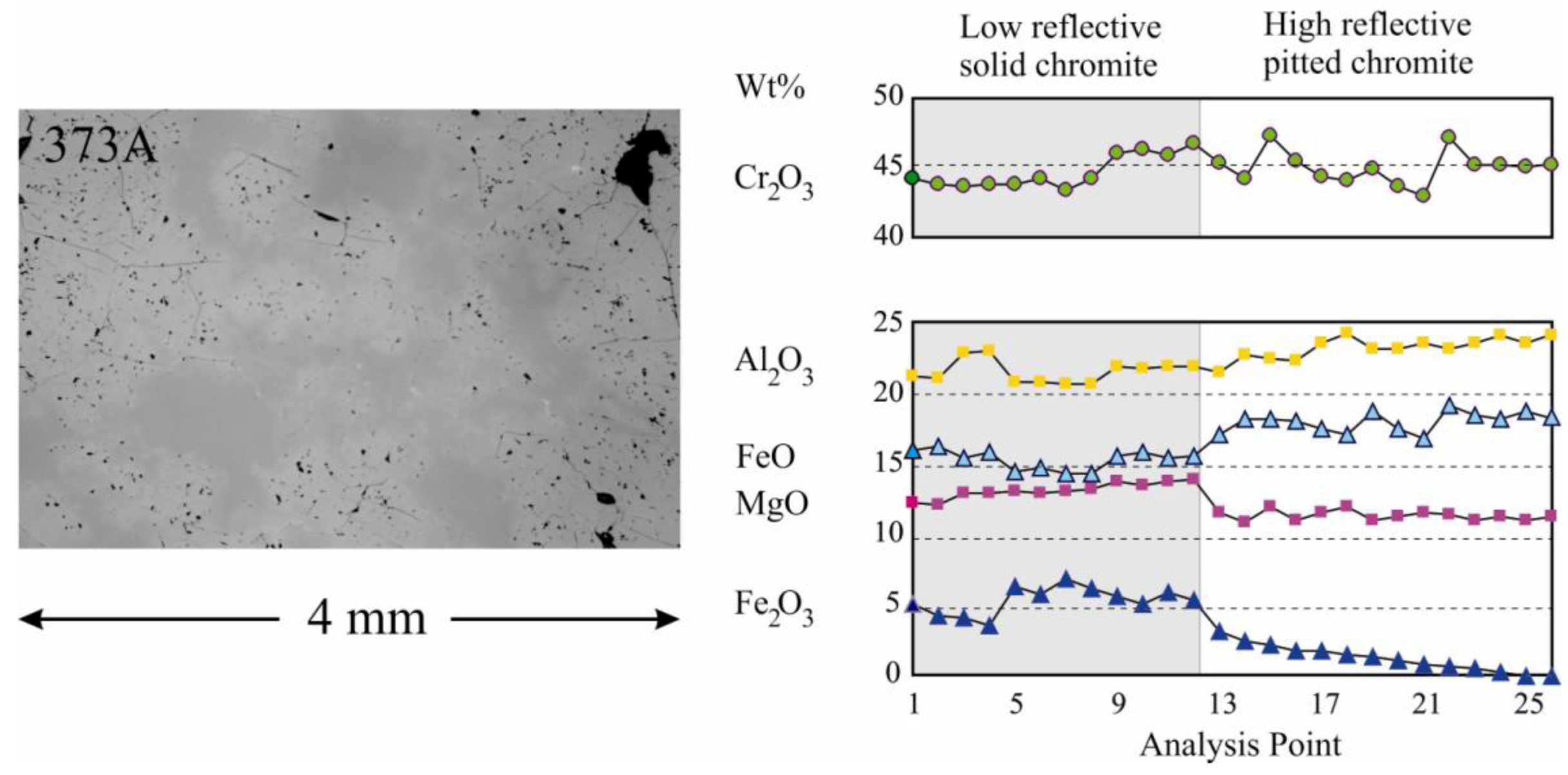
In the CHR-2 chromitites, chromite displays a complex zoning with a core composed of solid chromite, rimmed by a spinel phase densely pitted with pores (
Figure 6). The pores are filled with secondary silicate minerals (mainly kaolinite and smectite). The alteration pattern involves depletion of MgO and Fe
2O
3 in the pitted chromite and a general increase of Al
2O
3 and FeO, resulting in a slight increase of the reflectivity (
Figure 6). This alteration scheme showing a decrease of the Fe
2O
3/FeO ratio is contrary to that of the ferrian-chromite rims produced during serpentinization, and is ascribed to the different redox condition prevailing during lateritic weathering.
Figure 6.
Chromite alteration patterns in laterite hosted chromitite CHR-2. The dark, solid chromite may represent the primary magmatic composition. The more reflective, pitted chromite is believed to be a result of the deep chemical weathering under lateritic conditions.
Figure 6.
Chromite alteration patterns in laterite hosted chromitite CHR-2. The dark, solid chromite may represent the primary magmatic composition. The more reflective, pitted chromite is believed to be a result of the deep chemical weathering under lateritic conditions.
5. Mineralogy and Paragenesis of the Platinum-Group Minerals
A summary of the PGM recovered from the Niquelândia chromitite and dunite is given in
Table 1, while
Table 2 and
Table 3 provide a list of representative compositions obtained with an electron microprobe. The compositions of major PGM are presented in the relevant diagrams (
Figure 7).
