Current Paradigms of Tolerogenic Dendritic Cells and Clinical Implications for Systemic Lupus Erythematosus
Abstract
:1. Introduction
2. DC Subsets in Immune Tolerance
3. Phenotypic and Functional Signatures of Ex Vivo Generated tolDCs
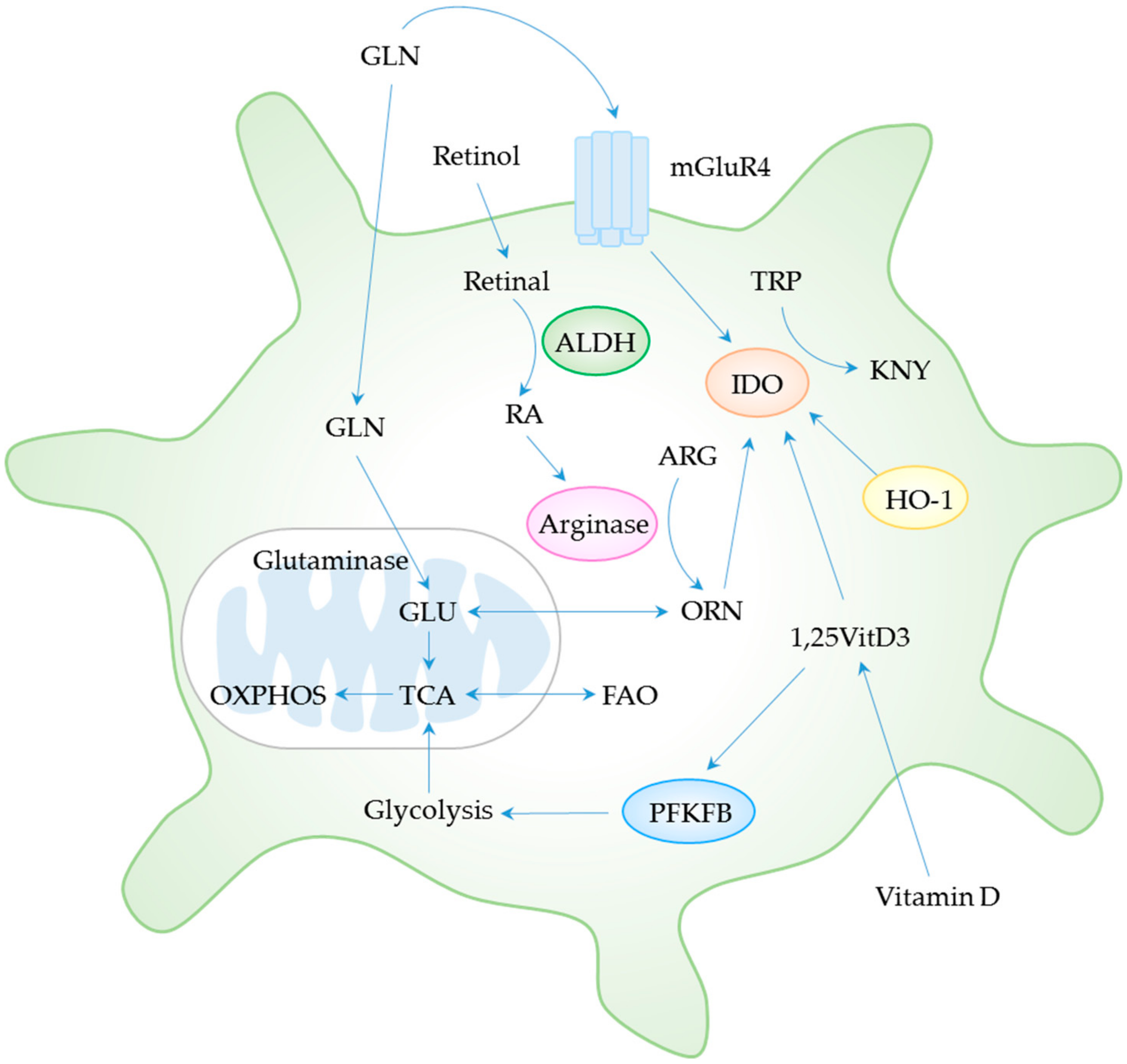
4. Metabolic Control of DC Tolerogenicity
4.1. Amino Acid Metabolism
4.2. Glycolysis and Fatty Acid Metabolism
4.3. Vitamin Metabolism
5. Clinical Implications of tolDCs for SLE
6. Conclusions and Perspectives
Author Contributions
Funding
Conflicts of Interest
References
- Merad, M.; Sathe, P.; Helft, J.; Miller, J.; Mortha, A. The dendritic cell lineage: Ontogeny and function of dendritic cells and their subsets in the steady state and the inflamed setting. Annu. Rev. Immunol. 2013, 31, 563–604. [Google Scholar] [CrossRef] [PubMed]
- Idoyaga, J.; Fiorese, C.; Zbytnuik, L.; Lubkin, A.; Miller, J.; Malissen, B.; Mucida, D.; Merad, M.; Steinman, R.M. Specialized role of migratory dendritic cells in peripheral tolerance induction. J. Clin. Investig. 2013, 123, 844–854. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Van de Ven, R.; Lindenberg, J.J.; Oosterhoff, D.; de Gruijl, T.D. Dendritic Cell Plasticity in Tumor-Conditioned Skin: CD14(+) Cells at the Cross-Roads of Immune Activation and Suppression. Front Immunol. 2013, 4, 403. [Google Scholar] [CrossRef] [PubMed]
- Huang, Q.; Liu, D.; Majewski, P.; Schulte, L.C.; Korn, J.M.; Young, R.A.; Lander, E.S.; Hacohen, N. The plasticity of dendritic cell responses to pathogens and their components. Science 2001, 294, 870–875. [Google Scholar] [CrossRef] [PubMed]
- Hackstein, H.; Thomson, A.W. Dendritic cells: Emerging pharmacological targets of immunosuppressive drugs. Nat. Rev. Immunol. 2004, 4, 24–34. [Google Scholar] [CrossRef]
- Svajger, U.; Rozman, P. Induction of Tolerogenic Dendritic Cells by Endogenous Biomolecules: An Update. Front. Immunol. 2018, 9, 2482. [Google Scholar] [CrossRef]
- Vander Lugt, B.; Riddell, J.; Khan, A.A.; Hackney, J.A.; Lesch, J.; DeVoss, J.; Weirauch, M.T.; Singh, H.; Mellman, I. Transcriptional determinants of tolerogenic and immunogenic states during dendritic cell maturation. J. Cell Biol. 2017, 216, 779–792. [Google Scholar] [CrossRef] [Green Version]
- Rutella, S.; Danese, S.; Leone, G. Tolerogenic dendritic cells: Cytokine modulation comes of age. Blood 2006, 108, 1435–1440. [Google Scholar] [CrossRef]
- Vendelova, E.; Ashour, D.; Blank, P.; Erhard, F.; Saliba, A.E.; Kalinke, U.; Lutz, M.B. Tolerogenic Transcriptional Signatures of Steady-State and Pathogen-Induced Dendritic Cells. Front. Immunol. 2018, 9, 333. [Google Scholar] [CrossRef]
- Berkun, Y.; Verbovetski, I.; Ben-Ami, A.; Paran, D.; Caspi, D.; Krispin, A.; Trahtemberg, U.; Gill, O.; Naparstek, Y.; Mevorach, D. Altered dendritic cells with tolerizing phenotype in patients with systemic lupus erythematosus. Eur. J. Immunol. 2008, 38, 2896–2904. [Google Scholar] [CrossRef]
- Obreque, J.; Vega, F.; Torres, A.; Cuitino, L.; Mackern-Oberti, J.P.; Viviani, P.; Kalergis, A.; Llanos, C. Autologous tolerogenic dendritic cells derived from monocytes of systemic lupus erythematosus patients and healthy donors show a stable and immunosuppressive phenotype. Immunology 2017, 152, 648–659. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.J.; Lo, Y.; Luk, D.; Lau, C.S.; Lu, L.; Mok, M.Y. Alternatively activated dendritic cells derived from systemic lupus erythematosus patients have tolerogenic phenotype and function. Clin. Immunol. 2015, 156, 43–57. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Esmaeili, S.A.; Mahmoudi, M.; Rezaieyazdi, Z.; Sahebari, M.; Tabasi, N.; Sahebkar, A.; Rastin, M. Generation of tolerogenic dendritic cells using Lactobacillus rhamnosus and Lactobacillus delbrueckii as tolerogenic probiotics. J. Cell Biochem. 2018, 119, 7865–7872. [Google Scholar] [CrossRef] [PubMed]
- Suwandi, J.S.; Nikolic, T.; Roep, B.O. Translating Mechanism of Regulatory Action of Tolerogenic Dendritic Cells to Monitoring Endpoints in Clinical Trials. Front. Immunol. 2017, 8, 1598. [Google Scholar] [CrossRef]
- Navarro-Barriuso, J.; Mansilla, M.J.; Martinez-Caceres, E.M. Searching for the Transcriptomic Signature of Immune Tolerance Induction-Biomarkers of Safety and Functionality for Tolerogenic Dendritic Cells and Regulatory Macrophages. Front. Immunol. 2018, 9, 2062. [Google Scholar] [CrossRef]
- Navarro-Barriuso, J.; Mansilla, M.J.; Naranjo-Gomez, M.; Sanchez-Pla, A.; Quirant-Sanchez, B.; Teniente-Serra, A.; Ramo-Tello, C.; Martinez-Caceres, E.M. Comparative transcriptomic profile of tolerogenic dendritic cells differentiated with vitamin D3, dexamethasone and rapamycin. Sci. Rep. 2018, 8, 14985. [Google Scholar] [CrossRef]
- Phillips, B.E.; Garciafigueroa, Y.; Trucco, M.; Giannoukakis, N. Clinical Tolerogenic Dendritic Cells: Exploring Therapeutic Impact on Human Autoimmune Disease. Front. Immunol. 2017, 8, 1279. [Google Scholar] [CrossRef]
- Eisenbarth, S.C. Dendritic cell subsets in T cell programming: Location dictates function. Nat. Rev. Immunol. 2019, 19, 89–103. [Google Scholar] [CrossRef]
- Poltorak, M.P.; Schraml, B.U. Fate mapping of dendritic cells. Front. Immunol. 2015, 6, 199. [Google Scholar] [CrossRef]
- Meredith, M.M.; Liu, K.; Darrasse-Jeze, G.; Kamphorst, A.O.; Schreiber, H.A.; Guermonprez, P.; Idoyaga, J.; Cheong, C.; Yao, K.H.; Niec, R.E.; et al. Expression of the zinc finger transcription factor zDC (Zbtb46, Btbd4) defines the classical dendritic cell lineage. J. Exp. Med. 2012, 209, 1153–1165. [Google Scholar] [CrossRef] [Green Version]
- Satpathy, A.T.; Kc, W.; Albring, J.C.; Edelson, B.T.; Kretzer, N.M.; Bhattacharya, D.; Murphy, T.L.; Murphy, K.M. Zbtb46 expression distinguishes classical dendritic cells and their committed progenitors from other immune lineages. J. Exp. Med. 2012, 209, 1135–1152. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Grajales-Reyes, G.E.; Iwata, A.; Albring, J.; Wu, X.; Tussiwand, R.; Kc, W.; Kretzer, N.M.; Briseno, C.G.; Durai, V.; Bagadia, P.; et al. Batf3 maintains autoactivation of Irf8 for commitment of a CD8alpha(+) conventional DC clonogenic progenitor. Nat. Immunol. 2015, 16, 708–717. [Google Scholar] [CrossRef] [PubMed]
- Breton, G.; Lee, J.; Zhou, Y.J.; Schreiber, J.J.; Keler, T.; Puhr, S.; Anandasabapathy, N.; Schlesinger, S.; Caskey, M.; Liu, K.; et al. Circulating precursors of human CD1c+ and CD141+ dendritic cells. J. Exp. Med. 2015, 212, 401–413. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Suzuki, S.; Honma, K.; Matsuyama, T.; Suzuki, K.; Toriyama, K.; Akitoyo, I.; Yamamoto, K.; Suematsu, T.; Nakamura, M.; Yui, K.; et al. Critical roles of interferon regulatory factor 4 in CD11bhighCD8alpha- dendritic cell development. Proc. Natl. Acad. Sci. USA 2004, 101, 8981–8986. [Google Scholar] [CrossRef] [PubMed]
- Cisse, B.; Caton, M.L.; Lehner, M.; Maeda, T.; Scheu, S.; Locksley, R.; Holmberg, D.; Zweier, C.; den Hollander, N.S.; Kant, S.G.; et al. Transcription factor E2-2 is an essential and specific regulator of plasmacytoid dendritic cell development. Cell 2008, 135, 37–48. [Google Scholar] [CrossRef] [PubMed]
- Fallarino, F.; Vacca, C.; Orabona, C.; Belladonna, M.L.; Bianchi, R.; Marshall, B.; Keskin, D.B.; Mellor, A.L.; Fioretti, M.C.; Grohmann, U.; et al. Functional expression of indoleamine 2,3-dioxygenase by murine CD8 alpha(+) dendritic cells. Int. Immunol. 2002, 14, 65–68. [Google Scholar] [CrossRef] [PubMed]
