Abstract
Elucidating the intracellular fate(s) of targeted toxins is of fundamental importance for their optimal use as anticancer drugs, since the biochemical targets of their enzymatic activity reside in the cell cytoplasm, as in the case of the plant ribosome inactivating proteins (RIP) saporin, ricin and of bacterial toxins. In this paper, we compared the cell surface binding and cytotoxic properties of the model RIP ricin to an immunotoxin constructed with a monoclonal antibody directed against the human T-cell marker CD7 covalently linked to saporin (CD7-SAP). Our results indicate that, despite the fact that internalization takes place via an apparently common entry route leading to the Golgi complex, surprisingly, the addition of an endoplasmic reticulum retrieval C-terminal signal (KDEL) to CD7-SAP does not potentiate its cytotoxicity. In addition, while ricin toxicity is clearly reduced by Brefeldin A under conditions where this fungal metabolite causes Golgi stack disruption, we paradoxically observed a potentiating effect by Brefeldin A on CD7-SAP cytotoxicity suggesting that this inhibitor interferes with retrograde route(s) other than the well established Trans-Golgi Network-ER retrograde route.
1. Introduction
Immunotoxins (ITx) are therapeutic agents composed of a targeting domain comprised of a monoclonal antibody or a growth-factor/ligand-binding domain linked to a toxin domain via a covalent bond or alternatively as recombinant fusion protein. The ITx targeting domain confers specificity against tumor cells expressing the target antigen(s). These therapeutic agents have previously been prepared using a variety of protein toxins [1,2,3] and different specific targeting domains. Several ITxs have undergone clinical trials to target various haematological malignancies in vivo. Some acute leukaemias, in particular T-ALL (T-cell Acute Lymphoblastic Leukaemia), over-express CD7 [4,5] also present on relapsed myeloid leukaemias and leukaemic progenitor cells from T-lineage leukaemia patients [6,7], but generally is not expressed on normal bone marrow stem cells. CD7 is a 40-kDa glycoprotein found primarily on T, NK, and pre-T cells whose putative functions in the immune system involve T and NK cell activation and/or adhesion. CD7 has, recently been identified as the cognate SECTM1 protein [8]. CD7 therefore represents a very good candidate target molecule in T-ALL for which ITxs can be constructed and specifically directed against neoplastic T-cells. In the recent past, a number of ITxs have been produced based on plant toxins such as Soapwort seed-extracted saporin [2]. Saporin from Saponaria officinalis belongs to the N-glycosidase family of plant ribosome inactivating proteins (RIPs) [1,9,10,11,12], that also includes the powerful ricin heterodimers from Ricinus communis seeds. The latter AB toxins or Type II toxins have a lectin-like cell-binding B domain that in the case of ricin mediates cell surface binding to exposed galactose residues on glyco-proteins/-lipids, resulting in binding to the majority of mammalian cells. Once internalized by a mammalian cell, ricin follows a Trans-Golgi to endoplasmic reticulum (ER) retrograde transport route and then mimics an unfolded polypeptide to exploit the ER-associated degradation (ERAD) pathway(s), dislocating misfolded polypeptides to the cytosol for degradation [9,13]. In this way, the A chain that possesses N-glycosidase catalytic activity gains access to the cytosol and acts on ribosomes by removing a single adenine residue (A4324 in rat) from a stem-loop universal structure in ribosomal 28S RNA [14,15]. This causes an irreversible block in protein synthesis followed by apoptotic cell death of the intoxicated cells [16,17,18,19]. The intracellular trafficking of type I RIPs (which may be viewed as an A toxic chain without a corresponding B subunit) remains largely elusive and ITx traffic may also be dependent on the particular targeting domain used to deliver the toxin moiety. Recently, we showedthat the intracellular transport route of saporin and of saporin-based chimaeras made with human urokinase domains bypasses the Golgi apparatus [20,21,22] while other experiments suggested an endosomal dislocation pathway for these toxins [20]. Furthermore, when analyzing entry of a transferrin-saporin conjugate in Glioblastoma multiforme tumour cells, we demonstrated that two different signaling /intracellular pathways could be used, one p53 dependent and the other independent from the presence of a fully active p53 [23], suggesting the possibility of an involvement of intracellular routes different from those described for chemical conjugates. In the study described here, we have characterized the binding, internalization and cytotoxic properties of an immunotoxin comprised of an anti CD7 monoclonal antibody coupled via a disulphide bond to the plant toxin saporin or to a recombinant KDEL-extended version of the same toxin We have compared their properties to those of the model toxin ricin in a view to engineering a fully recombinant molecule with an improved intracellular delivery route(s). By the use of an anti-CD7 antibody as the targeting moiety saporin is delivered to cell surface CD7, where binding is efficiently mediated via the antibody domain followed by receptor-mediated endocytosis. A main question to clarify here was if the dominant trafficking signal leads to passage through the Golgi complex, would the saporin-based ITx follow the same ricin intracellular route?
2. Experimental
2.1. Materials and Methods
All the reagents used were of analytical grade.
2.2. Saporin and SAPKDEL Purification
Saporin was purified from Saponaria officinalis seeds as previously described [24]. Recombinant SAPKDEL was purified from the pellet of a 1 L of culture of induced bacteria (BL21 SAPKDEL (pET11)) resuspended in 20 mM Phosphate buffer pH 6.5 supplemented with protease inhibitors (Pepstatin 1 μg/mL, Leupeptin 1 μg/mL, Aprotinin 10 μg/mL, PMSF 15 μg/mL) and DNase 1 μg/mL. The suspension was sonicated and then centrifuged at 7,000 g for 30 min at +4 °C, to eliminate cells debris. The supernatant was acidified at pH 5 with diluted acetic acid, to precipitate contaminant proteins with an acidic isoelectric point. This solution was centrifuged at 7,500 g for 60 min at +4 °C. This second supernatant was recovered, neutralized at pH 6.5 and dialyzed for 48–72 h, against 20 mM Phosphate buffer pH 6.5 and filtered through 0.22 μm filters before chromatographic separation.
The purified Saporin fractions were then analyzed by SDS-PAGE and HPLC chromatography on an ion exchange column (Resource S; Amersham Biosciences/GE Healthcare).
2.3. Fluorescent Labeling of Saporins and Ricin
Saporin (SAP) and SAPKDEL were labeled with tetrametylrhodamine isothiocyanate (TRITC) as previously described [25]. Briefly, the toxin was reacted with a 10-fold molar excess of TRITC at pH 7.0 in 10 mM phosphate buffer overnight at 4 °C. After the reaction the protein was purified by gel filtration to remove free TRITC and then further purified by ion exchange chromatography on Sepharose Fast Flow CM (Amersham Biosciences/GE Healthcare) and eluted with a linear NaCl gradient. Ricin was labeled with fluorescein isothiocyanate, as previously described [26]. The products of these labeling reactions are designated SAPT and RICINF, respectively.
2.4. Conjugation of Saporins to HB2 Antibody (Anti CD7)
Saporin (SAP), Saporin TRITC labeled (SAPT) and recombinant Saporin KDEL (SAPKDEL) were conjugated to the α-CD7 HB2 monoclonal antibodies, as previously described [27] with some modification by chemical cross-linking using the hetero-bifunctional reagent SPDP (Sulfosuccinimidyl 6-(3′-[2-pyridyldithio]-propionamido)hexanoate; PIERCE/Thermo Fisher Scientific) and the thiol introducing compound 2-Iminothiolane (Traut’s reagent; PIERCE/Thermo Fisher Scientific). The conjugated products (CD7-SAP, CD7-SAPT, CD7-SAPKDEL) were then purified by gel filtration on a G200 Sephadex column and by ion exchange chromatography on a Resource S column at pH 7.0 in 10 mM phosphate buffer, eluting with NaCl gradient.
