Surprisingly, the cited models [
8,
9,
10,
11,
12,
13,
14] illustrating formation of mollusk and coral skeletons are closely similar, although classical views admit a high level difference with respect to control exerted on their respective crystallization processes. Coral skeletons are considered as “biologically induced” structures (
i.e., almost uncontrolled), in contrast to mollusk shell built by “matrix mediated” biocrystals, whose full control by specifically secreted organic compounds is accepted [
23,
24]. Comparison of micrometer-scaled structural and chemical data allows a re-evaluation of this widely shared distinction.
2.1. Calcite Prisms in Bivalve Mollusks
As one of the earliest skeletal units ever observed, calcite prisms which build the outer layer of the
Pinna shell (Bivalve mollusk) deserve particular interest. Easily visible (with the naked eye or a simple lens) owing to their size and very simple three-dimensional arrangement, their remarkably simple morphological pattern (
Figure 1a–e) was already illustrated by Bowerbank on his
Figure 8 [
1,
14]. Crystallographic examination reinforces the concept of the prism as a simple crystal. Under cross-nicol illumination, transverse sections show color homogeneity for each prism (
Figure 1f–g), a change in color from prism to prism assessing the slight deviations of their
c axis from strict perpendicularity to shell surface. Such a regular spatial disposition makes this type of skeletal unit ideal material for a theoretical approach of biocrystallization (see [
25] for review).
Taking advantage of a possible lateral view of the prisms at the external edge of a growth lamella (
Figure 1b, arrow), the stratified mode of growth of the prisms is already apparent (
Figure 1i). Such a stratified structure of the calcite prisms was illustrated first by Grégoire [
26], but owing to the preparative process used, synchronism between mineralization layers of neighbor prisms was not visible. This important growth pattern is obvious, however, on wider sections perpendicular to the shell surface after etching (
Figure 2a–c). Although neighbor prisms are separated by strong organic envelopes, synchronism in formation of the superposed elementary mineralized layers of prisms is assessed by correlation between growth lines (
Figure 2a, arrows) and by the perfect similitude of their variations in thickness (
Figure 2a–c).
Regarding the surprising continuity of mineral layers through the strong polygonal organic envelopes of the prisms, the question arises of their individual but obviously synchronic development. Actually, growth of these envelopes is also a stepping process, as shown by Figures 1j and 2c–h. A one week decay of the honeycomb-like organic mesh in water resulted in partial hydrolysis and separation of the upper growth step of the envelopes (
Figure 1j). Sometimes, the stepping growth process of the prism envelopes is clearly visible on the inner side of envelopes (
Figure 2d) and even perfectly illustrated by the morphology of envelopes themselves in longitudinal sections (
Figure 2e–h).
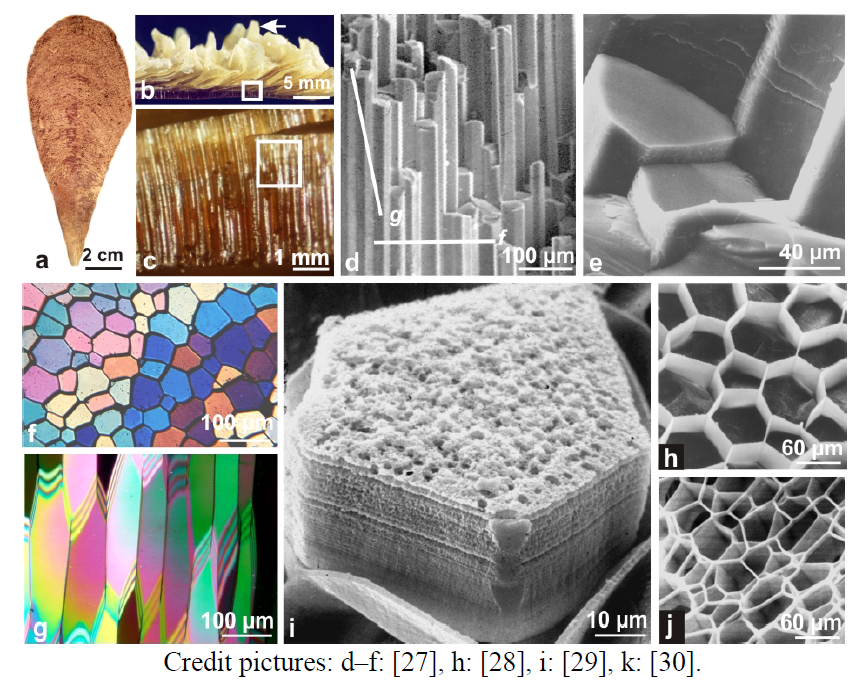
Figure 1.
