Dissecting the Entry Route of Saporin-based a-CD7 Immunotoxins in Human T-Cell Acute Lymphoblastic Leukaemia Cells
Abstract
:1. Introduction
2. Experimental
2.1. Materials and Methods
2.2. Saporin and SAPKDEL Purification
2.3. Fluorescent Labeling of Saporins and Ricin
2.4. Conjugation of Saporins to HB2 Antibody (Anti CD7)
2.5. Cell Culture and Cell Viability Experiments
2.6. Fluorescence and Confocal Microscopy
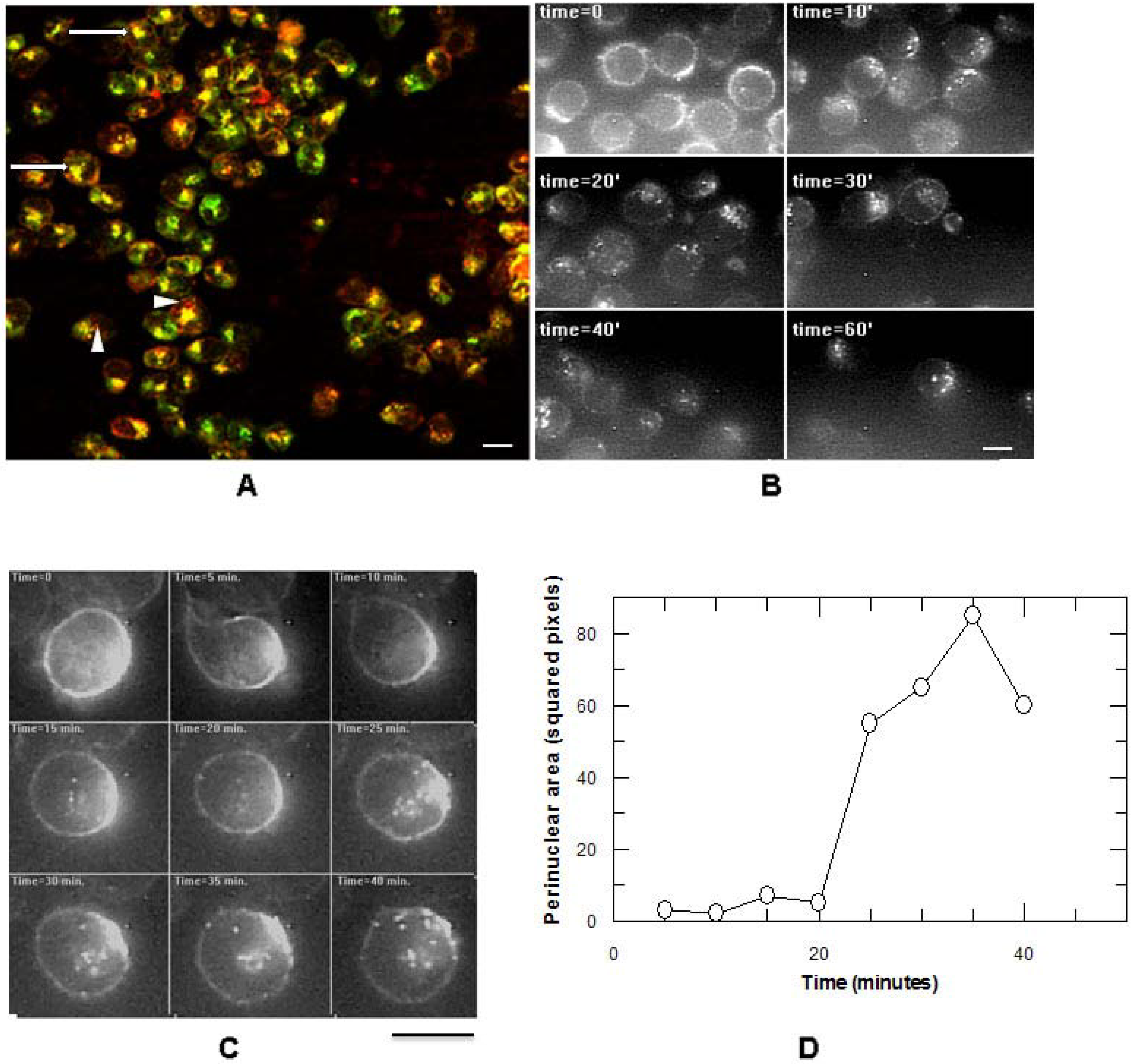
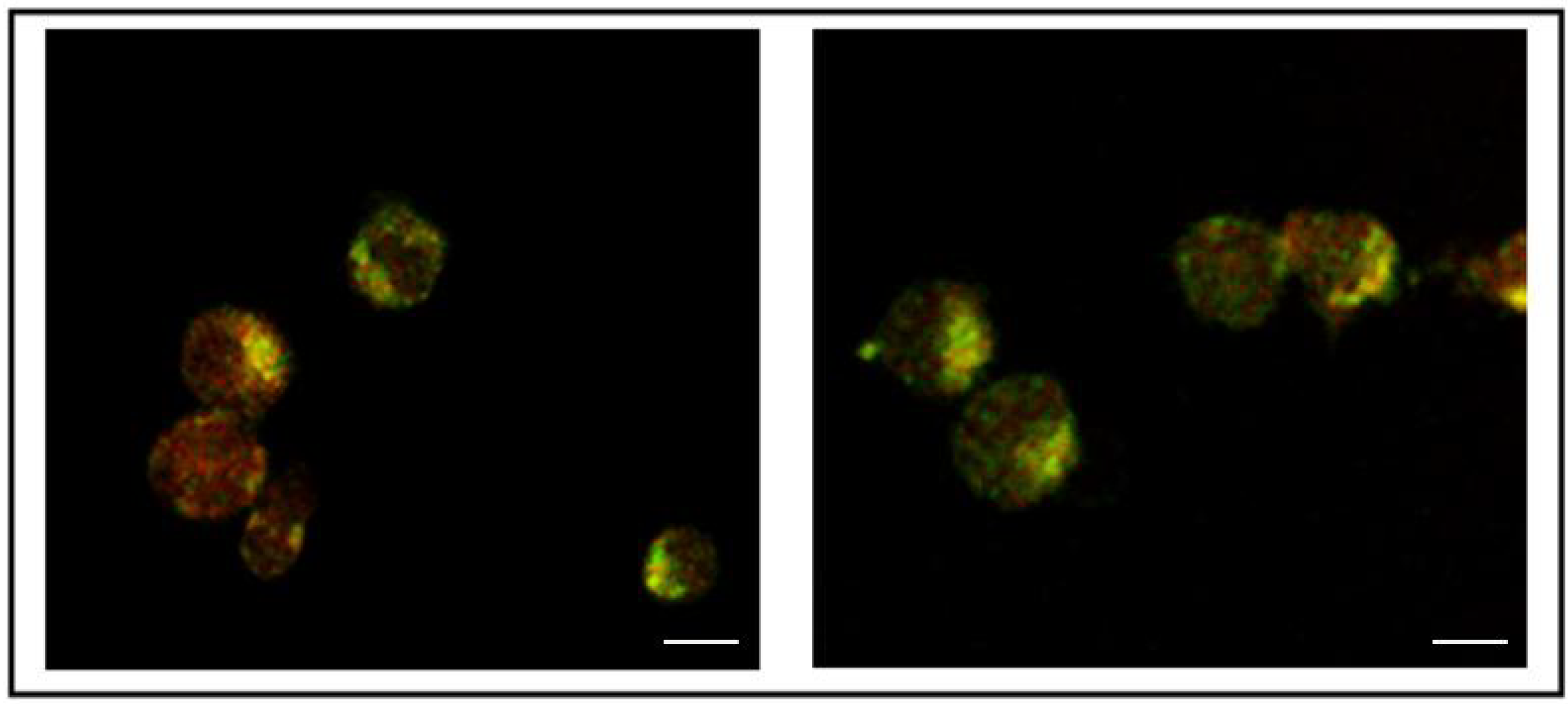
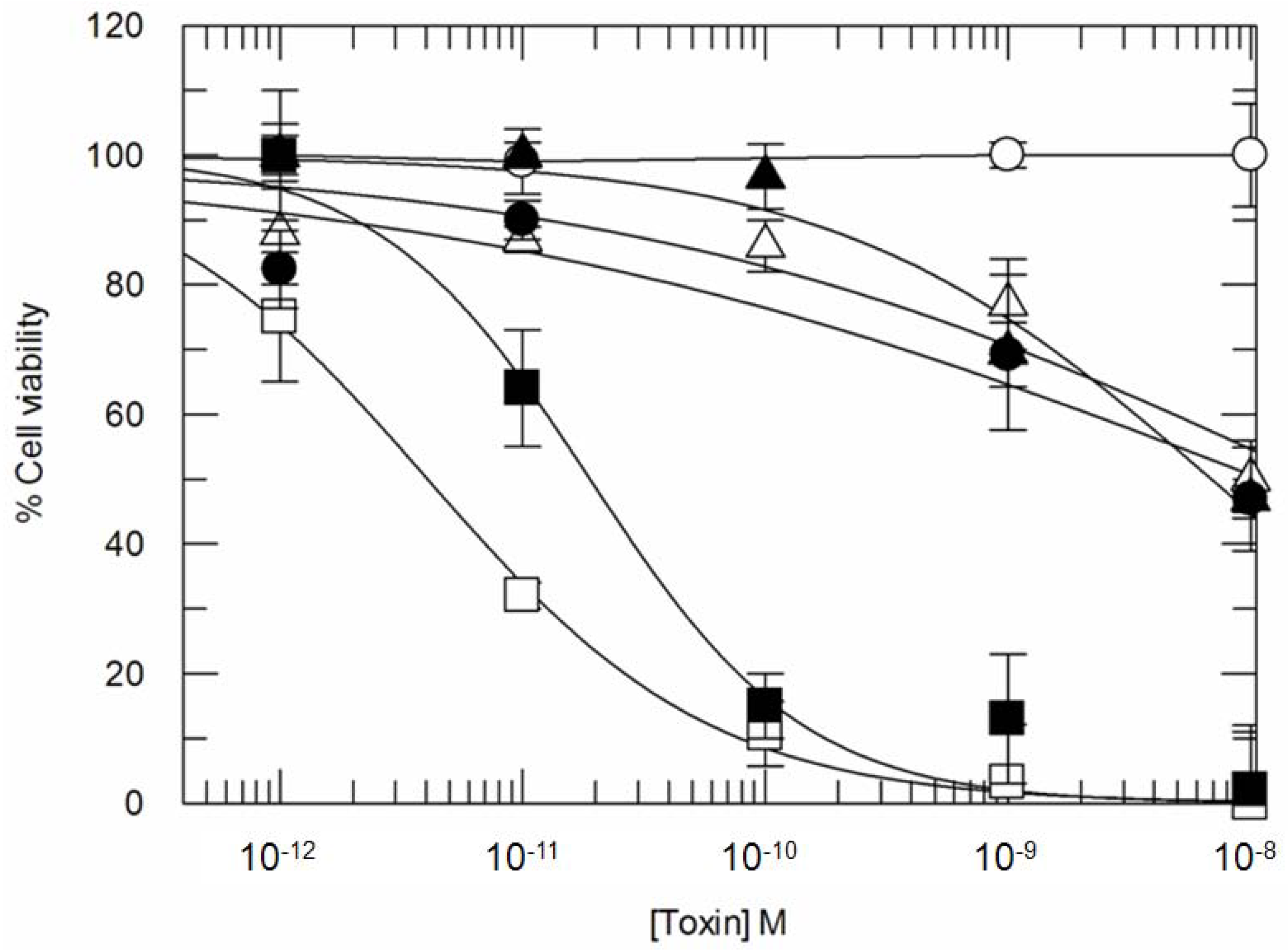
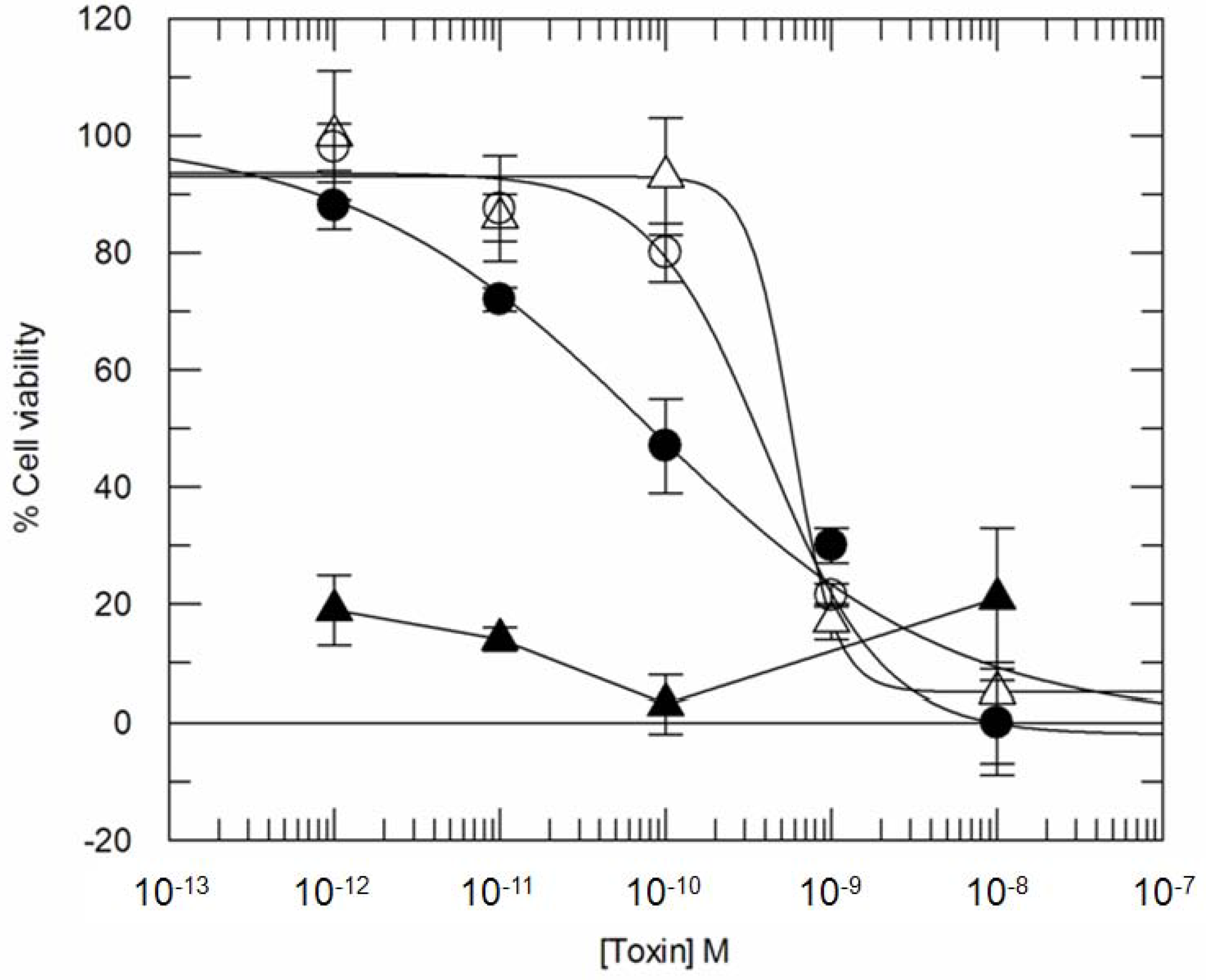
3. Results
3.2. Addition of KDEL to CD7-SAP does not Affect Its Retrograde Transport nor Its Cytotoxicity

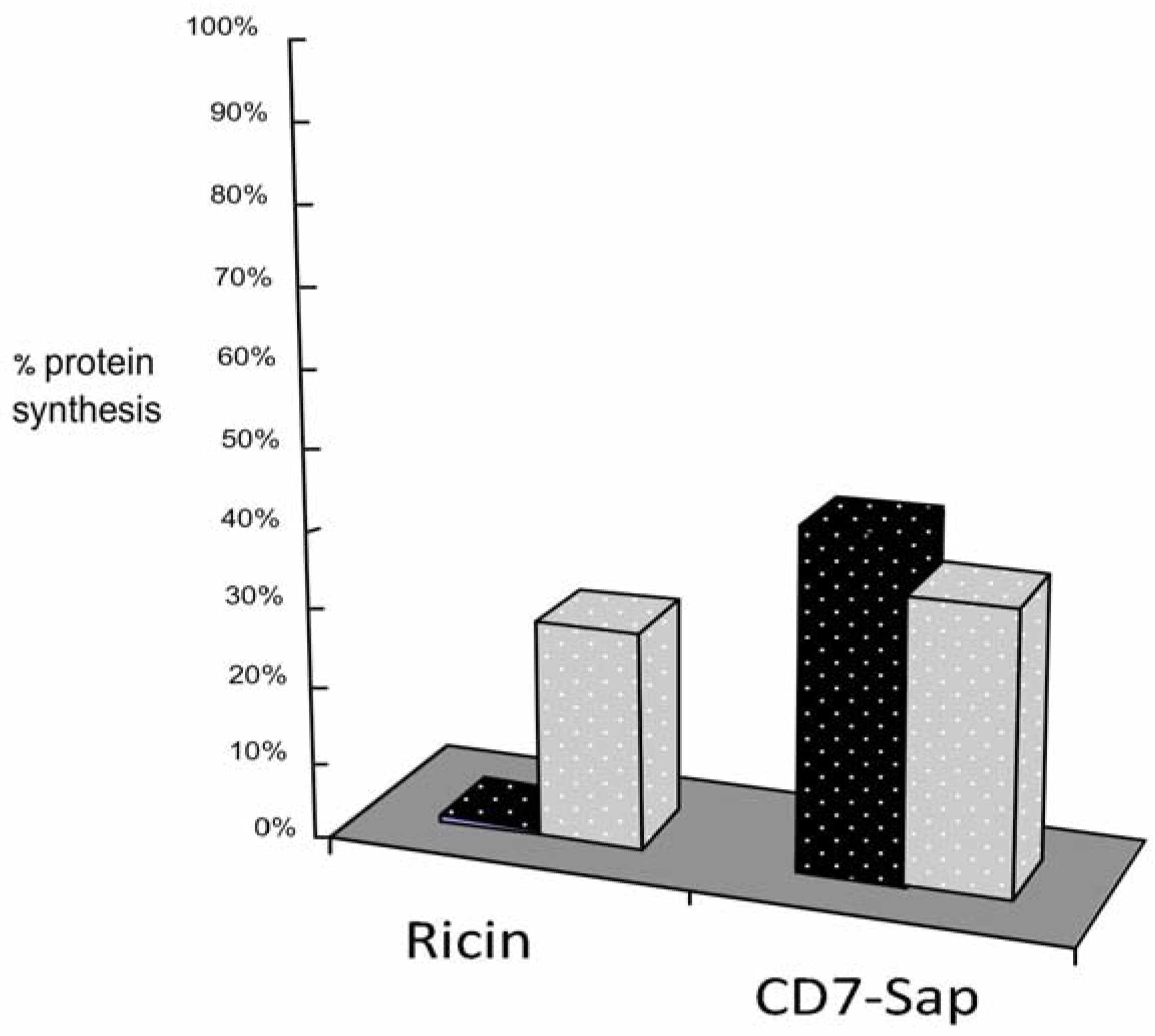
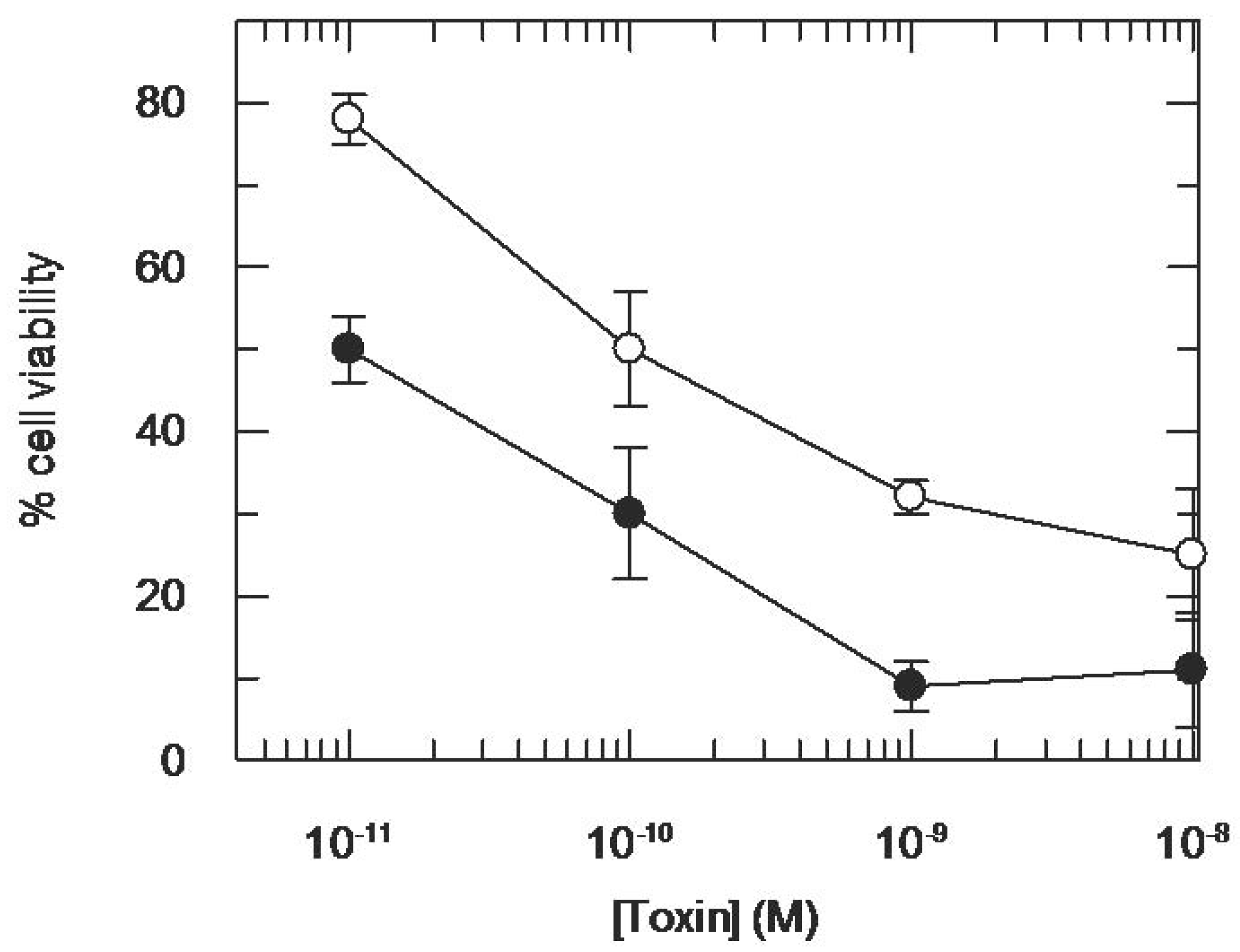
4. Discussion
5. Conclusions
Supplementary Materials
Supplementary Files
Supplementary File 1Acknowledgments
References
- Fabbrini, M.S.; Flavell, D.J.; Ippoliti, R. Plant protein toxins: Structure, function and biotechnological applications. In Bacterial Plant and Animal Toxins; Ascenzi, P., Polticelli, F., Visca, P., Eds.; Research Signpost: Trivandrum, India, 2003; pp. 69–99. [Google Scholar]
- Flavell, D.J. Saporin immunotoxins. Curr. Top. Microbiol. Immunol. 1998, 234, 57–61. [Google Scholar] [CrossRef]
- Kreitman, R.J.; Pastan, I. Immunotoxins in the treatment of hematologic malignancies. Curr. Drug Targets 2006, 7, 1301–1311. [Google Scholar] [CrossRef]