2.5. Cell Culture and Cell Viability Experiments
The CD7+ HSB-2 cell line was established originally from the peripheral blood of a paediatric patient with T-cell acute lymphoblastic leukaemia and was obtained from the European Collection of Cell Cultures (ECACC). The identity of the HSB-2 cell line was authenticated using the Identifier Plus® DNA profiling system (Applied Biosciences, Carlsbad, CA, USA) employing 15 short tandem repeat loci and the profiles obtained compared to those held on the ECACC database. Living cultures of HSB-2 were maintained at 37 °C in a 5% CO2 atmosphere in the logarithmic phase of growth, by passage at regular intervals, in antibiotic-free RPMI 1640 medium containing 10% FCS and supplemented with 2 mM glutamine, 2 mM sodium pyruvate.
HSB-2 cells were routinely cultured under 5% CO2 in a humidified incubator, in RPMI-10% FCS. On the day of the experiment, cells were seeded in 96-well plates at a density of 1 × 104 cells/mL and used the day after.
Cell viability was monitored over a period of 24–72 h in the above-indicated medium. In some experiments, pre-treatment of cells with Brefeldin-A (SIGMA Chemical Co., MO, USA) at the desired concentrations and was obtained by diluting the drug in the culture medium starting from an ethanol stock (1 mg/mL) and incubating cells before (or as in some experiments at the same time) the exposure to the various toxins and ITxs. HSB-2 cells were incubated with increasing concentrations of ricin, CD7-SAP and CD7-SAPKDEL made up as serial dilutions in tissue culture medium starting from PBS stocks.
At the end of the incubation period, we measured cell viability adding WST-1 reagent (Roche), a mitochondrial-sensitive probe, directly to the culture medium and reading absorbance ratio 440/650 nm. The data are reported as the % residual cell viability. Results (the experiment shown is representative of different experiments) are the mean of triplicate measures +/−SD.
IC50 values have been fitted according to the equation Y = 100% / 1 + (X/IC50)s where Y is the % cell viability and X is concentration of toxin used, using the program Grafit (Erithacus software).
In short time experiments, we measured 3H-leucine incorporation in treated or untreated HSB-2 cells. Results are reported as residual protein synthesis expressed as a percentage of the untreated control cells.
2.6. Fluorescence and Confocal Microscopy
HSB-2 cells (1 × 105 cells/mL) were treated with RICINF or CD7-SAPT (300 nM concentration each) for 1 h at 4 °C in PBS. After extensive washing with cold PBS, the cells were then transferred to 37 °C and further incubated for 5–60 min depending on the experiment. Cells were then fixed with 4% Para-formaldehyde in PBS and cyto-centrifuged onto a glass microscope slide. In some experiments cells were collected by cyto-spin without fixation after the incubation at 4 °C and then transferred onto a temperature controlled (37 °C) plate of a Zeiss-Axiophot fluorescence microscope and observed for fluorescence. Confocal images were obtained with a Sarastro 2000 confocal microscope.
3. Results
3.2. Addition of KDEL to CD7-SAP does not Affect Its Retrograde Transport nor Its Cytotoxicity
To further explore the possibility that the Golgi complex might be an intracellular compartment for routing this saporin-based ITx to the cytoplasm, we prepared a conjugated ITx by coupling the CD7 HB2 monoclonal antibody to a recombinant saporin (SAPKDEL) that has been modified by appending at its C-terminus the endoplasmic reticulum retrieval sequence KDEL which is normally recognized by KDEL-receptors cycling between the Golgi complex and the ER.
SAPKDEL behavior has been previously well characterized in Vero cells [22], but due to the chemical conjugation procedure used for the construction of the ITxs [27] we reasoned that the KDEL sequence may become a possible target of SPDP the hetero-bifunctional cross-liker used for ITX construction. Therefore, after the conjugation procedure, we first analyzed the availability of the four aminoacid sequence by testing the ITx containing SAPKDEL with a monoclonal antibody directed against the KDEL sequence. As shown in Figure 2, the main fractions eluted at 150 mM NaCl during the ion-exchange chromatography (Figure 2A, circled peak) contained a single protein with an apparent molecular weight of approximately 180 kDa, that would correspond to the expected MW of the CD7-SAPKDEL product (Figure 2B, fraction/lane3) following its conjugation. The ITx was recognized by both the anti saporin and anti-KDEL antibodies (Figure 2B), thus confirming that the cross-linking procedure did not affect the exposure of the KDEL sequence.
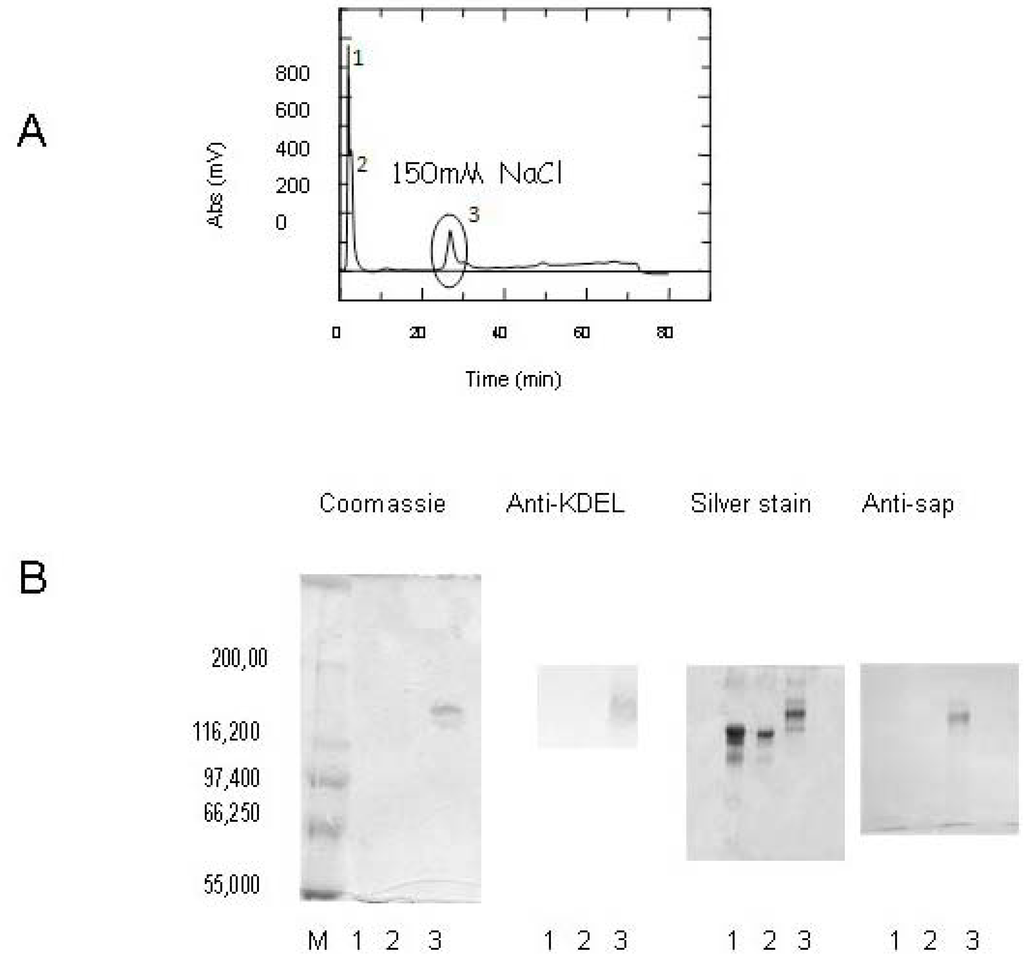
Figure 2.