Layered growth in the prisms of the mollusk pelecypod Pinna: the historical reference for biocrystals.a–e: The building units of the outer layer of the Pinna shell (a,b) are closely assembled linear polygonal prisms (c,d) with massive aspect of fractures (e); (f,g) Thin sections observed under the microscope between cross-nicols. Owing to the high refractive index of calcite, such primary colors are obtained far below the standard thickness: here about 5–6 micrometers. Note the homogeneity of color for each prism on the transverse section (f). Difference in color from prism to prism, results from variation of orientation of the crystallographic c axis (the elongation axis of the prism). When prisms are sectioned in length, color is more variable because obliquity of the sections with respect to elongation axis creates variations in the thickness with the possibility of superposed contributions of two prisms; (h) Strong acidic etching: the polygonal network of prism envelopes has been made free; (i) Layered growth visible without any preparative process of the lateral side of the mineralized unit. Note also the granular aspect of the growth surface; (j) After a one week decay in water the polygonal network corresponding to a single growth layer has been freed (and slightly displaced) from underlying envelopes.
Figure 1.
Layered growth in the prisms of the mollusk pelecypod Pinna: the historical reference for biocrystals.a–e: The building units of the outer layer of the Pinna shell (a,b) are closely assembled linear polygonal prisms (c,d) with massive aspect of fractures (e); (f,g) Thin sections observed under the microscope between cross-nicols. Owing to the high refractive index of calcite, such primary colors are obtained far below the standard thickness: here about 5–6 micrometers. Note the homogeneity of color for each prism on the transverse section (f). Difference in color from prism to prism, results from variation of orientation of the crystallographic c axis (the elongation axis of the prism). When prisms are sectioned in length, color is more variable because obliquity of the sections with respect to elongation axis creates variations in the thickness with the possibility of superposed contributions of two prisms; (h) Strong acidic etching: the polygonal network of prism envelopes has been made free; (i) Layered growth visible without any preparative process of the lateral side of the mineralized unit. Note also the granular aspect of the growth surface; (j) After a one week decay in water the polygonal network corresponding to a single growth layer has been freed (and slightly displaced) from underlying envelopes.
![Minerals 02 00011 g001]()
Credit pictures: d–f: [
27], h: [
28], i: [
29], k: [
30].
Figure 2.
Simultaneous growth of organic envelopes and mineralized components of the prisms. (a,d ) enzymatic etching (a,b) and acido-fixative etching (c) provide complementary evidence of a perfect synchronism in deposition of growth layers forming the prismatic outer part of the Pinna shells. Note that a stepping growth mode of envelopes is directly visible on their internal side. Figure 1j has shown that each growth layer can be separated from the others through soft hydrolysis process. (e–h) Calcitic prisms produced by Pinctada mantle also exhibit perfect synchronism in deposition (e,f). In addition, the stepping growth mode of envelopes is sometimes quite obviously illustrated, as here in the prismatic layer of a pearl bed (f,g). Using back scattered electrons, the high contrast between mineralized and purely organic parts emphasizes the morphological details resulting from the stepping growth mode (h); (i,j) Microprobe distribution maps of magnesium and sulfur on a polished section in a Pinna shell. Section almost perpendicular to shell surface: prisms are cut in their length but slightly obliquely; (k–l) Distribution of sulfated polysaccharides by synchrotron based X-ray absorption of sulfur (k) shows that prism envelopes (l: arrow) do not contain polysaccharides, although sulfur is abundant as shown by microprobe mapping (2j): envelopes are mostly protein made (see also Figure 3h). In addition the layered distribution provides additional evidence of the organic-mineral interplay within each growth cycle.
Figure 2.
Simultaneous growth of organic envelopes and mineralized components of the prisms. (a,d ) enzymatic etching (a,b) and acido-fixative etching (c) provide complementary evidence of a perfect synchronism in deposition of growth layers forming the prismatic outer part of the Pinna shells. Note that a stepping growth mode of envelopes is directly visible on their internal side. Figure 1j has shown that each growth layer can be separated from the others through soft hydrolysis process. (e–h) Calcitic prisms produced by Pinctada mantle also exhibit perfect synchronism in deposition (e,f). In addition, the stepping growth mode of envelopes is sometimes quite obviously illustrated, as here in the prismatic layer of a pearl bed (f,g). Using back scattered electrons, the high contrast between mineralized and purely organic parts emphasizes the morphological details resulting from the stepping growth mode (h); (i,j) Microprobe distribution maps of magnesium and sulfur on a polished section in a Pinna shell. Section almost perpendicular to shell surface: prisms are cut in their length but slightly obliquely; (k–l) Distribution of sulfated polysaccharides by synchrotron based X-ray absorption of sulfur (k) shows that prism envelopes (l: arrow) do not contain polysaccharides, although sulfur is abundant as shown by microprobe mapping (2j): envelopes are mostly protein made (see also Figure 3h). In addition the layered distribution provides additional evidence of the organic-mineral interplay within each growth cycle.