- Orabona, C.; Puccetti, P.; Vacca, C.; Bicciato, S.; Luchini, A.; Fallarino, F.; Bianchi, R.; Velardi, E.; Perruccio, K.; Velardi, A.; et al. Toward the identification of a tolerogenic signature in IDO-competent dendritic cells. Blood 2006, 107, 2846–2854. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jones, A.; Bourque, J.; Kuehm, L.; Opejin, A.; Teague, R.M.; Gross, C.; Hawiger, D. Immunomodulatory Functions of BTLA and HVEM Govern Induction of Extrathymic Regulatory T Cells and Tolerance by Dendritic Cells. Immunity 2016, 45, 1066–1077. [Google Scholar] [CrossRef] [Green Version]
- Price, J.D.; Hotta-Iwamura, C.; Zhao, Y.; Beauchamp, N.M.; Tarbell, K.V. DCIR2+ cDC2 DCs and Zbtb32 Restore CD4+ T-Cell Tolerance and Inhibit Diabetes. Diabetes 2015, 64, 3521–3531. [Google Scholar] [CrossRef] [Green Version]
- Uto, T.; Fukaya, T.; Takagi, H.; Arimura, K.; Nakamura, T.; Kojima, N.; Malissen, B.; Sato, K. Clec4A4 is a regulatory receptor for dendritic cells that impairs inflammation and T-cell immunity. Nat. Commun. 2016, 7, 11273. [Google Scholar] [CrossRef]
- Belladonna, M.L.; Grohmann, U.; Guidetti, P.; Volpi, C.; Bianchi, R.; Fioretti, M.C.; Schwarcz, R.; Fallarino, F.; Puccetti, P. Kynurenine pathway enzymes in dendritic cells initiate tolerogenesis in the absence of functional IDO. J. Immunol. 2006, 177, 130–137. [Google Scholar] [CrossRef] [PubMed]
- Coombes, J.L.; Siddiqui, K.R.; Arancibia-Carcamo, C.V.; Hall, J.; Sun, C.M.; Belkaid, Y.; Powrie, F. A functionally specialized population of mucosal CD103+ DCs induces Foxp3+ regulatory T cells via a TGF-beta and retinoic acid-dependent mechanism. J. Exp. Med. 2007, 204, 1757–1764. [Google Scholar] [CrossRef] [PubMed]
- Matteoli, G.; Mazzini, E.; Iliev, I.D.; Mileti, E.; Fallarino, F.; Puccetti, P.; Chieppa, M.; Rescigno, M. Gut CD103+ dendritic cells express indoleamine 2,3-dioxygenase which influences T regulatory/T effector cell balance and oral tolerance induction. Gut 2010, 59, 595–604. [Google Scholar] [CrossRef]
- Guilliams, M.; Crozat, K.; Henri, S.; Tamoutounour, S.; Grenot, P.; Devilard, E.; de Bovis, B.; Alexopoulou, L.; Dalod, M.; Malissen, B. Skin-draining lymph nodes contain dermis-derived CD103(-) dendritic cells that constitutively produce retinoic acid and induce Foxp3(+) regulatory T cells. Blood 2010, 115, 1958–1968. [Google Scholar] [CrossRef] [PubMed]
- Tordesillas, L.; Lozano-Ojalvo, D.; Dunkin, D.; Mondoulet, L.; Agudo, J.; Merad, M.; Sampson, H.A.; Berin, M.C. PDL2(+) CD11b(+) dermal dendritic cells capture topical antigen through hair follicles to prime LAP(+) Tregs. Nat. Commun. 2018, 9, 5238. [Google Scholar] [CrossRef]
- Yun, T.J.; Lee, J.S.; Machmach, K.; Shim, D.; Choi, J.; Wi, Y.J.; Jang, H.S.; Jung, I.H.; Kim, K.; Yoon, W.K.; et al. Indoleamine 2,3-Dioxygenase-Expressing Aortic Plasmacytoid Dendritic Cells Protect against Atherosclerosis by Induction of Regulatory T Cells. Cell Metab. 2016, 23, 852–866. [Google Scholar] [CrossRef]
- Chu, C.C.; Ali, N.; Karagiannis, P.; Di Meglio, P.; Skowera, A.; Napolitano, L.; Barinaga, G.; Grys, K.; Sharif-Paghaleh, E.; Karagiannis, S.N.; et al. Resident CD141 (BDCA3)+ dendritic cells in human skin produce IL-10 and induce regulatory T cells that suppress skin inflammation. J. Exp. Med. 2012, 209, 935–945. [Google Scholar] [CrossRef]
- Gregori, S.; Tomasoni, D.; Pacciani, V.; Scirpoli, M.; Battaglia, M.; Magnani, C.F.; Hauben, E.; Roncarolo, M.G. Differentiation of type 1 T regulatory cells (Tr1) by tolerogenic DC-10 requires the IL-10-dependent ILT4/HLA-G pathway. Blood 2010, 116, 935–944. [Google Scholar] [CrossRef]
- Comi, M.; Avancini, D.; Santoni de Sio, F.; Villa, M.; Uyeda, M.J.; Floris, M.; Tomasoni, D.; Bulfone, A.; Roncarolo, M.G.; Gregori, S. Coexpression of CD163 and CD141 identifies human circulating IL-10-producing dendritic cells (DC-10). Cell Mol. Immunol. 2019. [Google Scholar] [CrossRef]
- Kassianos, A.J.; Hardy, M.Y.; Ju, X.; Vijayan, D.; Ding, Y.; Vulink, A.J.; McDonald, K.J.; Jongbloed, S.L.; Wadley, R.B.; Wells, C.; et al. Human CD1c (BDCA-1)+ myeloid dendritic cells secrete IL-10 and display an immuno-regulatory phenotype and function in response to Escherichia coli. Eur. J. Immunol. 2012, 42, 1512–1522. [Google Scholar] [CrossRef]
- Tsoumakidou, M.; Tousa, S.; Semitekolou, M.; Panagiotou, P.; Panagiotou, A.; Morianos, I.; Litsiou, E.; Trochoutsou, A.I.; Konstantinou, M.; Potaris, K.; et al. Tolerogenic signaling by pulmonary CD1c+ dendritic cells induces regulatory T cells in patients with chronic obstructive pulmonary disease by IL-27/IL-10/inducible costimulator ligand. J. Allergy Clin. Immunol. 2014, 134, 944–954.e8. [Google Scholar] [CrossRef] [PubMed]
- Moseman, E.A.; Liang, X.; Dawson, A.J.; Panoskaltsis-Mortari, A.; Krieg, A.M.; Liu, Y.J.; Blazar, B.R.; Chen, W. Human plasmacytoid dendritic cells activated by CpG oligodeoxynucleotides induce the generation of CD4+CD25+ regulatory T cells. J. Immunol. 2004, 173, 4433–4442. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Liang, X.; Peterson, A.J.; Munn, D.H.; Blazar, B.R. The indoleamine 2,3-dioxygenase pathway is essential for human plasmacytoid dendritic cell-induced adaptive T regulatory cell generation. J. Immunol. 2008, 181, 5396–5404. [Google Scholar] [CrossRef] [PubMed]
- Steinman, R.M. Decisions about dendritic cells: Past, present, and future. Annu. Rev. Immunol. 2012, 30, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Durai, V.; Murphy, K.M. Functions of Murine Dendritic Cells. Immunity 2016, 45, 719–736. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Den Haan, J.M.; Lehar, S.M.; Bevan, M.J. CD8(+) but not CD8(-) dendritic cells cross-prime cytotoxic T cells in vivo. J. Exp. Med. 2000, 192, 1685–1696. [Google Scholar] [CrossRef]
- Edelson, B.T.; Kc, W.; Juang, R.; Kohyama, M.; Benoit, L.A.; Klekotka, P.A.; Moon, C.; Albring, J.C.; Ise, W.; Michael, D.G.; et al. Peripheral CD103+ dendritic cells form a unified subset developmentally related to CD8alpha+ conventional dendritic cells. J. Exp. Med. 2010, 207, 823–836. [Google Scholar] [CrossRef]
- Minoda, Y.; Virshup, I.; Leal Rojas, I.; Haigh, O.; Wong, Y.; Miles, J.J.; Wells, C.A.; Radford, K.J. Human CD141(+) Dendritic Cell and CD1c(+) Dendritic Cell Undergo Concordant Early Genetic Programming after Activation in Humanized Mice In Vivo. Front. Immunol. 2017, 8, 1419. [Google Scholar] [CrossRef]
- Bell, B.D.; Kitajima, M.; Larson, R.P.; Stoklasek, T.A.; Dang, K.; Sakamoto, K.; Wagner, K.U.; Kaplan, D.H.; Reizis, B.; Hennighausen, L.; et al. The transcription factor STAT5 is critical in dendritic cells for the development of TH2 but not TH1 responses. Nat. Immunol. 2013, 14, 364–371. [Google Scholar] [CrossRef] [Green Version]
- Gao, Y.; Nish, S.A.; Jiang, R.; Hou, L.; Licona-Limon, P.; Weinstein, J.S.; Zhao, H.; Medzhitov, R. Control of T helper 2 responses by transcription factor IRF4-dependent dendritic cells. Immunity 2013, 39, 722–732. [Google Scholar] [CrossRef]
- Sichien, D.; Lambrecht, B.N.; Guilliams, M.; Scott, C.L. Development of conventional dendritic cells: From common bone marrow progenitors to multiple subsets in peripheral tissues. Mucosal Immunol. 2017, 10, 831–844. [Google Scholar] [CrossRef] [PubMed]
