Sleep and Heat Related Changes in the Cognitive Performance of Underground Miners: A Possible Health and Safety Concern
Abstract
: This review describes some of the literature pertaining to sleep deprivation, shift working, and heat exposure. Consequences of each on human cognitive function, particularly with respect to vigilance and attentional capacity are reviewed. Individually, each of these factors is known to impair human cognition; however, we propose the possibility that for miners working in hot underground environments and who are assigned to rotating shifts, the combination may leave miners with significant degrees of fatigue and decreased ability to focus on tasks. We suggest that such decreased capacity for vigilance is a source of concern in an occupational health and safety context.1. Overview
Western societies have relatively recently undertaken a shift from an agrarian-based “wake at dawn-bed at dusk” pattern of activity to a 24 h/7 day per week system where virtually every service desired is available at any time during the day and night. On an evolutionary scale, this new pattern of human activity is nascent. In other words, our current habits have us operating out of phase with eons of biological development.
The literature reviewed here describes some of the consequences of altered sleeping behaviours for those who work shifts with a particular emphasis on those employed by the mining industry. We also describe some implications of poor sleep quality that may be exacerbated by the physical environments of the underground mining industry.
We will argue that the experience of shift working, increased degrees of “sleep debt”, (the difference between the amount of sleep an organism requires and the amount experienced) [1], the effects of increased ambient temperatures during work times, and sleep pathology might adversely affect miners' cognitive abilities. We will suggest that from an occupational health and safety perspective, such cognitive deficits may lead to situations that are dangerous for sleep deprived miners and those working around them.
This paper advances the hypothesis that miners will oftentimes be working in harsh physical environments whilst experiencing some degree of cognitive dysfunction arising from abnormal sleeping behaviours. Changes in normal sleeping behaviours may be the result of miners needing to work outside of normal daytime hours, needing to shift normal daily circadian patterns to accommodate rotating shifts, or sleep pathology. Fatigue induced cognitive changes arising from altered sleep patterns may be exacerbated by working in hot underground environments. Individually, or perhaps in interaction, these factors may leave miners with an increased risk for experiencing workplace accidents. We review here literature pertaining to the influence that these individual factors may have on human cognitive function before advancing our premise that such cognitive deficits are potentially contributing to unsafe workplaces in the mining sector.
2. Introduction
A fundamental reality for life on Earth is that it came to its current form with an immutable pattern of light and dark featuring prominently in its evolutionary development. This pattern of alternating light and dark has been an important feature of evolutionary processes from when the cosmos first formed. Accordingly, most, if not all, existing species have evolved a biology that responds to the planetary light/dark cycle.
Many responses to the planetary cycles are expressed at a behavioural level [2]. For example, rats are a nocturnal species, biologically predisposed to being active during darkness. So too are their predators, owls being an obvious example.
Some circadian patterned activities are endogenous synchronous activities that require daily resetting by sunlight such that the organism is entrained to the local light/dark conditions. Waking, eating, working, etc. are behavioural examples of circadian patterned activities. Circadian responses are also seen on molecular scales [3]. For example, clock genes are active during specific times of the day coding for biochemical responses that are necessary for the organism to respond to light or dark cued events. Transcriptional activators CLOCK and BMAL1 target the genes Period (Per) and Cryptochrome (Cry). These target genes form repressor complexes that inhibit the parent activators in a rhythmic fashion. While active, the target genes specifically modify metabolic events necessary for the organism to function in response to the light/dark cycle [4]. Clock gene activity is also related to some cognitive processes [5].
These biochemical events are described here to demonstrate that cellular processes programmed to the light/dark cycle exist. As suggested below, disturbances of this intricately balanced cellular activity may have serious consequences for human health and mental function [6]. The processes are the result of eons of evolutionary process, leaving humans as diurnal organisms that manifest biological imperatives on many different scales. Many human biological mechanisms are patterned to be active (or inactive) as a function of the light/dark cycle [7]. For the purposes of this review, we need only acknowledge that clock gene activity is important to the regulation and maintenance of the circadian pacemaker's control of these rhythms [4]. It is beyond the scope of this review to detail the neurobiology of entrainment and other circadian patterned cellular processes; however, excellent review articles detail the interactions between neurons of the suprachiasmatic nucleus and endocrine and other metabolic processes (For example, see Bass and Takahashi [4]).
We describe here two well-known biochemical systems that are influenced by sleep and which have subsequent implications for human cognitive function. This review argues that poor quality sleep for miners will decrease workplace safety as a result of altered vigilance and attentional capacity. Again, an extensive review of sleep neuroendocrinology is beyond the scope of this article; however, we overview these two systems to demonstrate the relationships between altered neurochemical systems and human cognition. Disruptions in these systems as a result of poor sleep may be a factor for occupational safety [8].
One important neuroendocrine system that assists in the resetting of biological mechanisms to local light conditions is the melatonin system. The pineal gland secretes melatonin [9] which is involved in the regulation the sleep-wake cycle [10,11]. Melatonin concentrations peak at night when light levels are low and act to inhibit the mechanisms of wakefulness [11]. Sleepers with normal sleep architecture, defined as a typical pattern of sleeping behaviours (see below) generally have a peak secretion of melatonin between 0100 and 0500 h [12,13].
Disruptions to melatonin rhythmicity can result in cognitive dysfunction [8]. In the absence of the evolutionarily defined peaking effect, homeostatic benefits of melatonin may be altered for those obliged to be awake during the night. Research has found that sleep efficiency (ratio of total sleep time to time in bed) is highest when the person sleeps during the night with endogenous melatonin release [14]. In contrast, when a person attempts to sleep during the circadian phase that has little melatonin release (daytime sleep), sleep efficiency is markedly reduced and the sleeper experiences more intervals of wakefulness [15].
Cortisol is another important hormone that is influenced by the sleep wake cycle. When the human cortisol system is disrupted, fatigue, depression, and immune system disorders often manifest. Those with disrupted cortisol regulation are also prone to obesity [16]. Cortisol levels are high during wakefulness and low during sleep. The lowest concentrations of cortisol are seen during the very earliest stages of light sleep and peak at the moment prior to our awakening in the morning, assuming that such awakening is biologically derived and is not as a function of external stimuli [17]. Cortisol secretion is also increased during times of stress [18]. Cortisol is implicated in the development of insomnia and its subsequent cognitive implications [19]. Thus, it may be that shift workers who experience abnormal cortisol secretions resulting from needing to work outside of endogenous circadian patterns may have diminished cognitive resources related to changes in cortisol metabolism.
These neurochemical systems are examples of the evidence that the human biology responsible for our sleep and waking behaviours has a fundamentally important relationship with the planetary light/dark cycle [3]. Irrespective of personal choices made by the individual to be active outside of the genetically determined circadian systems of the sleep/wake cycle, biochemical and cellular processes continue to operate on those patterns defined by evolutionary processes. As is described here, there are implications for biological mechanisms, some of which are potentially life threatening [20] when humans deemphasize the importance of their normal circadian rhythms.
2.1. The Human Sleep Cycle
While there have always been humans who were required to be alert during the night (those responsible for health or child care, military personnel, etc.) the societal shift to being active outside of daylight hours is directly attributable to Joseph Swan's (1828–1914) 1850s commercialization of the modern electric light bulb and Thomas Edison's (1847–1931) subsequent development of an efficient distribution system for electrical power in 1882. These milestones have permitted humans to actively engage in activities beyond daylight hours. Thus, as recently as 130 years ago, humans experienced opportunities to systematically participate in activities out of phase with the biological patterns that have been selected for over eons of evolution.
It is obvious that the extension of our activities into the dark phase has afforded humans with an enormous advantage in productivity and advancement; however, there is no disputing that those living in Western societies are behaving differently in terms of the behavioural and biological systems of our progenitors. Evidence is accumulating which suggests that there are enormous costs paid by some people who routinely undertake dark-phase activity [20].
To appreciate the implications of changes in sleep and circadian patterns arising from working outside of the normal light/dark cycle, an understanding of the normal patterns associated with human sleeping behaviours is necessary. This section reviews normal human sleep patterns and provides a primer of sleep and circadian medicine.
Readers will recognize the basic pattern of the sleep/wake cycle as a result of their own experiences. They know that under circumstances where there are no demands to function outside of the normal pattern of activity, every night humans are obliged to seek a warm, safe place where they can recline for, on average, 8 h. When humans sleep, they are effectively removed from a conscious perception of their external world, save for some sentinel functions that may serve protective purposes [21].
Less commonly understood is that sleep is not a homogenous state. During human sleep, there is a fundamental alteration in neurochemistry as a function of the person's specific sleep state. Humans are known to experience 2 different states of cortical activity when they are sleeping, each with its own underpinning neurophysiology [22].
The first brain state normally experienced as humans progress into a night's sleep is characterized by a reduced degree of brain activity. This state is called non-rapid eye movement (nREM) sleep [22] and it can be differentiated into subclasses using clinically relevant objective markers of electrophysiological activity. These subclasses include stage N1, a light stage of sleep that marks the transition between wakefulness and sleep, and N2 which is the sleep state that predominates throughout a typical night's sleep. The most profound reduction in brain activity during nREM sleep is during N3 sleep which is also known as slow wave sleep (SWS) [23]. SWS manifests on electroencephalogram (EEG) tracings with large jagged waveforms that are thought to reflect neural connections between the subcortical thalamic nuclei involved in sleeping behaviours and the cortex where most human cognitive functions are processed [24]. Some readers may note that this classification system of sleep states is unlike the familiar Rechtshaffen and Kales sleep scoring rules [25] used previously. The classification system used here replaces the “R and K rules” and our use of the new rules for staging and scoring sleep reflects current clinical practice [23].
The experience of SWS is important for humans as it is during this phase when important metabolic events like the secretion of growth hormones [26] are known to take place. Some memory functions are thought to be dependent on the experience of SWS [27].