- Cox, C.V.; Martin, H.M.; Kearns, P.R.; Virgo, P.; Evely, R.S.; Blair, A. Characterization of a progenitor cell population in childhood T-cell acute lymphoblastic leukemia. Blood 2007, 109, 674–682. [Google Scholar] [CrossRef]
- Tiftik, N.; Bolaman, Z.; Batun, S.; Ayyildiz, O.; Isikdogan, A.; Kadikoylu, G.; Muftuoglu, E. The importance of CD7 and CD56 antigens in acute leukaemias. Int. J. Clin. Pract. 2004, 58, 149–152. [Google Scholar] [CrossRef]
- Martín-Henao, G.A.; Quiroga, R.; Sureda, A.; García, J. CD7 expression on CD34+ cells from chronic myeloid leukaemia in chronic phase. Am. J. Hematol. 1999, 61, 178–186. [Google Scholar] [CrossRef]
- Hutchinson, R.; Bostrom, B.C.; Sather, H.N.; Reaman, G.H. Clinical features and treatment outcome of childhood T-lineage acute lymphoblastic leukemia according to the apparent maturational stage of T-lineage leukemic blasts: A Children’s Cancer Group study. J. Clin. Oncol. 1997, 15, 2214–2221. [Google Scholar]
- Lyman, S.D.; Escobar, S.; Rousseau, A.M.; Armstrong, A.; Fanslow, W.C. Identification of CD7 as a cognate of the human K12 (SECTM1) protein. J. Biol. Chem. 2000, 275, 3431–3437. [Google Scholar]
- de Virgilio, M.; Lombardi, A.; Caliandro, R.; Fabbrini, M. Ribosome-inactivating proteins: From plant defense to tumor attack. Toxins 2010, 2, 2699–2737. [Google Scholar] [CrossRef]