- Kashem, S.W.; Haniffa, M.; Kaplan, D.H. Antigen-Presenting Cells in the Skin. Annu. Rev. Immunol. 2017, 35, 469–499. [Google Scholar] [CrossRef] [PubMed]
- Kumamoto, Y.; Linehan, M.; Weinstein, J.S.; Laidlaw, B.J.; Craft, J.E.; Iwasaki, A. CD301b(+) dermal dendritic cells drive T helper 2 cell-mediated immunity. Immunity 2013, 39, 733–743. [Google Scholar] [CrossRef] [PubMed]
- Kashem, S.W.; Riedl, M.S.; Yao, C.; Honda, C.N.; Vulchanova, L.; Kaplan, D.H. Nociceptive Sensory Fibers Drive Interleukin-23 Production from CD301b+ Dermal Dendritic Cells and Drive Protective Cutaneous Immunity. Immunity 2015, 43, 515–526. [Google Scholar] [CrossRef] [Green Version]
- Robbins, S.H.; Walzer, T.; Dembele, D.; Thibault, C.; Defays, A.; Bessou, G.; Xu, H.; Vivier, E.; Sellars, M.; Pierre, P.; et al. Novel insights into the relationships between dendritic cell subsets in human and mouse revealed by genome-wide expression profiling. Genome Biol. 2008, 9, R17. [Google Scholar] [CrossRef]
- Schlitzer, A.; McGovern, N.; Teo, P.; Zelante, T.; Atarashi, K.; Low, D.; Ho, A.W.; See, P.; Shin, A.; Wasan, P.S.; et al. IRF4 transcription factor-dependent CD11b+ dendritic cells in human and mouse control mucosal IL-17 cytokine responses. Immunity 2013, 38, 970–983. [Google Scholar] [CrossRef]
- Bertola, A.; Ciucci, T.; Rousseau, D.; Bourlier, V.; Duffaut, C.; Bonnafous, S.; Blin-Wakkach, C.; Anty, R.; Iannelli, A.; Gugenheim, J.; et al. Identification of adipose tissue dendritic cells correlated with obesity-associated insulin-resistance and inducing Th17 responses in mice and patients. Diabetes 2012, 61, 2238–2247. [Google Scholar] [CrossRef]
- Collin, M.; Bigley, V. Human dendritic cell subsets: An update. Immunology 2018, 154, 3–20. [Google Scholar] [CrossRef]
- Kohli, K.; Janssen, A.; Forster, R. Plasmacytoid dendritic cells induce tolerance predominantly by cargoing antigen to lymph nodes. Eur. J. Immunol. 2016, 46, 2659–2668. [Google Scholar] [CrossRef] [Green Version]
- Baban, B.; Hansen, A.M.; Chandler, P.R.; Manlapat, A.; Bingaman, A.; Kahler, D.J.; Munn, D.H.; Mellor, A.L. A minor population of splenic dendritic cells expressing CD19 mediates IDO-dependent T cell suppression via type I IFN signaling following B7 ligation. Int. Immunol. 2005, 17, 909–919. [Google Scholar] [CrossRef] [Green Version]
- Zlotnikov-Klionsky, Y.; Nathansohn-Levi, B.; Shezen, E.; Rosen, C.; Kagan, S.; Bar-On, L.; Jung, S.; Shifrut, E.; Reich-Zeliger, S.; Friedman, N.; et al. Perforin-Positive Dendritic Cells Exhibit an Immuno-regulatory Role in Metabolic Syndrome and Autoimmunity. Immunity 2015, 43, 776–787. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Doebel, T.; Voisin, B.; Nagao, K. Langerhans Cells-The Macrophage in Dendritic Cell Clothing. Trends Immunol. 2017, 38, 817–828. [Google Scholar] [CrossRef] [PubMed]
- Clayton, K.; Vallejo, A.F.; Davies, J.; Sirvent, S.; Polak, M.E. Langerhans Cells-Programmed by the Epidermis. Front. Immunol. 2017, 8, 1676. [Google Scholar] [CrossRef] [PubMed]
- Seneschal, J.; Clark, R.A.; Gehad, A.; Baecher-Allan, C.M.; Kupper, T.S. Human epidermal Langerhans cells maintain immune homeostasis in skin by activating skin resident regulatory T cells. Immunity 2012, 36, 873–884. [Google Scholar] [CrossRef]
- King, J.K.; Philips, R.L.; Eriksson, A.U.; Kim, P.J.; Halder, R.C.; Lee, D.J.; Singh, R.R. Langerhans Cells Maintain Local Tissue Tolerance in a Model of Systemic Autoimmune Disease. J. Immunol. 2015, 195, 464–476. [Google Scholar] [CrossRef] [Green Version]
- Stagg, A.J. Intestinal Dendritic Cells in Health and Gut Inflammation. Front. Immunol. 2018, 9, 2883. [Google Scholar] [CrossRef] [Green Version]
- Coutant, F.; Miossec, P. Altered dendritic cell functions in autoimmune diseases: distinct and overlapping profiles. Nat. Rev. Rheumatol. 2016, 12, 703–715. [Google Scholar] [CrossRef]
- Schinnerling, K.; Garcia-Gonzalez, P.; Aguillon, J.C. Gene Expression Profiling of Human Monocyte-derived Dendritic Cells - Searching for Molecular Regulators of Tolerogenicity. Front. Immunol. 2015, 6, 528. [Google Scholar] [CrossRef]
- Garcia-Gonzalez, P.A.; Schinnerling, K.; Sepulveda-Gutierrez, A.; Maggi, J.; Hoyos, L.; Morales, R.A.; Mehdi, A.M.; Nel, H.J.; Soto, L.; Pesce, B.; et al. Treatment with Dexamethasone and Monophosphoryl Lipid A Removes Disease-Associated Transcriptional Signatures in Monocyte-Derived Dendritic Cells from Rheumatoid Arthritis Patients and Confers Tolerogenic Features. Front. Immunol. 2016, 7, 458. [Google Scholar] [CrossRef] [Green Version]
- Navarro-Barriuso, J.; Mansilla, M.J.; Quirant-Sanchez, B.; Ardiaca-Martinez, A.; Teniente-Serra, A.; Presas-Rodriguez, S.; Ten Brinke, A.; Ramo-Tello, C.; Martinez-Caceres, E.M. MAP7 and MUCL1 Are Biomarkers of Vitamin D3-Induced Tolerogenic Dendritic Cells in Multiple Sclerosis Patients. Front. Immunol. 2019, 10, 1251. [Google Scholar] [CrossRef]
- Greenwald, R.J.; Freeman, G.J.; Sharpe, A.H. The B7 family revisited. Annu. Rev. Immunol. 2005, 23, 515–548. [Google Scholar] [CrossRef] [PubMed]
- Tuettenberg, A.; Huter, E.; Hubo, M.; Horn, J.; Knop, J.; Grimbacher, B.; Kroczek, R.A.; Stoll, S.; Jonuleit, H. The role of ICOS in directing T cell responses: ICOS-dependent induction of T cell anergy by tolerogenic dendritic cells. J. Immunol. 2009, 182, 3349–3356. [Google Scholar] [CrossRef] [PubMed]
- Funes, S.C.; Manrique de Lara, A.; Altamirano-Lagos, M.J.; Mackern-Oberti, J.P.; Escobar-Vera, J.; Kalergis, A.M. Immune checkpoints and the regulation of tolerogenicity in dendritic cells: Implications for autoimmunity and immunotherapy. Autoimmun. Rev. 2019, 18, 359–368. [Google Scholar] [CrossRef]
- Laurent, S.; Carrega, P.; Saverino, D.; Piccioli, P.; Camoriano, M.; Morabito, A.; Dozin, B.; Fontana, V.; Simone, R.; Mortara, L.; et al. CTLA-4 is expressed by human monocyte-derived dendritic cells and regulates their functions. Hum. Immunol. 2010, 71, 934–941. [Google Scholar] [CrossRef] [PubMed]
- Yao, S.; Wang, S.; Zhu, Y.; Luo, L.; Zhu, G.; Flies, S.; Xu, H.; Ruff, W.; Broadwater, M.; Choi, I.H.; et al. PD-1 on dendritic cells impedes innate immunity against bacterial infection. Blood 2009, 113, 5811–5818. [Google Scholar] [CrossRef] [PubMed]
- Lim, T.S.; Chew, V.; Sieow, J.L.; Goh, S.; Yeong, J.P.; Soon, A.L.; Ricciardi-Castagnoli, P. PD-1 expression on dendritic cells suppresses CD8(+) T cell function and antitumor immunity. Oncoimmunology 2016, 5, e1085146. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Horuzsko, A. Expression and function of immunoglobulin-like transcripts on tolerogenic dendritic cells. Hum. Immunol. 2009, 70, 353–356. [Google Scholar] [CrossRef] [Green Version]
- Suss, G.; Shortman, K. A subclass of dendritic cells kills CD4 T cells via Fas/Fas-ligand-induced apoptosis. J. Exp. Med. 1996, 183, 1789–1796. [Google Scholar] [CrossRef]
- Bolland, S.; Ravetch, J.V. Spontaneous autoimmune disease in Fc(gamma)RIIB-deficient mice results from strain-specific epistasis. Immunity 2000, 13, 277–285. [Google Scholar] [CrossRef]
- Qin, D.; Wu, J.; Vora, K.A.; Ravetch, J.V.; Szakal, A.K.; Manser, T.; Tew, J.G. Fc gamma receptor IIB on follicular dendritic cells regulates the B cell recall response. J. Immunol. 2000, 164, 6268–6275. [Google Scholar] [CrossRef]