Table 1.
Mineralogy and texture of Platinum-group minerals(PGM) in chromitites and associated dunite of the Niquelândia layered intrusion.
Table 1.
Mineralogy and texture of Platinum-group minerals(PGM) in chromitites and associated dunite of the Niquelândia layered intrusion.
| mineral | ideal formula | morphology | common association | host-phase | occurrence |
|---|
| laurite | (Ru,Os,Ir)S2 | euhedral-subhedral | pentladite-silicates | chromite-serpentine | CHR-1,CHR-2, dunite |
| erlichmanite | (Os,Ru,Ir)S2 | euhedral-subhedral | silicates | chromite | CHR-1,CHR-2 |
| kashinite * | Ir2S3 | subhedral | singlephase | chromite | CHR-2 |
| iridosmine | Os-Ir | euhedral-subhedral | erlichmanite-singlephase | chromite | CHR-1,CHR-2 |
| unnamed Ir-Ni sulfide | (Ir,Ni)-S | bleb | pentladite | chromite | CHR-1 |
| undetermined Os-Ir sulfide | (Os,Ir)-S | bleb | pentladite | chromite | CHR-2 |
| irarsite | IrAsS | subhedral | laurite-iridosmine | chromite | CHR-2 |
| malanite | Cu(Pt,Ir)2S4 | subhedral | singlephase | chromite | CHR-2 |
| unnamed Ru-Fe oxide | Ru-O | rounded | laurite | Fe-chromite | CHR-2 |
| isoferroplatino-type * | Pt3Fe | anhedral (?) | Fe-hydroxide | Fe-chromite | CHR-2 |
| tetraferroplatino type | Pt(Fe,Ni,Cu) | anhedral | kaolinite, Fe-hydroxide | Fe-chromite | CHR-1,CHR-2 |
| tetraferroplatino-type | PtFe | rounded | singlephase | orthopyroxene | dunite |
| geversite | Pt(Sb,Bi)2 | euhedral | pentlandite-magnetite | interstitial sulfide bleb | dunite |
| sudburyite | (Pd,Ni)Sb | euhedral | pentlandite-magnetite | interstitial sulfide bleb | dunite |
| sobolevskite | PdSb | euhedral | pentlandite-magnetite | interstitial sulfide bleb | dunite |
| kotulskite | Pd(Te,Bi) | euhedral | pentlandite-magnetite | interstitial sulfide bleb | dunite |
| moncheite | PtTe2 | anhedral | Fe-chromite | accessory chromite | dunite |
| stumpflite | Pt(Sb,Bi) | anhedral | Fe-chromite | accessory chromite | dunite |
Table 2.
Electron microprobe analyses of laurite and erlichmanite from Niquelândia.
Table 2.
Electron microprobe analyses of laurite and erlichmanite from Niquelândia.
| Laurite | Os | Ir | Ru | Rh | Pt | Pd | Fe | Ni | Cu | S | As | total |
|---|
| 55-3 * | 23.80 | 7.68 | 31.50 | – | – | – | – | – | – | 32.26 | 1.89 | 97.13 |
| 211C-1a * | 15.04 | 3.15 | 43.77 | – | – | – | – | – | – | 36.25 | 1.50 | 99.71 |
| 609A-16* | 20.68 | 5.29 | 32.42 | 1.39 | – | 1.85 | 1.07 | 0.08 | 0.01 | 35.59 | 1.42 | 99.80 |
| 615A-2 * | 7.40 | 3.70 | 51.70 | – | 0.08 | – | – | 0.15 | – | 36.20 | – | 99.23 |
| 615F-6* | 13.18 | 6.03 | 40.53 | 0.83 | – | 2.03 | 1.48 | 0.19 | 0.12 | 34.69 | 1.68 | 100.76 |
| 373A-2 | 30.29 | 14.19 | 21.21 | 2.05 | – | – | – | 0.15 | – | 29.96 | 0.39 | 98.24 |
| 453A-1 | 31.38 | 6.80 | 28.20 | – | – | – | – | 0.17 | – | 31.26 | 1.17 | 98.98 |
| 454-5 | 24.00 | 5.50 | 32.90 | 0.29 | – | 1.30 | 1.10 | 0.16 | – | 33.70 | 0.74 | 99.69 |
| 454-9a | 31.10 | 6.40 | 20.10 | 1.80 | – | – | 5.31 | – | – | 33.50 | 1.40 | 99.61 |