- Hartley, M.R.; Lord, J.M. Cytotoxic ribosome-inactivating lectins from plants. Biochim. Biophys. Acta 2004, 1701, 1–14. [Google Scholar] [CrossRef]
- Stirpe, F. Ribosome-inactivating proteins. Toxicon 2004, 44, 371–383. [Google Scholar] [CrossRef]
- Stirpe, F.; Barbieri, L. Ribosome-inactivating proteins up to date. FEBS Lett. 1986, 195, 1–8. [Google Scholar]
- Lord, J.M.; Roberts, L.M.; Lencer, W.I. Entry of protein toxins into mammalian cells by crossing the endoplasmic reticulum membrane: Co-opting basic mechanisms of endoplasmic reticulum-associated degradation. Curr. Top. Microbiol. Immunol. 2005, 300, 149–168. [Google Scholar]
- Endo, Y.; Mitsui, K.; Motizuki, M.; Tsurugi, K. The mechanism of action of ricin and related toxic lectins on eukaryotic ribosomes. The site and the characteristics of the modification in 28 S ribosomal RNA caused by the toxins. J. Biol. Chem. 1987, 262, 5908–5912. [Google Scholar]
- Endo, Y.; Tsurugi, K. RNA N-glycosidase activity of ricin A-chain. Mechanism of action of the toxic lectin ricin on eukaryotic ribosomes. J. Biol. Chem. 1987, 262, 8128–8130. [Google Scholar]
- Gan, Y.H.; Peng, S.Q.; Liu, H.Y. Molecular mechanism of apoptosis induced by ricin in HeLa cells. Acta Pharmacol. Sin. 2000, 21, 243–248. [Google Scholar]