(A) ion-exchange chromatographic purification of CD7-SAPKDEL conjugate. The 3rd fraction marked by the circle corresponds to a 150 mM NaCl concentration in the linear gradient used for elution. This fraction is reported as sample 3 in the SDS-PAGE and blot analysis. (B) SDS-PAGE analysis of fractions obtained from the ion-exchange chromatography. Samples 1 and 2 correspond to the first peaks appearing in the elution gradient (see Panel A). The samples were stained either by Coomassie-blue or by silver staining, or analysed by western blotting using a monoclonal anti-KDEL or a polyclonal anti-SAP serum.
We therefore used CD7-SAPKDEL (fraction 3) in confocal microscopy experiments, in similar conditions as described above for CD7-SAP to visualize its passage through the Golgi complex.
Indeed, CD7-SAPKDEL well co-localized inside HSB-2 cells with NBD-ceramide, an established fluorescent marker of membranes of the Trans Golgi Network (TGN), as shown in Figure 3 and as revealed by the use of the same anti-KDEL antibody followed by an anti-mouse TRITC secondary antibody.
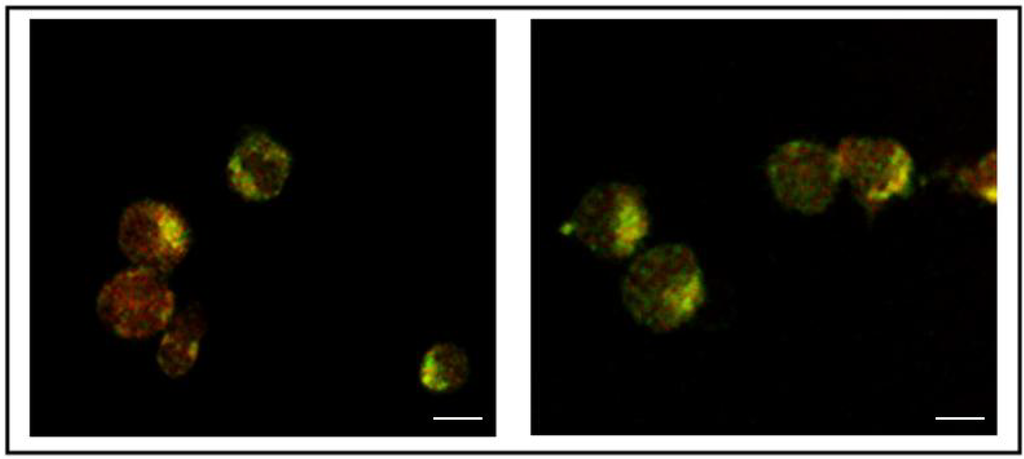
Figure 3.
Internalization of CD7-SAPKDEL ITx revealed by co-localization with C6-NBD-ceramide and immuno-staining with anti-KDEL and TRITC-anti mouse. Cells incubated with CD7-SAPKDEL were stained with the vital Golgi fluorescent marker NBD ceramide (Green staining) and after fixation with the anti-KDEL monoclonal antibody (Red staining). The two panels represent merged images of independent cells obtained by confocal microscopy. C6-NBD ceramide was added 10’ before the incubation together with the ITx (30’ time) followed by fixation of the cells and immune-localization. Please note that green or red fluorescence alone can be seen together with intensely fluorescent perinuclear structures which are due to yellow overlapping fluorescences likely representing the Golgi apparatus. The white bar represents 10 μm.
To verify whether HSB-2 cells are affected by both ricin and CD7-Saporin-based ITx treatment, cytotoxicity assays against these human T-ALL CD7+ cells were undertaken, for comparisons. All the toxins were tested in the absence or in the presence of Brefeldin A (BFA), a fungal metabolite known to inhibit ricin cytotoxicity by disrupting Golgi stacks. It should be stressed that in these experimental conditions, we had to greatly reduce the amounts of BFA normally employed to inhibit ricin cytotoxicity (5 μg/mL vs. 0.05 μg/mL) due to an intrinsic sensitivity of HSB-2 cells towards BFA.
Figure 4 shows the toxic effects of ricin and CD7-SAP ITxs after 24 h incubation. Ricin (open squares) was found to be extremely toxic at 24 h (IC50 estimated: 4 × 10−12 M) and, as expected, its toxicity was sensitive to Brefeldin A, with a corresponding 5-10-fold decrease in toxicity following BFA treatment of cells (IC50 increased to an estimated 1.9 × 10−11 M, black squares). The dose-related cytotoxicity of CD7-SAP (open circles) was not detected at the 24 h time point in this experiment. However, Brefeldin A treatment led to an apparent increase in CD7-SAP ITx cytotoxicity at 24 h, allowing for an approximate estimation of its IC50 to be made of between 10−8 and 10−9 M, under these experimental conditions.
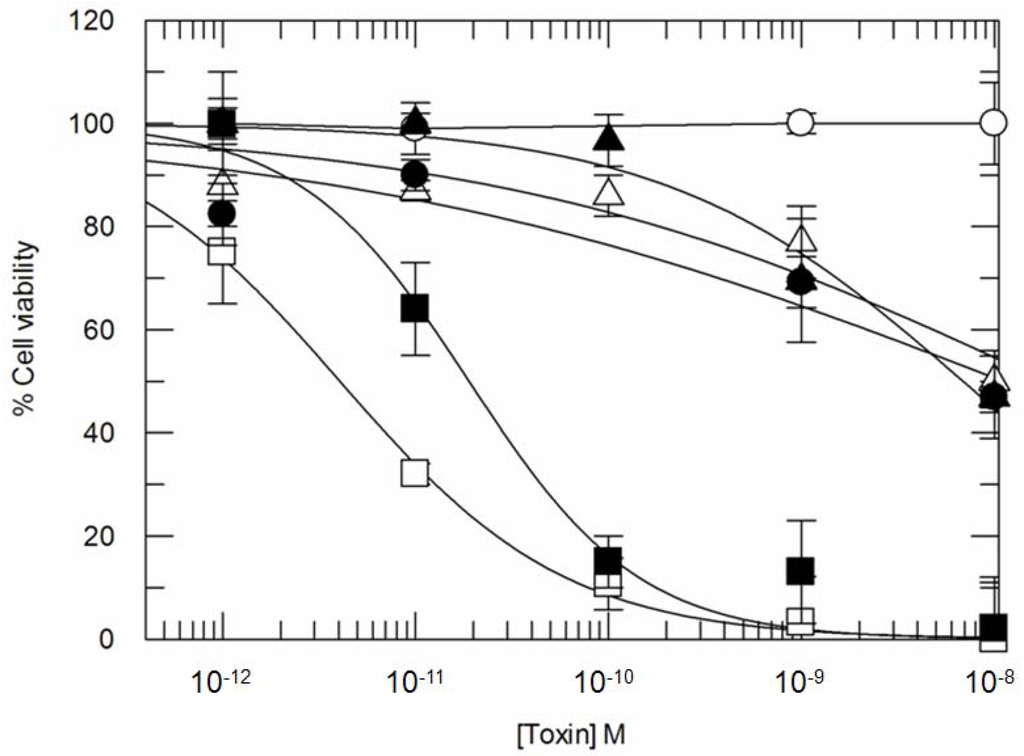
Figure 4.