The second brain state that arises during human sleep is called rapid eye movement (REM) sleep. In the timeline of medical research, the discovery of this brain state is new as it was only in the 1950s when researchers at the University of Chicago systematically investigated the observation that the eyes of sleeping humans move in a bizarre fashion during sleep [28]. Since the discovery of REM sleep, the scientific investigation of sleeping behaviours has yielded an enormous body of literature. We emphasize the nascent nature of the state of sleep research to illustrate that a scientific understanding of human sleep is preliminary. Hundreds of scientists across the globe explore sleeping behaviours; however, the function and purpose of sleep remains somewhat speculative [29].
Mammals that are experiencing REM sleep exhibit cortical neural activity that is quite similar to that of their wake state [28]. During REM sleep, a large amount of metabolic energy is expended in a process that the sleeper is largely unconscious of. A human that is awoken from REM sleep will likely reiterate some dream-based imagery; however, if the sleeper is not awoken, the cognitive recall of the dream content will pass from the sleeper's awareness immediately [30]. It is unlikely though that this expenditure of biochemical resources has the sole purpose of providing dream imagery to the human experiencing REM sleep; however, a parsimonious explanation for the several objectively observed phasic and tonic features of REM sleep has yet to be advanced [29].
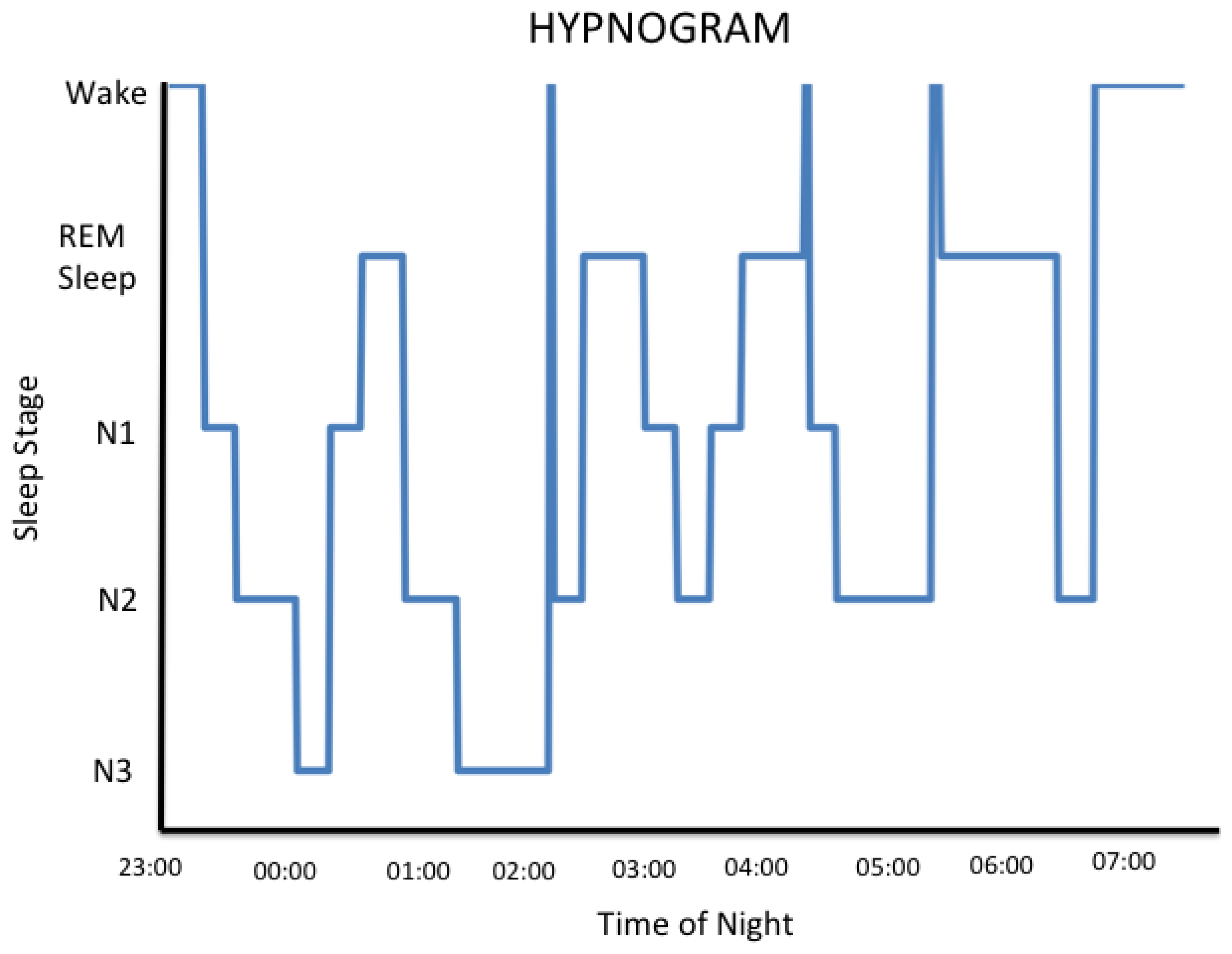
Thus, for the human, there are 3 specific brain states, each of which arises from their own unique neurophysiology: wake, nREM and REM sleep. For the human experiencing a routine night's sleep, the transitions between brain states are not linear. For the normal human brain that is entrained to and is responding to daylight, the wake state predominates and conscious awareness of the environment is established. During the night, each of the 3 brain states is expressed during sleep when consciousness of the external world is either greatly diminished or is absent [31]. The hypnogram below shows that sleeping humans experience epochs of each of the 3 brain states throughout the night but not in a linear order. The hypnogram reveals a cyclic pattern of the 3 brain states throughout the night. Humans enter their first REM sleep epoch approximately 90 minutes after they fall asleep and remain in REM sleep for approximately 20 minutes. The initial REM sleep interval is shorter than those that are experienced later in the night. Humans tend to have more nREM sleep earlier in the night. Note that there are intervals of wake that occur normally during a typical night's sleep; however, these are generally short and it is unlikely that the sleeper retains awareness of them [31].
When humans are prevented from sleeping in a manner dictated by their own biological mechanisms, they show increased sleepiness as a result of homeostatic processes [32]. Sleep debt is reduced when the sleep restricted person obtains recovery sleep. The degree to which an individual pays back that debt is proportional to the amount of sleep that was lost [33].
With respect to the hypotheses being advanced by this review, an understanding of the sleep architecture graphed in the hypnogram is important. The cycling of brain states has sophisticated neurochemical control mechanisms housed in phylogenetically ancient brain structures [34]. That is, the mechanisms serving homeostatic functions of the sleep cycle were organized in the human brain eons ago in response to the light/dark cycle and have since been consolidated by natural selection processes. Thus, the usual human sleep architecture is the result of millions of years of evolution and any homeostatic functions served by the experience of sleep are likely to be maximized when that neurobiology is expressed as is coded for by our evolutionary history.
Shift working miners will by necessity of workplace requirements be constantly performing outside of their normal homeostatic sleep wake drives [35]. Thus, employees who are beginning their shifts with a degree of sleep loss will simultaneously be faced with an increased drive to recover the lost sleep [36]. Irrespective of the cognitive changes that are directly attributable to truncated sleep time [37], the homeostatic drive to recovery sleep will also induce changes in cognitive function [38]. Organizational psychologists have realized the importance of recovery sleep [39].
2.2. Functions of Sleep
As described above, sleeping behaviours are processed in phylogenetically ancient neuroanatomical structures [34]. When we consider that in our evolutionary history, the act of assuming long periods of quiescence and dissociation from the external environments would have left our progenitors prone to predation, sleeping might appear to be maladaptive. However, since the experience of sleep-like responses is ubiquitous in the animal kingdom [40], and since there are severe and potentially life threatening consequences of sleep deprivation [41], it is clear that the experience of sleep serves some important metabolic and homeostatic functions. Natural selection processes suggest that sleep is of significant importance to the biology of human functioning.
Various hypotheses describing the function(s) of sleep have been advanced [29,42]. Many of these functions have been attributed to the general experience of sleep or to the experience of specific sleep states. For example, research has indicated that immune response efficiency is correlated with healthy sleeping [43]. Similarly, efficiency in thermoregulatory mechanisms has a basis in the neurophysiology of sleep [44]. Other functions that sleep is thought to be involved with include learning and memory formation [45-47], neocortical maintenance [48], energy conservation [49], and brain development [50]. It is beyond the scope of this review to detail all of the functions attributed to sleeping behaviours. We direct interested readers to excellent reviews authored by Frank [51] and Kilgore [52].
Our hypotheses are that sleep deprivation, working rotating shifts, pathology, and heat exposure may lead to unsafe workplaces with respect to potential lapses in vigilance and attention. We acknowledge that there are myriad of functions served by sleep and alterations of any of those may lead to outcomes that decrease workplace safety; however, here we focus on the cognitive benefits conferred as the result of healthy sleep experiences.
2.3. Sleep and Cognition
The literature describing the relationships between sleep and cognition is vast. The roles that sleep serves in various aspects of cognition have been investigated [52]. Previous literature suggests that the experience of sleep and/or specific sleep states factors into response time [53], short-term memory [54], and mathematical skills [55] among many others.
Executive functions such as innovation [56], multi-tasking [57], working memory (that which requires storage of information necessary for the performance of a given task) [58], impulsiveness [59], and verbal skills [60] are altered as a result of sleep disruptions. Further, chronically sleep deprived individuals demonstrate poor introspective understanding of the extent of their impairment [61]. Thus sleep-deprived individuals may show objectively measured declines in their executive functions; however, they have little insight into such deficits.
While any of these deficits might detrimentally influence worker productivity, from the perspective of workplace safety decreased attention and vigilance arising from sleep disruption is likely more important. It is not only sleep deprivation that may cause attention lapses. Alertness changes also with circadian phase, so, in addition to the experience of sleep disruption, the time when the subjects' alertness is measured is important [62]. It may be that when normal sleep architecture laid down by evolutionary processes is disrupted by changes in circadian patterns, another possible contributing factor for unsafe working environments is introduced.