- Rao, P.V.; Jayaraj, R.; Bhaskar, A.S.; Kumar, O.; Bhattacharya, R.; Saxena, P.; Dash, P.K.; Vijayaraghavan, R. Mechanism of ricin-induced apoptosis in human cervical cancer cells. Biochem. Pharmacol. 2005, 69, 855–865. [Google Scholar] [CrossRef]
- Sikriwal, D.; Ghosh, P.; Batra, J.K. Ribosome inactivating protein Saporin induces apoptosis through mitochondrial cascade, independent of translation inhibition. Int. J. Biochem. Cell Biol. 2008, 40, 2880–2888. [Google Scholar] [CrossRef]
- Williams, J.M.; Lea, N.; Lord, J.M.; Roberts, L.M.; Milford, D.V.; Taylor, C.M. Comparison of ribosome-inactivating proteins in the induction of apoptosis. Toxicol. Lett. 1997, 91, 121–127. [Google Scholar] [CrossRef]
- Geden, S.E.; Gardner, R.A.; Fabbrini, M.S.; Ohashi, M.; Phanstiel, I.O.; Teter, K. Lipopolyamine treatment increases the efficacy of intoxication with Saporin and an anticancer Saporin conjugate. FEBS J. 2007, 274, 4825–4836. [Google Scholar] [CrossRef]
- Ippoliti, R.; Lendaro, E.; Benedetti, P.; Torrisi, M.; Belleudi, F.; Carpani, D.; Soria, M.; Fabbrini, M. Endocytosis of a chimera between human pro-urokinase and the plant toxin Saporin: An unusual internalization mechanism. FASEB J. 2000, 14, 1335–1344. [Google Scholar] [CrossRef]