| 455-1 | 35.64 | 3.32 | 26.19 | 0.12 | – | | 0.91 | 0.55 | – | 31.25 | – | 97.99 |
| 455A-10 | 35.90 | 5.80 | 22.90 | 0.26 | – | 0.40 | 2.70 | 0.15 | 0.07 | 31.30 | 0.09 | 99.57 |
| 455A-8 | 31.00 | 5.10 | 28.70 | – | – | 0.40 | 1.80 | – | – | 32.70 | – | 99.70 |
| 456-15 | 29.88 | 4.30 | 30.40 | 0.29 | – | 0.71 | 1.40 | 0.12 | 0.54 | 32.10 | – | 99.74 |
| 456-5 | 38.40 | 6.10 | 23.10 | – | – | 0.55 | 0.80 | – | – | 30.20 | 0.28 | 99.43 |
| 456A-10a | 31.49 | 3.75 | 29.36 | 0.18 | – | – | 1.63 | 0.13 | – | 33.13 | 0.22 | 99.89 |
| 456A-10b | 31.50 | 2.60 | 29.80 | – | – | – | 2.90 | – | – | 32.80 | – | 99.60 |
| 617-1a | 0.65 | 44.40 | 8.77 | 2.26 | 1.97 | 0.24 | 1.05 | 0.17 | 0.02 | 13.86 | 25.71 | 99.10 |
| 617A-3 | 28.35 | 4.30 | 30.20 | 0.71 | – | 1.45 | – | – | – | 31.99 | 1.50 | 98.50 |
| 617A-4-1 | 27.31 | 2.53 | 31.90 | 0.66 | – | 1.48 | – | – | – | 31.93 | 1.33 | 97.14 |
| 658-1 | 15.36 | 6.78 | 40.56 | 0.25 | – | 0.55 | 0.80 | 0.29 | – | 33.45 | – | 98.04 |
| 659-3 | 27.61 | 6.75 | 29.54 | 0.04 | – | – | 0.57 | 0.11 | – | 32.68 | 1.34 | 98.64 |
| 659-6 | 26.78 | 5.24 | 31.78 | 0.31 | – | 1.30 | 1.27 | 0.33 | 0.09 | 31.56 | 0.14 | 98.80 |
| 659-7 | 31.70 | 3.90 | 30.45 | – | – | 1.45 | – | 0.12 | 0.13 | 30.87 | – | 98.62 |
| 659-9 | 18.92 | 5.13 | 39.39 | – | – | 1.86 | 1.35 | 0.07 | – | 33.81 | 0.07 | 100.64 |
| 660-4a | 19.00 | 5.11 | 40.48 | – | – | 1.54 | 1.02 | 0.05 | 0.04 | 33.64 | – | 100.88 |
| 660-5b | 33.85 | 6.08 | 25.11 | 0.05 | – | 0.88 | 1.32 | 0.17 | 0.16 | 29.86 | – | 97.48 |
| 660As-5 | 17.41 | 7.52 | 40.71 | 0.73 | – | – | – | 0.12 | – | 33.13 | – | 99.62 |
| 660As-7 | 31.20 | 4.65 | 32.75 | 0.41 | – | – | – | – | – | 31.75 | – | 100.76 |
| 660Es-2b | 22.12 | 8.87 | 38.08 | – | – | – | 0.95 | 0.15 | – | 31.13 | 0.50 | 101.80 |
| 660-1a | 15.54 | 8.27 | 37.62 | 0.15 | – | – | 1.82 | 0.41 | – | 35.67 | – | 99.48 |
| 660-1b | 23.29 | 7.61 | 32.19 | 0.25 | – | 1.39 | 1.98 | 0.19 | – | 31.37 | – | 98.27 |
| 660-1c | 24.19 | 4.95 | 30.25 | – | – | 1.47 | 1.85 | 0.37 | 0.14 | 33.99 | 0.28 | 97.46 |
| 609B-1* | 57.02 | 13.96 | 0.92 | 0.12 | – | – | 1.02 | 0.07 | – | 23.54 | 1.77 | 98.42 |
| 373A-7a | 56.40 | 13.40 | 1.40 | – | 0.09 | – | – | – | – | 25.70 | 0.23 | 97.22 |
| 456-9 | 50.23 | 6.83 | 11.65 | – | – | 0.91 | 1.26 | 0.09 | – | 26.82 | 0.48 | 98.26 |
| 456A-4a | 47.43 | 3.33 | 16.75 | 0.15 | – | – | 0.39 | 0.11 | – | 29.77 | 0.08 | 98.01 |
| 660-10 | 30.49 | 21.13 | 14.67 | – | – | 0.68 | 1.26 | 0.11 | – | 25.74 | 4.85 | 98.93 |
| 660Bs-10 | 66.07 | 9.99 | 0.41 | – | – | – | 1.24 | – | – | 21.84 | – | 99.55 |
| 660Ds-6 | 38.67 | 5.09 | 26.12 | 0.06 | – | – | 0.89 | 0.04 | – | 29.10 | – | 99.97 |
| 660Es-1a | 33.07 | 4.98 | 30.47 | – | – | – | 1.25 | – | 0.06 | 29.90 | – | 99.73 |
Table 3.
Electron microprobe compositions of PGM sulfides, sulfarsenides, alloys and bismuth-tellurides from Niquelândia.
Table 3.
Electron microprobe compositions of PGM sulfides, sulfarsenides, alloys and bismuth-tellurides from Niquelândia.