From the perspective of occupational health and safety, miners who are having lapses in attention or who are unable to focus on a tedious task as a result of sleep restriction or fatigue might be susceptible to making errors that could lead to accidents and increased workplace mortality [63]. While it would be difficult to evaluate post hoc if a lapse in worker's vigilance due to sleep disruption was responsible for a specific personal injury or one that caused injury to a nearby coworker, fatigue and drowsiness have been reported as factors in many notorious incidents including the Challenger shuttle explosion [64], the Exxon Valdez oil spill [65], and the Union Carbide gas leak in Bhopal [66], among others.
2.4. Consequences of Sleep Deprivation
Sleep deprivation leads to dysfunction measurable on a number of dimensions ranging from cellular responses [67,68] to societal outcomes [69]. Anywhere on that continuum of sleep deprivation induced changes are issues that pertain to the thesis proposed here: miners who do not acquire adequate sleep are more likely to develop physical and cognitive deficits that could lead to workplace accidents. Some of the consequences of reduced sleep are described here.
Animal and clinical literature describes the serious consequences of moderate sleep deprivation. The focus of this review is on the implications for cognition that sleep alterations have; however, there are many somatic results too. We briefly describe some of these here as these physical outcomes may potentially induce subjective complaints of sleepiness and subsequent cognitive changes. As examples, chronically sleep deprived humans have an increased prevalence of metabolic disorders [70], neuroendocrine problems [71], reduced immune responses [72], and increased oxidative stress [73]. Type II diabetes and obesity [74] are more likely in the population of sleep deprived individuals, as are mood disorders [75], decreased sociability, and somatic complaints [76].
Perhaps the most obvious outcome of sleep deprivation is fatigue. Fatigue can be considered as either a mental or physical process [77]. Fatigue manifests with muscular weakness, exhaustion, and lethargy; however, there are also cognitive deficits that arise when the individual is fatigued. These include anxiety, irritability, attentional lapses, and listlessness [78]. Either physical or mental fatigue is potentially dangerous in an active mine site where much physical activity is expected; however, we focus here on the possibility of sleep deprivation induced cognitive deficits that can lead miners to have decreased vigilance and focus.
The scientific literature makes it clear that obtaining good quality sleep is a crucial factor for alertness and attentional capacity [79,80]. Subjects who were sleep deprived reported problems with higher order cognitive processing such that deficits in some types of memory encoding [81,82], decision-making [83], and divergent operations [84] were observed. The type of fatigue that may follow successive periods of poor quality sleep has negative consequences for alertness and vigilance ability [85]. Learning and focusing capacity is similarly reduced [86]. With respect to those who are required to operate heavy equipment or engage in other potentially dangerous activities, one of the most serious consequences of poor quality sleep is the increased likelihood of experiencing microsleeps. Microsleeps are intervals when a person falls asleep up to 30 seconds while ostensibly claiming an awake state [87]. When sleep deprivation is of an extended nature, people will often involuntarily initiate eye movements that are indicative of an nREM sleep state [88]. Concurrent with those eye movements are lapses in attention [89]. Obviously, such microsleeps represent a danger to those operating heavy equipment or those who are in proximity to it.
A common tool that evaluates attention and concentration is the psychomotor vigilance task (PVT). For the PVT, subjects respond to a signal that is presented on a variable schedule for an interval ranging from 5 (shortened version) to 10 minutes. Response times and attentional lapses (defined as responses that take longer than some predefined period) are recorded [90]. Extended reaction times are indicative of sleep loss [53]. Poor performance on the PVT is related to disrupted or fragmented SWS suggesting that vigilant attention is affected when SWS sleep is of poor quality [91]. Interestingly, older adults are more resistant to the effects of sleep deprivation on performance of the PVT [92].
The effects of sleep deprivation in humans have been studied using other objective methods. Diminished brain activity arising from sleepiness is reported in the neuroimaging literature [93]. For example, many of the cognitive deficits arising from poor sleep as described above are correlated with reductions in metabolic rates and blood flow in the human thalamus and cortex [94,95]. The prefrontal cortex is the neuroanatomical locus for several important higher order cognitive functions such as attention, executive processing, and performance of tasks requiring divergent thinking [96]. These areas of the human brain show decreased metabolic activity when the subject is sleep deprived [97]. Similarly, EEG data suggests a reduction in arousal when the subject is sleepy [98].
The physiological observations of sleep deprivation suggest causality for many of the cognitive disruptions arising from fatigue and restricted sleeping. While many of the studies reviewed here include manipulations of acute sleep deprivation, the danger to miners arising from such extreme sleep deprivation is less likely than it is from the cumulative effects of poor sleep [91]. It is important to note that the cumulative effects from several successive nights of poor sleep results in cognitive performances much like those that arise from a night of total sleep deprivation [61]. Chronic fatigue is likely for individuals who have accumulated sleep deprivation. Interestingly, subjects operating under conditions of sleep deprivation have little introspective understanding of the cognitive consequences of poor sleep. For example, subjects experiencing 6 or less hours of sleep for several nights reported feeling moderately sleepy; however, their objectively observed cognitive performances were markedly reduced. Thus, subjects accumulated cognitive dysfunction over days; however, they had little insight into the reductions of their attention and vigilance functions [61].
Partial sleep deprivation or sleep restriction studies also suggest that poor quality of sleep results in decreased cognitive performance. Dose response curves for subjects experiencing 4 h of sleep per day for 4 days showed levels of performance on alertness and memory tasks like those who had experienced 2 nights of total sleep deprivation. Belenky et al. (2003) [85] showed that when subjects had their normal sleep time shortened by as little as 2 h per night over a week long interval, the subjects performed some tests of vigilance and attentional ability as though they were totally deprived of sleep for 24 h. PVT results suggest that for otherwise healthy subjects who were restricted to less than 4 h of sleep over 5 consecutive nights had measurable deficits on attention and vigilance skills [99]. Thus, cognitive declines were similarly accumulated in direct relationship to fatigue induced by accumulated sleep restriction. It is important to note that even as the sleep restricted miner may be able to pay down their sleep debt that accrues over several nights of truncated sleep on their off time, the cognitive consequences of excessive sleep loss are not mitigated by a single night of recovery sleep [85].
A particularly compelling way for describing cognitive changes arising from sleep deprivation equates the extent of sleep deprivation with performances like those who have consumed alcoholic beverages. Blood alcohol levels (BAL) provide a measure that most readers will understand as a result of wide media attention to the problem of driving while under the influence of alcohol. Comparing altered performance on tasks due to sleep deprivation performance to that like those with increased BAL gives salience to the nature of the sleep deprivation based performance. Studies that have used this approach have indicated that remaining awake for 19 consecutive hours yields results on some cognitive and reaction time tasks like those who have a BAL of 0.05%. Extending the awake time to 24 consecutive hours leaves individuals with response times similar to those with a BAL of 0.1% [100]. BAL of this magnitude exceeds the legal limit for operating a motor vehicle in most legal jurisdictions in North America. Studies conducted in simulators [100] and in field studies [101] suggest that on some measures, workers performed as though they had BAL of 0.05 or higher when just moderately fatigued [102].
Thus, poor quality of sleep clearly affects human cognitive functions. The research described to here related only the implications of shortened sleep times. Other sleeping-behaviour related phenomena might also induce fatigue in miners who are working. Miners are routinely obliged to alter their biologically defined patterns of circadian behaviours. We now consider the consequences for cognition resulting from such alterations.
3. Shiftworking
Shift working is a fact of life for many employees. For Western workplaces, it is estimated that 20% of employees are obliged to work shifts [103]. Working rotating shifts is certainly an expectation of those employed as underground miners. Irrespective of any personal choices made by those in our “24/7” society to spend less time asleep than might be biologically optimum, practical requirements of working shifts truncates time in bed. Miners (and other shift workers) may be required to start their work interval near 0700 h. In order to be at work on time, it is likely that the miner will need to arise very early to allow for commuting time, etc. For example, a miner scheduled to work a day shift might need to awaken near 5:00 am. To acquire 8 h of sleep, the average time spent asleep for humans per night, the miner would need to retire at 2100 h. While there are probably some adults who are able to sleep this early, it is likely that many would find it difficult to sleep at that time either due to societal or family pressures to be awake or the circadian driven propensity for sleep initiation prohibits it [104].
Similarly, those scheduled to work night shifts may be reporting for duty after a reduced opportunity for sleep. Working night shifts often results in workers having poor personal habits that exacerbate sleep related issues. For example, poor diet, substance abuse, smoking, and diminished exercising are correlated with working night shifts [105] and each of these factors is related to decreased sleep quality [106,107]. For those working night shift, the risk of accidents increases, particularly at the end of the shift [108].
Even while the night shift worker may be tired from their previous shift, they might still need to respond to events during regular hours. Timetables for circumstances pertaining to parenting, medical appointments, banking, etc. may require that the shift worker attend to such events even though they have been awake for several hours and/or they will need to be awake for several more hours to come.
These workplace requirements of miners who work shifts leave them in a state of sleep deprivation with all of the inherent cognitive consequences described in the previous sections [109]. While working shifts induces a degree of sleep deprivation, there are other potential causes of cognitive impairment in shift working miners. Even when the night shift working miner is able to acquire sleep during daylight hours, circadian misalignment of the opportunity to sleep and the biologically defined time of increased sleep drive often have night shift workers complaining of insomnia whilst being unusually fatigued [85]. Irrespective of the fatigue that may be so induced, daytime sleep obtained by a night shift working employee might be of decreased quality and quantity [110]. Sleep obtained during the day tends to have reduced sleep efficiency [110] and sleep efficiency is positively correlated with human cognitive function [111].
The fact that some employees are required to work rotating shifts is in and of itself problematic. Consequences of working rotating shifts include insomnia [112], an increased prevalence of some types of cancer [113] and cardiovascular disease [114], or shift workers may have adverse outcomes during pregnancy [115], among others. Analysis of EEG recordings taken from rotating shift workers indicate that their day time sleep is 1–4 h shorter than the sleep obtained when the worker is on day shifts [116,117]. The stage of sleep that is most reduced under these circumstances is “N2” sleep [118,119]. Older literature may refer to this stage of sleep as “stage 2 sleep” reflecting a past approach to scoring clinical sleep studies [25]. N2 or stage 2 sleep is the predominant sleep stage of nREM sleep (see Figure 1 above). As described above, the neurophysiology associated with stage N2 sleep has been shown to be important for some cognitive processes. For example, it is associated with some types of learning [120,121].