Twenty-four hour Cytotoxicity of CD7-SAP and CD7-SAPKDEL compared to ricin in the presence or in the absence of Brefeldin A (BFA). Cells were incubated with BFA at 0.05 μg/mL 30 min before addition of each toxin and then further incubated for 24 h. Percent cell viability refers to control untreated cells. Samples have been performed in triplicate. Symbols are as follows: □ ricin, ■ ricin + BFA, ○ CD7-SAP, ● CD7-SAP + BFA, ∆ CD7-SAPKDEL, ▲ CD7-SAPKDEL + BFA.
We next examined whether the presence of a C-terminal KDEL would affect the potency of our ITx. As shown in Figure 4 (open triangles), CD7-SAPKDEL exerts again a relatively poor intoxication effect (estimated IC50 values comparable to CD7-SAP between 10−8 M and 10−9 M) when cells were exposed for only 24 h, while its cytotoxicity was apparently not significantly affected by the presence of Brefedin A (black triangles).
Since it is well established [31,32,33] that ITxs may require a long delay in time before exerting their cytotoxic activity, we then also tested their activity against HSB-2 cells after 72 h of incubation.
Under these experimental conditions, ricin at all concentrations killed the majority of the HSB-2 cells and therefore we couldn’t evaluate in parallel ricin cytotoxicity at 72 h.
At 72 h the viability of ITx-treated cells is clearly affected by CD7-SAP (Figure 5, open circles) in a dose-dependent manner and after 72 h of incubation the estimated IC50 value is approximately 2 × 10−9 M. BFA treatment, surprisingly, induces a potentiation of about one log (IC50 approximately 2 × 10−10 M) at 72 h for CD7-SAP ITx (Figure 5, closed circles). After 72 h of incubation (Figure 5, open triangles), potency of the CD7-SAPKDEL immunotoxin is roughly comparable to the cognate CD7-SAP with estimated IC50 values of about 2 × 10−9 M. Intriguingly, we now could also observe a substantial increase of cytotoxicity for CD7-SAPKDEL in the presence of Brefeldin A at 72 h (Figure 5, closed triangles), a phenomenon that could be observed before only for some ricin-A-chain based immunotoxins [34]. Importantly, as compared to free saporin under the same experimental conditions (data not shown, IC50 estimated to be 10−6 M), both CD7-SAP and CD7-SAPKDEL ITxs are at least 4 orders of magnitude more effective than the unconjugated toxin, as would be expected.
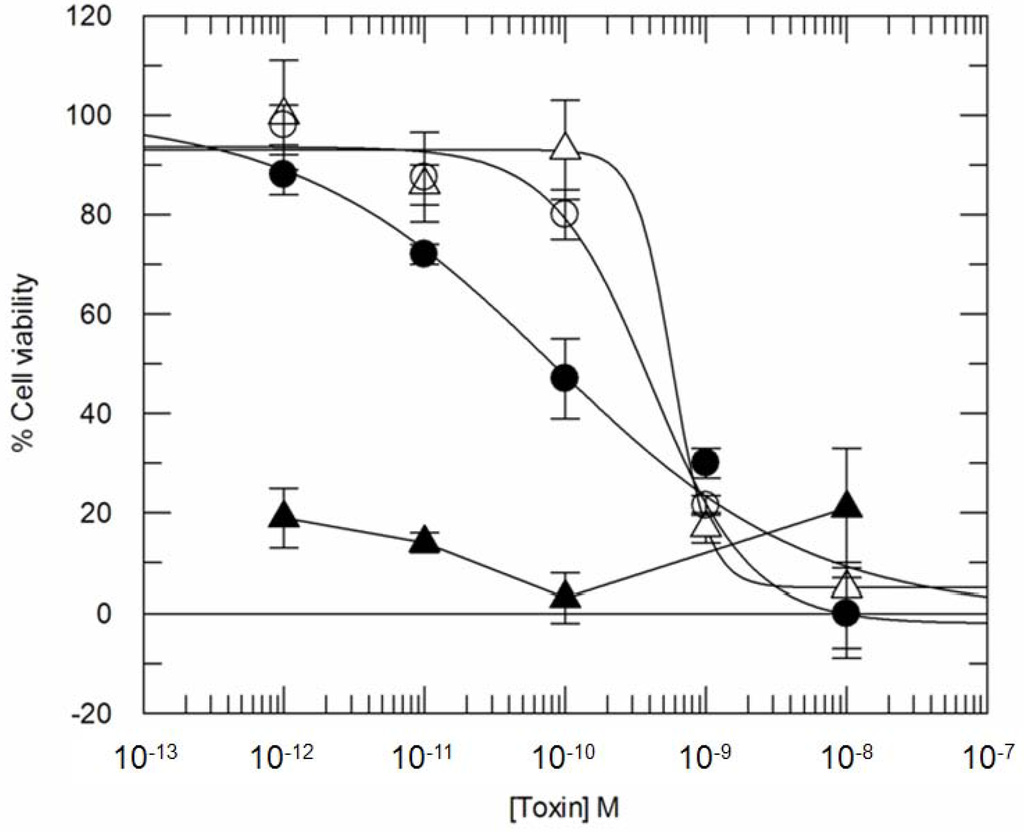
Figure 5.
Seventy-two hour Cytotoxicity of ITxs CD7-SAP and CD7-SAPKDEL compared to ricin either in the presence or in the absence of BFA. Cells were pre-incubated with BFA at 0.05 μg/mL 30 min before the addition of toxins and then further incubated for 72 h. Percent cell viability refers to control untreated cells. Samples have been performed in triplicate. Symbols refer to: ○ CD7-SAP, ● CD7-SAP + BFA, ∆ CD7-SAPKDEL, ▲ CD7-SAPKDEL + BFA
Comparisons of the ability to inhibit protein synthesis by the model toxin ricin and ITx were also performed on HSB-2 cells after shorter incubation periods, as shown in Figure 6. The toxin concentrations used in this experiment were, as stated above, different (10−10 M ricin and 10−8 M D7-SAP, respectively), to compensate for the differences in onset of cellular cytotoxicity by each [31,32,33], and also to take into account, our previous kinetic observations for each. Figure 6 refers to a protein synthesis inhibition assay conducted after 4 hours exposure of HSB-2 cells to CD7-SAP or ricin. Ricin is markedly more cytotoxic than CD7-SAP under these experimental conditions. As previously reported, ricin cytotoxicity can be inhibited [34] by Brefeldin A (at 5 μg/mL) while in the same conditions, conversely, CD7-SAP cytotoxicity is scarcely influenced by the same Brefeldin A concentration.
To further characterize intracellular compartments other than the Golgi, possibly involved in CD7-SAP-mediated toxicity, we tested whether CD7-SAP, if passing through endo-lysosomal compartments, could be sensitive to the presence of chloroquine, a known potentiating agent of ITxs (causing endosomal alkalinisation) [35]. Indeed, as shown in Figure 7 the addition of Chloroquine , did increase the potency of CD7-SAP by approximately one order of magnitude.