| Os | Ir | Ru | Rh | Pt | Pd | Fe | Ni | Cu | S | As | Bi | Te | Sb | total |
|---|
| Malanite |
| 617A-5a-2 | 3.80 | 29.91 | – | 0.91 | 31.45 | 0.21 | 2.99 | – | 10.46 | 21.12 | – | – | – | – | 100.85 |
| 617A-5a-3 | – | 31.45 | – | 1.90 | 30.35 | 0.48 | 1.90 | 0.12 | 11.31 | 21.76 | 0.08 | – | – | – | 99.35 |
| Irarsite |
| 617A-5b-4 | – | 53.50 | 0.62 | 4.58 | 0.79 | 1.28 | 2.40 | – | 0.70 | 11.11 | 25.22 | – | – | – | 100.20 |
| 660Es-2a | 1.54 | 61.20 | 1.79 | 0.49 | – | – | 0.76 | – | – | 9.84 | 24.34 | – | – | – | 99.96 |
| Os-Ir-Ru alloys |
| 373A-7b | 76.61 | 21.55 | 0.33 | – | – | – | – | – | – | 0.19 | – | – | – | – | 98.68 |
| 455A-4 | 79.40 | 17.30 | 0.50 | – | – | – | 2.50 | – | – | – | – | – | – | – | 99.70 |
| 615E-5* | 90.70 | 3.39 | – | 0.08 | – | – | 2.18 | – | – | – | – | – | – | – | 96.35 |
| 617-1b | 55.29 | 22.70 | 17.22 | 0.57 | 3.74 | – | 0.82 | – | – | – | – | – | – | – | 100.34 |
| 660-9-3 | 49.43 | 43.67 | 0.30 | – | – | – | 5.17 | 0.75 | – | – | – | – | – | – | 99.32 |
| 661-1b | 27.72 | 64.00 | – | – | – | – | 6.46 | 1.46 | – | 0.58 | – | – | – | – | 100.22 |
| Pt-Fe Alloys |
| 55-1* | 0.13 | 0.17 | – | 0.30 | 73.40 | 0.57 | 23.20 | 0.48 | 0.06 | – | – | – | – | – | 98.31 |
| 453-2 | – | – | – | – | 73.34 | – | 25.32 | 1.07 | – | – | – | – | – | – | 99.73 |
| 660-4b | – | – | – | 0.32 | 76.25 | – | 16.31 | 6.22 | – | – | – | – | – | – | 99.10 |
| 661-1a | – | 2.46 | – | 0.25 | 63.23 | – | 27.33 | 6.53 | – | – | – | – | – | – | 99.80 |
| Pt- and Pd-bearing tellurides, bismuthides, and antimonides |
| 209I-10a* | – | – | – | – | 0.60 | 35.87 | 0.65 | 2.53 | – | – | – | 37.13 | 16.48 | 6.70 | 99.96 |
| 209I-10b-1* | – | – | – | 0.30 | 26.90 | 0.17 | – | 5.80 | – | – | – | 19.63 | 44.10 | 1.30 | 98.20 |
| 209I-10b-2* | – | 12.76 | – | – | 33.76 | 1.31 | 0.76 | 2.90 | – | – | – | 5.62 | 11.25 | 27.43 | 95.79 |
| 209I-5* | – | 13.95 | – | 0.43 | 28.10 | – | 1.27 | 2.09 | – | – | – | 1.60 | 7.00 | 45.45 | 99.89 |
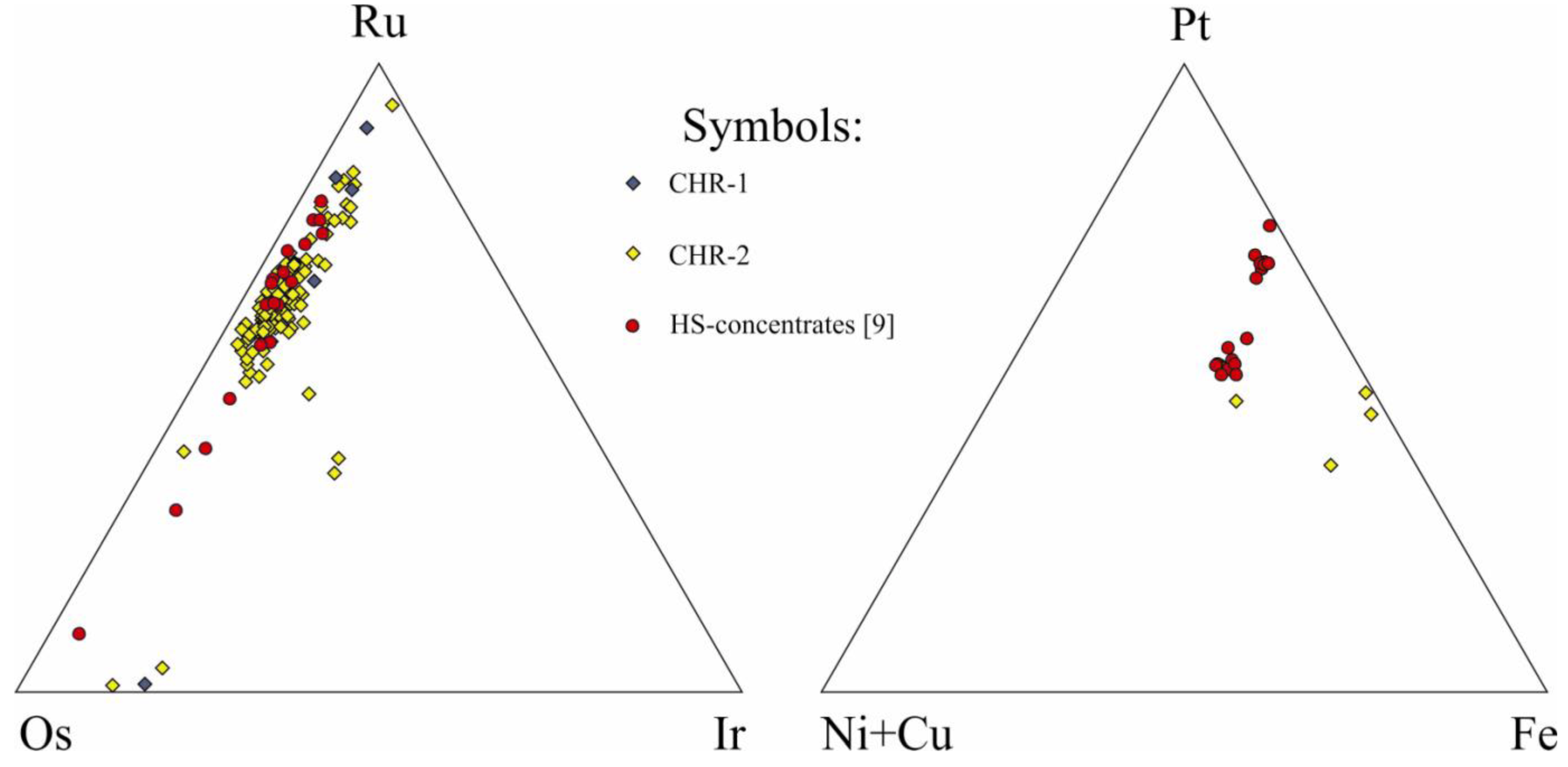
Figure 7.