Thus, the possibility of a worker beginning their shifts with some degree of fatigue is high. Individuals working night shift are more likely to be sleepy during their shifts. Objective evidence of an increased sleepiness on the night shift is found in EEG data collected from train operators [122]. Approximately 25% of the subjects assessed showed brain wave patterns that were indicative of sleep. Further, these subjects showed an increased tendency to express slow eye movements in the early morning, another physiological manifestation of sleep. Decreased cognitive capacity was observed during these objectively determined hallmarks of sleep. For example, train operators were more likely to overlook a warning signal [122]. Similar findings were seen in a sample of truckers [123] and long-haul aircrew [124]. Some process workers were observed to actually fall asleep during their night shifts even though they self-reported sleepiness but not the experience of sleeping [117]. Medical interns working in hospital environments overnight showed EEG determined sleep intrusions and attentional lapses in the early morning [125]. Simulator studies using truckers [126] and power station operators [127] as subjects corroborate the field study data. Clearly, there are cognitive consequences associated with working night shifts.
Space considerations preclude a review of the biological and cellular consequences of working rotating shifts; however, interested readers are directed to an excellent review by Bass and Takahashi [4]. It is important to note though, some researchers have identified the experience of shift working as a health hazard. For example, the International Agency for Research on Cancer has stated that shift working is probably carcinogenic [128].
Objective data measuring performance confirms that shift workers, particularly those working at night, are susceptible to alterations in their cognitive abilities while at work [85]. The length of the shift being worked may also contribute to fatigue-induced cognitive changes [129]. When shift workers are engaged in 12 h shifts there is only a 12 h interval between successive night shifts. With commute times, societal and family obligations, and biological asynchrony with sleep drives, there are approximately 6 h of available sleep time between shifts. Sleep obtained during that 6 h period is likely to be of reduced quality [130]. As described above, sleep debt can accumulate and since miners are likely to be assigned 3 to 4 consecutive shifts, cumulative effects on cognition will likely manifest [131]. Extended duration shifts are related to an increase in the number of workplace accidents, fatigue, decreased vigilance and altered quality of sleep [132]. Decreased productivity and performance as well as increased reaction times have been recorded in those who work extended duration shifts [133]. Sleep loss due to the length of working hour requirements can predict impaired cognitive performance and subsequent increases in accident and error rates [89].
Of increasing concern to employers is the safety of their employees when they drive home at the end of their shifts. Such employees are at significant risk of being involved in a motor vehicle accident relative to the general populace [134]. Those who work at night are particularly at risk for crashing while driving after their shift [102]. As described previously, research equating BAL and driving performance could be interpreted as having these shift workers driving whilst impaired.
The issue of fatigue while driving has been the focus of much research. Of particular interest has been the relationship between fatigue, sleep deprivation, and accident rates in the transportation sector [135]. With respect to drivers who work overnight, there is a pronounced increase in the rate of road accidents involving trucks on the night shift [136,137]. The American National Transportation Safety Board reports that between 30 and 40% of all American trucking accidents are as a result of driver fatigue [138]. Total sleep time in the preceding 24 h and disrupted sleep patterns arising from split-shifts were identified as the most influential factors in truck accidents. Given the reliance on heavy machinery in modern mining workplaces, these data have bearing on occupational health and safety concerns for underground operations throughout the night.
In addition to the effects of acute or accumulated sleep deprivation, employees who work extended duration shifts are more likely to be involved in motor vehicle collisions [102]. A regression analysis revealed that for every extended duration shift an employee worked per month, there was an 9.1% increase in the crash rate overall and a 16.2% increase in the likelihood of crashing during the commute home after such a shift [139].
With respect to risk management, sleepy shift workers who are driving home after a shift may expose their employers to liability issues. In the United States, fatigued drivers who were involved in personal injury car accidents have been held jointly responsible with their employers for injuries to others [140].
In summary, the literature reviewed here raises the hypothesis that shift working miners' attention and vigilance is reduced as a result of their normal employment requirements. Their sleep is shortened either due to a need for early arousal to be at work on time or because of societal and biological factors interfering with sleeping opportunities during the day leaving them fatigued during work times. That they are expected to work rotating shifts in and of itself induces fatigue. It is clear that fatigue induces decreased vigilance and focus. Thus, it may be that employees who are performing outside of that evolutionarily defined sleep patterns or who are obliged to truncate their sleep time in order to attend to their workplace experience consequences attributable to such disruptions in normal sleep architecture that may result in personal injury to themselves or their coworkers.
4. Sleep Pathology
We assume that like the rest of Western society, mining sector employees show increased propensity for adverse lifestyle based health issues [141]. This section introduces yet another possible sleep related factor that may influence miners' vigilance and attentional capacity during work times: neurological consequences arising from sleep based medical pathology.
Many of the employees in the mining industry are middle aged or older. Biologically defined sleep patterns change as function of aging, leaving the older worker with worse sleep than when they were younger [142]. Further, older workers are more likely to have health issues that affect their sleep. For example, some medications alter sleep architecture [143,144]. Similarly, there is a well established trend to obesity and hypertension in North American society reflecting poor lifestyle choices [141]. In addition to these, smoking [145], alcohol use [146], and sedentary lifestyles [147] all contribute to poor quality of sleep and inherently reduced sleep efficiency. Objective evidence of increased obesity, smoking, addiction, or other sleep restricting medical phenomena in the population of shift working miners might be difficult to obtain and generalize to other mine sites; however, we assume that shift working miners present as a cross section of society. Accordingly, we suggest that as with the general population, incidence of these pathologies is increasing in the population of miners. Thus, in addition to causes of fatigue like shift working and/or sleep time restrictions as described above, miners may also experience reduced sleep efficiency due to medical reasons with subsequent cognitive dysfunction.
Respiratory problems during sleep (obstructive sleep apnea (OSA), for example) are under diagnosed within the general population. Some estimate that the prevalence rate of undiagnosed OSA might be as high as 80% [148]. OSA manifests with many, sometimes hundreds, of small disruptions to the normal sleep architecture due to the sufferer's repeated need to arouse such that they can inhale and subsequently, oxygenate themselves [149]. Two specific concerns arise from this maladaptive respiratory pattern with respect to daytime cognitive function. First, as described elsewhere in this review, phenomena that alter normal sleep architecture with subsequent decreases in sleep efficiency are related to cognitive dysfunction [85]. Second, the repeated decreases in blood oxygenation that occur during an apneic event may induce cumulative cellular damage within the brain [150]. Jackson, Howard, and Barnes [151] described untreated OSA patients as experiencing decreased attentional and vigilance function as a result of these nightly neurological arousals.
Neuroimaging data suggests that OSA might be causal to cellular damage leading to some of the cognitive changes associated with the syndrome. One study showed that neuronal cell bodies (grey matter) in the hippocampus, a structure vitally important for learning and memory [152,153] are reduced in numbers as a function of severity of OSA symptoms [154]. Microlesions within the brain have also been correlated with OSA [155].
Clearly, the consequences of untreated sleep pathology may be an important occupational health and safety concern as well as a matter of great personal concern for those middle aged miners who may, like the general population, have increased propensity for OSA.
5. Heat Exposure
The literature reviewed to this point has strongly suggested that miners, and other shift workers, may have decreased cognitive functions as a result of shortened sleep time, being required to work rotating shifts, having sleep pathology, or any combination of these. Previous literature has also suggested that sleep related cognitive dysfunction would become progressively worse during the course of a specific shift [108] and over the few days that the miner might be assigned to that shift before getting a series of days off [78]. We review here yet another factor that may induce fatigue and subsequent loss of attentional capacity and decreased vigilance or that might exacerbate the dysfunction arising from poor sleep: the workplace environment itself.
Underground miners are routinely exposed to environments that are dusty, damp, noisy, and hot. Miners are expected to undertake a significant degree of physical activity during the course of the shift. Underground mining environments can be as warm as 51 °C [156]. These environmental factors can induce fatigue and the types of cognitive changes that have been previously described [157].
Core body temperature measures can be elevated during work in a hot environment. Increased core temperatures are related to poor performance on some cognitive tasks [158]. Recent evidence shows that the consequences of heat stress are task dependent. Tasks that require higher order neural processing tend to be sensitive to heat exposure while routine and undemanding tasks are somewhat immune to increases in heat exposure [159]. Thus, when miners are required to respond to novel circumstances, heat exposure induced fatigue may interfere with their ability to react quickly.
Interestingly, acclimation to an environment with reductions in core body temperatures reduces heat exposure induced cognitive dysfunction [160]. These observations suggest a hypothesis that shorter shifts over more days might result in increased safety within a mining environment. As described above, extended duration shifts are more likely to induce workplace fatigue. Allowing an acclimatization effect over several days and reducing the number of hours the miner is expected to work may mitigate potential causes of fatigue and subsequent cognitive dysfunction; however, such a timetable may be unpopular with some miners, particularly younger employees [161].
Mining is a physically demanding undertaking. As such and without consideration of increased ambient temperatures, when performing physical tasks, an increased metabolic demand ensues resulting in raised core body temperatures with subsequent cognitive deficits as related above [162]. Such elevated metabolic activity requires increased heat dissipation to achieve homeostatic thermoregulation. Bleeding of heat from the actively working miner is problematic. The most effective strategy of maintaining heat balance is by diaphoresis (sweating). The cooling effect of sweating results from the evaporation of sweat after it is produced. Evaporation of sweat produced while working underground is hampered as a function of the heavy safety apparel that miners don for each shift [163].
Thus, miners who are engaged in their normal workplace activities experience increased metabolically derived heat gain, poor opportunities to dissipate that heat through diaphoretic processes, and they are acquiring more heat in transfer from increased ambient temperatures. These combined causes of increased core body temperature are likely to result in cognitive impairment that will worsen over the course of miners' extended duration shifts.
It is worth noting that there are circadian thermoregulatory processes. Thus, in addition to the processes of thermal homeostasis described above, there are times during the 24 h day when biological predisposition to increased metabolic activity occurs [164]. It may be that these cyclic changes in metabolic responses will contribute to some of the cognitive deficits observed in shift working employees at certain times of the day.