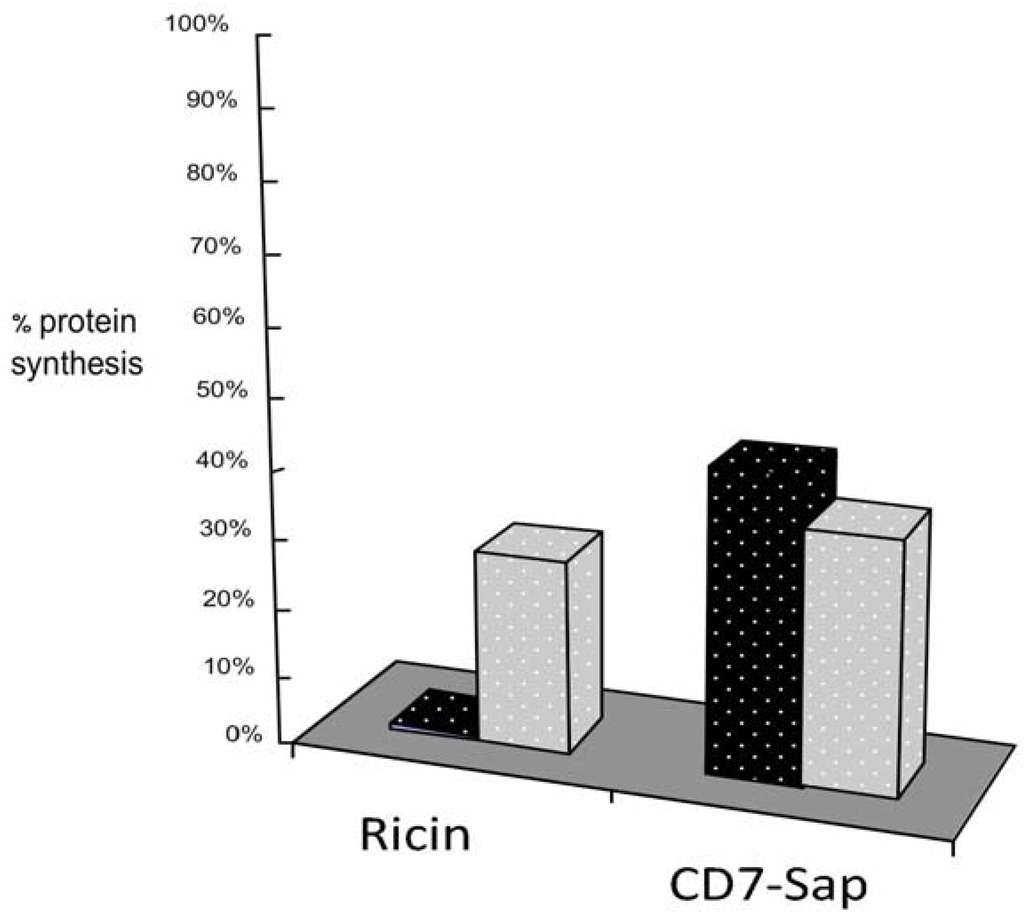
Figure 6.
Four-hour protein synthesis inhibition assay induced by CD7-SAP ITx or ricin in the presence (5 μg/mL) or in the absence of BFA. Cytotoxicities were measured after 4 h incubation with Ricin (10−10 M) and CD7-SAP (10−8 M) by the incorporation of 3H-leucine in the presence or in the absence of BFA. Data are reported as percent protein synthesis with respect to control untreated cells. Black histogram bars represent cells exposed to toxins in the absence of BFA, light grey histogram bars represent cells exposed to toxins following BFA treatment.
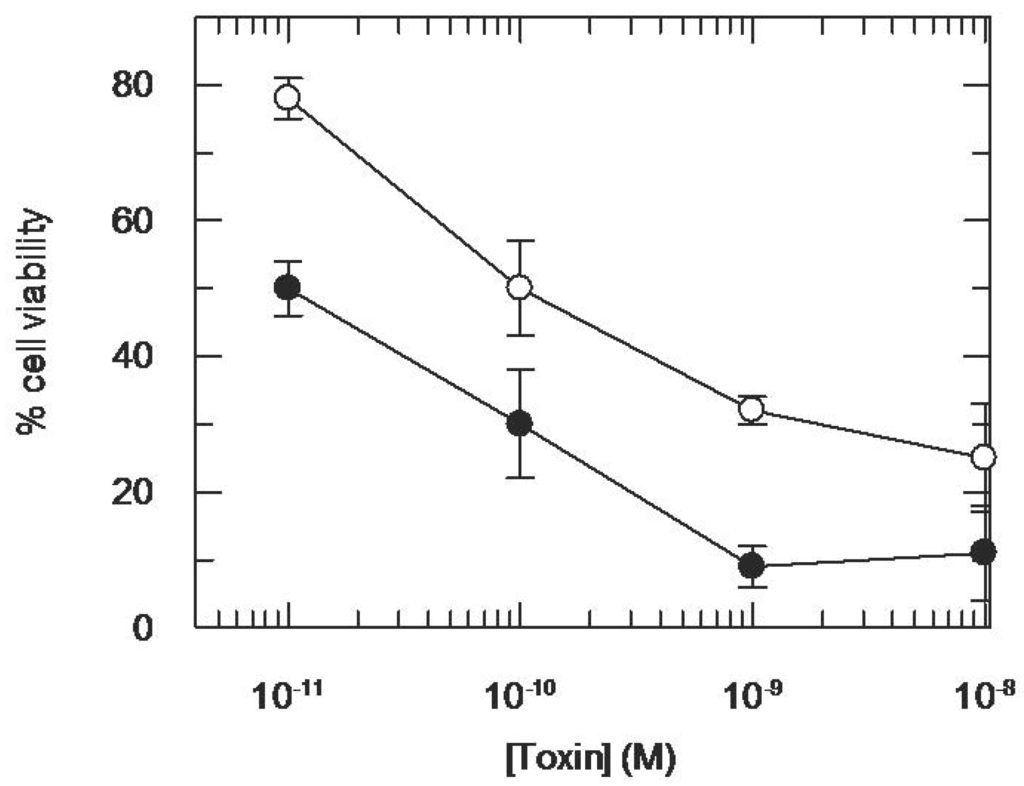
Figure 7.
Seventy-two hour Cytotoxicity of CD7-SAP for HSB-2 cells after incubation in the presence or in the absence of Chloroquine (Chl, 5 μM). HSB-2 cells were incubated with CD7-SAP for 72 h in the presence or in the absence of 5 μM chloroquine. Percent cellular viability refers to control untreated cells. Measures have been performed in quadruplicate cultures. Symbols are as follows: ○ CD7-SAP, ● CD7-SAP+ Chl.
