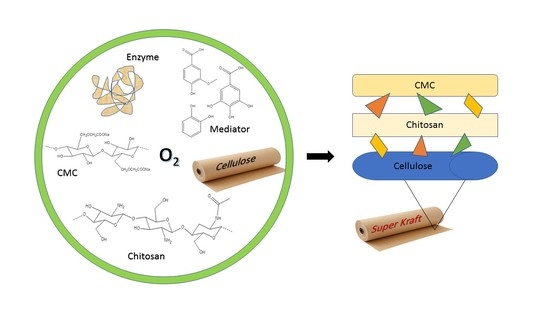
Effect of Laccase-Mediated Biopolymer Grafting on Kraft Pulp Fibers for Enhancing Paper’s Mechanical Properties
Abstract
:1. Introduction
2. Materials and Methods
2.1. Lacasse Biografting
2.2. Paper Properties Evaluation
2.2.1. Mechanical Tests
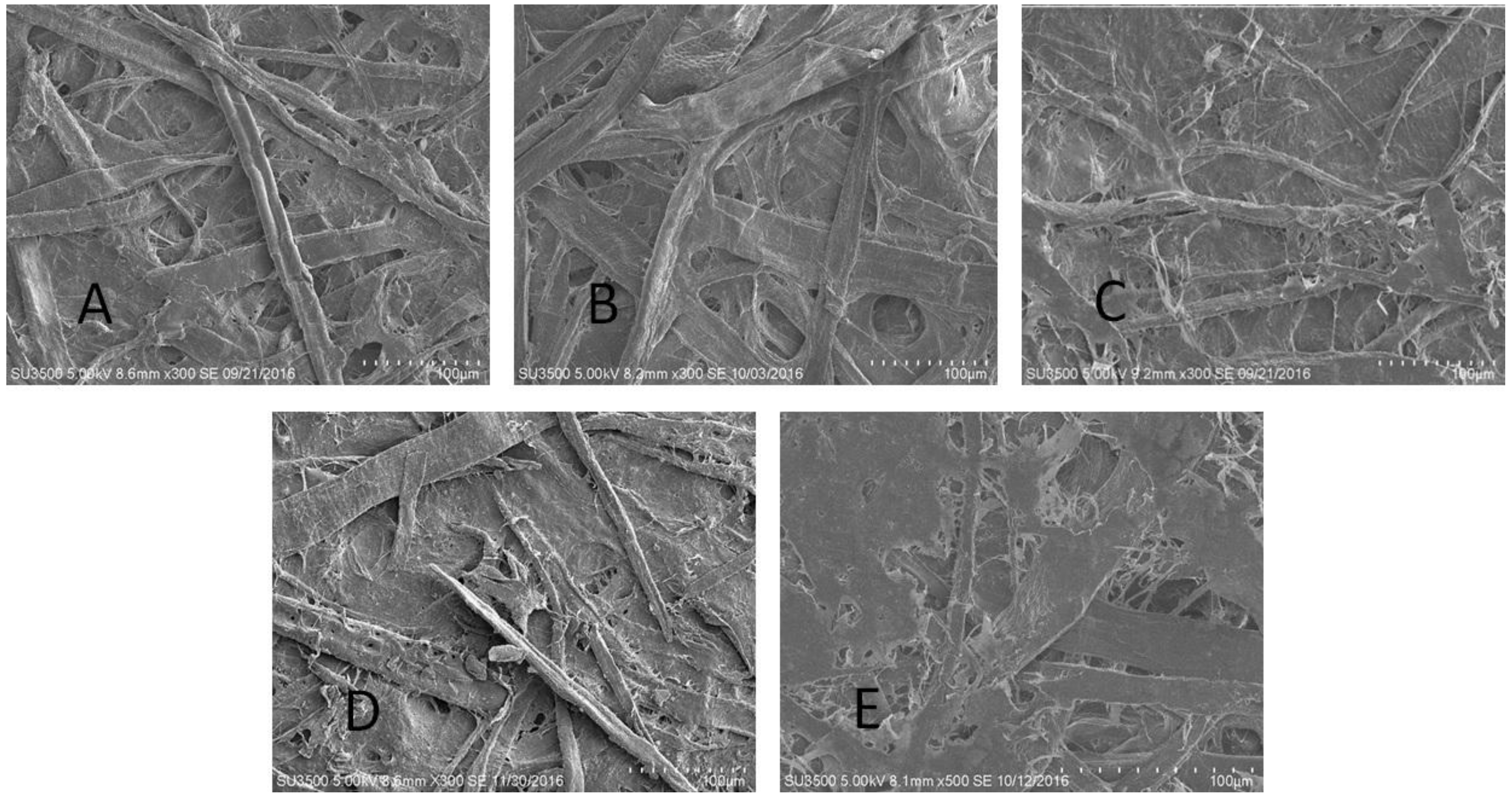
2.2.2. Scanning Electron Microscopy (SEM)–Energy Dispersive Spectrometry (EDS)
2.2.3. Confocal Laser Scanning Microscopy (CLSM)
2.2.4. Fourier-Transformed Infrared Spectroscopy (FTIR)
2.2.5. Nuclear Magnetic Resonance (13C NMR)
3. Results
3.1. Lacasse Biografting and Mechanical Properties
3.2. Paper Characterization
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Aracri, E.; Fillat, A.; Colom, F.J.; Gutierrez, A.; del Rio, C.J.; Martinez, T.A.; Vidal, T. Enzymatic grafting of simple phenols on flax and sisal pulp fibres using laccases. Bioresour. Technol. 2010, 101, 8211–8216. [Google Scholar] [CrossRef] [PubMed]
- Aracri, E.; Roncero, M.B.; Vidal, T. Studying the effects of laccase-catalysed grafting of ferulic acid on sisal pulp fibers. Bioresour. Technol. 2011, 102, 7555–7560. [Google Scholar] [CrossRef] [PubMed]
- Elegir, G.; Kindl, A.; Sadocco, P.; Orlandi, M. Development of antimicrobial cellulose packaging through laccase-mediated grafting of phenolic compounds. Enzym. Microb. Technol. 2008, 43, 84–92. [Google Scholar] [CrossRef]
- Widsten, P.; Kandelbauer, A. Laccase applications in the forest products industry: A review. Enzym. Microb. Technol. 2008, 42, 293–307. [Google Scholar] [CrossRef]
- Bourbonnais, R.; Paice, M. Oxidation of non-phenolic substrates: an expanded role for laccase in lignin biodegradation. FEBS Lett. 1990, 267, 99–102. [Google Scholar] [CrossRef]
- Kunamneni, A.; Camarero, S.; Garcia-Burgos, C.; Plou, F.J.; Ballesteros, A.; Alcalde, M. Engineering and Applications of fungal laccases for organic synthesis. Microb. Cell Fact. 2008, 7, 32. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sealey, J.; Ragauskas, A.J. Investigation of Laccase/NHydroxybenzotriazole Delignification of Kraft Pulp. J. Wood Chem. Technol. 1998, 18, 403–416. [Google Scholar] [CrossRef]