- Desai, D.D.; Harbers, S.O.; Flores, M.; Colonna, L.; Downie, M.P.; Bergtold, A.; Jung, S.; Clynes, R. Fc gamma receptor IIB on dendritic cells enforces peripheral tolerance by inhibiting effector T cell responses. J. Immunol. 2007, 178, 6217–6226. [Google Scholar] [CrossRef] [PubMed]
- Torres-Aguilar, H.; Aguilar-Ruiz, S.R.; Gonzalez-Perez, G.; Munguia, R.; Bajana, S.; Meraz-Rios, M.A.; Sanchez-Torres, C. Tolerogenic dendritic cells generated with different immunosuppressive cytokines induce antigen-specific anergy and regulatory properties in memory CD4+ T cells. J. Immunol. 2010, 184, 1765–1775. [Google Scholar] [CrossRef] [PubMed]
- Gueguen, C.; Bouley, J.; Moussu, H.; Luce, S.; Duchateau, M.; Chamot-Rooke, J.; Pallardy, M.; Lombardi, V.; Nony, E.; Baron-Bodo, V.; et al. Changes in markers associated with dendritic cells driving the differentiation of either TH2 cells or regulatory T cells correlate with clinical benefit during allergen immunotherapy. J. Allergy Clin. Immunol. 2016, 137, 545–558. [Google Scholar] [CrossRef] [PubMed]
- Szeles, L.; Keresztes, G.; Torocsik, D.; Balajthy, Z.; Krenacs, L.; Poliska, S.; Steinmeyer, A.; Zuegel, U.; Pruenster, M.; Rot, A.; et al. 1,25-dihydroxyvitamin D3 is an autonomous regulator of the transcriptional changes leading to a tolerogenic dendritic cell phenotype. J. Immunol. 2009, 182, 2074–2083. [Google Scholar] [CrossRef] [PubMed]
- Aguilar, H.; Alvarez-Errico, D.; Garcia-Montero, A.C.; Orfao, A.; Sayos, J.; Lopez-Botet, M. Molecular characterization of a novel immune receptor restricted to the monocytic lineage. J. Immunol. 2004, 173, 6703–6711. [Google Scholar] [CrossRef] [PubMed]
- Voss, O.H.; Tian, L.; Murakami, Y.; Coligan, J.E.; Krzewski, K. Emerging role of CD300 receptors in regulating myeloid cell efferocytosis. Mol. Cell Oncol. 2015, 2, e964625. [Google Scholar] [CrossRef] [Green Version]
- Tian, L.; Choi, S.C.; Murakami, Y.; Allen, J.; Morse, H.C., 3rd; Qi, C.F.; Krzewski, K.; Coligan, J.E. p85alpha recruitment by the CD300f phosphatidylserine receptor mediates apoptotic cell clearance required for autoimmunity suppression. Nat. Commun. 2014, 5, 3146. [Google Scholar] [CrossRef]
- Lee, S.M.; Nam, Y.P.; Suk, K.; Lee, W.H. Immune receptor expressed on myeloid cells 1 (IREM-1) inhibits B cell activation factor (BAFF)-mediated inflammatory regulation of THP-1 cells through modulation of the activities of extracellular regulated kinase (ERK). Clin. Exp. Immunol. 2010, 161, 504–511. [Google Scholar] [CrossRef]
- Ueno, K.; Urai, M.; Izawa, K.; Otani, Y.; Yanagihara, N.; Kataoka, M.; Takatsuka, S.; Abe, M.; Hasegawa, H.; Shimizu, K.; et al. Mouse LIMR3/CD300f is a negative regulator of the antimicrobial activity of neutrophils. Sci. Rep. 2018, 8, 17406. [Google Scholar] [CrossRef]
- Anderson, A.E.; Swan, D.J.; Sayers, B.L.; Harry, R.A.; Patterson, A.M.; von Delwig, A.; Robinson, J.H.; Isaacs, J.D.; Hilkens, C.M. LPS activation is required for migratory activity and antigen presentation by tolerogenic dendritic cells. J. Leukoc. Biol. 2009, 85, 243–250. [Google Scholar] [CrossRef]
- Garcia-Gonzalez, P.; Morales, R.; Hoyos, L.; Maggi, J.; Campos, J.; Pesce, B.; Garate, D.; Larrondo, M.; Gonzalez, R.; Soto, L.; et al. A short protocol using dexamethasone and monophosphoryl lipid A generates tolerogenic dendritic cells that display a potent migratory capacity to lymphoid chemokines. J. Transl. Med. 2013, 11, 128. [Google Scholar] [CrossRef] [PubMed]
- De Laere, M.; Derdelinckx, J.; Hassi, M.; Kerosalo, M.; Oravamaki, H.; Van den Bergh, J.; Berneman, Z.; Cools, N. Shuttling Tolerogenic Dendritic Cells across the Blood-Brain Barrier In Vitro via the Introduction of De Novo C-C Chemokine Receptor 5 Expression Using Messenger RNA Electroporation. Front. Immunol. 2017, 8, 1964. [Google Scholar] [CrossRef] [PubMed]
- Nikolic, T.; Woittiez, N.J.C.; van der Slik, A.; Laban, S.; Joosten, A.; Gysemans, C.; Mathieu, C.; Zwaginga, J.J.; Koeleman, B.; Roep, B.O. Differential transcriptome of tolerogenic versus inflammatory dendritic cells points to modulated T1D genetic risk and enriched immune regulation. Genes Immun. 2017, 18, 176–183. [Google Scholar] [CrossRef] [PubMed]
- Azzaoui, I.; Yahia, S.A.; Chang, Y.; Vorng, H.; Morales, O.; Fan, Y.; Delhem, N.; Ple, C.; Tonnel, A.B.; Wallaert, B.; et al. CCL18 differentiates dendritic cells in tolerogenic cells able to prime regulatory T cells in healthy subjects. Blood 2011, 118, 3549–3558. [Google Scholar] [CrossRef]
- Rapp, M.; Wintergerst, M.W.M.; Kunz, W.G.; Vetter, V.K.; Knott, M.M.L.; Lisowski, D.; Haubner, S.; Moder, S.; Thaler, R.; Eiber, S.; et al. CCL22 controls immunity by promoting regulatory T cell communication with dendritic cells in lymph nodes. J. Exp. Med. 2019, 216, 1170–1181. [Google Scholar] [CrossRef] [Green Version]
- Perrier, P.; Martinez, F.O.; Locati, M.; Bianchi, G.; Nebuloni, M.; Vago, G.; Bazzoni, F.; Sozzani, S.; Allavena, P.; Mantovani, A. Distinct transcriptional programs activated by interleukin-10 with or without lipopolysaccharide in dendritic cells: induction of the B cell-activating chemokine, CXC chemokine ligand 13. J. Immunol. 2004, 172, 7031–7042. [Google Scholar] [CrossRef]
- Omari, K.M.; Lutz, S.E.; Santambrogio, L.; Lira, S.A.; Raine, C.S. Neuroprotection and remyelination after autoimmune demyelination in mice that inducibly overexpress CXCL1. Am. J. Pathol. 2009, 174, 164–176. [Google Scholar] [CrossRef]
- Karim, H.; Kim, S.H.; Lapato, A.S.; Yasui, N.; Katzenellenbogen, J.A.; Tiwari-Woodruff, S.K. Increase in chemokine CXCL1 by ERbeta ligand treatment is a key mediator in promoting axon myelination. Proc. Natl. Acad. Sci. USA 2018, 115, 6291–6296. [Google Scholar] [CrossRef]
- Hasegawa, H.; Matsumoto, T. Mechanisms of Tolerance Induction by Dendritic Cells In Vivo. Front. Immunol. 2018, 9, 350. [Google Scholar] [CrossRef]
- Kautz-Neu, K.; Kostka, S.L.; Dinges, S.; Iwakura, Y.; Udey, M.C.; von Stebut, E. A role for leukocyte-derived IL-1RA in DC homeostasis revealed by increased susceptibility of IL-1RA-deficient mice to cutaneous leishmaniasis. J. Investig. Dermatol. 2011, 131, 1650–1659. [Google Scholar] [CrossRef]
- Teh, B.K.; Yeo, J.G.; Chern, L.M.; Lu, J. C1q regulation of dendritic cell development from monocytes with distinct cytokine production and T cell stimulation. Mol. Immunol. 2011, 48, 1128–1138. [Google Scholar] [CrossRef] [PubMed]
- Mascarell, L.; Airouche, S.; Berjont, N.; Gary, C.; Gueguen, C.; Fourcade, G.; Bellier, B.; Togbe, D.; Ryffel, B.; Klatzmann, D.; et al. The regulatory dendritic cell marker C1q is a potent inhibitor of allergic inflammation. Mucosal Immunol. 2017, 10, 695–704. [Google Scholar] [CrossRef] [PubMed]
- Doyen, V.; Rubio, M.; Braun, D.; Nakajima, T.; Abe, J.; Saito, H.; Delespesse, G.; Sarfati, M. Thrombospondin 1 is an autocrine negative regulator of human dendritic cell activation. J. Exp. Med. 2003, 198, 1277–1283. [Google Scholar] [CrossRef] [PubMed]
- Grimbert, P.; Bouguermouh, S.; Baba, N.; Nakajima, T.; Allakhverdi, Z.; Braun, D.; Saito, H.; Rubio, M.; Delespesse, G.; Sarfati, M. Thrombospondin/CD47 interaction: A pathway to generate regulatory T cells from human CD4+ CD25- T cells in response to inflammation. J. Immunol. 2006, 177, 3534–3541. [Google Scholar] [CrossRef] [PubMed]