4. Discussion
The primary goal of this work was to investigate the trafficking of saporin-based anti-CD7 ITxs in order to identify if appending a KDEL sequence to the C-terminus of saporin would improve the efficiency of cytosolic delivery to target cytosolic ribosomes [9]. Based on the kinetics of CD7-SAP internalization and its partial co-localization with ricin, we further investigated the effects of appending a KDEL sequence to the C-terminus of saporin by genetic engineering of recombinant saporin reasoning that if the ITx route passed through the Trans Golgi Network-to-ER retrogade pathway, we could predict an increase in ITx performance, due to its potential interaction with KDEL receptors, normally cycling between the Golgi and ER membranes which in the case of ricin results in translocation from the ER lumen to the cytosol via sec67 in the translocon. We have therefore also analyzed the cytotoxicity of two different immunotoxins, made by chemically cross-linking a monoclonal antibody directed against the human CD7 molecule expressed on the surface of malignant T-cells to either the native SO6 isoform of the type I ribosome inactivating protein saporin, or to an ITx constructed with recombinant saporin with a KDEL sequence appended to the C-terminus peptide extended (SAPKDEL).
When we further analyzed the kinetics of internalization of the CD7-SAP we found that it was similar to that of the model toxin ricin but that the saporin ITx took considerably longer to intoxicate HSB-2 cells than ricin ashas also been previously demonstrated for other immunotoxins [36,37]. This delay in the onset of toxicity has been attributed to a variety of reasons that include differences in the rate of cellular uptake, the intracellular delivery route(s) to cytoplasm or in the need for an activation step necessary for to the release of free toxin.
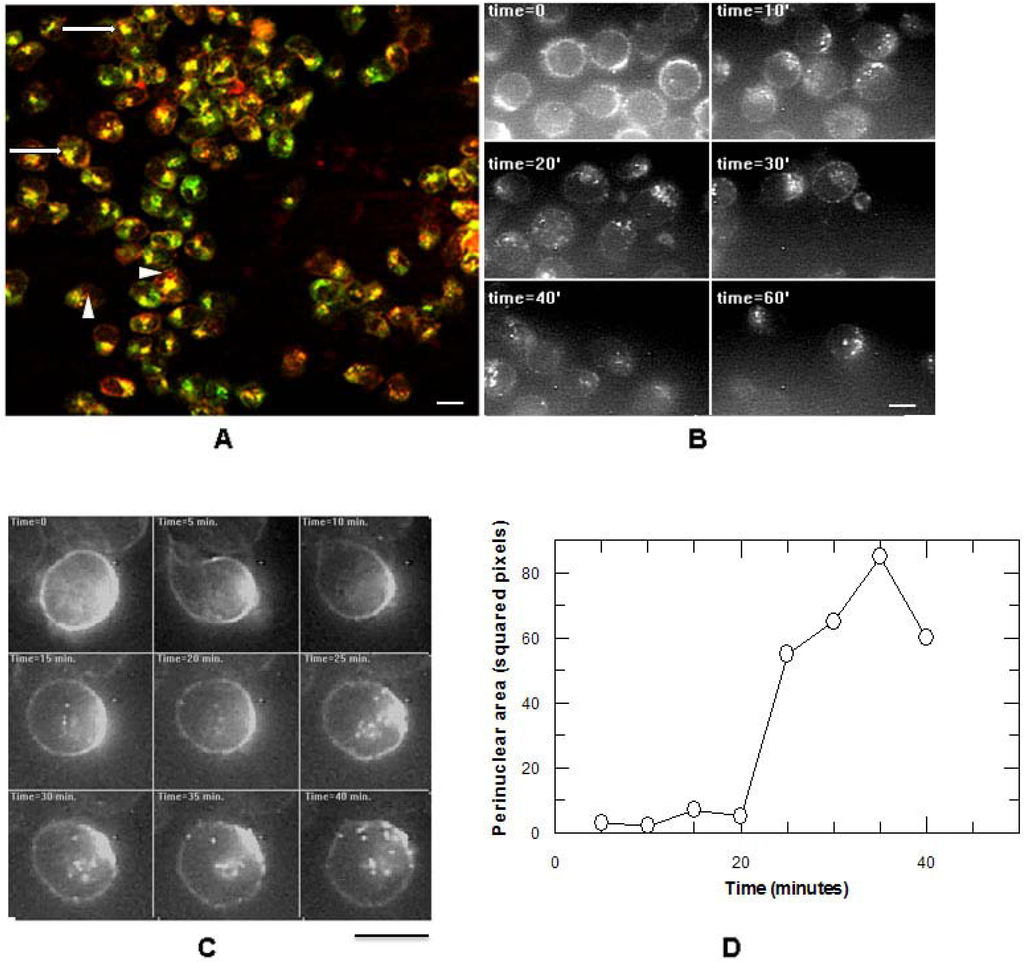
We have demonstrated here that CD7-SAP efficiently binds the cellular membrane of target HSB-2 cells and is internalized at a rate comparable to that of ricin. After40 min, we could visualize a massive accumulation of ITx within the perinuclear zone, and after 60 min CD7-SAP partially co-localized with ricin inside perinuclear structures that have previously been demonstrated to be the Golgi apparatus [26]. One major difference between ITx and ricin uptake is lag phase of about 20 min for ITx compared to only a few minutes in the case of ricinThis is likely because there are multiple molecular structures on the plasma membrane surface of target cells that display exposed galactose residues that therefore bind ricin B chain as opposed to the single CD7 molecular structure for the ITx. Ricin can therefore potentially utilize multiple endocytic paths to gain access to the cytoplasm [38] and can thus, can intoxicate cells within a much shorter period after internalization. The question we ask is might the intracellular fate differ between ricin and CD7-SAP, despite their passage through the Golgi?
The CD7-SAP ITx binds to only a single target molecule and its cytotoxic activity was maximal after 72 h incubation. Intoxication by CD7-SAP was also found to be sensitive to the action of chloroquine, a chemical agent that disrupts proton gradients across the endosomal membrane thereby increasing the pH within acidic vesicular compartments. Furthermore, in contrast to what is observed for ricin, the activity of CD7-SAP regardless of whether or not it is carrying the KDEL sequence, cannot be inhibited by treatment with Brefeldin-A, a fungal metabolite that disrupts the integrity of Golgi apparatus thus blocking the toxicity of ricin [39]. The BFA effect on ricin cytotoxicity has been directly connected to its retrograde transport pathway [40] and explains the potential role that KDEL receptors have in retrieving back to the ER, KDEL-ricin A chain, from where the unfolded A chain translocates to the cytosol via the Sec61 translocon [41].
Several lines of evidence suggest to us, that saporin and saporin-conjugates would normally be not able to reach the Golgi complex and thus, do not follow the same route as ricin to reach the cytosol [20,21,22,25]. In the case of the type I RIP saporin, endosomal /acidic compartments are in our view the most likely sites from where translocation of this toxin to the cytoplasm occurs. We were therefore surprised to observe for the first time in the present study a saporin ITx whose intracellular localization was at least partially overlapping with that of ricin, presumably at the level of the Golgi complex.
However, despite this novel observation, the cognate CD7-SAP bearing an appended KDEL sequence did not show any potentiation of its toxicity as compared to the parental ITx. Furthermore the cytotoxicity of CD7-SAPKDEL for target HSB-2 cells like the parental ITx was not affected by BFA.