- Vago, R.; Marsden, C.; Lord, J.; Ippoliti, R.; Flavell, D.; Flavell, S.; Ceriotti, A.; Fabbrini, M. Saporin and ricin A chain follow different intracellular routes to enter the cytosol of intoxicated cells. FEBS J. 2005, 272, 4983–4995. [Google Scholar] [CrossRef]
- Cimini, A.; Mei, S.; Benedetti, E.; Laurenti, G.; Koutris, I.; Cinque, B.; Cifone, M.G.; Galzio, R.; Pitari, G.; Leandro, L.D.; et al. Distinct cellular responses induced by Saporin and a trasferrin-Saporin conjugate in two different human glioblastoma cell lines. J. Cell. Physiol. 2012, 227, 939–51. [Google Scholar] [CrossRef]
- Barbieri, L.; Bolognesi, A.; Stirpe, F. Purification and conjugation of type 1 ribosome-inactivating proteins. Methods Mol. Biol. 2001, 166, 71–85. [Google Scholar]
- Ippoliti, R.; Lendaro, E.; D’Agostino, I.; Fiani, M.L.; Guidarini, D.; Vestri, S.; Benedetti, P.A.; Brunori, M. A chimeric Saporin-transferrin conjugate compared to ricin toxin: Role of the carrier in intracellular transport and toxicity. FASEB J. 1995, 9, 1220–1225. [Google Scholar]
- Lendaro, E.; Ippoliti, R.; Bellelli, A.; Brunori, M.; Evangelista, V.; Guidarini, D.; Benedetti, P.A. Intracellular dynamics of ricin followed by fluorescence microscopy on living cells reveals a rapid accumulation of the dimeric toxin in the Golgi apparatus. FEBS Lett. 1994, 344, 99–104. [Google Scholar] [CrossRef]