![Minerals 02 00011 g002]()
Credit pictures: a–c: [
28]; e–g: [
30]; k–l: [
31].
Clearly, formation of the organic polygonal network is a part of the formation of each growth layer. With respect to timing of the growth layer formation, it is a reasonable assumption that envelope formation may occur at the beginning of the cycle, before crystallization of the mineral phase (the reverse formation order, the insertion of the polygonal envelopes within an already completed mineral phase seems difficult, from an obvious mechanical reason). Consequently, prism shapes depend on the regularity in superposition of the polygonal networks through the repeated micrometer-thick mineralization cycles. It must be noted that such a simple observation emphasizes the key-role of envelopes in the shaping process of the prisms, providing a counter argument to the commonly claimed “crystal growth competition” mechanisms as the origin of skeletal microstructures [
32,
33]. Little is known about biochemical complexity of prism envelopes (see [
34] for review). Built mostly (if not exclusively) by proteins (see below
Figure 3h), any change in biochemical composition of the set of molecules involved in the self-assembly process may result in morphological variation in the shape of the polygonal units. In some case this change in prismatic spatial organization of the polygons is clearly visible. Thus, modification of this self-assembly process due to compositional changes in the involved protein cluster will result in shape variations of the biomineral (
Figure 5.9) [
30].
Variations in chemical compositions of the superposed growth layers also illustrate the layered mode of growth for
the prisms [
31]. Electron microprobe mapping of magnesium (
Figure 2i) or sulfur (
Figure 2j) on wide surfaces of shell sections clearly shows that variations in the chemical composition of the prisms remain strictly linked to growth layering all along shell formation. Additionally, these variations are distinct from one element to another one. Obviously (
Figure 2i) changes of the magnesium content in the superposed layers are independent from variations affecting sulfur. This strict and permanent correlation between structural and chemical patterns implies a cyclical biomineralization mechanism whose secretion activity is synchronized at the animal level, or more precisely the whole mineralizing surface of the mantle producing the prismatic layer of the shell.
Attention must be drawn to the possibility (and importance) of identifying the chemical form (coordination) under which a given element is present in the growth layers. The example of sulfur is demonstrative: the distribution of sulfur in
Figure 2j (microprobe mapping) shows a high content of sulfur (as a chemical element) in the envelopes of the prisms. Studying sulfur distribution using X-ray synchrotron allows specifying a given oxidation state by selecting energy of the XANES absorption peak (e.g., 2.4825 eV as in sulfated polysaccharides).
Data (
Figure 2k–l) show that in the mineralizing organic phase of the prisms the sulfur is in the same oxidation state as in polysaccharides. No such polysaccharide-sulfur is visible in the section of the envelopes, which is a first indication that they are mostly built by proteins.
With respect to the model of crystallization within a liquid-filled crystallization chamber, the layered mode of growth of the prisms is remarkable from two respects:
- thickness of each growth layer is identical across the whole mineralizing surface of the growing prisms. The thickness of Ca-carbonate deposition does not depend as much on crystal orientation as it does on the faces of freely growing crystals in saturated solutions. In biogenic crystals this thickness is related to the secretion activity of the mineralizing epithelium.
- chemical composition of a given growth layer is strictly limited to a single layer, clearly distinct from previous and following ones. It must be taken into account that periodicity of the secretion process is sometimes very high. For instance growth layers in the calcite prisms of the gastropod
Concholepas are deposited in up to 40 cycles a day, when nutriment availability is optimal [
35]. Formation of micro-thick layers with such specific and rapidly varying chemical compositions is hardly compatible with large liquid volumes as pictured in current crystallization chamber models.
2.2. Evidence of a Coordinated Stepping Growth Mode in Coral Fibers
Coral skeletons (Scleractinia corals but not corals belonging to Alcyonaria or Octocorals) are mostly built by crystal-like fibers (always aragonite in modern corals) generally arranged in variously diverging fan-systems (
Figure 4a–d). A long time before Lowenstam’s classical statement of a “biologically induced crystallization” [
23], such pictures had led Bryan and Hill [
36] to suggest that a “spherulitic crystallization” was the main mechanism responsible for development of coral fibers viewed as “single crystals of orthorhombic aragonite”.