The coupling of metabolic circadian responses to increased ambient temperatures found in an underground mine, sleep deprivation, shift working, and pathological conditions leading to altered sleep architecture suggests a cumulative explanation for decreased attention and vigilance in the mining sector. Given the reliance of the mining industry on heavy moving equipment and other dangerous activities such as planting and detonating explosives, hot, sleepy, and cognitively impaired miners are a potential danger to themselves and others.
6. Conclusions
Further research is necessary to determine the extent to which underground miners are experiencing fatigue whilst working. It remains unknown if combinations of early morning awakening or sleep restriction during daytime sleep, sleep pathology, or working in hot environments decrease cognitive function for physically active miners during working hours. If there are fatigue induced neurocognitive outcomes, an opportunity for decreased worker safety exists. Modern mines likely have multiple redundant safety checks built into their procedures that act to prevent large-scale disasters; however, several mining disasters have occurred recently [165,166]. It is unknown yet, and perhaps it may never be known if employee fatigue contributed to these mining accidents. It is not unreasonable to expect that engineers and designers developed safety checks for NASA, mariners, and nuclear power stations to prevent disaster yet events such as the Challenger explosion, the grounding of the Exxon Valdez, and the near meltdown at Chernobyl are emblazoned on the public psyche. Fatigue on the part of employees directly contributed to these events. Clearly, it is in the best interests of the mining industry and its employees to fully explore this putative danger with an eye to preventing future mining disasters.
References
- Dinges, D.F.; Pack, F.; Williams, K.; Gillen, K.A.; Powell, J.W.; Ott, G.E.; Aptowicz, C.; Pack, A.I. Cumulative sleepiness, mood disturbance, and psychomotor vigilance performance decrements during a week of sleep restricted to 4–5 hours per night. Sleep 1997, 20, 267–277. [Google Scholar]
- Czeisler, C.A.; Jewett, M.E. Human Circadian Physiology: Interaction of the Behavioral Rest-Activity Cycle with the Output of the Endogenous Circadian Pacemaker. In Handbook of Sleep Disorders; Thorpy, M.J., Ed.; Marcel Dekker: New York, NY, USA, 1990. [Google Scholar]
- Rutter, J.; Reick, M.; McKnight, S.L. Metabolism and the control of circadian rhythms. Annu. Rev. Biochem. 2002, 71, 307–331. [Google Scholar]
- Bass, J.; Takahashi, J.S. Circadian integration of metabolism and energetics. Science 2010, 330, 1349–1354. [Google Scholar]
- Kyriacou, C.P.; Hastings, M.H. Circadian clocks: Genes, sleep, and cognition. Trends Cogn. Sci. 2010, 14, 259–267. [Google Scholar]
- Czeisler, C.A.; Gooley, J.J. Sleep and circadian rhythms in humans. Cold Spring Harb. Symp. Quant. Biol. 2007, 72, 579–597. [Google Scholar]
- Lockley, S.W.; Brainard, G.C.; Czeisler, C.A. High sensitivity of the human circadian melatonin rhythm to resetting by short wavelength light. J. Clin. Endocr. Metab. 2003, 88, 4502–4505. [Google Scholar]
- Wright, K.P., Jr.; Hull, J.T.; Hughes, R.J.; Ronda, J.M.; Czeisler, C.A. Sleep and wakefulness out of phase with internal biological time impairs learning in humans. J. Cogn. Neurosci. 2006, 18, 508–521. [Google Scholar]
- Lavie, P. Melatonin: Role in gating nocturnal rise in sleep propensity. J. Biol. Rhythms. 1997, 12, 657–665. [Google Scholar]
- Shochat, T.; Haimov, I.; Lavie, P. Melatonin—The key to the gate of sleep. Ann. Med. 1998, 30, 109–114. [Google Scholar]
- Lavie, P. Sleep-wake as a biological rhythm. Annu. Rev. Psychol. 2001, 52, 277–303. [Google Scholar]
- Brown, G.M.; Young, S.N.; Gauthier, S.; Tsui, H.; Grota, L.J. Melatonin in human cerebrospinal fluid in daytime; Its origin and variation with age. Life Sci. 1979, 25, 929–936. [Google Scholar]
- Vijayalaxmi; Thomas, C.R., Jr.; Reiter, R.J.; Herman, T.S. Melatonin: From basic research to cancer treatment clinics. J. Clin. Oncol. 2002, 20, 2575–2601. [Google Scholar]
- Wyatt, J.K.; Ritz-De Cecco, A.; Czeisler, C.A.; Dijk, D.J. Circadian temperature and melatonin rhythms, sleep, and neurobehavioral function in humans living on a 20-h day. Am. J. Physiol. 1999, 277, R1152–R1163. [Google Scholar]
- Dijk, D.J.; Shanahan, T.L.; Duffy, J.F.; Ronda, J.M.; Czeisler, C.A. Variation of electroencephalographic activity during non-rapid eye movement and rapid eye movement sleep with phase of circadian melatonin rhythm in humans. J. Physiol. 1997, 505, 851–858. [Google Scholar]
- Kudielka, B.M.; Buchtal, J.; Uhde, A.; Wust, S. Circadian cortisol profiles and psychological self-reports in shift workers with and without recent change in the shift rotation system. Biol. Psychol. 2007, 74, 92–103. [Google Scholar]
- Hennig, J.; Kieferdorf, P.; Moritz, C.; Huwe, S.; Netter, P. Changes in cortisol secretion during shiftwork: Implications for tolerance to shiftwork? Ergonomics 1998, 41, 610–621. [Google Scholar]
- Putignano, P.; Dubini, A.; Toja, P.; Invitti, C.; Bonfanti, S.; Redaelli, G.; Zappulli, D.; Cavagnini, F. Salivary cortisol measurement in normal-weight, obese and anorexic women: Comparison with plasma cortisol. Eur. J. Endocrinol. 2001, 145, 165–171. [Google Scholar]
- Riemann, D.; Kloepfer, C.; Berger, M. Functional and structural brain alterations in insomnia: Implications for pathophysiology. Eur. J. Neurosci. 2009, 29, 1754–1760. [Google Scholar]
- Erren, T.C.; Morfeld, P.; Stork, J.; Knauth, P.; von Mulmann, M.J.; Breitstadt, R.; Muller, U.; Emmerich, M.; Piekarski, C. Shift work, chronodisruption and cancer?—The IARC 2007 challenge for research and prevention and 10 theses from the cologne colloquium 2008. Scand. J. Work Environ. Health 2009, 35, 74–79. [Google Scholar]
- Hennevin, E.; Maho, C.; Hars, B.; Dutrieux, G. Learning-induced plasticity in the medial geniculate nucleus is expressed during paradoxical sleep. Behav. Neurosci. 1993, 107, 1018–1030. [Google Scholar]
- Jouvet, M. Neurophysiology of the states of sleep. Physiol. Rev. 1967, 47, 117–177. [Google Scholar]
- Iber, C.A.-I.; Chesson, S.A.; Quan, S.F. The AASM Manual for the Scoring of Sleep and Associated Events: Rules, Terminology and Technical Specifications; American Academy of Sleep Medicine: Westchester, NY, USA, 2007. [Google Scholar]
- Greene, R.W.; Frank, M.G. Slow wave activity during sleep: Functional and therapeutic implications. Neuroscientist 2010, 16, 618–633. [Google Scholar]
- Rechtschaffen, A.; Kales, A. A Manual of Standardized Terminology, Techniques, and Scoring System for Sleep Stages of Human Subjects; US Department of Health, Education, and Welfare: Bethesda, MD, USA, 1968. [Google Scholar]
- Takahashi, Y.; Kipnis, D.M.; Daughaday, W.H. Growth hormone secretion during sleep. J. Clin. Invest. 1968, 47, 2079–2090. [Google Scholar]
- Power, A.E. Slow-wave sleep, acetylcholine, and memory consolidation. Proc. Natl. Acad. Sci. USA 2004, 101, 1795–1796. [Google Scholar]
- Aserinsky, E.; Kleitman, N. Regularly occurring periods of eye motility, and concomitant phenomena, during sleep. Science 1953, 118, 273–274. [Google Scholar]
- Rechtschaffen, A. Current perspectives on the function of sleep. Perspect. Biol. Med. 1998, 41, 359–390. [Google Scholar]
- Hobson, J.A.; McCarley, R.W. The brain as a dream state generator: An activation-synthesis hypothesis of the dream process. Am. J. Psychiatry 1977, 134, 1335–1348. [Google Scholar]
- Hirshkowitz, M.; Moore, C.A.; Hamilton, C.R., 3rd; Rando, K.C.; Karacan, I. Polysomnography of adults and elderly: Sleep architecture, respiration, and leg movement. J. Clin. Neurophysiol. 1992, 9, 56–62. [Google Scholar]
- Borbely, A.A.; Achermann, P. Sleep homeostasis and models of sleep regulation. J. Biol. Rhythms. 1999, 14, 557–568. [Google Scholar]
- Saper, C.B.; Scammell, T.E.; Lu, J. Hypothalamic regulation of sleep and circadian rhythms. Nature 2005, 437, 1257–1263. [Google Scholar]
- Reinoso-Suarez, F.; de Andres, I.; Rodrigo-Angulo, M.L.; Garzon, M. Brain structures and mechanisms involved in the generation of REM sleep. Sleep Med. Rev. 2001, 5, 63–77. [Google Scholar]
- Boivin, D.B.; Tremblay, G.M.; James, F.O. Working on atypical schedules. Sleep Med. 2007, 8, 578–589. [Google Scholar]
- Drake, C.L.; Roehrs, T.; Richardson, G.; Walsh, J.K.; Roth, T. Shift work sleep disorder: Prevalence and consequences beyond that of symptomatic day workers. Sleep 2004, 27, 1453–1462. [Google Scholar]