- Flavell, D.J.; Boehm, D.A.; Noss, A.; Flavell, S.U. Comparison of the potency and therapeutic efficacy of the anti-CD7 immunotoxin HB2-Saporin constructed with one or two Saporin moieties per immunotoxin molecule. Br. J. Cancer 1997, 75, 1035–1043. [Google Scholar] [CrossRef]
- Wu, Y.N.; Gadina, M.; Tao-Cheng, J.H.; Youle, R.J. Retinoic acid disrupts the Golgi apparatus and increases the cytosolic routing of specific protein toxins. J. Cell Biol. 1994, 125, 743–753. [Google Scholar] [CrossRef]
- Simpson, J.C.; Dascher, C.; Roberts, L.M.; Lord, J.M.; Balch, W.E. Ricin cytotoxicity is sensitive to recycling between the endoplasmic reticulum and the Golgi complex. J. Biol. Chem. 1995, 270, 20078–20083. [Google Scholar]
- Grimmer, S.; Iversen, T.G.; van Deurs, B.; Sandvig, K. Endosome to Golgi transport of ricin is regulated by cholesterol. Mol. Biol. Cell 2000, 11, 4205–4216. [Google Scholar]
- Braham, K.; Junqua, S.; Tursz, T.; Le Pecq, J.B.; Lipinski, M. Kinetic analysis of choriocarcinoma cell intoxication induced by ricin and ricin A chain immunotoxin. Cancer Res. 1988, 48, 806–811. [Google Scholar]
- Godal, A.; Fodstad, O.; Pihl, A. Kinetics of uptake and degradation of an abrin immunotoxin by melanoma cells and studies of the rates of cellular intoxication. Int. J. Cancer 1988, 42, 400–404. [Google Scholar] [CrossRef]
- Kim, Y.W. Kinetic analysis of cytotoxicity. Cancer. Treat. Res. 1988, 37, 405–415. [Google Scholar] [CrossRef]