Etching of polished surface of a fibrous fan-system of coral skeleton (
Figure 4e–f) immediately disproves the Bryan and Hill concept of fibers freely growing to form spherulites as it occurs in purely chemically saturated solutions. Contrastingly, the growth pattern of fibers exhibits close similarity to the growth mode of the
Pinna calcite prisms as seen above: far from individual and independent development of fibers,
Figure 3f clearly shows that incremental growth is strictly coordinated between neighboring fibers. Additionally, thickness of the growth layers is identical on all fibers of the fan system, irrespective of their individual crystallographic orientation. Undoubtedly, the biologically controlled deposition mechanism imposes its specific rules to the growth of the fan system as a whole. Looking at etched and polished sections of entire coral septa (
Figure 4g–n) shows that synchronism of the stepping growth is even ensured on much wider mineralizing surfaces.
At the whole septum level, we can find structural evidences of a fully controlled mineralization of the skeleton in corals. When species-specific morphologies of the septa have to be obtained, the basal ectoderm of the polyp is able to modulate the thickness of the growth layers (
Figure 4m–n). Microstructural evidence of this global control of the polyp on the corallite structure is a non-surprising result for coral taxonomists who were long familiar with close relationship between septum morphology, fibrous spatial organization and coral taxonomy [
6,
7,
37].
Although fully illustrated in two examples only, this result establishes a full similitude in the growth mode between two skeletal units that were (and still frequently are) considered as representatives of two distinct types of biomineralization: the “biologically induced” coral fiber and the “matrix mediated” calcite prism of the Pinna shell.
Conclusion 1: From a structural view-point (micrometer scale), both calcite prisms of Pinnaand aragonite fibers of Scleractinia corals exhibit evidence of a coordinated stepping growth mode. These skeletal units are built by superposition of mineralized layers basically continuous between neighboring units. Variations in thickness of these growth layers, strictly similar on the whole mineralizing area, provide demonstrative argument for a well synchronized metabolism of the mineralizing cell layer.
Extensive investigation [
30] suggests that a layered growth pattern is a very common mineralization method. Control of Ca-carbonate crystallization on wide areas but on thicknesses limited to the micrometer range seems the first level of a “common strategy” adopted by the various phyla creating a calcareous skeleton. Initially based on simple geometric evidences, this conclusion finds substantial support when considering the distribution of biochemical compounds permanently associated to the Ca-carbonate skeletal units (see figure 5 below). With respect to the use of calcareous skeletons as environmental archives, it is worth noting that each of these growth layers is the actual environment recording unit [
38], an interesting conclusion owing to the continuous improvement of spatial resolution of analytical instruments.
Figure 3.
Figure 3. Layered growth of the
Pinctada shell at the micrometer scale: correspondence between mineralogical and biochemical layers. (a–d) View of the internal surface. The aragonite (A) and calcite (C) deposition areas (a: internal view; b; shell section) are separated by a thin organic sheet (c–d) which progressively covers the upper surface of the prisms (d: arrows). Consequently, prism growth is immediately interrupted (in contrast to scheme e, for instance); (e) Sections perpendicular to shell surface. In this model of common liquid chamber (redrawn from [
12]) no explanation is given on how the mantle can control the simultaneous deposition of calcite and aragonite at the
d point; (f–i) The calcite-aragonite transition viewed on section perpendicular to shell surface. SEM secondary electron (f) shows the early deposition of aragonite (arrows) whereas SEM back scattered electrons imaging of this sector (g) reveals the general layering by emphasizing the contrast between organic and mineralized materials; h–i: X-ray absorption maps of protein sulfur (h) and polysaccharide sulfur (i) by synchrotron XANES mapping on the same sector. Note the strict correlation between distribution of mineral phases (f–g) and inversion of protein and polysaccharide proportions between the two mineral components. Prism envelopes and covering organic layer are practically pure protein (deep red on 4h); (j) Laser confocal view of the calcite aragonite transition in a
Pinctada shell. The daily growth doublets (
dgd) are well visible through fluorescence at 633 nm helium-neon laser illumination (Doc. A. Ball, NHM London, UK). Note their perfect parallelism even at the transition step, where last calcite is deposited on the right (C) of the growth layer whereas first aragonite components are produced on left side (A); (k,l) Stepping development of nacre. After closure of the calcite compartment (C) by the organic sheet, non-granular and fibrous aragonite (A) is produced first. It becomes true nacre only when parallel lamination occurs (arrows); k: occurrence of nacreous layering: nitrogen mapping using nanoSIMS (doc. A. Meibom, MNHN, Paris, France); l: equivalent area viewed by BSE imaging: progressive occurrence of organic layering is also well visible; (m–p) Stepping development of nacreous tablets. Deposition and growth of nacreous tablets is simultaneously controlled on several layers, following a long recognized staircase scheme (m–o). Deposited at the forefront of each layer, the nacreous tablet centers (n:
ntc), are progressively surrounded by concentric layers (p) (doc.: H. Mutvei).