- Call, H.P.; Mucke, I. History, overview and applications of mediated lignolytic systems, especially laccase-mediator-systems (Lignozym(R)-process). J. Biotechnol. 1997, 53, 163–202. [Google Scholar] [CrossRef]
- Rathner, R.; Petz, S.; Tasnádi, G.; Koller, M.; Ribitsch, V. Monitoring the kinetics of biocatalytic removal of the endocrine disrupting compound 17a-ethinylestradiol from differently polluted wastewater bodies. J. Environ. Chem. Eng. 2017, 5, 1920–1926. [Google Scholar] [CrossRef]
- Baiocco, P.; Barreca, A.M.; Fabbrini, M.; Galli, C.; Gentili, P. Promoting laccase activity towards non phenolic substrates: A mechanistic investigation with some laccase mediator systems. Org. Biomol. Chem. 2003, 1, 191–197. [Google Scholar] [CrossRef] [PubMed]
- Gutierrez, A.; Rencoret, J.; Ibarra, D.; Molina, S.; Camarero, S.; Romero, J.; del Río, J.C.; Martínez, A.T. Removal of lipophilic extractives from paper pulp by laccase and lignin-derived phenols as natural mediators. Environ. Sci. Technol. 2007, 41, 4124–4129. [Google Scholar] [CrossRef] [PubMed]
- Cañas, A.; Camarero, S. Laccases and their natural mediators: Biotechnological tools for sustainable eco-friendly processes. Biothechnol. Adv. 2010, 28, 694–705. [Google Scholar] [CrossRef] [PubMed]
- Soares, G.; Amorim, P.; Costa-Ferreira, M. Use of laccase together with redox mediators to decolourize Remazol Brilliant Blue R. J. Biotechnol. 2001, 89, 123–129. [Google Scholar] [CrossRef]
- Camarero, S.; Ibarra, D.; Martínez, M.; Martínes, A. Lignin-Derived Compounds as Efficient Laccase Mediators for Decolorization of Different Types of Recalcitrat Dyes. Appl. Environ. Microbiol. 2005, 71, 1775–1784. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez, S.; Toca, J. Industrial and biothechnological applications of laccases: A review. Biotechnol. Adv. 2006, 24, 500–513. [Google Scholar] [CrossRef] [PubMed]