- Alhola, P.; Polo-Kantola, P. Sleep deprivation: Impact on cognitive performance. Neuropsychiatr. Dis. Treat. 2007, 3, 553–567. [Google Scholar]
- Akerstedt, T.; Wright, K.P. Sleep loss and fatigue in shift work and shift work disorder. Sleep Med. Clin. 2009, 4, 257–271. [Google Scholar]
- Scott, B.A.; Judge, T.A. Insomnia, emotions, and job satisfaction: A multilevel study. J. Manag. 2006, 32, 622–645. [Google Scholar]
- Summa, K.C.; Turek, F.W. The genetics of sleep: Insight from rodent models. Sleep Med. Clin. 2011, 6, 141–154. [Google Scholar]
- Cappuccio, F.P.; D'Elia, L.; Strazzullo, P.; Miller, M.A. Sleep duration and all-cause mortality: A systematic review and meta-analysis of prospective studies. Sleep 2010, 33, 585–592. [Google Scholar]
- Siegel, J.M. Clues to the functions of mammalian sleep. Nature 2005, 437, 1264–1271. [Google Scholar]
- Lange, T.; Dimitrov, S.; Born, J. Effects of sleep and circadian rhythm on the human immune system. Ann. N. Y. Acad. Sci. 2010, 1193, 48–59. [Google Scholar]
- Krauchi, K.; Deboer, T. The interrelationship between sleep regulation and thermoregulation. Front. Biosci. 2010, 15, 604–625. [Google Scholar]
- Legault, G.; Smith, C.T.; Beninger, R.J. Scopolamine during the paradoxical sleep window impairs radial arm maze learning in rats. Pharmacol. Biochem. Behav. 2004, 79, 715–721. [Google Scholar]
- Stickgold, R. Sleep-dependent memory consolidation. Nature 2005, 437, 1272–1278. [Google Scholar]
- Legault, G.; Delay, S.; Madore, A. Identification of a rapid eye movement sleep window for learning of the win-shift radial arm maze task for male sprague-dawley rats. J. Sleep Res. 2010, 19, 508–515. [Google Scholar]
- Tononi, G.; Cirelli, C. Sleep and synaptic homeostasis: A hypothesis. Brain Res. Bull. 2003, 62, 143–150. [Google Scholar]
- Berger, R.J.; Phillips, N.H. Energy conservation and sleep. Behav. Brain Res. 1995, 69, 65–73. [Google Scholar]
- Frank, M.G. Sleep and developmental plasticity not just for kids. Prog. Brain Res. 2011, 193, 221–232. [Google Scholar]
- Frank, M.G. The mystery of sleep function: Current perspectives and future directions. Rev. Neurosci. 2006, 17, 375–392. [Google Scholar]
- Killgore, W.D. Effects of sleep deprivation on cognition. Prog. Brain Res. 2010, 185, 105–129. [Google Scholar]
- Cain, S.W.; Silva, E.J.; Chang, A.M.; Ronda, J.M.; Duffy, J.F. One night of sleep deprivation affects reaction time, but not interference or facilitation in a stroop task. Brain Cogn. 2011, 76, 37–42. [Google Scholar]
- Greneche, J.; Krieger, J.; Bertrand, F.; Erhardt, C.; Maumy, M.; Tassi, P. Short-term memory performances during sustained wakefulness in patients with obstructive sleep apnea-hypopnea syndrome. Brain Cogn. 2011, 75, 39–50. [Google Scholar]
- Frey, D.J.; Badia, P.; Wright, K.P., Jr. Inter- and intra-individual variability in performance near the circadian nadir during sleep deprivation. J. Sleep Res. 2004, 13, 305–315. [Google Scholar]
- Harrison, Y.; Horne, J.A. One night of sleep loss impairs innovative thinking and flexible decision making. Organ. Behav. Hum. Decis. Process. 1999, 78, 128–145. [Google Scholar]
- Haavisto, M.L.; Porkka-Heiskanen, T.; Hublin, C.; Harma, M.; Mutanen, P.; Muller, K.; Virkkala, J.; Sallinen, M. Sleep restriction for the duration of a work week impairs multitasking performance. J. Sleep Res. 2010, 19, 444–454. [Google Scholar]
- Prilipko, O.; Huynh, N.; Schwartz, S.; Tantrakul, V.; Kim, J.H.; Peralta, A.R.; Kushida, C.; Paiva, T.; Guilleminault, C. Task positive and default mode networks during a parametric working memory task in obstructive sleep apnea patients and healthy controls. Sleep 2011, 34, A293–A301. [Google Scholar]
- Bedard, M.A.; Montplaisir, J.; Richer, F.; Rouleau, I.; Malo, J. Obstructive sleep apnea syndrome: Pathogenesis of neuropsychological deficits. J. Clin. Exp. Neuropsychol. 1991, 13, 950–964. [Google Scholar]
- Ferrie, J.E.; Shipley, M.J.; Akbaraly, T.N.; Marmot, M.G.; Kivimaki, M.; Singh-Manoux, A. Change in sleep duration and cognitive function: Findings from the whitehall II study. Sleep 2011, 34, 565–573. [Google Scholar]
- Van Dongen, H.P.; Maislin, G.; Mullington, J.M.; Dinges, D.F. The cumulative cost of additional wakefulness: Dose-response effects on neurobehavioral functions and sleep physiology from chronic sleep restriction and total sleep deprivation. Sleep 2003, 26, 117–126. [Google Scholar]
- Czeisler, C.A.; Weitzman, E.; Moore-Ede, M.C.; Zimmerman, J.C.; Knauer, R.S. Human sleep: Its duration and organization depend on its circadian phase. Science 1980, 210, 1264–1267. [Google Scholar]
- Williamson, A.; Lombardi, D.A.; Folkard, S.; Stutts, J.; Courtney, T.K.; Connor, J.L. The link between fatigue and safety. Accid. Anal. Prev. 2011, 43, 498–515. [Google Scholar]
- Mitler, M.M.; Carskadon, M.A.; Czeisler, C.A.; Dement, W.C.; Dinges, D.F.; Graeber, R.C. Catastrophes, sleep, and public policy: Consensus report. Sleep 1988, 11, 100–109. [Google Scholar]
- National Transport Safety Board. Grounding of the US Tankship Exxon Valdez on Bligh Reef, Prince William Sound near Valdez, Alaska, March 24, 1989; National Transportation Safety Board: Maritime Accident Report; Washington, DC, USA, 1990. [Google Scholar]
- Dinges, D.F. An overview of sleepiness and accidents. J. Sleep Res. 1995, 4, 4–14. [Google Scholar]
- Huang, W.; Ramsey, K.M.; Marcheva, B.; Bass, J. Circadian rhythms, sleep, and metabolism. J. Clin. Invest. 2011, 121, 2133–2141. [Google Scholar]
- Poe, G.R.; Walsh, C.M.; Bjorness, T.E. Cognitive neuroscience of sleep. Prog. Brain Res. 2010, 185, 1–19. [Google Scholar]
- Bixler, E. Sleep and society: An epidemiological perspective. Sleep Med. 2009, 10, S3–S6. [Google Scholar]
- Levy, P.; Bonsignore, M.R.; Eckel, J. Sleep, sleep-disordered breathing and metabolic consequences. Eur. Respir. J. 2009, 34, 243–260. [Google Scholar]
- Zirlik, S.; Hauck, T.; Fuchs, F.S.; Neurath, M.F.; Konturek, P.C.; Harsch, I.A. Leptin, obestatin and apelin levels in patients with obstructive sleep apnoea syndrome. Med. Sci. Monit. 2011, 17, CR159–CR164. [Google Scholar]
- Imeri, L.; Opp, M.R. How (and why) the immune system makes us sleep. Nat. Rev. Neurosci. 2009, 10, 199–210. [Google Scholar]
- Brown, M.K.; Naidoo, N. The upr and the anti-oxidant response: Relevance to sleep and sleep loss. Mol. Neurobiol. 2010, 42, 103–113. [Google Scholar]
- Patel, S.R.; Hu, F.B. Short sleep duration and weight gain: A systematic review. Obesity 2008, 16, 643–653. [Google Scholar]
- Harvey, A.G. Sleep and circadian functioning: Critical mechanisms in the mood disorders? Annu. Rev. Clin. Psychol. 2011, 7, 297–319. [Google Scholar]
- Haack, M.; Mullington, J.M. Sustained sleep restriction reduces emotional and physical well-being. Pain 2005, 119, 56–64. [Google Scholar]
- Brake, D.J.; Bates, G.P. Fatigue in industrial workers under thermal stress on extended shift lengths. Occup. Med. (Lond.) 2001, 51, 456–463. [Google Scholar]
- Raediker, B.; Janssen, D.; Schomann, C.; Nachreiner, F. Extended working hours and health. Chronobiol. Int. 2006, 23, 1305–1316. [Google Scholar]
- Dijk, D.J.; Duffy, J.F.; Czeisler, C.A. Circadian and sleep/wake dependent aspects of subjective alertness and cognitive performance. J. Sleep Res. 1992, 1, 112–117. [Google Scholar]
- Smith, M.E.; McEvoy, L.K.; Gevins, A. The impact of moderate sleep loss on neurophysiologic signals during working-memory task performance. Sleep 2002, 25, 784–794. [Google Scholar]
- Smith, C. Sleep states and memory processes in humans: Procedural versus declarative memory systems. Sleep Med. Rev. 2001, 5, 491–506. [Google Scholar]
- Yoo, S.S.; Hu, P.T.; Gujar, N.; Jolesz, F.A.; Walker, M.P. A deficit in the ability to form new human memories without sleep. Nat. Neurosci. 2007, 10, 385–392. [Google Scholar]
- Harrison, Y.; Horne, J.A. The impact of sleep deprivation on decision making: A review. J. Exp. Psychol. 2000, 6, 236–249. [Google Scholar]
- Wimmer, F.; Hoffmann, R.F.; Bonato, R.A.; Moffitt, A.R. The effects of sleep deprivation on divergent thinking and attention processes. J. Sleep Res. 1992, 1, 223–230. [Google Scholar]
- Belenky, G.; Wesensten, N.J.; Thorne, D.R.; Thomas, M.L.; Sing, H.C.; Redmond, D.P.; Russo, M.B.; Balkin, T.J. Patterns of performance degradation and restoration during sleep restriction and subsequent recovery: A sleep dose-response study. J. Sleep Res. 2003, 12, 1–12. [Google Scholar]