Figure 3.
Figure 3. Layered growth of the
Pinctada shell at the micrometer scale: correspondence between mineralogical and biochemical layers. (a–d) View of the internal surface. The aragonite (A) and calcite (C) deposition areas (a: internal view; b; shell section) are separated by a thin organic sheet (c–d) which progressively covers the upper surface of the prisms (d: arrows). Consequently, prism growth is immediately interrupted (in contrast to scheme e, for instance); (e) Sections perpendicular to shell surface. In this model of common liquid chamber (redrawn from [
12]) no explanation is given on how the mantle can control the simultaneous deposition of calcite and aragonite at the
d point; (f–i) The calcite-aragonite transition viewed on section perpendicular to shell surface. SEM secondary electron (f) shows the early deposition of aragonite (arrows) whereas SEM back scattered electrons imaging of this sector (g) reveals the general layering by emphasizing the contrast between organic and mineralized materials; h–i: X-ray absorption maps of protein sulfur (h) and polysaccharide sulfur (i) by synchrotron XANES mapping on the same sector. Note the strict correlation between distribution of mineral phases (f–g) and inversion of protein and polysaccharide proportions between the two mineral components. Prism envelopes and covering organic layer are practically pure protein (deep red on 4h); (j) Laser confocal view of the calcite aragonite transition in a
Pinctada shell. The daily growth doublets (
dgd) are well visible through fluorescence at 633 nm helium-neon laser illumination (Doc. A. Ball, NHM London, UK). Note their perfect parallelism even at the transition step, where last calcite is deposited on the right (C) of the growth layer whereas first aragonite components are produced on left side (A); (k,l) Stepping development of nacre. After closure of the calcite compartment (C) by the organic sheet, non-granular and fibrous aragonite (A) is produced first. It becomes true nacre only when parallel lamination occurs (arrows); k: occurrence of nacreous layering: nitrogen mapping using nanoSIMS (doc. A. Meibom, MNHN, Paris, France); l: equivalent area viewed by BSE imaging: progressive occurrence of organic layering is also well visible; (m–p) Stepping development of nacreous tablets. Deposition and growth of nacreous tablets is simultaneously controlled on several layers, following a long recognized staircase scheme (m–o). Deposited at the forefront of each layer, the nacreous tablet centers (n:
ntc), are progressively surrounded by concentric layers (p) (doc.: H. Mutvei).
![Minerals 02 00011 g003]()
Credit pictures. a–c: [
39]; e: [
12]; f–i: [
40]; j: [
30]; k: [
40]; o: [
41]; p: [
42].
Figure 4.
Figure 4. Structural evidence for an overall control on the growth of coral fibers. (a–d) From skeleton morphology to fiber fan-systems in skeleton of a coral Porites; (e,f) Ultra-thin section of fan systems (cross-nicol polarized light) and etching of the same surface: continuity of growth layers emphasizes the common stepping growth mode for neighbor fibers. Note the regular thickness in deposition of a given growth layer whatever the crystallographic orientation of the substrate; (g–i) A polished and etched surface in the skeleton of a Pavona coral shows that the stepping growth is fully coordinated at the septum level: perfect continuity of the growth lines; (j–l) Similar growth-mode in the septum of a Favia corallite; (m,n ) The specific morphology of septa in a given coral species is obtained by localized modulation of the thickness of the growth layers, as illustrated by the etched-surface of this Favia stelligera septum.
Figure 4.
Figure 4. Structural evidence for an overall control on the growth of coral fibers. (a–d) From skeleton morphology to fiber fan-systems in skeleton of a coral Porites; (e,f) Ultra-thin section of fan systems (cross-nicol polarized light) and etching of the same surface: continuity of growth layers emphasizes the common stepping growth mode for neighbor fibers. Note the regular thickness in deposition of a given growth layer whatever the crystallographic orientation of the substrate; (g–i) A polished and etched surface in the skeleton of a Pavona coral shows that the stepping growth is fully coordinated at the septum level: perfect continuity of the growth lines; (j–l) Similar growth-mode in the septum of a Favia corallite; (m,n ) The specific morphology of septa in a given coral species is obtained by localized modulation of the thickness of the growth layers, as illustrated by the etched-surface of this Favia stelligera septum.
Credit pictures: e–f: [
38]; i–n: [
30].