- Leonowicz, A.; Szklarz, G.; Wojtas-Wasilewska, M. The effect of fungal laccase on fractionated lignosulphonates (Peritan Na). Phytochemistry 1985, 24, 393–396. [Google Scholar] [CrossRef]
- Aracri, E.; Colom, J.F.; Vidal, T. Application of laccase-natural mediator systems to sisal pulp: An effective approach to biobleaching or functionalizing pulp fibres? Bioresour. Technol. 2009, 100, 5911–5916. [Google Scholar] [CrossRef] [PubMed]
- Chandra, R.P.; Felby, C.; Ragauskas, A.J. Improving laccase-facilitated grafting of 4-hydroxybenzoic acid to high-kappa kraft pulps. J. Wood Chem. Technol. 2005, 24, 69–81. [Google Scholar] [CrossRef]
- Holten-Andersen, N.; Harrington, M.J.; Birkedal, H.; Lee, B.P.; Messersmith, P.B.; Lee, K.Y.C.; Waite, J.H. pH-induced metal-lignad cross-links inspired by mussel yield self-healing polymer networks with near-covalent elastic moduli. Proc. Natl. Acad. Sci. USA 2011, 108, 2651–2655. [Google Scholar] [CrossRef] [PubMed]
- Barrett, D.G.; Fullenkamp, D.E.; He, L.; Holten-Andersen, N.; Lee, K.Y.C.; Messersmith, P.B. pH-Based Regulation of Hydrogel Mechanical properties Through Mussel-Inspired Chemistry and Processing. Adv. Funct. Mat. 2013, 23, 1111–1119. [Google Scholar] [CrossRef] [PubMed]
- Mattson, K.M.; Latimer, A.A.; McGrath, A.J.; Lynd, N.A.; Lundberg, P.; Hudson, Z.M.; Hawker, C.J. A Facile Synthesis of Catechol-Functionalized Poly(ethylene oxide) Block and Random Copolymers. J. Polym. Sci. Pol. Chem. 2015, 53, 2685–2692. [Google Scholar] [CrossRef]
- Shin, M.; Ryu, J.H.; Park, J.P.; Kim, K.; Yang, J.W.; Lee, H. DNA/Tannic Acid Hybrid Gel Exhibiting Biodegradability, Extensibility, Tissue Adhesiveness, and Hemostatic Ability. Adv. Funct. Mat. 2015, 25, 1270–1278. [Google Scholar] [CrossRef]