- Curcio, G.; Ferrara, M.; De Gennaro, L. Sleep loss, learning capacity and academic performance. Sleep Med. Rev. 2006, 10, 323–337. [Google Scholar]
- American Academy of Sleep Medicine. International Classification of Sleep Disorders, Revised: Diagnostic and Coding Manual; American Academy of Sleep Medicine: Chicago, IL, USA, 2001. [Google Scholar]
- Cajochen, C.; Khalsa, S.B.; Wyatt, J.K.; Czeisler, C.A.; Dijk, D.J. EEG and ocular correlates of circadian melatonin phase and human performance decrements during sleep loss. Am. J. Physiol. 1999, 277, R640–R649. [Google Scholar]
- Lockley, S.W.; Cronin, J.W.; Evans, E.E.; Cade, B.E.; Lee, C.J.; Landrigan, C.P.; Rothschild, J.M.; Katz, J.T.; Lilly, C.M.; Stone, P.H.; et al. Effect of reducing interns' weekly work hours on sleep and attentional failures. N. Engl. J. Med. 2004, 351, 1829–1837. [Google Scholar]
- Wilkinson, R.T.; Houghton, D. Field test of arousal: A portable reaction timer with data storage. Hum. Factors 1982, 24, 487–493. [Google Scholar]
- Bonnet, M.H.; Arand, D.L. Clinical effects of sleep fragmentation versus sleep deprivation. Sleep Med. Rev. 2003, 7, 297–310. [Google Scholar]
- Bonnet, M.H. The effect of sleep fragmentation on sleep and performance in younger and older subjects. Neurobiol. Aging 1989, 10, 21–25. [Google Scholar]
- Dang-Vu, T.T.; Schabus, M.; Desseilles, M.; Sterpenich, V.; Bonjean, M.; Maquet, P. Functional neuroimaging insights into the physiology of human sleep. Sleep 2010, 33, 1589–1603. [Google Scholar]
- Thomas, M.; Sing, H.; Belenky, G.; Holcomb, H.; Mayberg, H.; Dannals, R.; Wagner, H.; Thorne, D.; Popp, K.; Rowland, L.; et al. Neural basis of alertness and cognitive performance impairments during sleepiness. I. Effects of 24 h of sleep deprivation on waking human regional brain activity. J. Sleep Res. 2000, 9, 335–352. [Google Scholar]
- Goel, N.; Rao, H.; Durmer, J.S.; Dinges, D.F. Neurocognitive consequences of sleep deprivation. Semin. Neurol. 2009, 29, 320–339. [Google Scholar]
- Robbins, T.W. Dissociating executive functions of the prefrontal cortex. Philos. Trans. R. Soc. Lond. B Biol. Sci. 1996, 351, 1463–1470. [Google Scholar]
- Muzur, A.; Pace-Schott, E.F.; Hobson, J.A. The prefrontal cortex in sleep. Trends Cogn. Sci. 2002, 6, 475–481. [Google Scholar]
- Smith, S.J.; Keefe, F.J.; Caldwell, D.S.; Romano, J.; Baucom, D. Gender differences in patient-spouse interactions: A sequential analysis of behavioral interactions in patients having osteoarthritic knee pain. Pain 2004, 112, 183–187. [Google Scholar]
- Banks, S.; Van Dongen, H.P.; Maislin, G.; Dinges, D.F. Neurobehavioral dynamics following chronic sleep restriction: Dose-response effects of one night for recovery. Sleep 2010, 33, 1013–1026. [Google Scholar]
- Dawson, D.; Reid, K. Fatigue, alcohol and performance impairment. Nature 1997, 388. [Google Scholar] [CrossRef]
- Arnedt, J.T.; Owens, J.; Crouch, M.; Stahl, J.; Carskadon, M.A. Neurobehavioral performance of residents after heavy night call vs. after alcohol ingestion. J. Am. Med. Assoc. 2005, 294, 1025–1033. [Google Scholar]
- Scott, L.D.; Hwang, W.T.; Rogers, A.E.; Nysse, T.; Dean, G.E.; Dinges, D.F. The relationship between nurse work schedules, sleep duration, and drowsy driving. Sleep 2007, 30, 1801–1807. [Google Scholar]
- Hossain, J.L.; Reinish, L.W.; Heslegrave, R.J.; Hall, G.W.; Kayumov, L.; Chung, S.A.; Bhuiya, P.; Jovanovic, D.; Huterer, N.; Volkov, J.; et al. Subjective and objective evaluation of sleep and performance in daytime versus nighttime sleep in extended-hours shift-workers at an underground mine. J. Occup. Environ. Med. 2004, 46, 212–226. [Google Scholar]
- Dijk, D.J.; Czeisler, C.A. Contribution of the circadian pacemaker and the sleep homeostat to sleep propensity, sleep structure, electroencephalographic slow waves, and sleep spindle activity in humans. J. Neurosci. 1995, 15, 3526–3538. [Google Scholar]
- Knutsson, A.; Boggild, H. Shiftwork and cardiovascular disease: Review of disease mechanisms. Rev. Environ. Health 2000, 15, 359–372. [Google Scholar]
- St-Onge, M.P.; Roberts, A.L.; Chen, J.; Kelleman, M.; O'Keeffe, M.; Roychoudhury, A.; Jones, P.J. Short sleep duration increases energy intakes but does not change energy expenditure in normal-weight individuals. Am. J. Clin. Nutr. 2011, 94, 410–416. [Google Scholar]
- Boggild, H.; Knutsson, A. Shift work, risk factors and cardiovascular disease. Scand. J. Work Environ. Health 1999, 25, 85–99. [Google Scholar]
- Axelsson, J.; Kecklund, G.; Akerstedt, T.; Lowden, A. Effects of alternating 8- and 12-hour shifts on sleep, sleepiness, physical effort and performance. Scand. J. Work Environ. Health 1998, 24, 62–68. [Google Scholar]
- Ferguson, S.A.; Paech, G.M.; Dorrian, J.; Roach, G.D.; Jay, S.M. Performance on a simple response time task: Is sleep or work more important for miners? Appl. Ergon. 2011, 42, 210–213. [Google Scholar]
- Niu, S.F.; Chung, M.H.; Chen, C.H.; Hegney, D.; O'Brien, A.; Chou, K.R. The effect of shift rotation on employee cortisol profile, sleep quality, fatigue, and attention level: A systematic review. J. Nurs. Res. 2011, 19, 68–81. [Google Scholar]
- Yu, J.M.; Tseng, I.J.; Yuan, R.Y.; Sheu, J.J.; Liu, H.C.; Hu, C.J. Low sleep efficiency in patients with cognitive impairment. Acta Neurol. Taiwan 2009, 18, 91–97. [Google Scholar]
- Akerstedt, T.; Wright, K.P. Sleep loss and fatigue in shift work and shift work disorder. Sleep Med. Clin. 2009, 4, 257–271. [Google Scholar]
- Stevens, R.G. Working against our endogenous circadian clock: Breast cancer and electric lighting in the modern world. Mutat. Res. 2009, 680, 106–108. [Google Scholar]
- Szosland, D. Shift work and metabolic syndrome, diabetes mellitus and ischaemic heart disease. Int. J. Occup. Med. Environ. Health 2010, 23, 287–291. [Google Scholar]
- Mahoney, M.M. Shift work, jet lag, and female reproduction. Int. J. Endocrinol. 2010, 2010, 813764:1–813764:9. [Google Scholar]
- Foret, J.; Benoit, O. Sleep patterns of workers on rotating shifts. Electroencephalogr. Clin. Neurophysiol. 1974, 37, 377–344. [Google Scholar]
- Torsvall, L.; Akerstedt, T.; Gillander, K.; Knutsson, A. Sleep on the night shift: 24-hour EEG monitoring of spontaneous sleep/wake behavior. Psychophysiology 1989, 26, 352–358. [Google Scholar]
- Webb, W.B.; Agnew, H.W., Jr. Sleep: Effects of a restricted regime. Science 1965, 150, 1745–1747. [Google Scholar]
- Akerstedt, T.; Gillberg, M. A dose-response study of sleep loss and spontaneous sleep termination. Psychophysiology 1986, 23, 293–297. [Google Scholar]
- Fogel, S.M.; Smith, C.T. Learning-dependent changes in sleep spindles and stage 2 sleep. J. Sleep Res. 2006, 15, 250–255. [Google Scholar]
- Fogel, S.M.; Smith, C.T. The function of the sleep spindle: A physiological index of intelligence and a mechanism for sleep-dependent memory consolidation. Neurosci. Biobehav. R. 2011, 35, 1154–1165. [Google Scholar]
- Torsvall, L.; Akerstedt, T. Sleepiness on the job: Continuously measured EEG changes in train drivers. Electroencephalogr. Clin. Neurophysiol. 1987, 66, 502–511. [Google Scholar]
- Kecklund, G.; Akerstedt, T. Sleepiness in long distance truck driving: An ambulatory EEG study of night driving. Ergonomics 1993, 36, 1007–1017. [Google Scholar]
- Rosekind, M.R.; Graeber, R.C.; Dinges, D.F.; Connel, L.J.; Rountree, M.S.; Gillen, K. Crew Factors in Flight Operations IX: Effects of Planned Cockpit Rest on Crew Performance and Alertness in Longhaul Operations; NASA Ames Research Center: Moffett Field, CA, USA, 1995. [Google Scholar]
- Landrigan, C.P.; Rothschild, J.M.; Cronin, J.W.; Kaushal, R.; Burdick, E.; Katz, J.T.; Lilly, C.M.; Stone, P.H.; Lockley, S.W.; Bates, D.W.; et al. Effect of reducing interns' work hours on serious medical errors in intensive care units. N. Engl. J. Med. 2004, 351, 1838–1848. [Google Scholar]
- Gillberg, M.; Kecklund, G.; Akerstedt, T. Sleepiness and performance of professional drivers in a truck simulator—Comparisons between day and night driving. J. Sleep Res. 1996, 5, 12–15. [Google Scholar]
- Gillberg, M.; Kecklund, G.; Goransson, B.; Akerstedt, T. Operator performance and signs of sleepiness during day and night work in a simulated thermal power plant. Int. J. Ind. Ergonom. 2003, 31, 101–109. [Google Scholar]
- International Working Group on the Evaluation of Carcinogenic Risks to Humans. In Painting, Firefighting, and Shiftwork; IARC: Lyon, France, 2007.
