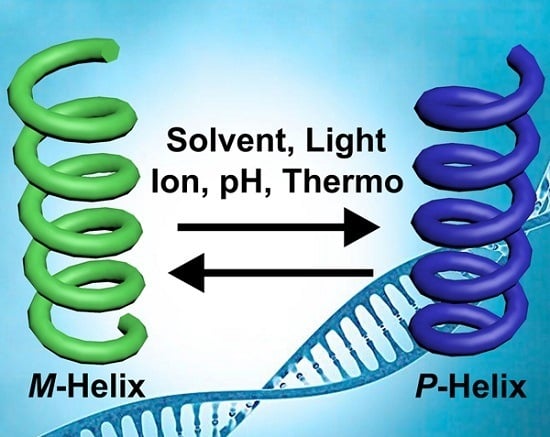
Stimuli-Directed Helical Chirality Inversion and Bio-Applications
Abstract
:1. Introduction
2. Stimuli-Directed Helical Chirality Inversion
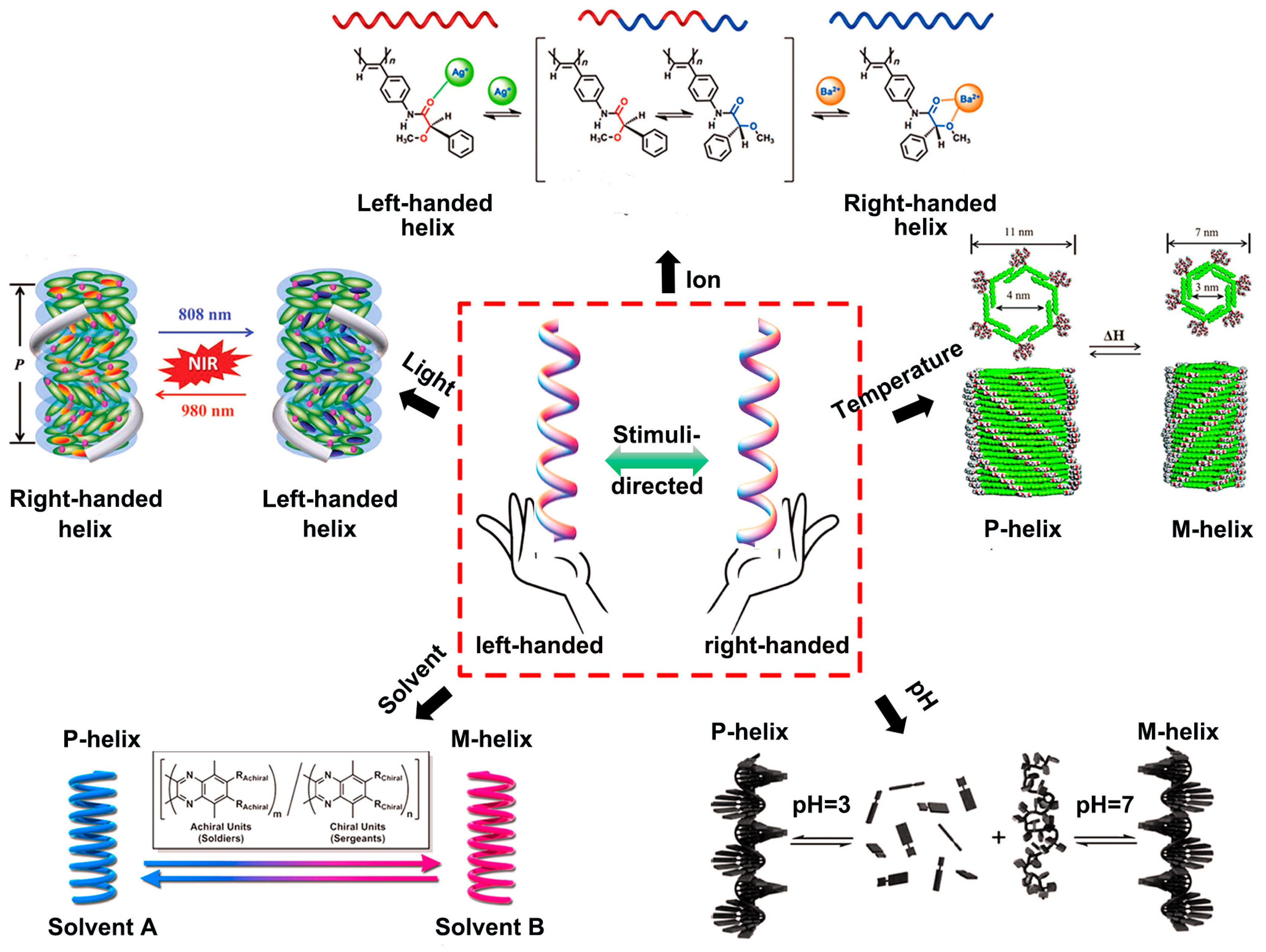
2.1. Solvent-Directed Helical Chirality Inversion