- Oh, D.X.; Kim, S.; Lee, D.; Hwang, D.S. Tunicate-mimetic nanofibrous hydrogel adhesive with improved wet adhesion. Acta Biomater. 2015, 20, 104–112. [Google Scholar] [CrossRef] [PubMed]
- Zhan, K.; Kim, C.; Sung, K.; Ejima, H.; Yoshie, N. Tunicate-Inspired Gallol Polymers for Underwater Adhesive: A Comparative Study of Catechol and Gallol. Biomacromolecules 2017, 18, 2959–2966. [Google Scholar] [CrossRef] [PubMed]
- Felby, C.; Nielsen, B.; Olesen, P.; Skibsted, L. Identification and quantification of radical reaction intermediates by electron spin resonance spectrometry of laccase-catalyzed oxidation of wood fibers from beech. Appl. Microbiol. Biotechnol. 1997, 48, 459–464. [Google Scholar] [CrossRef]
- Chandra, R.P.; Lehtonen, L.K.; Ragauskas, A.J. Modification of high lignin content kraft pulps with laccase to improve paper strength properties. 1. Laccase treatment in the presence of gallic acid. Biotechnol. Prog. 2004, 20, 255–261. [Google Scholar] [CrossRef] [PubMed]
- Chandra, R.P.; Ragauskas, A.J. Modification of High-Lignin Kraft Pulps with Laccase. Part 2. Xylanase-Enhanced Strength Benefits. Biotechnol. Prog. 2005, 21, 1302–1306. [Google Scholar] [CrossRef] [PubMed]
- Chandra, R.P.; Ragauskas, A.J. Evaluating laccase-facilitated coupling of phenolic acids to high-yield kraft pulps. Enzym. Microb. Technol. 2002, 30, 855–861. [Google Scholar] [CrossRef]
- Espoo, J.B.; Rajamaki, H.M.; Rajamaki, S.P.; Helsinki, L.V.; Tolkkinen., S.G.; Espoo, T.O.; Espoo, A.S. Process for Producing a Fibrous Product. U.S. Patent 2007/0131362 A1, 14 June 2007. [Google Scholar]
- Buckman Laboratories Inc. Mater Safety Data Sheet; Buckman Laboratories Inc.: S.A. de C.V., Mexico, 2009. [Google Scholar]
- Camarero, S.; Ibarra, D.; Martínez, Á.; Romero, J.; Gutiérrez, A.; del Rio, J. Paper pulp delignification using laccase and natural mediators. Enzym. Microb. Technol. 2007, 40, 1264–1271. [Google Scholar] [CrossRef] [Green Version]