2.3. Concomitant Depositions of Distinct Ca-Carbonate Polymorphs with Specific Crystallization Patterns and Compositions: A Major Obstacle to the “Liquid Crystallization Chamber” Model
Independently of their taxonomic position (i.e., place in the evolutionary tree of life) most of the organisms producing calcareous skeletons built a microstructural sequence made by superposition of two (sometimes three) distinct types of Ca-carbonate built by specific microstructural units. When only calcite or aragonite is used, this sequence comprises two morphologically distinct types of skeletal units as exemplified by many Bivalve mollusks such as commercial oysters (Ostrea, Crassostrea). Scleractinian corals illustrate this microstructural diversification of aragonite skeletons as well as some Pelecypod families such as Trigoniidae or Unionidae. Modern gastropod families exhibit aragonite shells with highly differentiated microstructure, whereas ancient gastropod families such as Haliotidae and Patellidae use both polymorphs in various proportions.
Considering models of crystallization in a “common chamber”, a number of contradictory pieces of evidence are produced by a careful examination of these sequential skeletons. The correlation between structural and biochemical data is specifically demonstrative when calcite and aragonite are simultaneously involved as in the shells of Pelecypods (Bivalve mollusks) belonging to the Pteriidae family, among which Pinctada, the “pearl oyster”, is the most studied. With an outer layer made of calcite prisms and a thick inner layer built by aragonite in the form of nacreous tablets, this large shell appears as a perfect illustration of the classical model of Bivalve shell, and (by extension) a model for all mollusk shells.
A close examination of the contact area between calcite and aragonite on the internal side of a valve (
Figure 3a–d) shows that the very first aragonite components, on the form of granular round spots (
Figure 3c–d) are not directly deposited onto the surface of the calcite prisms, but on a thin organic sheet progressively covering the prism growth surfaces (
Figure 3d: arrows). Redrawn from the Volkmer’s model [
12],
Figure 3e drives attention to the question of how a simultaneous and precisely localized precipitation of calcite (C) and aragonite (A) can be obtained by a mechanism of saturation within a common liquid chamber. Attention must be paid to the absence of permanent anatomical separation between the two mineralizing domains in the Pteriomorphids (the family to which the genus
Pinctada belongs), a point that is correctly drawn in the Volkmer’s scheme.
The biochemical control on deposition of the mineral phase of the two main shell components (prismatic calcite and nacreous aragonite) is illustrated by Figures 4f–l. In addition to previous structural evidence, these various physical characterizations applied to the transitional area between calcite and aragonite domains show that specific organic components are associated to each of them.
With respect to the overall shell growth process, the layered pattern obtained by laser confocal fluorescence (
Figure 3j) is of particular importance. This picture shows the perfect parallelism of the superposed mineralizing layers and the continuity between calcite and aragonite mineralizing areas in a given growth layer. Thickness increasing and simultaneous lateral expansion of the shell result from a single stepping process common for both the calcite and aragonite domains. Every growth layer comprises both calcite and aragonite deposition areas as assessed by the biochemical mapping: obviously, these clusters of mineralizing molecules, each of them specific to prisms or nacre have never been dispersed within a liquid crystallization chamber, but deposited in such a way that a very precise limit between the two domains is permanently maintained through the lateral expansion of the shell.
Initiated at the growing edge of the mantle, prisms grow in length by superposition of mineralized layers, up to coverage of their growth surface by the organic sheet (
Figure 3b). Evolution of aragonite is more complex. Granular or fibrous aragonite is first deposited at the forefront of the aragonite area (
Figure 3c–d and f–g). Later,
i.e., more internally with respect to the calcite/aragonite limit, or above when looking at transversal sections (f–i and k–l) the first nacreous tablet centers appear (
Figure 3n:
ntc) whose specific biochemical composition is now established [
43]. Development of nacreous tablets follows the Erben’s morphological sketch (
Figure 3o) [
41], but from a structural view point is also a layered stepping process (
Figure 3p), as evidenced by Mutvei [
42].