Ternary plot of Ru-Os-Ir sulfide and Pt-Fe alloy PGM associated with the chromitites of Niquelândia.
Figure 7.
Ternary plot of Ru-Os-Ir sulfide and Pt-Fe alloy PGM associated with the chromitites of Niquelândia.
In the CHR-1 horizon, PGM have been found in both the chromitite and the adjacent dunite with different mineral assemblages in each case. The chromitite contains predominantly Ru-Os-Ir sulfides (mainly laurite) and alloys, mostly occurring included in fresh chromite. The PGM may occur as single phase or composite grains with Ni-sulfides (pentlandite, millerite) and silicate (clinopyroxene), or form minute spots within small blebs of pentlandite (
Figure 8). The dunite contains Pt-Fe alloy included in orthopyroxene and laurite associated altered olivine (55-1, 55-3,
Figure 9). Composite PGM of Pt-Pd-Bi-Te-Sb occur associated with partially-oxidized interstitial sulfide, or with the alteration rim of accessory chromite grains (
Figure 9, 209I-10a, 209I-10b).
Sulfides of the laurite-erlichmanite series are the most common PGM in the CHR-2 chromitites. A few grains associated with phlogopite and amphibole respectively (
Figure 10, 659-7 and 659-9) were found included in solid, low reflective chromite. Most of the other examples occur as anhedral to sub-euhedral crystals within the pitted high reflective chromite, sometimes associated with other PGM sulfides, alloys, or sulf-arsenides (
Figure 11). In some case (
Figure 12), sulfides of the laurite-erlichmanite series show a particular association with hematite, and rutile (456As-10a, 456As-10b), or are included in vermicular ilmenite at the boundary of large chromite grains (660-1). This type of laurite displays clear chemical zoning showing an irregular increase in Os towards the rim.
The study of concentrates obtained by hydroseparation (HS) has shown the widespread occurrence of various types of Pt-Fe alloys (with Pt
3Fe to Pt (Fe, Ni, Cu) stoichiometries) in chromitite samples from the CHR-2 horizon, characterized by a relatively coarse-grain size between 20 and 80 µm [
9]. Such large grains were not encountered
in situ, during the microscopic study of polished sections and the reason for the discrepancy is explained by the authors. Here we report new findings of Pt-Fe alloys after those described by [
7] and give details of their unusual occurrence. Irregular grains of Pt-Fe alloys occur in secondary assemblages consisting of Fe-hydroxides and ilmenite associated with kaolinite lamellae (
Figure 13). Electron microprobe composition is consistent with a tetraferroplatinum-type stoichiometry PtFe, with abundant Ni and Cu substitution, whereas the isoferroplatinum-type compositions Pt
3Fe encountered in the concentrates have not been observed
in situ.