- Williams, M.A.; Bauer, S.; Lu, W.; Guo, J.; Walter, S.; Bushnell, T.P.; Lillehoj, E.P.; Georas, S.N. Deletion of the mucin-like molecule muc1 enhances dendritic cell activation in response to toll-like receptor ligands. J. Innate Immun. 2010, 2, 123–143. [Google Scholar] [CrossRef]
- Mellor, A.L.; Munn, D.H. IDO expression by dendritic cells: tolerance and tryptophan catabolism. Nat. Rev. Immunol. 2004, 4, 762–774. [Google Scholar] [CrossRef] [PubMed]
- Li, X.L.; Li, H.; Zhang, M.; Xu, H.; Yue, L.T.; Zhang, X.X.; Wang, S.; Wang, C.C.; Li, Y.B.; Dou, Y.C.; et al. Exosomes derived from atorvastatin-modified bone marrow dendritic cells ameliorate experimental autoimmune myasthenia gravis by up-regulated levels of IDO/Treg and partly dependent on FasL/Fas pathway. J. Neuroinflammation 2016, 13, 8. [Google Scholar] [CrossRef] [Green Version]
- Acovic, A.; Simovic Markovic, B.; Gazdic, M.; Arsenijevic, A.; Jovicic, N.; Gajovic, N.; Jovanovic, M.; Zdravkovic, N.; Kanjevac, T.; Harrell, C.R.; et al. Indoleamine 2,3-dioxygenase-dependent expansion of T-regulatory cells maintains mucosal healing in ulcerative colitis. Therap. Adv. Gastroenterol. 2018, 11, 1756284818793558. [Google Scholar] [CrossRef]
- Cauwels, A.; Van Lint, S.; Catteeuw, D.; Pang, S.; Paul, F.; Rogge, E.; Verhee, A.; Prinz, M.; Kley, N.; Uze, G.; et al. Targeting interferon activity to dendritic cells enables in vivo tolerization and protection against EAE in mice. J. Autoimmun. 2019, 97, 70–76. [Google Scholar] [CrossRef]
- Sotero-Esteva, W.D.; Wolfe, D.; Ferris, M.; Taylor, M.W. An indoleamine 2,3-dioxygenase-negative mutant is defective in stat1 DNA binding: Differential response to IFN-gamma and IFN-alpha. J. Interferon Cytokine Res. 2000, 20, 623–632. [Google Scholar] [CrossRef]
- Grohmann, U.; Bianchi, R.; Belladonna, M.L.; Silla, S.; Fallarino, F.; Fioretti, M.C.; Puccetti, P. IFN-gamma inhibits presentation of a tumor/self peptide by CD8 alpha- dendritic cells via potentiation of the CD8 alpha+ subset. J. Immunol 2000, 165, 1357–1363. [Google Scholar] [CrossRef] [PubMed]
- Worah, K.; Mathan, T.S.M.; Vu Manh, T.P.; Keerthikumar, S.; Schreibelt, G.; Tel, J.; Duiveman-de Boer, T.; Skold, A.E.; van Spriel, A.B.; de Vries, I.J.M.; et al. Proteomics of Human Dendritic Cell Subsets Reveals Subset-Specific Surface Markers and Differential Inflammasome Function. Cell Rep. 2016, 16, 2953–2966. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ginhoux, F.; Liu, K.; Helft, J.; Bogunovic, M.; Greter, M.; Hashimoto, D.; Price, J.; Yin, N.; Bromberg, J.; Lira, S.A.; et al. The origin and development of nonlymphoid tissue CD103+ DCs. J. Exp. Med. 2009, 206, 3115–3130. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jaensson, E.; Uronen-Hansson, H.; Pabst, O.; Eksteen, B.; Tian, J.; Coombes, J.L.; Berg, P.L.; Davidsson, T.; Powrie, F.; Johansson-Lindbom, B.; et al. Small intestinal CD103+ dendritic cells display unique functional properties that are conserved between mice and humans. J. Exp. Med. 2008, 205, 2139–2149. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lippens, C.; Duraes, F.V.; Dubrot, J.; Brighouse, D.; Lacroix, M.; Irla, M.; Aubry-Lachainaye, J.P.; Reith, W.; Mandl, J.N.; Hugues, S. IDO-orchestrated crosstalk between pDCs and Tregs inhibits autoimmunity. J. Autoimmun. 2016, 75, 39–49. [Google Scholar] [CrossRef] [PubMed]
- Manlapat, A.K.; Kahler, D.J.; Chandler, P.R.; Munn, D.H.; Mellor, A.L. Cell-autonomous control of interferon type I expression by indoleamine 2,3-dioxygenase in regulatory CD19+ dendritic cells. Eur. J. Immunol. 2007, 37, 1064–1071. [Google Scholar] [CrossRef] [PubMed]
- Chauveau, C.; Remy, S.; Royer, P.J.; Hill, M.; Tanguy-Royer, S.; Hubert, F.X.; Tesson, L.; Brion, R.; Beriou, G.; Gregoire, M.; et al. Heme oxygenase-1 expression inhibits dendritic cell maturation and proinflammatory function but conserves IL-10 expression. Blood 2005, 106, 1694–1702. [Google Scholar] [CrossRef] [Green Version]
- Wong, T.H.; Chen, H.A.; Gau, R.J.; Yen, J.H.; Suen, J.L. Heme Oxygenase-1-Expressing Dendritic Cells Promote Foxp3+ Regulatory T Cell Differentiation and Induce Less Severe Airway Inflammation in Murine Models. PLoS ONE 2016, 11, e0168919. [Google Scholar] [CrossRef]
- Campbell, N.K.; Fitzgerald, H.K.; Malara, A.; Hambly, R.; Sweeney, C.M.; Kirby, B.; Fletcher, J.M.; Dunne, A. Naturally derived Heme-Oxygenase 1 inducers attenuate inflammatory responses in human dendritic cells and T cells: relevance for psoriasis treatment. Sci. Rep. 2018, 8, 10287. [Google Scholar] [CrossRef]
- Park, D.J.; Agarwal, A.; George, J.F. Heme oxygenase-1 expression in murine dendritic cell subpopulations: effect on CD8+ dendritic cell differentiation in vivo. Am. J. Pathol 2010, 176, 2831–2839. [Google Scholar] [CrossRef]
- Jung, I.D.; Lee, J.S.; Lee, C.M.; Noh, K.T.; Jeong, Y.I.; Park, W.S.; Chun, S.H.; Jeong, S.K.; Park, J.W.; Son, K.H.; et al. Induction of indoleamine 2,3-dioxygenase expression via heme oxygenase-1-dependant pathway during murine dendritic cell maturation. Biochem. Pharmacol. 2010, 80, 491–505. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.; Bazer, F.W.; Davis, T.A.; Kim, S.W.; Li, P.; Marc Rhoads, J.; Carey Satterfield, M.; Smith, S.B.; Spencer, T.E.; Yin, Y. Arginine metabolism and nutrition in growth, health and disease. Amino Acids 2009, 37, 153–168. [Google Scholar] [CrossRef] [PubMed]
- Caldwell, R.W.; Rodriguez, P.C.; Toque, H.A.; Narayanan, S.P.; Caldwell, R.B. Arginase: A Multifaceted Enzyme Important in Health and Disease. Physiol. Rev. 2018, 98, 641–665. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mondanelli, G.; Bianchi, R.; Pallotta, M.T.; Orabona, C.; Albini, E.; Iacono, A.; Belladonna, M.L.; Vacca, C.; Fallarino, F.; Macchiarulo, A.; et al. A Relay Pathway between Arginine and Tryptophan Metabolism Confers Immunosuppressive Properties on Dendritic Cells. Immunity 2017, 46, 233–244. [Google Scholar] [CrossRef]
- Harlin, H.; Meng, Y.; Peterson, A.C.; Zha, Y.; Tretiakova, M.; Slingluff, C.; McKee, M.; Gajewski, T.F. Chemokine expression in melanoma metastases associated with CD8+ T-cell recruitment. Cancer Res. 2009, 69, 3077–3085. [Google Scholar] [CrossRef]
- Panfili, E.; Mondanelli, G.; Orabona, C.; Bianchi, R.; Gargaro, M.; Fallarino, F.; Puccetti, P.; Grohmann, U.; Volpi, C.; Belladonna, M.L. IL-35Ig-expressing dendritic cells induce tolerance via Arginase 1. J. Cell Mol. Med. 2019, 23, 3757–3761. [Google Scholar] [CrossRef]
- Dunand-Sauthier, I.; Irla, M.; Carnesecchi, S.; Seguin-Estevez, Q.; Vejnar, C.E.; Zdobnov, E.M.; Santiago-Raber, M.L.; Reith, W. Repression of arginase-2 expression in dendritic cells by microRNA-155 is critical for promoting T cell proliferation. J. Immunol. 2014, 193, 1690–1700. [Google Scholar] [CrossRef]
- McGovern, N.; Shin, A.; Low, G.; Low, D.; Duan, K.; Yao, L.J.; Msallam, R.; Low, I.; Shadan, N.B.; Sumatoh, H.R.; et al. Human fetal dendritic cells promote prenatal T-cell immune suppression through arginase-2. Nature 2017, 546, 662–666. [Google Scholar] [CrossRef]
- Cynober, L.A. Plasma amino acid levels with a note on membrane transport: Characteristics, regulation, and metabolic significance. Nutrition 2002, 18, 761–766. [Google Scholar] [CrossRef]
- Cruzat, V.; Macedo Rogero, M.; Noel Keane, K.; Curi, R.; Newsholme, P. Glutamine: Metabolism and Immune Function, Supplementation and Clinical Translation. Nutrients 2018, 10, 1564. [Google Scholar] [CrossRef]