We recently demonstrated that both an unconjugated KDEL saporin and ATF-SAPKDEL, a chimeric toxin made fusing the Amino-Terminal Fragment (ATF) of urokinase to saporin showed no increase in cytotoxicity towards Vero cells as compared to their wt versions [22]. The CD7-SAPKDEL ITx used in the present study, also behaves similarly, showing after 72 h of incubation a similar cytotoxicity to that of the parent CD7-SAP ITx on HSB-2 cells, both with IC50 values around 2 × 10−9 M. Interestingly, both ITxs paradoxically show an increase in their respective toxicities towards HSB-2 cells when the incubation is undertaken for 72 h in the presence of sub-optimal Brefeldin-A concentrations. This is particularly evident for the CD7-SAPKDEL ITx. One explanation is that under sub-optimal conditions, BFA may act as a sensitizing agent for target HSB-2 cells to saporin-based immunotoxins via a mechanism that is currently unknown but which may simply relate to an accumulation of toxin in endosomal compartments thus increasing the chances of translocation of toxin from here to the cytosol. The Golgi stacks or other subcellular membrane organelles in HSB-2 cells might be affected differently by this drug, possibly suggesting that under our experimental conditions a possible rerouting to the ER would occur which would not normally take place during saporin or saporin-conjugate intracellular trafficking in the absence of any drug treatment.
Activation of the small soluble GTPase Arf1 is mediated by guanine nucleotide exchange factors (GEFs) that convert Arf1 to its active, GTP-bound form. BFA at high concentrations inactivates freezes Arf1–GDPcomplexes, preventing Arf1 activation, blocking membrane trafficking from the ER and Golgi apparatus thus cause Golgi disassembly [42]. However, at lower concentrations BFA might also have pleiotropic effects on other GEfs and/or might also perturb other intracellular /recycling pathways. Nevertheless, the fact that at the low BFA concentrations employed in this study, CD7-SAPKDEL appears to be more affected by the BFA treatment suggesting that interaction with recycled KDEL-receptors could be taking place under these particular conditions.
5. Conclusions
In conclusion, sensitization of target HSB-2 cells to CD7-SAP by BFA very likely depends on an intracellular pathway that differs from the one described for ricin. This is despite the fact that ricin and saporin initially partially co-migrate to a perinuclear region identified as the Golgi apparatus. In fact, the much longer time lag observed for CD7-SAP intoxication of HSB-2 cells compared with the considerably shorter time for ricin would also indicate that pathway(s) with differing cytosolic escape efficiencies from the lumen of vesicular compartments are followed by each toxin. The addition of a KDEL retrieval sequence to CD7-SAPKDEL did not increase ITx cytotoxicity compared with the parental CD7-SAP ITx. This would again indicate that saporin-containing ITxs, may normally exploit other intracellular compartments (different from the ER) for their cytosolic dislocation. Further characterization of saporin interactions with intracellular membranes will be needed to shed further light on the nature of this pathway(s) which may suggest novel strategies to improve saporin-based ITx killing efficiency.
Supplementary Materials
Supplementary materials can be accessed at: http://www.mdpi.com/2073-4468/2/1/50/s1.
Supplementary Files
Supplementary File 1Acknowledgments
This work was partially supported by the Italian PRIN project (to R.I.), by Leukaemia Busters charity (to R.I, M.S.F, D.J.F., S.U.F.), by the Mi.SE (Ministero per lo Sviluppo Economico, Italia, Accordo MiSE/ICE/CRUI, to R.I.) and from the University of L’Aquila (to R.I.). We wish to thank Marco Colombatti, Università di Verona, for the kind gift of ricin.
References
- Fabbrini, M.S.; Flavell, D.J.; Ippoliti, R. Plant protein toxins: Structure, function and biotechnological applications. In Bacterial Plant and Animal Toxins; Ascenzi, P., Polticelli, F., Visca, P., Eds.; Research Signpost: Trivandrum, India, 2003; pp. 69–99. [Google Scholar]
- Flavell, D.J. Saporin immunotoxins. Curr. Top. Microbiol. Immunol. 1998, 234, 57–61. [Google Scholar] [CrossRef]
- Kreitman, R.J.; Pastan, I. Immunotoxins in the treatment of hematologic malignancies. Curr. Drug Targets 2006, 7, 1301–1311. [Google Scholar] [CrossRef]
- Cox, C.V.; Martin, H.M.; Kearns, P.R.; Virgo, P.; Evely, R.S.; Blair, A. Characterization of a progenitor cell population in childhood T-cell acute lymphoblastic leukemia. Blood 2007, 109, 674–682. [Google Scholar] [CrossRef]
- Tiftik, N.; Bolaman, Z.; Batun, S.; Ayyildiz, O.; Isikdogan, A.; Kadikoylu, G.; Muftuoglu, E. The importance of CD7 and CD56 antigens in acute leukaemias. Int. J. Clin. Pract. 2004, 58, 149–152. [Google Scholar] [CrossRef]
- Martín-Henao, G.A.; Quiroga, R.; Sureda, A.; García, J. CD7 expression on CD34+ cells from chronic myeloid leukaemia in chronic phase. Am. J. Hematol. 1999, 61, 178–186. [Google Scholar] [CrossRef]
- Hutchinson, R.; Bostrom, B.C.; Sather, H.N.; Reaman, G.H. Clinical features and treatment outcome of childhood T-lineage acute lymphoblastic leukemia according to the apparent maturational stage of T-lineage leukemic blasts: A Children’s Cancer Group study. J. Clin. Oncol. 1997, 15, 2214–2221. [Google Scholar]
- Lyman, S.D.; Escobar, S.; Rousseau, A.M.; Armstrong, A.; Fanslow, W.C. Identification of CD7 as a cognate of the human K12 (SECTM1) protein. J. Biol. Chem. 2000, 275, 3431–3437. [Google Scholar]
- de Virgilio, M.; Lombardi, A.; Caliandro, R.; Fabbrini, M. Ribosome-inactivating proteins: From plant defense to tumor attack. Toxins 2010, 2, 2699–2737. [Google Scholar] [CrossRef]
- Hartley, M.R.; Lord, J.M. Cytotoxic ribosome-inactivating lectins from plants. Biochim. Biophys. Acta 2004, 1701, 1–14. [Google Scholar] [CrossRef]
- Stirpe, F. Ribosome-inactivating proteins. Toxicon 2004, 44, 371–383. [Google Scholar] [CrossRef]
- Stirpe, F.; Barbieri, L. Ribosome-inactivating proteins up to date. FEBS Lett. 1986, 195, 1–8. [Google Scholar]
- Lord, J.M.; Roberts, L.M.; Lencer, W.I. Entry of protein toxins into mammalian cells by crossing the endoplasmic reticulum membrane: Co-opting basic mechanisms of endoplasmic reticulum-associated degradation. Curr. Top. Microbiol. Immunol. 2005, 300, 149–168. [Google Scholar]