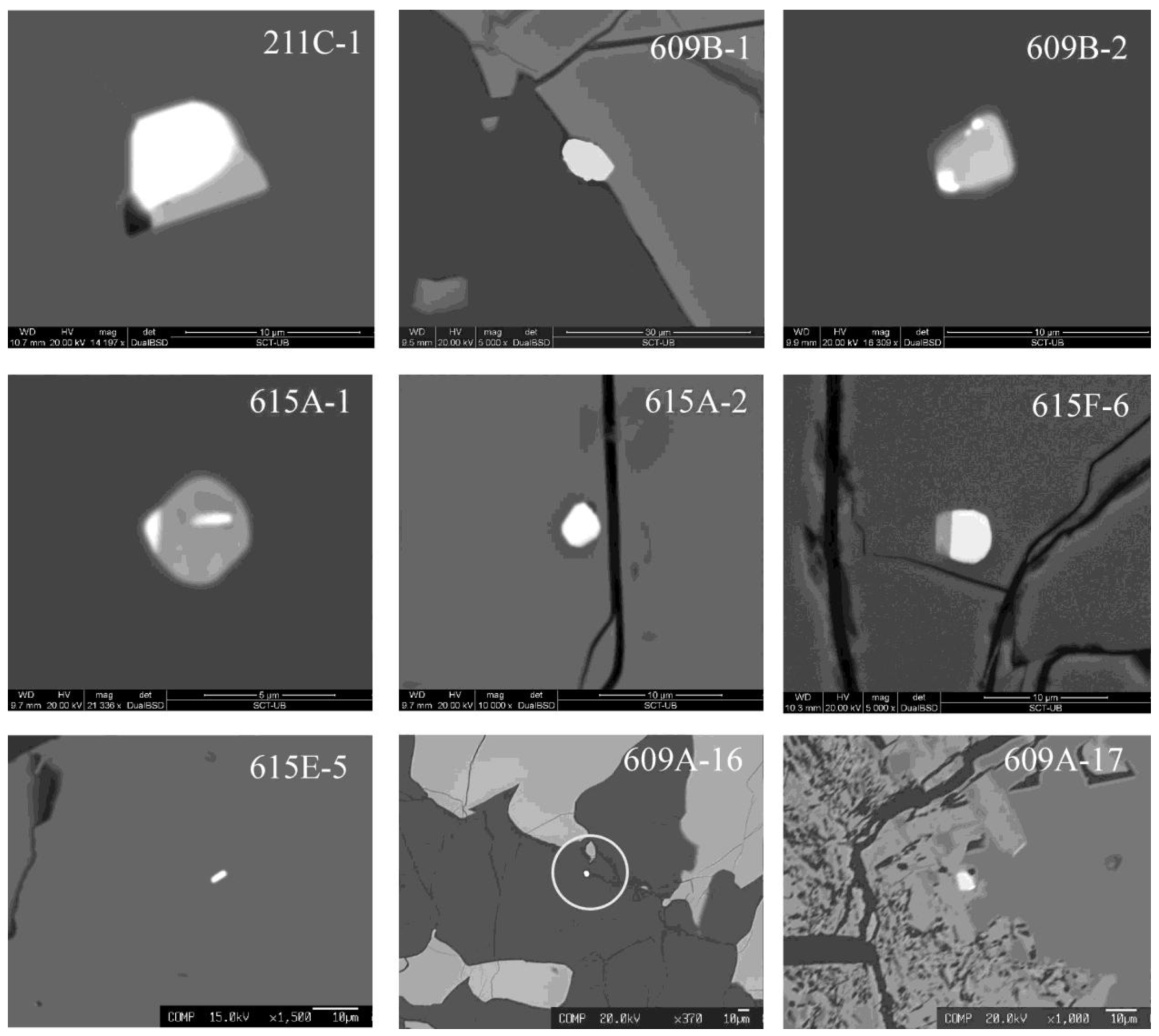
Figure 8.
Paragenetic assemblages of PGM associated with the CHR-1 chromitite (BSE images). 211C-1: Laurite + pentlandite + cpx composite inclusion in fresh chromite; 609B-1: Erlichmanite at the contact between chromite and serpentine (dark grey); 609B-2: Pentlandite + pyrrhotite + Ir-Os-(S) (white spots) composite inclusion in fresh chromite; 615A-1: Pentlandite + pyrrhotite + Ir-(S) (border white patch) + Os-(S) (horizontal white rod) composite inclusion in fresh chromite; 615A-2: Sub-euhedral laurite included in fresh chromite; 615F-6: Laurite + pentlandite composite inclusion in fresh chromite; 615E-5: Native Os included in fresh chromite; 609A-16: Euhedral laurite in the serpentine matrix of the CHR-1 chromitite; 609A-17: Pentlandite + Ir-sulfide in the alteration rim of a large chromite grain.
Figure 8.
Paragenetic assemblages of PGM associated with the CHR-1 chromitite (BSE images). 211C-1: Laurite + pentlandite + cpx composite inclusion in fresh chromite; 609B-1: Erlichmanite at the contact between chromite and serpentine (dark grey); 609B-2: Pentlandite + pyrrhotite + Ir-Os-(S) (white spots) composite inclusion in fresh chromite; 615A-1: Pentlandite + pyrrhotite + Ir-(S) (border white patch) + Os-(S) (horizontal white rod) composite inclusion in fresh chromite; 615A-2: Sub-euhedral laurite included in fresh chromite; 615F-6: Laurite + pentlandite composite inclusion in fresh chromite; 615E-5: Native Os included in fresh chromite; 609A-16: Euhedral laurite in the serpentine matrix of the CHR-1 chromitite; 609A-17: Pentlandite + Ir-sulfide in the alteration rim of a large chromite grain.
Figure 9.
Paragenetic assemblages of PGM in the dunite adjacent to CHR-1 chromitite (BSE images). 55-1: Pt-Fe alloy included in opx; 55-2: Laurite in serpentine veins after olivine, in close proximity to pentlandite + native Cu after chalcopyrite (large grain); 209I-10a: Partially euhedral sobolevskite-kotulskite-sudburyite type PGM associated with pentlandite veined with magnetite, interstitial to serpentinized olivine and accessory chromite in dunite; 209I-10b: Composite grain of moncheite type and stumpflite type PGM attached to the rim of porous ferrian-chromite coating accessory chromite in dunite.
Figure 9.
Paragenetic assemblages of PGM in the dunite adjacent to CHR-1 chromitite (BSE images). 55-1: Pt-Fe alloy included in opx; 55-2: Laurite in serpentine veins after olivine, in close proximity to pentlandite + native Cu after chalcopyrite (large grain); 209I-10a: Partially euhedral sobolevskite-kotulskite-sudburyite type PGM associated with pentlandite veined with magnetite, interstitial to serpentinized olivine and accessory chromite in dunite; 209I-10b: Composite grain of moncheite type and stumpflite type PGM attached to the rim of porous ferrian-chromite coating accessory chromite in dunite.