- Nachreiner, F.; Akkermann, S.; Hanecke, K. Fatal accident risk as a function of hours into work. In Shiftwork in the 21st Century; Hornberger, S., Knauth, S., Costa, G., Folkard, S., Eds.; Peter Lang: Frankfurt, Germany, 2000; pp. 19–24. [Google Scholar]
- Roach, G.D.; Reid, K.J.; Dawson, D. The amount of sleep obtained by locomotive engineers: Effects of break duration and time of break onset. Occup. Environ. Med. 2003, 60, e17:1–e17:5. [Google Scholar]
- Folkard, S.; Tucker, P. Shift work, safety and productivity. Occup. Med. (Lond.) 2003, 53, 95–101. [Google Scholar]
- Smith, L.; Folkard, S.; Tucker, P.; Macdonald, I. Work shift duration: A review comparing eight hour and 12 hour shift systems. Occup. Environ. Med. 1998, 55, 217–229. [Google Scholar]
- Scott, L.D.; Rogers, A.E.; Hwang, W.T.; Zhang, Y. Effects of critical care nurses' work hours on vigilance and patients' safety. Am. J. Crit. Care 2006, 15, 30–37. [Google Scholar]
- Robb, G.; Sultana, S.; Ameratunga, S.; Jackson, R. A systematic review of epidemiological studies investigating risk factors for work-related road traffic crashes and injuries. Inj. Prev. 2008, 14, 51–58. [Google Scholar]
- Harris, W. Fatigue, Circadian Rhythm and Truck Accidents. In Vigilance; Mackie, R.R., Ed.; Plenum Press: New York, NY, USA, 1977; pp. 133–146. [Google Scholar]
- Hamelin, P. Lorry driver's time habits in work and their involvement in traffic accidents. Ergonomics 1987, 30, 1323–1333. [Google Scholar]
- Langlois, P.H.; Smolensky, M.H.; Hsi, B.P.; Weir, F.W. Temporal patterns of reported single-vehicle car and truck accidents in Texas, U.S.A. during 1980–1983. Chronobiol Int 1985, 2, 131–140. [Google Scholar]
- National Transportation and Safety Board. In Factors That Affect Fatigue in Heavy Truck Accidents; National Transportation Safety Board: Washington, DC, USA, 1995.
- Lockley, S.W.; Barger, L.K.; Ayas, N.T.; Rothschild, J.M.; Czeisler, C.A.; Landrigan, C.P. Effects of health care provider work hours and sleep deprivation on safety and performance. Jt. Comm. J. Qual. Patient Saf. 2007, 33, 7–18. [Google Scholar]
- Mountain, S.A.; Quon, B.S.; Dodek, P.; Sharpe, R.; Ayas, N.T. The impact of housestaff fatigue on occupational and patient safety. Lung 2007, 185, 203–209. [Google Scholar]
- Flegal, K.M.; Carroll, M.D.; Kuczmarski, R.J.; Johnson, C.L. Overweight and obesity in the United States: Prevalence and trends, 1960–1994. Int. J. Obes. Relat. Metab. Disord. 1998, 22, 39–47. [Google Scholar]
- Vitiello, M.V.; Larsen, L.H.; Moe, K.E. Age-related sleep change: Gender and estrogen effects on the subjective-objective sleep quality relationships of healthy, noncomplaining older men and women. J. Psychosom. Res. 2004, 56, 503–510. [Google Scholar]
- Dimsdale, J.E.; Norman, D.; DeJardin, D.; Wallace, M.S. The effect of opioids on sleep architecture. J. Clin. Sleep Med. 2007, 3, 33–36. [Google Scholar]
- Mayers, A.G.; Baldwin, D.S. Antidepressants and their effect on sleep. Hum. Psychopharmacol 2005, 20, 533–559. [Google Scholar]
- Jaehne, A.; Loessl, B.; Barkai, Z.; Riemann, D.; Hornyak, M. Effects of nicotine on sleep during consumption, withdrawal and replacement therapy. Sleep Med. Rev. 2009, 13, 363–377. [Google Scholar]
- Arnedt, J.T.; Rohsenow, D.J.; Almeida, A.B.; Hunt, S.K.; Gokhale, M.; Gottlieb, D.J.; Howland, J. Sleep following alcohol intoxication in healthy, young adults: Effects of sex and family history of alcoholism. Alcohol. Clin. Exp. Res. 2011, 35, 870–878. [Google Scholar]
- Reid, K.J.; Baron, K.G.; Lu, B.; Naylor, E.; Wolfe, L.; Zee, P.C. Aerobic exercise improves self-reported sleep and quality of life in older adults with insomnia. Sleep Med. 2010, 11, 934–940. [Google Scholar]
- Young, T.; Evans, L.; Finn, L.; Palta, M. Estimation of the clinically diagnosed proportion of sleep apnea syndrome in middle-aged men and women. Sleep 1997, 20, 705–706. [Google Scholar]
- Guilleminault, C.; Tilkian, A.; Dement, W.C. The sleep apnea syndromes. Annu. Rev. Med. 1976, 27, 465–484. [Google Scholar]
- Murray, B.J. Brain death by a thousand hypoxic cuts in sleep. Am. J. Resp. Crit. Care Med. 2007, 175, 528–529. [Google Scholar]
- Jackson, M.L.; Howard, M.E.; Barnes, M. Cognition and daytime functioning in sleep-related breathing disorders. Prog. Brain Res. 2011, 190, 53–68. [Google Scholar]
- Morris, R.G.; Garrud, P.; Rawlins, J.N.; O'Keefe, J. Place navigation impaired in rats with hippocampal lesions. Nature 1982, 297, 681–683. [Google Scholar]
- Morgado-Bernal, I. Learning and memory consolidation: Linking molecular and behavioral data. Neuroscience 2011, 176, 12–19. [Google Scholar]
- Macey, P.M.; Henderson, L.A.; Macey, K.E.; Alger, J.R.; Frysinger, R.C.; Woo, M.A.; Harper, R.K.; Yan-Go, F.L.; Harper, R.M. Brain morphology associated with obstructive sleep apnea. Am. J. Resp. Crit. Care Med. 2002, 166, 1382–1387. [Google Scholar]
- Minoguchi, K.; Yokoe, T.; Tazaki, T.; Minoguchi, H.; Oda, N.; Tanaka, A.; Yamamoto, M.; Ohta, S.; O'Donnell, C.P.; Adachi, M. Silent brain infarction and platelet activation in obstructive sleep apnea. Am. J. Resp. Crit. Care Med. 2007, 175, 612–617. [Google Scholar]
- Ramani, R.V. Underground Mine Ventilation. Available online: http://accessscience.com/content/Underground-mine-ventilation/YB990895 (accessed on 19 April 2011).
- McMorris, T.; Swain, J.; Smith, M.; Corbett, J.; Delves, S.; Sale, C.; Harris, R.C.; Potter, J. Heat stress, plasma concentrations of adrenaline, noradrenaline, 5-hydroxytryptamine and cortisol, mood state and cognitive performance. Int. J. Psychophysiol. 2006, 61, 204–215. [Google Scholar]
- Simmons, S.E.; Saxby, B.K.; McGlone, F.P.; Jones, D.A. The effect of passive heating and head cooling on perception, cardiovascular function and cognitive performance in the heat. Eur. J. Appl. Physiol. 2008, 104, 271–280. [Google Scholar]
- Pilcher, J.J.; Nadler, E.; Busch, C. Effects of hot and cold temperature exposure on performance: A meta-analytic review. Ergonomics 2002, 45, 682–698. [Google Scholar]
- Radakovic, S.S.; Maric, J.; Surbatovic, M.; Radjen, S.; Stefanova, E.; Stankovic, N.; Filipovic, N. Effects of acclimation on cognitive performance in soldiers during exertional heat stress. Mil. Med. 2007, 172, 133–136. [Google Scholar]
- Keller, S.M. Effects of extended work shifts and shift work on patient safety, productivity, and employee health. AAOHN J. 2009, 57, 497–502. [Google Scholar]
- Gagge, A.P.; Gonzalex, R.R. Mechanisms of heat exchange: Biophysics and physiology. In Handbook of Physiology, Section 4. Environmental Physiology; Fregly, M.J., Blatteis, C.M., Eds.; American Physiological Society: Bethesda, MD, USA, 1996; pp. 45–84. [Google Scholar]
- Jay, O.; Kenny, G.P. Heat exposure in the Canadian workplace. Am. J. Ind. Med. 2010, 53, 842–853. [Google Scholar]
- Smith, R.E. Circadian variations in human thermoregulatory responses. J. Appl. Physiol. 1969, 26, 554–560. [Google Scholar]
- Becker, B.; Heyman, D.; Cooper, M.; Lehren, A.W. No survivors found afer West Virginia mine disaster. N. Y. Times 2010, A1. [Google Scholar]
- Fotheringham, A. Clippings from the Westray file. Macleans 1992, 105, 15–64. [Google Scholar]
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Legault, G. Sleep and Heat Related Changes in the Cognitive Performance of Underground Miners: A Possible Health and Safety Concern. Minerals 2011, 1, 49-72. https://doi.org/10.3390/min1010049
Legault G. Sleep and Heat Related Changes in the Cognitive Performance of Underground Miners: A Possible Health and Safety Concern. Minerals. 2011; 1(1):49-72. https://doi.org/10.3390/min1010049
Chicago/Turabian StyleLegault, Glenn. 2011. "Sleep and Heat Related Changes in the Cognitive Performance of Underground Miners: A Possible Health and Safety Concern" Minerals 1, no. 1: 49-72. https://doi.org/10.3390/min1010049