- Ko, H.M.; Oh, S.H.; Bang, H.S.; Kang, N.I.; Cho, B.H.; Im, S.Y.; Lee, H.K. Glutamine protects mice from lethal endotoxic shock via a rapid induction of MAPK phosphatase-1. J. Immunol. 2009, 182, 7957–7962. [Google Scholar] [CrossRef] [PubMed]
- Ayush, O.; Lee, C.H.; Kim, H.K.; Im, S.Y.; Cho, B.H.; Lee, H.K. Glutamine suppresses DNFB-induced contact dermatitis by deactivating p38 mitogen-activated protein kinase via induction of MAPK phosphatase-1. J. Investig. Dermatol. 2013, 133, 723–731. [Google Scholar] [CrossRef] [PubMed]
- Ockenga, J.; Borchert, K.; Stuber, E.; Lochs, H.; Manns, M.P.; Bischoff, S.C. Glutamine-enriched total parenteral nutrition in patients with inflammatory bowel disease. Eur. J. Clin. Nutr. 2005, 59, 1302–1309. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, H.T.; Lin, C.S.; Pan, S.C.; Wu, T.H.; Lee, C.S.; Chang, D.M.; Tsai, C.Y.; Wei, Y.H. Alterations of oxygen consumption and extracellular acidification rates by glutamine in PBMCs of SLE patients. Mitochondrion 2019, 44, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Fallarino, F.; Volpi, C.; Fazio, F.; Notartomaso, S.; Vacca, C.; Busceti, C.; Bicciato, S.; Battaglia, G.; Bruno, V.; Puccetti, P.; et al. Metabotropic glutamate receptor-4 modulates adaptive immunity and restrains neuroinflammation. Nat. Med. 2010, 16, 897–902. [Google Scholar] [CrossRef]
- Volpi, C.; Mondanelli, G.; Pallotta, M.T.; Vacca, C.; Iacono, A.; Gargaro, M.; Albini, E.; Bianchi, R.; Belladonna, M.L.; Celanire, S.; et al. Allosteric modulation of metabotropic glutamate receptor 4 activates IDO1-dependent, immunoregulatory signaling in dendritic cells. Neuropharmacology 2016, 102, 59–71. [Google Scholar] [CrossRef]
- Guda, P.; Guda, C.; Subramaniam, S. Reconstruction of pathways associated with amino acid metabolism in human mitochondria. Genomics Proteomics Bioinf. 2007, 5, 166–176. [Google Scholar] [CrossRef]
- Malinarich, F.; Duan, K.; Hamid, R.A.; Bijin, A.; Lin, W.X.; Poidinger, M.; Fairhurst, A.M.; Connolly, J.E. High mitochondrial respiration and glycolytic capacity represent a metabolic phenotype of human tolerogenic dendritic cells. J. Immunol. 2015, 194, 5174–5186. [Google Scholar] [CrossRef]
- Sun, Y.; Oravecz-Wilson, K.; Bridges, S.; McEachin, R.; Wu, J.; Kim, S.H.; Taylor, A.; Zajac, C.; Fujiwara, H.; Peltier, D.C.; et al. miR-142 controls metabolic reprogramming that regulates dendritic cell activation. J. Clin. Investig. 2019, 129, 2029–2042. [Google Scholar] [CrossRef]
- Herber, D.L.; Cao, W.; Nefedova, Y.; Novitskiy, S.V.; Nagaraj, S.; Tyurin, V.A.; Corzo, A.; Cho, H.I.; Celis, E.; Lennox, B.; et al. Lipid accumulation and dendritic cell dysfunction in cancer. Nat. Med. 2010, 16, 880–886. [Google Scholar] [CrossRef] [Green Version]
- Mucida, D.; Park, Y.; Kim, G.; Turovskaya, O.; Scott, I.; Kronenberg, M.; Cheroutre, H. Reciprocal TH17 and regulatory T cell differentiation mediated by retinoic acid. Science 2007, 317, 256–260. [Google Scholar] [CrossRef] [PubMed]
- Bhatt, S.; Qin, J.; Bennett, C.; Qian, S.; Fung, J.J.; Hamilton, T.A.; Lu, L. All-trans retinoic acid induces arginase-1 and inducible nitric oxide synthase-producing dendritic cells with T cell inhibitory function. J. Immunol. 2014, 192, 5098–5108. [Google Scholar] [CrossRef] [PubMed]
- Dawicki, W.; Li, C.; Town, J.; Zhang, X.; Gordon, J.R. Therapeutic reversal of food allergen sensitivity by mature retinoic acid-differentiated dendritic cell induction of LAG3(+)CD49b(-)Foxp3(-) regulatory T cells. J. Allergy Clin. Immunol. 2017, 139, 1608–1620.e1603. [Google Scholar] [CrossRef]
- Adorini, L. Tolerogenic dendritic cells induced by vitamin D receptor ligands enhance regulatory T cells inhibiting autoimmune diabetes. Ann. NY Acad. Sci. 2003, 987, 258–261. [Google Scholar] [CrossRef] [PubMed]
- Farias, A.S.; Spagnol, G.S.; Bordeaux-Rego, P.; Oliveira, C.O.; Fontana, A.G.; de Paula, R.F.; Santos, M.P.; Pradella, F.; Moraes, A.S.; Oliveira, E.C.; et al. Vitamin D3 induces IDO+ tolerogenic DCs and enhances Treg, reducing the severity of EAE. CNS Neurosci. Ther. 2013, 19, 269–277. [Google Scholar] [CrossRef]
- Correale, J.; Ysrraelit, M.C.; Gaitan, M.I. Vitamin D-mediated immune regulation in multiple sclerosis. J. Neurol. Sci. 2011, 311, 23–31. [Google Scholar] [CrossRef] [PubMed]
- Vanherwegen, A.S.; Eelen, G.; Ferreira, G.B.; Ghesquiere, B.; Cook, D.P.; Nikolic, T.; Roep, B.; Carmeliet, P.; Telang, S.; Mathieu, C.; et al. Vitamin D controls the capacity of human dendritic cells to induce functional regulatory T cells by regulation of glucose metabolism. J. Steroid Biochem. Mol. Biol. 2019, 187, 134–145. [Google Scholar] [CrossRef] [PubMed]
- Kaliyaperumal, A.; Michaels, M.A.; Datta, S.K. Antigen-specific therapy of murine lupus nephritis using nucleosomal peptides: Tolerance spreading impairs pathogenic function of autoimmune T and B cells. J. Immunol. 1999, 162, 5775–5783. [Google Scholar]
- Kang, H.K.; Liu, M.; Datta, S.K. Low-dose peptide tolerance therapy of lupus generates plasmacytoid dendritic cells that cause expansion of autoantigen-specific regulatory T cells and contraction of inflammatory Th17 cells. J. Immunol. 2007, 178, 7849–7858. [Google Scholar] [CrossRef]
- Elmann, A.; Sharabi, A.; Dayan, M.; Zinger, H.; Ophir, R.; Mozes, E. Altered gene expression in mice with lupus treated with edratide, a peptide that ameliorates the disease manifestations. Arthritis Rheum. 2007, 56, 2371–2381. [Google Scholar] [CrossRef]
- Sthoeger, Z.M.; Sharabi, A.; Molad, Y.; Asher, I.; Zinger, H.; Dayan, M.; Mozes, E. Treatment of lupus patients with a tolerogenic peptide, hCDR1 (Edratide): Immunomodulation of gene expression. J. Autoimmun. 2009, 33, 77–82. [Google Scholar] [CrossRef] [PubMed]
- Urowitz, M.B.; Isenberg, D.A.; Wallace, D.J. Safety and efficacy of hCDR1 (Edratide) in patients with active systemic lupus erythematosus: Results of phase II study. Lupus Sci. Med. 2015, 2, e000104. [Google Scholar] [CrossRef] [PubMed]
- Sharabi, A.; Mozes, E. The suppression of murine lupus by a tolerogenic peptide involves foxp3-expressing CD8 cells that are required for the optimal induction and function of foxp3-expressing CD4 cells. J. Immunol 2008, 181, 3243–3251. [Google Scholar] [CrossRef] [PubMed]
- Sela, U.; Sharabi, A.; Dayan, M.; Hershkoviz, R.; Mozes, E. The role of dendritic cells in the mechanism of action of a peptide that ameliorates lupus in murine models. Immunology 2009, 128, e395–e405. [Google Scholar] [CrossRef]
- Grohmann, U.; Orabona, C.; Fallarino, F.; Vacca, C.; Calcinaro, F.; Falorni, A.; Candeloro, P.; Belladonna, M.L.; Bianchi, R.; Fioretti, M.C.; et al. CTLA-4-Ig regulates tryptophan catabolism in vivo. Nat. Immunol. 2002, 3, 1097–1101. [Google Scholar] [CrossRef]
- Munn, D.H.; Sharma, M.D.; Mellor, A.L. Ligation of B7-1/B7-2 by human CD4+ T cells triggers indoleamine 2,3-dioxygenase activity in dendritic cells. J. Immunol. 2004, 172, 4100–4110. [Google Scholar] [CrossRef]
- Huang, C.; Zhang, L.; Ling, F.; Wen, S.; Luo, Y.; Liu, H.; Liu, J.; Zheng, W.; Liang, M.; Sun, J.; et al. Effect of immune tolerance induced by immature dendritic cells and CTLA4-Ig on systemic lupus erythematosus: An in vivo study. Exp. Ther. Med. 2018, 15, 2499–2506. [Google Scholar] [CrossRef]