Figure 10.
Paragenetic assemblages of Ru-Os-Ir PGM in the CHR-2 chromitite (BSE images). 660As-7: Euhedral grain of laurite with included pentlandite in chromite 2; 660As-5: Porous crystal of laurite + ferrianchromite located along crack in chromite 2; 660-2: Sub-euhedral erlichmanite + kaolinite included in chromite 2; 660-4a: Euhedral laurite + serpentine included in chromite; 659-7: Composite inclusion of laurite + phlogopite in chromite 1; 659-9: Composite inclusion of sub-euhedral erlichmanite + Ni-sulfide + amphibole in chromite 1; 660-5b: Sub-euhedral laurite + Fe-oxide + serpentine included in chromite 2; 659-6: Composite inclusion of laurite + Ir-sulfide + millerite + serpentine in chromite 2; 659-3: Composite inclusion of laurite + hematite + serpentine in chromite 2.
Figure 10.
Paragenetic assemblages of Ru-Os-Ir PGM in the CHR-2 chromitite (BSE images). 660As-7: Euhedral grain of laurite with included pentlandite in chromite 2; 660As-5: Porous crystal of laurite + ferrianchromite located along crack in chromite 2; 660-2: Sub-euhedral erlichmanite + kaolinite included in chromite 2; 660-4a: Euhedral laurite + serpentine included in chromite; 659-7: Composite inclusion of laurite + phlogopite in chromite 1; 659-9: Composite inclusion of sub-euhedral erlichmanite + Ni-sulfide + amphibole in chromite 1; 660-5b: Sub-euhedral laurite + Fe-oxide + serpentine included in chromite 2; 659-6: Composite inclusion of laurite + Ir-sulfide + millerite + serpentine in chromite 2; 659-3: Composite inclusion of laurite + hematite + serpentine in chromite 2.
Figure 11.
Paragenetic assemblages of Ru-Os-Ir PGM in the CHR-2 chromitite (BSE images). 373A-7: Euhedral composite grain of erlichmanite + iridosmine + pentlandite (dark crystal in the sulfide), included in chromite 2. 660Es-2: Partially altered composite grain of laurite + irarsite in fractured chromite 2. 617-1: Composite grain consisting of irarsite (low) + Os-alloy (middle) + laurite (upper) included in chromite 2.
Figure 11.
Paragenetic assemblages of Ru-Os-Ir PGM in the CHR-2 chromitite (BSE images). 373A-7: Euhedral composite grain of erlichmanite + iridosmine + pentlandite (dark crystal in the sulfide), included in chromite 2. 660Es-2: Partially altered composite grain of laurite + irarsite in fractured chromite 2. 617-1: Composite grain consisting of irarsite (low) + Os-alloy (middle) + laurite (upper) included in chromite 2.
Figure 12.
Paragenetic assemblages of Ru-Os-Ir PGM in the CHR-2 chromitite (BSE images). 660-1: Three laurite grains included in ilmenite; the laurite is strongly zoned showing Os-enriched rim. 456A-10a: Euhedral laurite associated with hematite included in chromite 1. 456A-10b: Sub-euhedral laurite associated with rutile in chromite 1.
Figure 12.
Paragenetic assemblages of Ru-Os-Ir PGM in the CHR-2 chromitite (BSE images). 660-1: Three laurite grains included in ilmenite; the laurite is strongly zoned showing Os-enriched rim. 456A-10a: Euhedral laurite associated with hematite included in chromite 1. 456A-10b: Sub-euhedral laurite associated with rutile in chromite 1.
Figure 13.
Paragenetic assemblages of Pt-Fe and Os-Ir alloys in the CHR-2 chromitite (BSE images). 373A-1: Large kaolinite lamella veined with Fe-hydroxides; a small grain of Pt-Fe alloy is located at the contact between kaolinite and a composite aggregate of Fe-hydroxide; an ilmenite crystal located along the fracture is visible in the NE corner of the large-field picture. 660-4: Pt-Fe alloy located in ilmenite at the contact with enclosing chromite 1; ilmenite occurs in a fracture with kaolinite and Fe-hydroxide; 661-1: Composite grain of Pt-Fe (light grey) and Os-Ir (white) alloys in Fe-hydroxide at the rim of a large kaolinite lamella; kaolinite is spotted and coated with botryoidal Fe-hydroxide; 660-9: Large grain of Os-Ir alloy at the rim of a large aggregate of Fe-hydroxide, located at the junction of fractures in chromite 1.
Figure 13.
Paragenetic assemblages of Pt-Fe and Os-Ir alloys in the CHR-2 chromitite (BSE images). 373A-1: Large kaolinite lamella veined with Fe-hydroxides; a small grain of Pt-Fe alloy is located at the contact between kaolinite and a composite aggregate of Fe-hydroxide; an ilmenite crystal located along the fracture is visible in the NE corner of the large-field picture. 660-4: Pt-Fe alloy located in ilmenite at the contact with enclosing chromite 1; ilmenite occurs in a fracture with kaolinite and Fe-hydroxide; 661-1: Composite grain of Pt-Fe (light grey) and Os-Ir (white) alloys in Fe-hydroxide at the rim of a large kaolinite lamella; kaolinite is spotted and coated with botryoidal Fe-hydroxide; 660-9: Large grain of Os-Ir alloy at the rim of a large aggregate of Fe-hydroxide, located at the junction of fractures in chromite 1.