- Hudson, T.H.; Grillo, F.G. Brefeldin-A enhancement of ricin A-chain immunotoxins and blockade of intact ricin, modeccin, and abrin. J. Biol. Chem. 1991, 266, 18586–18592. [Google Scholar]
- Wu, M. Enhancement of immunotoxin activity using chemical and biological reagents. Br. J. Cancer 1997, 75, 1347–1355. [Google Scholar] [CrossRef]
- Casellas, P.; Bourrie, B.J.; Gros, P.; Jansen, F.K. Kinetics of cytotoxicity induced by immunotoxins. Enhancement by lysosomotropic amines and carboxylic ionophores. J. Biol. Chem. 1984, 259, 9359–9364. [Google Scholar]
- Sung, C.; Wilson, D.; Youle, R.J. Comparison of protein synthesis inhibition kinetics and cell killing induced by immunotoxins. J. Biol. Chem. 1991, 266, 14159–14162. [Google Scholar]
- Sandvig, K.; van Deurs, B. Delivery into cells: Lessons learned from plant and bacterial toxins. Gene Ther. 2005, 12, 865–872. [Google Scholar] [CrossRef]
- Yoshida, T.; Chen, C.C.; Zhang, M.S.; Wu, H.C. Disruption of the Golgi apparatus by brefeldin A inhibits the cytotoxicity of ricin, modeccin, and Pseudomonas toxin. Exp. Cell. Res. 1991, 192, 389–395. [Google Scholar] [CrossRef]
- Wesche, J.; Rapak, A.; Olsnes, S. Dependence of ricin toxicity on translocation of the toxin A-chain from the endoplasmic reticulum to the cytosol. J. Biol. Chem. 1999, 274, 34443–34449. [Google Scholar]