- Group, A.T. Treatment of lupus nephritis with abatacept: the Abatacept and Cyclophosphamide Combination Efficacy and Safety Study. Arthritis Rheumatol. 2014, 66, 3096–3104. [Google Scholar] [CrossRef]
- Chamberlain, C.; Colman, P.J.; Ranger, A.M.; Burkly, L.C.; Johnston, G.I.; Otoul, C.; Stach, C.; Zamacona, M.; Dorner, T.; Urowitz, M.; et al. Repeated administration of dapirolizumab pegol in a randomised phase I study is well tolerated and accompanied by improvements in several composite measures of systemic lupus erythematosus disease activity and changes in whole blood transcriptomic profiles. Ann. Rheum. Dis. 2017, 76, 1837–1844. [Google Scholar] [CrossRef]
- Boumpas, D.T.; Furie, R.; Manzi, S.; Illei, G.G.; Wallace, D.J.; Balow, J.E.; Vaishnaw, A.; on behalf of the BG9588 Lupus Nephritis Trial Group. A short course of BG9588 (anti-CD40 ligand antibody) improves serologic activity and decreases hematuria in patients with proliferative lupus glomerulonephritis. Arthritis Rheum. 2003, 48, 719–727. [Google Scholar] [CrossRef]
- Barthels, C.; Ogrinc, A.; Steyer, V.; Meier, S.; Simon, F.; Wimmer, M.; Blutke, A.; Straub, T.; Zimber-Strobl, U.; Lutgens, E.; et al. CD40-signalling abrogates induction of RORgammat(+) Treg cells by intestinal CD103(+) DCs and causes fatal colitis. Nat. Commun. 2017, 8, 14715. [Google Scholar] [CrossRef] [PubMed]
- Pellerin, A.; Otero, K.; Czerkowicz, J.M.; Kerns, H.M.; Shapiro, R.I.; Ranger, A.M.; Otipoby, K.L.; Taylor, F.R.; Cameron, T.O.; Viney, J.L.; et al. Anti-BDCA2 monoclonal antibody inhibits plasmacytoid dendritic cell activation through Fc-dependent and Fc-independent mechanisms. EMBO Mol. Med. 2015, 7, 464–476. [Google Scholar] [CrossRef] [PubMed]
- Gardet, A.; Pellerin, A.; McCarl, C.A.; Diwanji, R.; Wang, W.; Donaldson, D.; Franchimont, N.; Werth, V.P.; Rabah, D. Effect of in vivo Hydroxychloroquine and ex vivo Anti-BDCA2 mAb Treatment on pDC IFNalpha Production From Patients Affected With Cutaneous Lupus Erythematosus. Front. Immunol. 2019, 10, 275. [Google Scholar] [CrossRef] [PubMed]
- Furie, R.; Werth, V.P.; Merola, J.F.; Stevenson, L.; Reynolds, T.L.; Naik, H.; Wang, W.; Christmann, R.; Gardet, A.; Pellerin, A.; et al. Monoclonal antibody targeting BDCA2 ameliorates skin lesions in systemic lupus erythematosus. J. Clin. Investig. 2019, 129, 1359–1371. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Smith, N.; Rodero, M.P.; Bekaddour, N.; Bondet, V.; Ruiz-Blanco, Y.B.; Harms, M.; Mayer, B.; Badder-Meunier, B.; Quartier, P.; Bodemer, C.; et al. Control of TLR7-mediated type I IFN signaling in pDCs through CXCR4 engagement-A new target for lupus treatment. Sci. Adv. 2019, 5, eaav9019. [Google Scholar] [CrossRef] [PubMed]
- Byun, S.H.; Lee, J.H.; Jung, N.C.; Choi, H.J.; Song, J.Y.; Seo, H.G.; Choi, J.; Jung, S.Y.; Kang, S.; Choi, Y.S.; et al. Rosiglitazone-mediated dendritic cells ameliorate collagen-induced arthritis in mice. Biochem. Pharmacol. 2016, 115, 85–93. [Google Scholar] [CrossRef] [PubMed]
- Zhao, F.; Xiao, C.; Evans, K.S.; Theivanthiran, T.; DeVito, N.; Holtzhausen, A.; Liu, J.; Liu, X.; Boczkowski, D.; Nair, S.; et al. Paracrine Wnt5a-beta-Catenin Signaling Triggers a Metabolic Program that Drives Dendritic Cell Tolerization. Immunity 2018, 48, 147–160.e147. [Google Scholar] [CrossRef]
- Shinde, R.; Hezaveh, K.; Halaby, M.J.; Kloetgen, A.; Chakravarthy, A.; da Silva Medina, T.; Deol, R.; Manion, K.P.; Baglaenko, Y.; Eldh, M.; et al. Apoptotic cell-induced AhR activity is required for immunological tolerance and suppression of systemic lupus erythematosus in mice and humans. Nat. Immunol. 2018, 19, 571–582. [Google Scholar] [CrossRef]
- Quintana, F.J.; Murugaiyan, G.; Farez, M.F.; Mitsdoerffer, M.; Tukpah, A.M.; Burns, E.J.; Weiner, H.L. An endogenous aryl hydrocarbon receptor ligand acts on dendritic cells and T cells to suppress experimental autoimmune encephalomyelitis. Proc. Natl. Acad. Sci. USA 2010, 107, 20768–20773. [Google Scholar] [CrossRef] [Green Version]
- Nguyen, N.T.; Kimura, A.; Nakahama, T.; Chinen, I.; Masuda, K.; Nohara, K.; Fujii-Kuriyama, Y.; Kishimoto, T. Aryl hydrocarbon receptor negatively regulates dendritic cell immunogenicity via a kynurenine-dependent mechanism. Proc. Natl. Acad. Sci. USA 2010, 107, 19961–19966. [Google Scholar] [CrossRef] [Green Version]
- Wang, H.; Li, T.; Chen, S.; Gu, Y.; Ye, S. Neutrophil Extracellular Trap Mitochondrial DNA and Its Autoantibody in Systemic Lupus Erythematosus and a Proof-of-Concept Trial of Metformin. Arthritis Rheumatol. 2015, 67, 3190–3200. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ferreira, G.B.; Vanherwegen, A.S.; Eelen, G.; Gutierrez, A.C.F.; Van Lommel, L.; Marchal, K.; Verlinden, L.; Verstuyf, A.; Nogueira, T.; Georgiadou, M.; et al. Vitamin D3 Induces Tolerance in Human Dendritic Cells by Activation of Intracellular Metabolic Pathways. Cell Rep. 2015, 10, 711–725. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shoenfeld, Y.; Isenberg, D.A.; Rauch, J.; Madaio, M.P.; Stollar, B.D.; Schwartz, R.S. Idiotypic cross-reactions of monoclonal human lupus autoantibodies. J. Exp. Med. 1983, 158, 718–730. [Google Scholar] [CrossRef]
- Sthoeger, Z.; Sharabi, A.; Asher, I.; Zinger, H.; Segal, R.; Shearer, G.; Elkayam, O.; Mozes, E. The tolerogenic peptide hCDR1 immunomodulates cytokine and regulatory molecule gene expression in blood mononuclear cells of primary Sjogren’s syndrome patients. Clin. Immunol. 2018, 192, 85–91. [Google Scholar] [CrossRef] [PubMed]
- Koshy, M.; Berger, D.; Crow, M.K. Increased expression of CD40 ligand on systemic lupus erythematosus lymphocytes. J. Clin. Investig. 1996, 98, 826–837. [Google Scholar] [CrossRef]
- Yellin, M.J.; D’Agati, V.; Parkinson, G.; Han, A.S.; Szema, A.; Baum, D.; Estes, D.; Szabolcs, M.; Chess, L. Immunohistologic analysis of renal CD40 and CD40L expression in lupus nephritis and other glomerulonephritides. Arthritis Rheum. 1997, 40, 124–134. [Google Scholar] [CrossRef]
- Elgueta, R.; Benson, M.J.; de Vries, V.C.; Wasiuk, A.; Guo, Y.; Noelle, R.J. Molecular mechanism and function of CD40/CD40L engagement in the immune system. Immunol. Rev. 2009, 229, 152–172. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dzionek, A.; Sohma, Y.; Nagafune, J.; Cella, M.; Colonna, M.; Facchetti, F.; Gunther, G.; Johnston, I.; Lanzavecchia, A.; Nagasaka, T.; et al. BDCA-2, a novel plasmacytoid dendritic cell-specific type II C-type lectin, mediates antigen capture and is a potent inhibitor of interferon alpha/beta induction. J. Exp. Med. 2001, 194, 1823–1834. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Harden, J.L.; Anderson, C.D.; Egilmez, N.K. Tolerogenic Phenotype of IFN-gamma-Induced IDO+ Dendritic Cells Is Maintained via an Autocrine IDO-Kynurenine/AhR-IDO Loop. J. Immunol. 2016, 197, 962–970. [Google Scholar] [CrossRef]
- Wculek, S.K.; Khouili, S.C.; Priego, E.; Heras-Murillo, I.; Sancho, D. Metabolic Control of Dendritic Cell Functions: Digesting Information. Front. Immunol. 2019, 10, 775. [Google Scholar] [CrossRef] [Green Version]

| Dendritic Cell Subsets | Surface Markers | Transcription Factors | Features of DC Subsets with Tolerogenic Functions | Mechanisms Involved in Regulatory T Cell Induction | Ref. |
|---|---|---|---|---|---|
| Mouse | |||||
| 1. Conventional DCs | |||||
| 1.1 Lymphoid DCs | |||||