- Ibarra, D.; Camarero, S.; Romero, J.; Martínez, M.; Martínez, A. Integrating laccase–mediator treatment into an industrial-type sequence for totally chlorine-free bleaching of eucalypt kraft pulp. J. Chem. Technol. Biotechnol. 2006, 81, 1159–1165. [Google Scholar] [CrossRef]
- Roth, S.; Spiess, A.C. Laccases for biorefinery applications: A critical review on challenges and perspectives. Bioprocess Biosyst. Eng. 2015, 38, 2285–2313. [Google Scholar] [CrossRef] [PubMed]
- Morozova, O.V.; Shumakovich, G.P.; Gorbacheva, M.A.; Shleev, S.V.; Yaropolov, A.I. “Blue” Laccases. J. Biochem. 2007, 72, 1136–1150. [Google Scholar] [CrossRef]
- Fabbrini, M.; Galli, C.; Gentili, P. Comparing the catalytic efficiency of some mediators of laccase. J. Mol. Catal. B Enzym. 2002, 16, 231–240. [Google Scholar] [CrossRef]
- Galli, C.; Gentili, P. Chemical messengers: Mediated oxidations with the enzyme laccasey. J. Phys. Organ. Chem. 2004, 17, 973–977. [Google Scholar] [CrossRef]
- Kilmartin, P. Electrochemical Detection of Natural Antioxidants: Principles and Protocols. Antioxid. Redox Signal. 2001, 3, 941–955. [Google Scholar] [CrossRef] [PubMed]
- Lund, M.; Felby, C. Wet strength improvement of unbleached kraft pulp through laccase catalyzed oxidation. Enzym. Microb. Technol. 2001, 28, 760–765. [Google Scholar] [CrossRef]
- Cannatelli, M.D.; Ragauskas, A.J. Conversion of lignin into value-added materials and chemicals via laccase-assisted copolymerization. Appl. Microbiol. Biotechnol. 2016, 100, 8685–8691. [Google Scholar] [CrossRef] [PubMed]
- Vila, C.; Barneto, A.G.; Fillat, A.; Vidal, T.; Ariza, J. Use of thermogravimetric analysis to monitor the effects of natural laccase mediators on flax pulp. Bioresour. Technol. 2011, 102, 6554–6561. [Google Scholar] [CrossRef] [PubMed]
- Crouvisier-Urion, K.; Bodart, P.R.; Winckler, P.; Raya, J.; Gougeon, R.D.; Cayot, P.; Domenek, S.; Debeaufort, F.; Karbowiak, T. Biobased Composite Films from Chitosan and Lignin: Antioxidant Activity Related to Structure and Moisture. ACS Sustain. Chem. Eng. 2016, 4, 6371–6381. [Google Scholar] [CrossRef]