- Endo, Y.; Mitsui, K.; Motizuki, M.; Tsurugi, K. The mechanism of action of ricin and related toxic lectins on eukaryotic ribosomes. The site and the characteristics of the modification in 28 S ribosomal RNA caused by the toxins. J. Biol. Chem. 1987, 262, 5908–5912. [Google Scholar]
- Endo, Y.; Tsurugi, K. RNA N-glycosidase activity of ricin A-chain. Mechanism of action of the toxic lectin ricin on eukaryotic ribosomes. J. Biol. Chem. 1987, 262, 8128–8130. [Google Scholar]
- Gan, Y.H.; Peng, S.Q.; Liu, H.Y. Molecular mechanism of apoptosis induced by ricin in HeLa cells. Acta Pharmacol. Sin. 2000, 21, 243–248. [Google Scholar]
- Rao, P.V.; Jayaraj, R.; Bhaskar, A.S.; Kumar, O.; Bhattacharya, R.; Saxena, P.; Dash, P.K.; Vijayaraghavan, R. Mechanism of ricin-induced apoptosis in human cervical cancer cells. Biochem. Pharmacol. 2005, 69, 855–865. [Google Scholar] [CrossRef]
- Sikriwal, D.; Ghosh, P.; Batra, J.K. Ribosome inactivating protein Saporin induces apoptosis through mitochondrial cascade, independent of translation inhibition. Int. J. Biochem. Cell Biol. 2008, 40, 2880–2888. [Google Scholar] [CrossRef]
- Williams, J.M.; Lea, N.; Lord, J.M.; Roberts, L.M.; Milford, D.V.; Taylor, C.M. Comparison of ribosome-inactivating proteins in the induction of apoptosis. Toxicol. Lett. 1997, 91, 121–127. [Google Scholar] [CrossRef]
- Geden, S.E.; Gardner, R.A.; Fabbrini, M.S.; Ohashi, M.; Phanstiel, I.O.; Teter, K. Lipopolyamine treatment increases the efficacy of intoxication with Saporin and an anticancer Saporin conjugate. FEBS J. 2007, 274, 4825–4836. [Google Scholar] [CrossRef]
- Ippoliti, R.; Lendaro, E.; Benedetti, P.; Torrisi, M.; Belleudi, F.; Carpani, D.; Soria, M.; Fabbrini, M. Endocytosis of a chimera between human pro-urokinase and the plant toxin Saporin: An unusual internalization mechanism. FASEB J. 2000, 14, 1335–1344. [Google Scholar] [CrossRef]
- Vago, R.; Marsden, C.; Lord, J.; Ippoliti, R.; Flavell, D.; Flavell, S.; Ceriotti, A.; Fabbrini, M. Saporin and ricin A chain follow different intracellular routes to enter the cytosol of intoxicated cells. FEBS J. 2005, 272, 4983–4995. [Google Scholar] [CrossRef]
- Cimini, A.; Mei, S.; Benedetti, E.; Laurenti, G.; Koutris, I.; Cinque, B.; Cifone, M.G.; Galzio, R.; Pitari, G.; Leandro, L.D.; et al. Distinct cellular responses induced by Saporin and a trasferrin-Saporin conjugate in two different human glioblastoma cell lines. J. Cell. Physiol. 2012, 227, 939–51. [Google Scholar] [CrossRef]
- Barbieri, L.; Bolognesi, A.; Stirpe, F. Purification and conjugation of type 1 ribosome-inactivating proteins. Methods Mol. Biol. 2001, 166, 71–85. [Google Scholar]
- Ippoliti, R.; Lendaro, E.; D’Agostino, I.; Fiani, M.L.; Guidarini, D.; Vestri, S.; Benedetti, P.A.; Brunori, M. A chimeric Saporin-transferrin conjugate compared to ricin toxin: Role of the carrier in intracellular transport and toxicity. FASEB J. 1995, 9, 1220–1225. [Google Scholar]
- Lendaro, E.; Ippoliti, R.; Bellelli, A.; Brunori, M.; Evangelista, V.; Guidarini, D.; Benedetti, P.A. Intracellular dynamics of ricin followed by fluorescence microscopy on living cells reveals a rapid accumulation of the dimeric toxin in the Golgi apparatus. FEBS Lett. 1994, 344, 99–104. [Google Scholar] [CrossRef]
- Flavell, D.J.; Boehm, D.A.; Noss, A.; Flavell, S.U. Comparison of the potency and therapeutic efficacy of the anti-CD7 immunotoxin HB2-Saporin constructed with one or two Saporin moieties per immunotoxin molecule. Br. J. Cancer 1997, 75, 1035–1043. [Google Scholar] [CrossRef]
- Wu, Y.N.; Gadina, M.; Tao-Cheng, J.H.; Youle, R.J. Retinoic acid disrupts the Golgi apparatus and increases the cytosolic routing of specific protein toxins. J. Cell Biol. 1994, 125, 743–753. [Google Scholar] [CrossRef]
- Simpson, J.C.; Dascher, C.; Roberts, L.M.; Lord, J.M.; Balch, W.E. Ricin cytotoxicity is sensitive to recycling between the endoplasmic reticulum and the Golgi complex. J. Biol. Chem. 1995, 270, 20078–20083. [Google Scholar]
- Grimmer, S.; Iversen, T.G.; van Deurs, B.; Sandvig, K. Endosome to Golgi transport of ricin is regulated by cholesterol. Mol. Biol. Cell 2000, 11, 4205–4216. [Google Scholar]
- Braham, K.; Junqua, S.; Tursz, T.; Le Pecq, J.B.; Lipinski, M. Kinetic analysis of choriocarcinoma cell intoxication induced by ricin and ricin A chain immunotoxin. Cancer Res. 1988, 48, 806–811. [Google Scholar]
- Godal, A.; Fodstad, O.; Pihl, A. Kinetics of uptake and degradation of an abrin immunotoxin by melanoma cells and studies of the rates of cellular intoxication. Int. J. Cancer 1988, 42, 400–404. [Google Scholar] [CrossRef]
- Kim, Y.W. Kinetic analysis of cytotoxicity. Cancer. Treat. Res. 1988, 37, 405–415. [Google Scholar] [CrossRef]
- Hudson, T.H.; Grillo, F.G. Brefeldin-A enhancement of ricin A-chain immunotoxins and blockade of intact ricin, modeccin, and abrin. J. Biol. Chem. 1991, 266, 18586–18592. [Google Scholar]
- Wu, M. Enhancement of immunotoxin activity using chemical and biological reagents. Br. J. Cancer 1997, 75, 1347–1355. [Google Scholar] [CrossRef]
- Casellas, P.; Bourrie, B.J.; Gros, P.; Jansen, F.K. Kinetics of cytotoxicity induced by immunotoxins. Enhancement by lysosomotropic amines and carboxylic ionophores. J. Biol. Chem. 1984, 259, 9359–9364. [Google Scholar]
- Sung, C.; Wilson, D.; Youle, R.J. Comparison of protein synthesis inhibition kinetics and cell killing induced by immunotoxins. J. Biol. Chem. 1991, 266, 14159–14162. [Google Scholar]
- Sandvig, K.; van Deurs, B. Delivery into cells: Lessons learned from plant and bacterial toxins. Gene Ther. 2005, 12, 865–872. [Google Scholar] [CrossRef]
- Yoshida, T.; Chen, C.C.; Zhang, M.S.; Wu, H.C. Disruption of the Golgi apparatus by brefeldin A inhibits the cytotoxicity of ricin, modeccin, and Pseudomonas toxin. Exp. Cell. Res. 1991, 192, 389–395. [Google Scholar] [CrossRef]
- Wesche, J.; Rapak, A.; Olsnes, S. Dependence of ricin toxicity on translocation of the toxin A-chain from the endoplasmic reticulum to the cytosol. J. Biol. Chem. 1999, 274, 34443–34449. [Google Scholar]
- Wales, R.; Roberts, L.M.; Lord, J.M. Addition of an endoplasmic reticulum retrieval sequence to ricin A chain significantly increases its cytotoxicity to mammalian cells. J. Biol. Chem. 1993, 268, 23986–23990. [Google Scholar]
- Robineau, S.; Chabre, M.; Antonny, B. Binding site of Brefeldin A at the interface between the small G-protein ADP ribosylation factor 1 (ARF1) and the nucleotide exchange factor Sec7 domain. Proc. Natl. Acad. Sci. USA 2000, 97, 9913–9918. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).