2.4. Coral Skeletons are also Built by Superposition of Two Distinct Mineralization Areas
A dual crystallization process can be illustrated also in the skeletons of scleractinian corals (
Figure 5). In addition to aragonite fibers that form the main part of the corallites, a second crystallization domain exists, without exception, also producing aragonite crystals. This latter is made of randomly oriented tiny crystals [
44]. Viewed from upper surface of the corallites, spatial distribution of the microgranular domains is very diverse but typical for each coral genus and biologically controlled, as shown by comparative molecular biology [
37]. This is the reason why architecture of the coral septa is a first-rank taxonomical character at the genus and family level. Actually, the microgranular domain is always in median position in the coral septa, sandwiched between the lateral fibrous layers (
Figure 5h–i and 5l–n). A remarkable agreement exists between these geometrical data and the pioneering observations of Vandermeulen and Watabe [
45] on the development of mineralization in the post-larval growth of
Pocillopora. A few hours after larval settlement,
skeleton formation occurs first as a purely granular mineralization producing the first cycle of juvenile septa and circular wall. This early skeletal stage is followed by an additional layer of fibers on both sides of the granular framework. In spite of the complex morphological and structural evolution that has occurred in the coral Scleractinia since their origin in the Triassic time, extensive molecular investigation has shown that such a two-step mineralization process was preserved by scleractinian corals through formation of their evolutionary lineages.
The biochemical characterization of the organic components associated to the skeletal elements fully confirms the duality of the skeletal microstructure in corals. Synchrotron-based XANES mapping allows simultaneous characterization of the high polysaccharide contents (
Figure 5c–f) of the distal mineralization areas [
46]. Actually, these granular mineralization areas are the continuation of the first mineralization phase observed by Vandermeulen and Watabe. Simultaneously, the banding pattern of polysaccharide distribution (
Figure 5f: arrows) is also made visible, polysaccharide layers corresponding to the successive calcification processes and stepping growth mode of fibers.
Thus, in addition to simultaneous crystallization of morphologically distinct aragonite skeletal units, a chemical property of coral skeletons demonstrates spectacularly the inadequacy of the common crystallization volume (named “sub-ectodermal space” in corals). Strongly different isotope fractionations or minor element concentrations between the two mineralizing areas have been firmly established by different technical approaches. It was firstly recognized on deep-sea corals, taking advantage of their remarkably simple organization.
Desmophyllum a solitary coral (
Figure 5j–k) has a size large enough to use the classical drilling method in order to separately collect calcareous material from the median mineralizing area and compare it to the lateral fibrous structures (
Figure 5l–m). Using this method, Adkins [
47] found a considerable difference between the δ 18 O of these distinct mineralizing areas. Also recognized in
Lophelia (another deep-sea coral) by using SIMS measurements [
48] these properties were generalized to a wide diversity of Scleractinia [
49] then extended to minor element concentrations [
50]. Since this early evidence, equivalent results have been obtained each time that instruments with spatial resolution compatible to the sizes of the distinct crystallization areas have been used.
Figure 5.
Figure 5. Dual crystallization and environmental recording in coral skeletons. (a–d) The two mineralization areas in coral skeletons are illustrated here by a lateral view of a septum of a Colpophyllia coral (from Guadalupe, Caribbean archipelago): spines of the growing edge (a: arrows) produce short lateral branches appearing as granules of the septum sides (a: double arrows); SEM view (b) show the long recognized specific microstructure of these growing edge areas (b: g) well distinct from fibrous tissues (b: f = fibers seen from their distal tips); (c–e) Synchrotron-based XANES mapping in a Montastrea corallite. c: Morphology of the corallite; d: polished surface on the sample holder; e: studied surface under UV illumination to locate the distal mineralization areas (microgranular); f: distribution of sulfur from sulfated polysaccharides by XANES method: note the higher content in polysaccharides of the median mineralizing areas and the clearly visible banding pattern in the fibrous area (black arrows). ma: microgranular area, fa: fibrous area; (g) Etching made after XANES mapping reveals the skeletal microstructure of the corresponding area: tiny microgranules of aragonite (ma) are visible in the median (=distal) area, as well as the stepping growth mode of fibers on both sides. (h,i) Scheme of the stepping growth-mode of a coral septum, viewed from a section perpendicular to the septal plane. (j–m) Difference in δ18O between the two distinct mineralizing domains (median area made of tiny granular crystals and fibers, on both sides of the median area) in the septum of a Desmophyllum; (n–o) Either continuous (n: section in a septum of a Favia corallite) or discontinuous as in the skeletons of the Porites colonies (o), a significant differences in isotopic fractionation or minor element partitioning is always found between microgranular and fibrous mineralizing areas. In 5o, ellipses exemplify how sampling positioning may result in important dispersion of the measured values depending on the proportions of the two distinct mineralizing areas involved in the measurements.
Figure 5.