| Resident cDC1 | CD11chi, CD8α+, CD11b−, Clec9a+, XCR1+, CD24+ | Zbtb46, BATF3, ID2, IRF8 | IDO+ cDC1 (induced via IFN-γ-inducible signaling) | IDO | [26,27] |
| BTLA+CD205+ cDC1 | BTLA and TGF-β | [28] | |||
| Resident cDC2 | CD11chi, CD8α−, CD11b+, DCIR2+, SIRPα+ | Zbtb46, IRF4 | No information | DCIR2 | [29,30] |
| IFN-γ-stimulated cDC2 | Kynurenine (IDO-independent) | [31] | |||
| 1.2 Non-lymphoid DCs | |||||
| Migratory cDC1 | CD11c+, CD103+, CD11b−, Clec9a+, XCR1+, CD24+ | Zbtb46, BATF3, ID2, IRF8 | Mucosal cDC1 | TGF-β, retinoic acid, IDO | [32,33] |
| Migratory cDC2 | CD11c+, CD103−, CD11b+, CCR2+, SIRPα+, CD301b+, CX3CR1+ | Zbtb46, IRF4 | Retinoic acid-producing cDC2 | ALDH, retinoic acid | [34] |
| PD-L2+ cDC2 | No information | [35] | |||
| 2. Non-conventional DCs | |||||
| pDC | CD11clo, B220+, Siglec-H+, BST2+ | E2-2 | IDO+ pDC | IDO | [36] |
| Human | |||||
| 1. Conventional DCs | |||||
| Myeloid cDC1 | CD141+, XCR1+, Clec9a+ | Zbtb46, BATF3, ID2, IRF8 | CD141+CD163+ DC-10 | IL-10, ILT4/HLA-G, IDO | [37,38,39] |
| Myeloid cDC2 | CD1c+, CD11b+, DCIR2+, SIRPα+, CX3CR1+ | Zbtb46, IRF4 | TLR4+ cDC2 | IDO, IL-10 | [40] |
| Pulmonary cDC2 | IL-10, IL-27, ICOSL | [41] | |||
| 2. Non-conventional DCs | |||||
| pDC | CD45RA+, CD123+, CD303+, CD304+ | E2-2 | IDO+ pDC | IDO | [36] |
| TLR9L-stimulated pDC | IDO | [42,43] | |||
| Drug/Biologics | Phenotype(s) of tolDCs | Function(s) of tolDCs | Study Model | Ref. |
|---|---|---|---|---|
| Ex vivo induction of tolDCs | ||||
| Vitamin D + Dexamethazone | High IL-10 | Treg induction | In vitro model in MoDCs from SLE patients | [12] |
| Rosiglitazone + Dexamethasone | Immature or semi-mature Resistant to autologous apoptotic cell-induced maturation | Inhibition of allogeneic CD4+ T cell response | In vitro model in MoDCs from SLE patients | [11] |
| L. delbrueckii L. rhamnosus | Semi-mature Increased IDO and IL-10 | - | In vitro model in MoDCs from SLE patients | [13] |
| In vivoinduction of tolDCs | ||||
| Tolerogenic nucleosomal peptide | Increased TGF-β | Treg expansion Contraction of Th17 Attenuation of autoreactive T cell and B cell | SNF1 mice | [148,149] |
| hCDR1 | Decreased IL-1β and TNF-α Increased IDO and TGF-β | Treg expansion Attenuation of autoreactive T and B cells | BWF1 mice Clinical trial in active SLE patients | [150,151,152,153,154] |
| CTLA4-Ig | Increased IDO | Treg induction Attenuation of autoreactive T and B cells | MRL-Faslpr mice Clinical trial in SLE patients | [155,156,157,158] |
| Anti-CD40L | - | Treg induction | DC-LMP1/CD40 mice (autoimmune colitis) Clinical trial in SLE patients | [159,160,161] |
| Anti-BCDA-2 | Decreased type I IFN in pDCs | - | In vitro model in pDCs from patients with CLE Clinical trial in SLE patients | [162,163,164] |
| CXCR4 agonist | Decreased type I IFN in pDCs | - | In vitro model in pDCs from SLE patients Pristane-induced lupus like model | [165] |
| PPARγ agonist | Immature or semi-matureIncreased FAO and IDO | Treg activation and induction | CIA mice Clinical trial in SLE patients | [166,167] |
| AhR agonist | Increased IL-10, kynurenins and retinoic acid Maintain IDO expression | Treg differentiation and expansion | In vitro model in MoM from patients with SLE and active SLE MRL-Faslpr mice Clinical trial in lupus arthritis and nephritis patients | [168,169,170] |
| AMPK activator | Increased OXPHOS and FAO | - | In vitro model in Human MoDCs Clinical trial in SLE patient as add-on treatment to treat lupus flare | [171,172] |
| Drug/ Biologics | Sponsors/ Clinical Trial No. | Year | Study Population | Summary of Results | Challenges/ Future Development | Ref. |
|---|---|---|---|---|---|---|
| Edratide (hCDR1) | Teva Pharmaceutical industries/ NCT00203151 | Sep 2005–Feb 2007 | Phase II trial in Patients with active SLE (N = 340) | -The primary endpoints based solely on SLEDAI-2K and AMS were not met. -Improved BILAG, the Composite SLE Responder Index | -Need more clinical study to confirm result | The Prelude study |
| Abatacept (CTLA4-Ig) | NIAID/NCT00774852 | Nov 2008–June 2014 | Phase II add-on trial in active lupus nephritis (N = 134) | -No statistically significant differences in % complete response at 24 or 52 weeks | -Might need to test with higher dose | The Access Trial Group [158] |
| Bristol-Myers Squibb/ NCT01714817 | Jan 2013–May 2018 | 2-year long-term extension phase III trial in 405 patients with active class III or IV lupus nephritis | -No statistically significant differences in % complete renal response which is primary outcome -Earlier and more robust response | -Might be useful in different subset of SLE patients | The Allure trial | |
| BG9588 (Anti-CD40L Antibody) | NIAMS/ NCT00001789 | June 1999–May 2000 | Phase I trial in lupus nephritis patients (N = 28) | -Terminated due to thromboembolic events -A short course of treatment showed some clinical improvement | -Additional studies will be needed to solve the issue of drug safety before evaluate its long-term effects | [160] |
| BIIB059 (humanized mAb to BDCA2) | Biogen/ NCT02106897 | April 2014–May 2016 | First in human trial Healthy control (N = 54), SLE with active skin lesions (N = 12) | -Favorable safety and PK profiles -Decreased expression of IFN response genes in blood -Reduced immune infiltrates in skin lesions | -Support the important role of pDC as target for skin manifestation | [164] |
| Biogen/ NCT02847598 | Oct 2016–Aug 2019 | Phase II clinical trial in SLE patients with active skin manifestations and CLE patients (N = 264) | No result posted yet | |||
| Thiazolidinediones (TZD) (PPARγ agonist) | NIAMS/ NCT02338999 | Jan 015 Status: Active | SLE patients, Plan to enroll 88 participants | No result posted yet | ||
| Laquinimod (AhR agonist) | Teva Pharmaceutical industries/ NCT01085084 | July 2010–Dec 2012 | Phase II clinical trial in SLE with active lupus arthritis (N = 82) | No result posted yet | ||
| Teva Pharmaceutical industries/ NCT01085097 | July2010–Dec2012 | Phase II clinical trial in SLE with active lupus nephritis (N = 47) | -62.5% of patients with active lupus nephritis received 0.5mg/day of laquinimod achieved renal response, compared to 33.3% of patients with placebo at 24 weeks | -Larger trial is needed to confirm result | https://www.tevapharm.com/ | |
| Metformin (AMPK activator) | Renji Hospital/ NCT02741960 | May2016–Dec2018 | Proof of concept trial of add-on metformin to conventional immunosuppressants in lupus flares in SLE patients (N = 113) | -Metformin as an add-on can reduce clinical flares, prednisone exposure, and body weight | -Promising choice, larger trial is needed to confirm result | [171] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ritprajak, P.; Kaewraemruaen, C.; Hirankarn, N. Current Paradigms of Tolerogenic Dendritic Cells and Clinical Implications for Systemic Lupus Erythematosus. Cells 2019, 8, 1291. https://doi.org/10.3390/cells8101291
Ritprajak P, Kaewraemruaen C, Hirankarn N. Current Paradigms of Tolerogenic Dendritic Cells and Clinical Implications for Systemic Lupus Erythematosus. Cells. 2019; 8(10):1291. https://doi.org/10.3390/cells8101291
Chicago/Turabian StyleRitprajak, Patcharee, Chamraj Kaewraemruaen, and Nattiya Hirankarn. 2019. "Current Paradigms of Tolerogenic Dendritic Cells and Clinical Implications for Systemic Lupus Erythematosus" Cells 8, no. 10: 1291. https://doi.org/10.3390/cells8101291