- Thakur, K.; Kalia, S.; Kaith, B.S.; Pathania, D.; Kumar, A. Surface functionalization of coconut fibers by enzymatic biografting of syringaldehyde for the development of biocomposites. RSC Adv. 2015, 5, 76844–76851. [Google Scholar] [CrossRef]
- Thakur, K.; Kalia, S.; Pathania, D.; Kumar, A.; Sharmac, N.; Schauer, C.L. Surface functionalization of lignin constituent of coconut fibers via laccase-catalyzed biografting for development of antibacterial and hydrophobic properties. J. Clean. Prod. 2016, 113, 176–182. [Google Scholar] [CrossRef]
- Wysokowski, M.; Klapiszewski, Ł.; Moszyński, D.; Bartczak, P.; Szatkowski, T.; Majchrzak, I.; Siwińska-Stefańska, K.; Bazhenov, V.V.; Jesionowski, T. Modification of Chitin with Kraft Lignin and Development of New Biosorbents for Removal of Cadmium(II) and Nickel(II) Ions. Mar. Drugs 2014, 12, 2245–2268. [Google Scholar] [CrossRef] [PubMed]


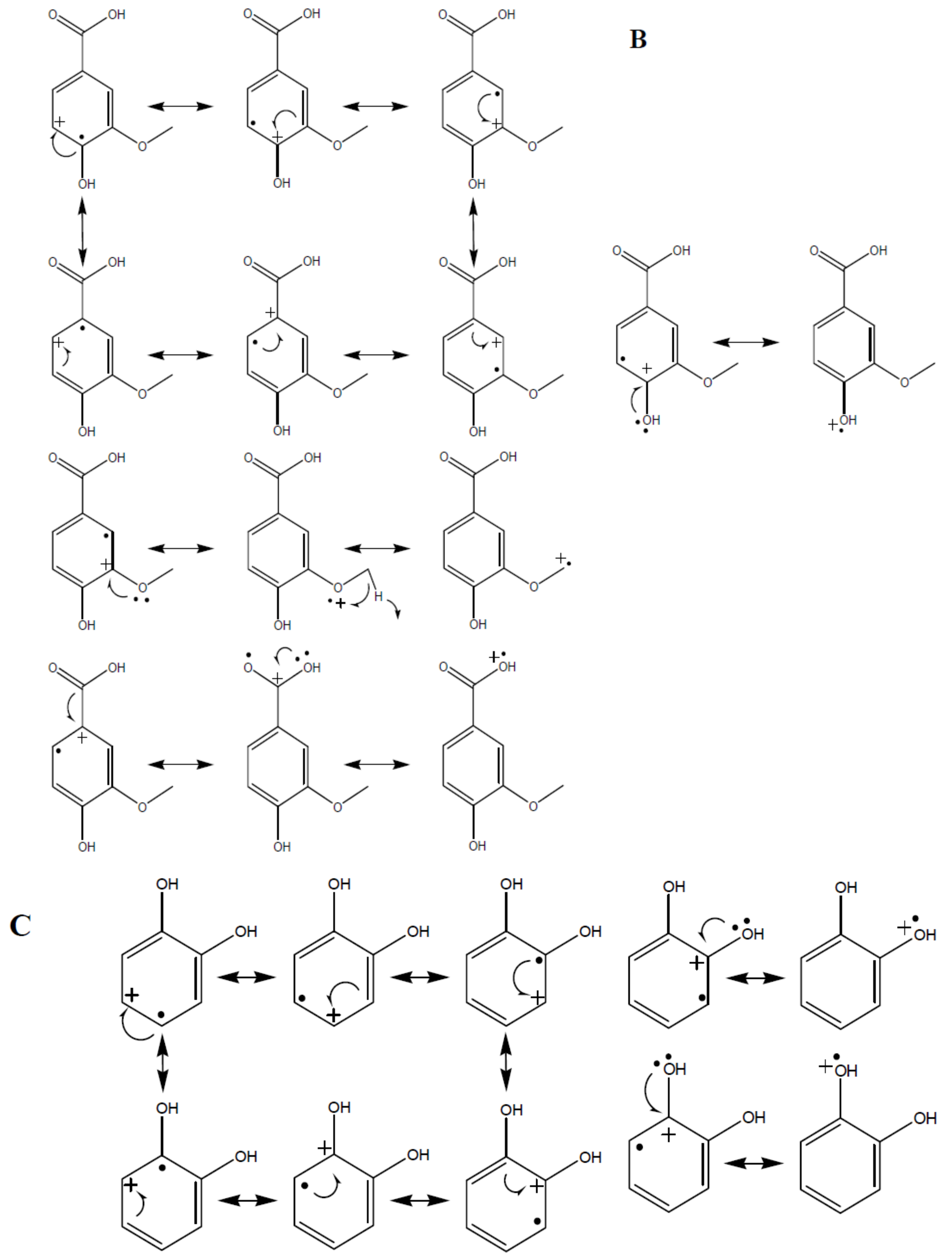

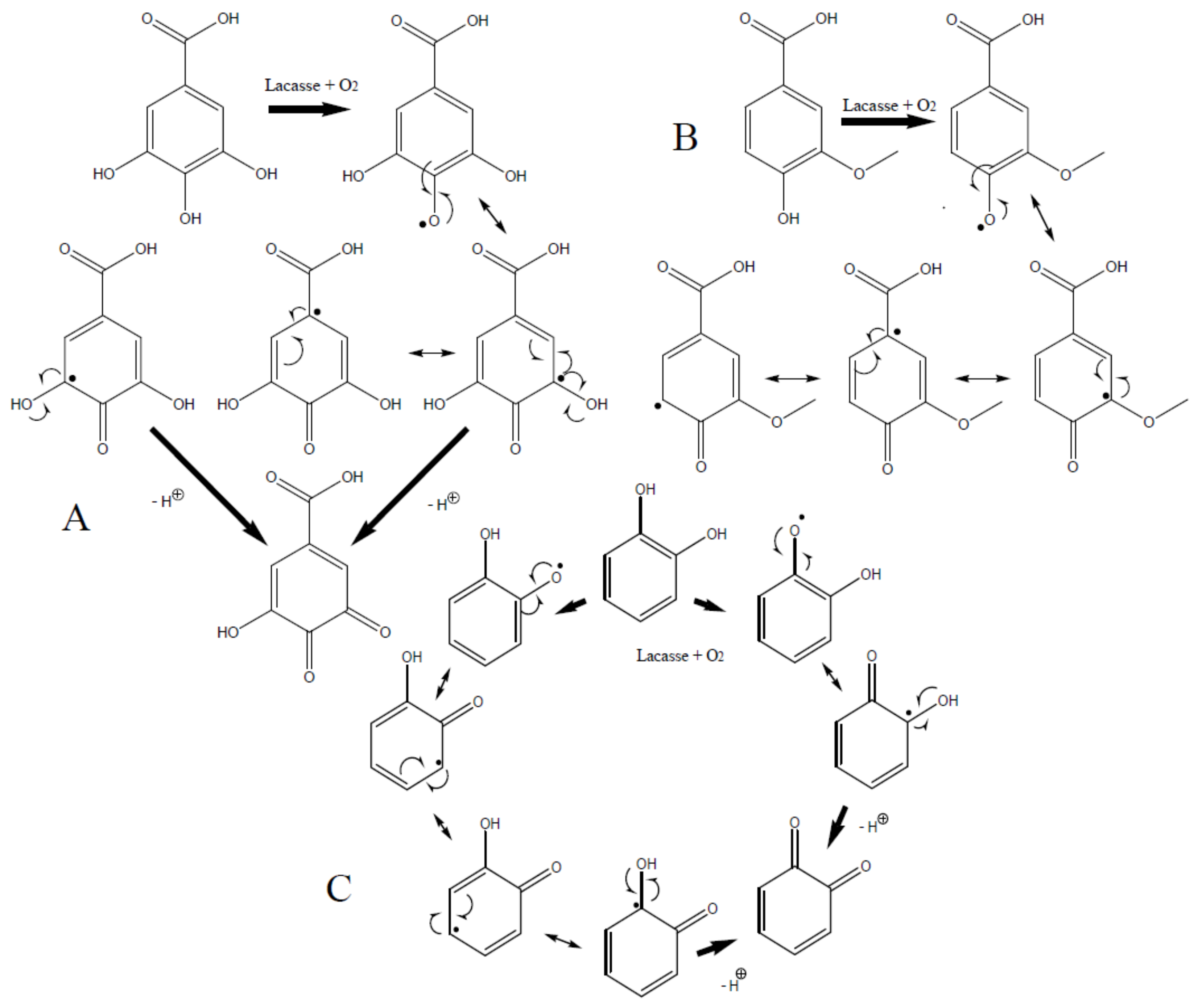
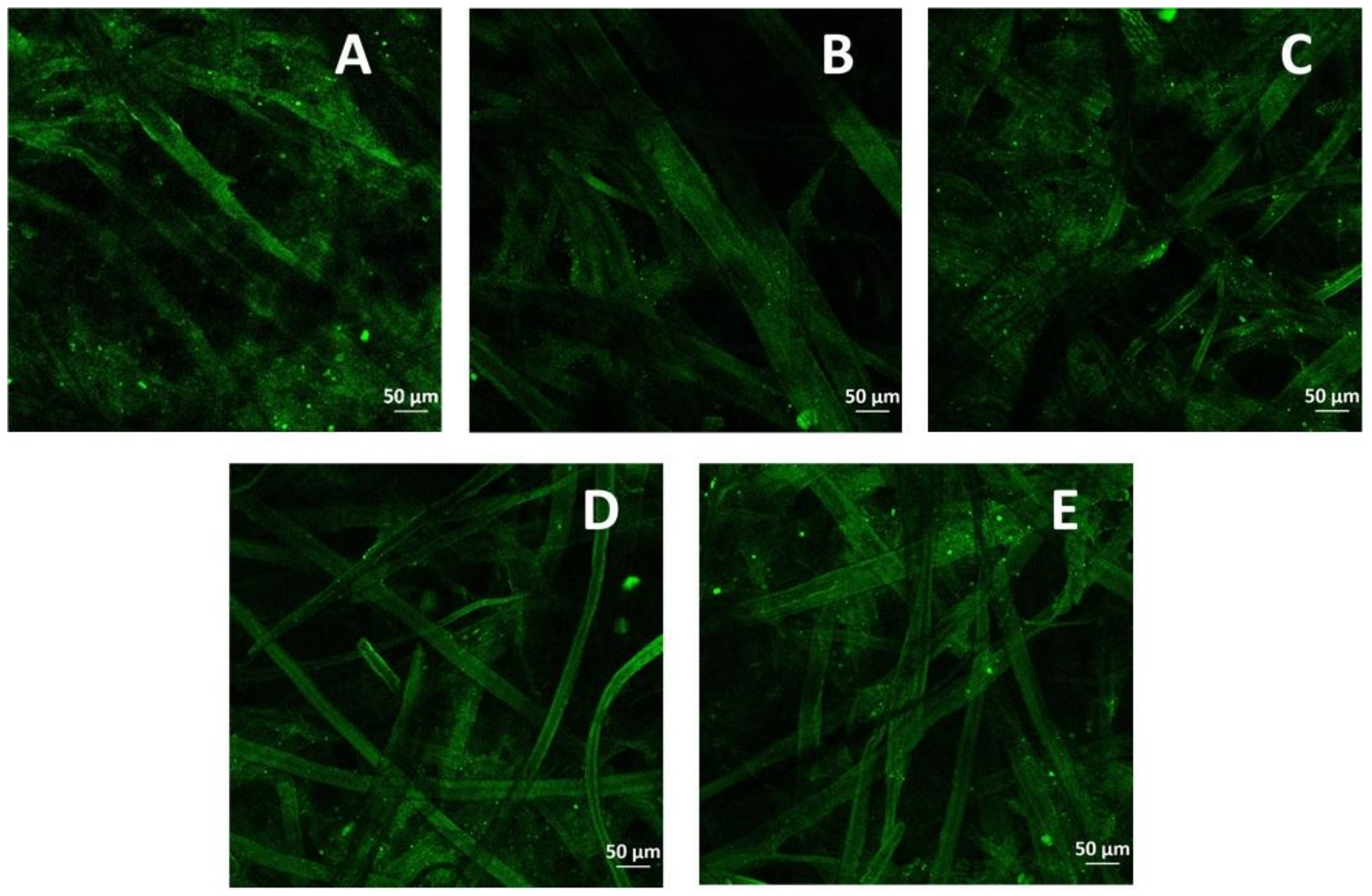
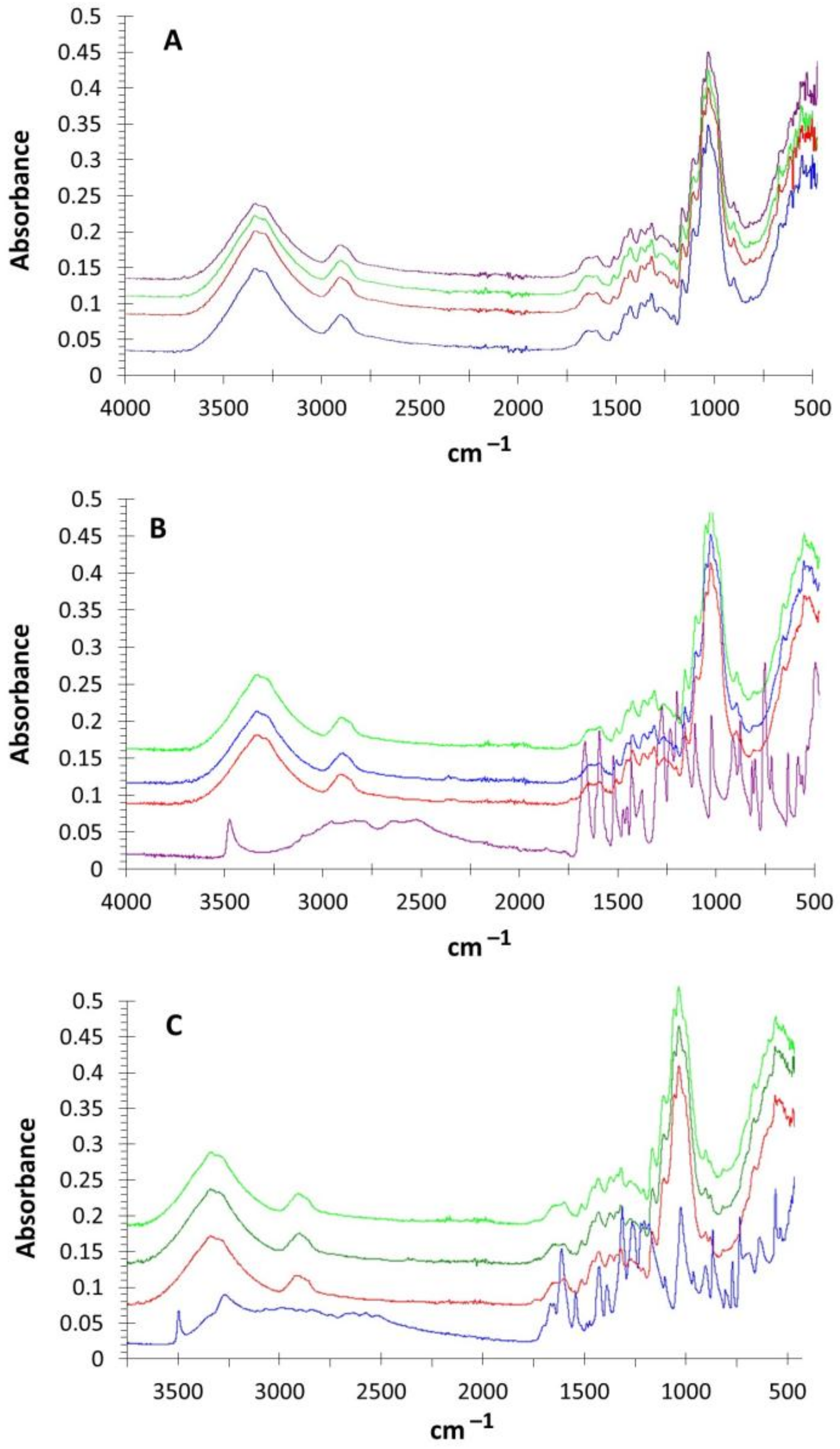
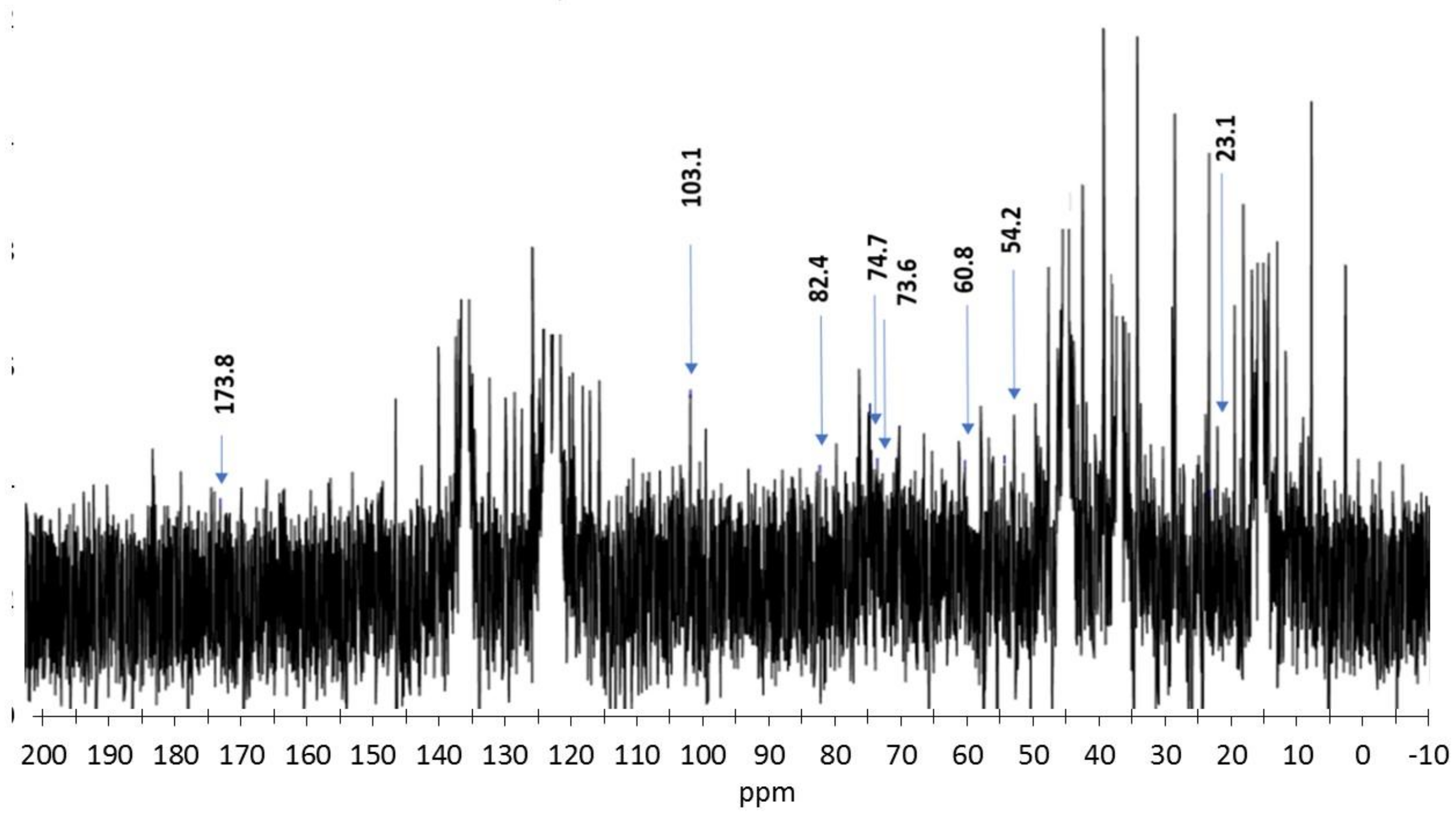
| Mediator | CPX * × 103 | CMT * lb/6 Pulg | Ring crush lb/6 Pulg | Mullen lb/6 Pulg2 | Tension kg/15 mm | ΔE (mV) | Thermal Profile |
|---|---|---|---|---|---|---|---|
| Gallic acid | 0 | 50.03 | 35.14 | 59.93 | 11.10 | 42.2 | −5% HA * |
| Gallic acid | 5 | 52.53 | 35.87 | 92.45 | 8.48 | −44.4 | +4% CF * |
| Vanillic acid | 0 | 49.01 | 27.44 | 75.10 | 9.33 | 66 | +4% CF |
| Vanillic acid | 5 | 57.08 | 43.07 | 77.16 | 10.17 | 4.4 | |
| Catechol | 0 | 46.38 | 35.93 | 65.83 | 11.24 | 41.6 | No change |
| Catechol | 5 | 64.88 | 34.13 | 71.47 | 8.74 | −21.3 | |
| Blank | 0 | 41.0 | 24.8 | 41 | 5.70 | NA * | Reference |
| Functional Group | Vila, 2011 [40] | Functional Group | Thakur, 2015 [42] | Functional Group | Thakur, 2016 [43] |
|---|---|---|---|---|---|