- Wales, R.; Roberts, L.M.; Lord, J.M. Addition of an endoplasmic reticulum retrieval sequence to ricin A chain significantly increases its cytotoxicity to mammalian cells. J. Biol. Chem. 1993, 268, 23986–23990. [Google Scholar]
- Robineau, S.; Chabre, M.; Antonny, B. Binding site of Brefeldin A at the interface between the small G-protein ADP ribosylation factor 1 (ARF1) and the nucleotide exchange factor Sec7 domain. Proc. Natl. Acad. Sci. USA 2000, 97, 9913–9918. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Giansanti, F.; Giordani, V.; Vago, R.; Flavell, D.J.; Flavell, S.U.; Fabbrini, M.S.; Ippoliti, R. Dissecting the Entry Route of Saporin-based a-CD7 Immunotoxins in Human T-Cell Acute Lymphoblastic Leukaemia Cells. Antibodies 2013, 2, 50-65. https://doi.org/10.3390/antib2010050
Giansanti F, Giordani V, Vago R, Flavell DJ, Flavell SU, Fabbrini MS, Ippoliti R. Dissecting the Entry Route of Saporin-based a-CD7 Immunotoxins in Human T-Cell Acute Lymphoblastic Leukaemia Cells. Antibodies. 2013; 2(1):50-65. https://doi.org/10.3390/antib2010050
Chicago/Turabian StyleGiansanti, Francesco, Valeria Giordani, Riccardo Vago, David J. Flavell, Sopsamorn U. Flavell, Maria Serena Fabbrini, and Rodolfo Ippoliti. 2013. "Dissecting the Entry Route of Saporin-based a-CD7 Immunotoxins in Human T-Cell Acute Lymphoblastic Leukaemia Cells" Antibodies 2, no. 1: 50-65. https://doi.org/10.3390/antib2010050