Figure 5. Dual crystallization and environmental recording in coral skeletons. (a–d) The two mineralization areas in coral skeletons are illustrated here by a lateral view of a septum of a Colpophyllia coral (from Guadalupe, Caribbean archipelago): spines of the growing edge (a: arrows) produce short lateral branches appearing as granules of the septum sides (a: double arrows); SEM view (b) show the long recognized specific microstructure of these growing edge areas (b: g) well distinct from fibrous tissues (b: f = fibers seen from their distal tips); (c–e) Synchrotron-based XANES mapping in a Montastrea corallite. c: Morphology of the corallite; d: polished surface on the sample holder; e: studied surface under UV illumination to locate the distal mineralization areas (microgranular); f: distribution of sulfur from sulfated polysaccharides by XANES method: note the higher content in polysaccharides of the median mineralizing areas and the clearly visible banding pattern in the fibrous area (black arrows). ma: microgranular area, fa: fibrous area; (g) Etching made after XANES mapping reveals the skeletal microstructure of the corresponding area: tiny microgranules of aragonite (ma) are visible in the median (=distal) area, as well as the stepping growth mode of fibers on both sides. (h,i) Scheme of the stepping growth-mode of a coral septum, viewed from a section perpendicular to the septal plane. (j–m) Difference in δ18O between the two distinct mineralizing domains (median area made of tiny granular crystals and fibers, on both sides of the median area) in the septum of a Desmophyllum; (n–o) Either continuous (n: section in a septum of a Favia corallite) or discontinuous as in the skeletons of the Porites colonies (o), a significant differences in isotopic fractionation or minor element partitioning is always found between microgranular and fibrous mineralizing areas. In 5o, ellipses exemplify how sampling positioning may result in important dispersion of the measured values depending on the proportions of the two distinct mineralizing areas involved in the measurements.
![Minerals 02 00011 g005]()
Credit pictures: c–g: [
46]; n–o: [
49].
In addition to their importance in paleo-environmental studies (measurements must be made on microstructurally homogenous areas), these differences in isotopic fractionations and minor element concentrations between the distinct crystallization areas of a given coral skeleton is also demonstrative of the inadequacy of the “common liquid chamber” (or “sub-ectodermal space” model for crystallization of the skeleton units. A spatial distribution of chemical or isotopic properties between the two mineralizing domains of the skeleton (both aragonite in this case) is not possible if crystallization has occurred within the classically hypothesized “common liquid chamber”.
Conclusively, such a close correlation between mineralogy, microstructure, biochemistry of mineralizing matrices up to minor element concentrations and isotope fractionation of the mineral phase fully disprove the hypothesis of crystallization occurring within a common liquid chamber located between the mineralizing organ and the growing mineral surface. In other words, this implies that during the mineralizing phase, a close contact must exist between skeletal surface and the epithelial cell layer. These structural and chemical converging results fully confirm the Clode and Marshall’s observation of a close contact between coral skeleton and the overlying basal epithelium of the polyp [
51].
Conclusion 2: Structural and chemical investigations bring converging results disproving the possibility of a common liquid-filled chamber between the mineralizing cell layer and the mineralized surface of the growing skeletons.
Remarkably, two similar conclusions were drawn several decades ago by independent investigators. Miles Crenshaw, who made important contributions to the knowledge of organic components involved in crystallization of biogenic Ca-carbonates was for a long time working on the composition of the “extra-pallial fluid”. Considering his results and those of co-workers using similar analytical approaches, he wrote a singular statement fully diverging from the common opinion: between the mineralizing organ and the inner shell “the transfer of material is essentially direct” [
52] (p. 120).
Some years before, Wilbur (
Figure 1) [
53] (p. 244) had presented an equivalent view under a graphic form: a precise sketch of spatial relationships between mantle and shell of a prismatic/ nacreous bivalve mollusk. In contrast to the above cited schemes (all more recent than Wilbur’s one), the outer mantle epithelium is drawn very close to the whole surface of the inner shell. Actually, Wilbur also used the expression extra-pallial fluid and was interpreting the shell structure in term of alternating organic and mineral layers (
Figure 2) [
53]. However, in Wilbur’s figure, the space dedicated to “precipitation” was about 1 micrometer in thickness, which is typically the thickness of the organic gel at the moment where crystallization occurs. A third (and even older) example is found in the 1933 study of Kessel [
54] of the structure of a gastropod shell (
Vivipara viviparus) in which the layered structure of the shell is interpreted as evidence for a cyclic mineralization process producing the most geometrically complex and well ordered microstructure (crossed-lamellar), the formation of which as a freely growing process within a liquid chamber cannot be conceived without a tight control of the mineralizing organ. Remarkably, it was also in the 80s that Johnston, after a careful investigation of coral skeletons, wrote that the proposed models involving organic compounds in biomineralization or experimental approaches were carried out “in ignorance of this material’s spatial distribution and micro-architecture within the skeleton” [
55,
56]. It is also a major change in biomineralization studies that newer methods of observation and measurements now offer a positive perspective for investigating this key question.