| Amorphous cellulose | 897 cm−1 | Vibration C=O in different spatial direction | 944 cm−1 | Stretching C–H out of plane | 852 cm−1 |
| Amorphous cellulose | 1273 cm−1 | Stretching C–O by grafting | 1244 cm−1 | Asymmetric stretch ether C–O–C | 1034 cm−1 |
| Crystalline cellulose | 1370 cm−1 | C=O vibration in different spatial direction | 1327 cm−1 | Carbonyl stretch C=O | 1276 cm−1 |
| Crystalline cellulose | 1430 cm−1 | C=O vibration in different spatial direction | 1449 cm−1 | Methoxy group C–O–CH3 | 1379 cm−1 |
| Lignin | 1460 cm−1 | Aromatic skeletal vibrations | 1449 cm−1 | Methoxy group C–O–CH3 | 1466 cm−1 |
| Lignin | 1510cm−1 | Aromatic skeletal vibrations | 1512 cm−1 | Aromatic stretching C–C bond | 1516 cm−1 |
| Lignin | 1600 cm−1 | Double substitution p-aromatic ring | 1602 cm−1 | Carbonyl stretching C–C bond | 1600 cm−1 |
| Stretching C–H | 2900 cm−1 | Double substitution p-aromatic ring | 1628 cm−1 | C=O by phenolic insertion | 1665 cm−1 |
| Face, Iα | 3267 cm−1 | Carbonyl group C=O | 1673 cm−1 | C=O by phenolic insertion | 1691 cm−1 |
| Vibrations O–H | 3290 cm−1 | Stretching C–H | 2924 cm−1 | Carboxyl group | 2500–3000cm−1 |
| Vibrations O–H | 3333 cm−1 | Vibrations O–H | 3380 cm−1 |
| Signal no. | Sample ppm | Lignin Characterization Reference, ppm | Functional Group Assignment |
|---|---|---|---|
| 1 | 173.82 | 173.6–173.8 | N–C–O (C8) in chitosan |
| 2 | 146.99 | 146.8 | C4 in non-esterified lignin guayaquil |
| 3 | 140.49 | 140.8–140.4 | Structure lignin A(4), B(1) and D(1) |
| 4 | 137.89 | 138.2 | C1 from esterified lignin syrigil |
| 5 | 132.76 | 132.4–132.6 | C5 in ether bond 5-5 |
| 6 | 128.98 | 129.3 | Cβ in lignin aryl group–CH=CH–CHO |
| 7 | 127.89 | 128.0 | Cα y Cβ in lignin aryl group–CH=CH–CH2OH |
| 8 | 126.24 | 125.9 | C5 in non-esterified lignin bond 5-5 no |
| 9 | 120.68 | 120.5–120.7 | A6, B6 from lignin |
| 10 | 120.14 | 119.4 | C6 in a guayaquil lignin unit |
| 11 | 118.65 | 118.4 | C6 in a guayaquil lignin unit |
| 12 | 116.11 | 116.5 | B5 from lignin |