Similar to Pt-Fe alloys, other alloys of the Os-Ir-Ru type occur associated with secondary Fe-hydroxides, goethite and limonite (
Figure 13, 661-1, 660-9). Finally, a large grain of Ru-oxide was encountered included in the pitted chromite of CHR-2. The grain consists of a Ru-O compound with minor Mn and Ni, just slightly more reflective than the surrounding chromite (see the optical and BSE images in
Figure 14). The grain contains two bright lamellae of Ru-Os sulfide, possibily corresponding to laurite.
Figure 14.
Paragenetic assemblages of PGE oxide in the CHR-2 chromitite (optical and BSE images). 660As-12: Large grain of Ru-Ir oxide with impurities of Mn, Ni and As; the internal white patches are relicts of Os-rich laurite.
Figure 14.
Paragenetic assemblages of PGE oxide in the CHR-2 chromitite (optical and BSE images). 660As-12: Large grain of Ru-Ir oxide with impurities of Mn, Ni and As; the internal white patches are relicts of Os-rich laurite.
6. Constraints on the Origin of PGM in the Niquelândia Chromitites
As remarked by [
7], chromitites of the Niquelândia layered intrusions contain two genetically different populations of PGM: (1) Primary PGM formed during fractionation of chromitite and dunite, at high-temperature; and (2) secondary PGM derived from
in-situ alteration of primary PGM, or formed by mobilization of PGE during serpentinization and lateritic weathering.
Primary PGM in the chromitite-dunite assemblage of CHR-1 show a well defined order of crystallization. The early PGM are Ru-Os-Ir sulfides and alloys. They crystallized at high temperature and were mechanically trapped within growing chromite with small amounts of silicate and sulfide.
Despite the presence of sulfides in the assemblage, entrapment of these PGM is believed to have occurred before sulfur saturation of the melt [
12]. During formation of the chromitite, Pt and Pd (with some IPGE) remained in the silicate melt. They precipitated later, collecting within the immiscible sulfide liquid segregated from the silicate magma after the main chromitite event. The sporadic occurrence of Pt-Fe alloys encapsulated in the orthopyroxene and laurite in serpentinized olivine suggests that some PGE were removed from the silicate melt at some stage of the dunite fractional crystallization when the melt was sulfur-undersaturated [
7].
PGM inclusions were protected from serpentinization by the host chromite. Chromite is more resistant to alteration than silicates and sulfides. In contrast, PGM located in the rims of chromite grains or in the silicate gangue were exposed to alteration, and could have undergone secondary remobilization.
The origin of the PGM associated with the interstitial sulfides and ferrian-chromite in the CHR-1 dunite (
Figure 9) is uncertain. They may represent relicts of primary Pt-Pd-Bi-Te-Sb minerals partially eroded by chemically aggressive solutions, during serpentinization, or even PGM precipitated from the solution itself, as reported for similar Pt-Pd phases in the altered silicate matrix of the Campo Formoso chromitites [
13]. In one case, the euhedral shape of the partially eroded Pt-Bi-Te grain associated with the sulfide (
Figure 9, 209I-10b) would suggest that it is a relict of a primary phase surviving alteration.
In general, it is difficult to identify the primary or secondary origin of the Os-Ir-Ru PGM in the CHR-2 chromitite. However, based on their texture and morphology, most of the Ru-Os-Ir sulfides, sulfarsenides and alloys included in the CHR-2 chromitite represent primary phases that survived alteration of the enclosing chromite, due to their higher chemical stability compared with the chromite. Notably, if the Ir-Os sulfarsenides represent primary phases we have to admit an increase of the As/S activity ratio in the CHR-2 chromite-forming system compared with CHR-1 that could have taken place in response to As-S fractionation due to differentiation. This question is unresolved. Some Ru-Os-Ir sulfides, however, display evidence of chemical corrosion (660As-5,
Figure 10), others are intimately associated with secondary silicates: serpentine (659-3, 659-6, 660-5b, 660-4a,
Figure 10) or kaolinite (660-2,
Figure 10) and therefore, their primary or secondary origin is unclear. The laurite inclusions in vermicular ilmenite (660-1,
Figure 12) would appear to have formed during precipitation of ilmenite, a process that could have occurred at relatively low-temperature as a result of Ti loss during chromite alteration. The close association of the tetraferroplatinum type alloys with kaolinite plus Fe-hydroxides is a clear indication that the Pt-PGM were deposited during low temperature alteration. The primary isoferroplatinum type alloys recovered from HS concentrates, might represent the primary source for Pt remobilization. The Pt is re-deposited at the interface between kaolinite and Fe hydroxides, possibly representing a local redox micro-boundary necessary to trigger Pt precipitation (Bowles, 1986). The evidence that PGE were mobilized during the lateritization process is provided by the finding of PGE oxides associated with altered chromitite. The internal texture of the PGM in
Figure 14 clearly indicates that the Ru-oxide is derived from alteration of a laurite precursor whose relict is still preserved. As shown in other occurrences elsewhere [
14], alteration of laurite involves removing of S and Os, which are replaced by O and small amounts of base metals (Fe, Mn, Ni).