| 13 | 103.14 | 102.7–105.7 | C1 chitosan glucosidic bond |
| 14 | 82.46 | 80.9–85.7 | C4 in chitosan glucosidic ring |
| 15 | 76.79 | 76.5–77.1 | Cα in β-1 (1,2 disyringilpropane-1,3-diol acetate) from lignin |
| 16 | 75.37 | 75.5 | Cα in guayaquil-O-aryl from lignin |
| 17 | 74.73 | 74.7 | C5 chitosan glucosidic bond |
| 18 | 73.61 | 73.1–75.7 | C3 chitosan glucosidic bond |
| 19 | 60.84 | 59.6–60.8 | C6 chitosan glucosidic bond |
| 20 | 54.23 | 55.2–57.6 | C2 in chitosan glucosidic ring bonded to amine/amide |
| 21 | 39.74 | 41.0–41.2 | Cα in β–β lignin open bond |
| 22 | 29.38 | 29.2 | CH2 in lignin aliphatic lateral chain |
| 23 | 23.12 | 22.8–23.3 | N–CH3 (C7) in chitosan |
| 24 | 19.88 | 20.5 | CH3 acetoxy from lignin |
| 25 | 13.43 | 14.0 | γCH3 n-propyl from lignin side chain |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ballinas-Casarrubias, L.; Villanueva-Solís, L.; Espinoza-Hicks, C.; Camacho-Dávila, A.; Castillo, H.A.P.; Pérez, S.B.; Villa, E.D.; Hernández, M.D.D.; González-Sánchez, G. Effect of Laccase-Mediated Biopolymer Grafting on Kraft Pulp Fibers for Enhancing Paper’s Mechanical Properties. Polymers 2017, 9, 570. https://doi.org/10.3390/polym9110570
Ballinas-Casarrubias L, Villanueva-Solís L, Espinoza-Hicks C, Camacho-Dávila A, Castillo HAP, Pérez SB, Villa ED, Hernández MDD, González-Sánchez G. Effect of Laccase-Mediated Biopolymer Grafting on Kraft Pulp Fibers for Enhancing Paper’s Mechanical Properties. Polymers. 2017; 9(11):570. https://doi.org/10.3390/polym9110570
Chicago/Turabian StyleBallinas-Casarrubias, Lourdes, Luis Villanueva-Solís, Carlos Espinoza-Hicks, Alejandro Camacho-Dávila, Hilda Amelia Piñón Castillo, Samuel B. Pérez, Eduardo Duarte Villa, Miguel De Dios Hernández, and Guillermo González-Sánchez. 2017. "Effect of Laccase-Mediated Biopolymer Grafting on Kraft Pulp Fibers for Enhancing Paper’s Mechanical Properties" Polymers 9, no. 11: 570. https://doi.org/10.3390/polym9110570