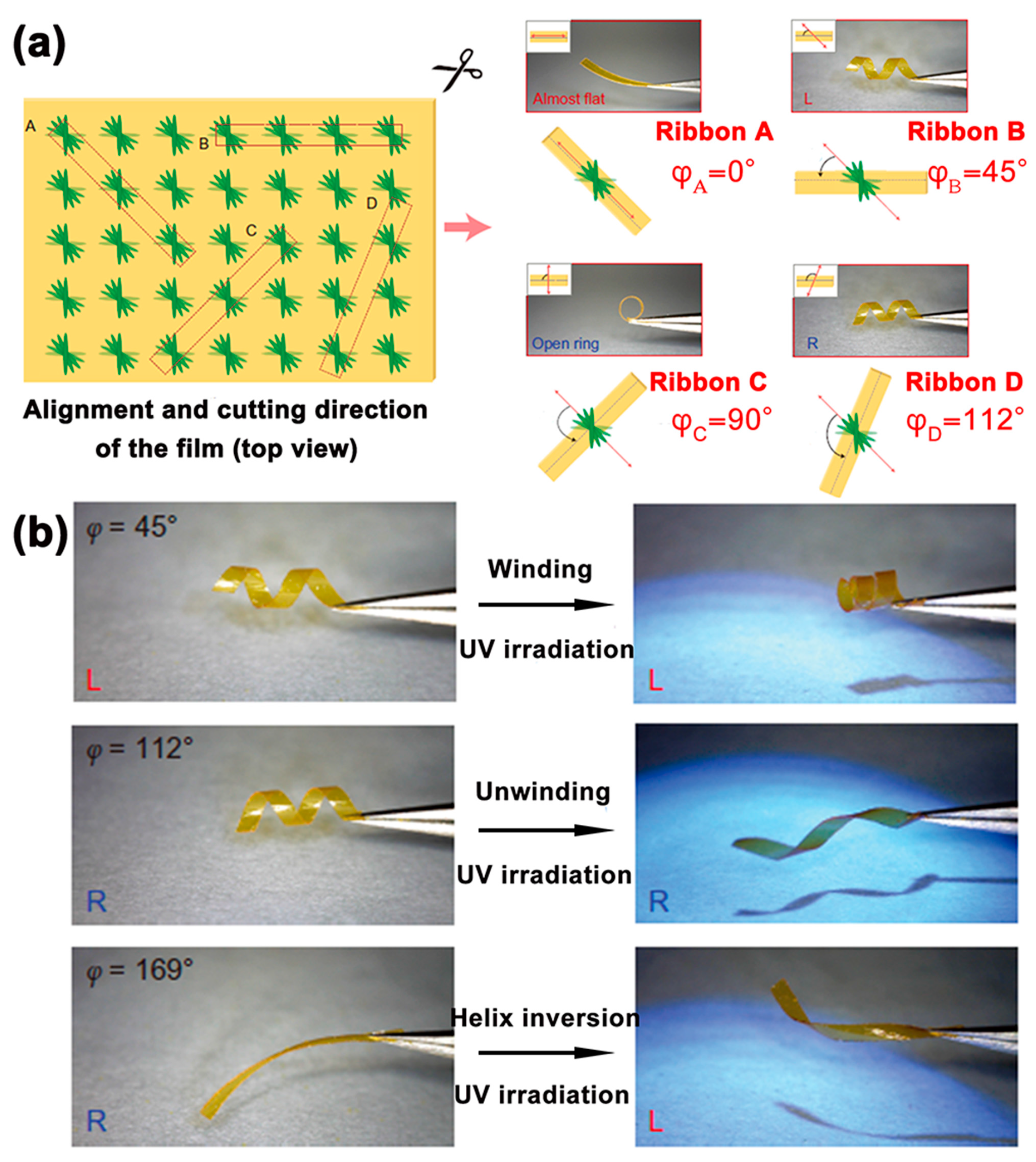
2.2. Photo-Directed Helical Chirality Inversion
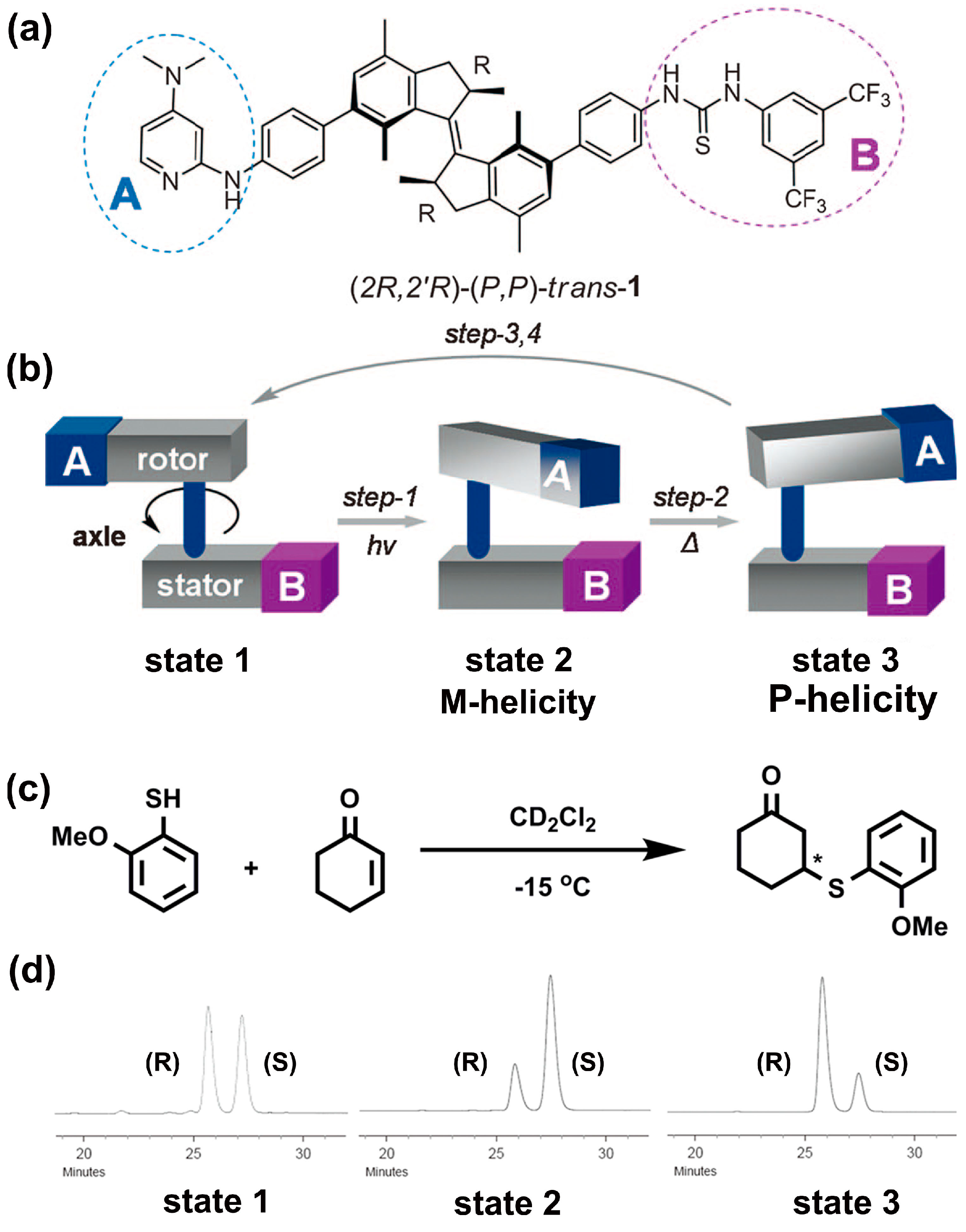
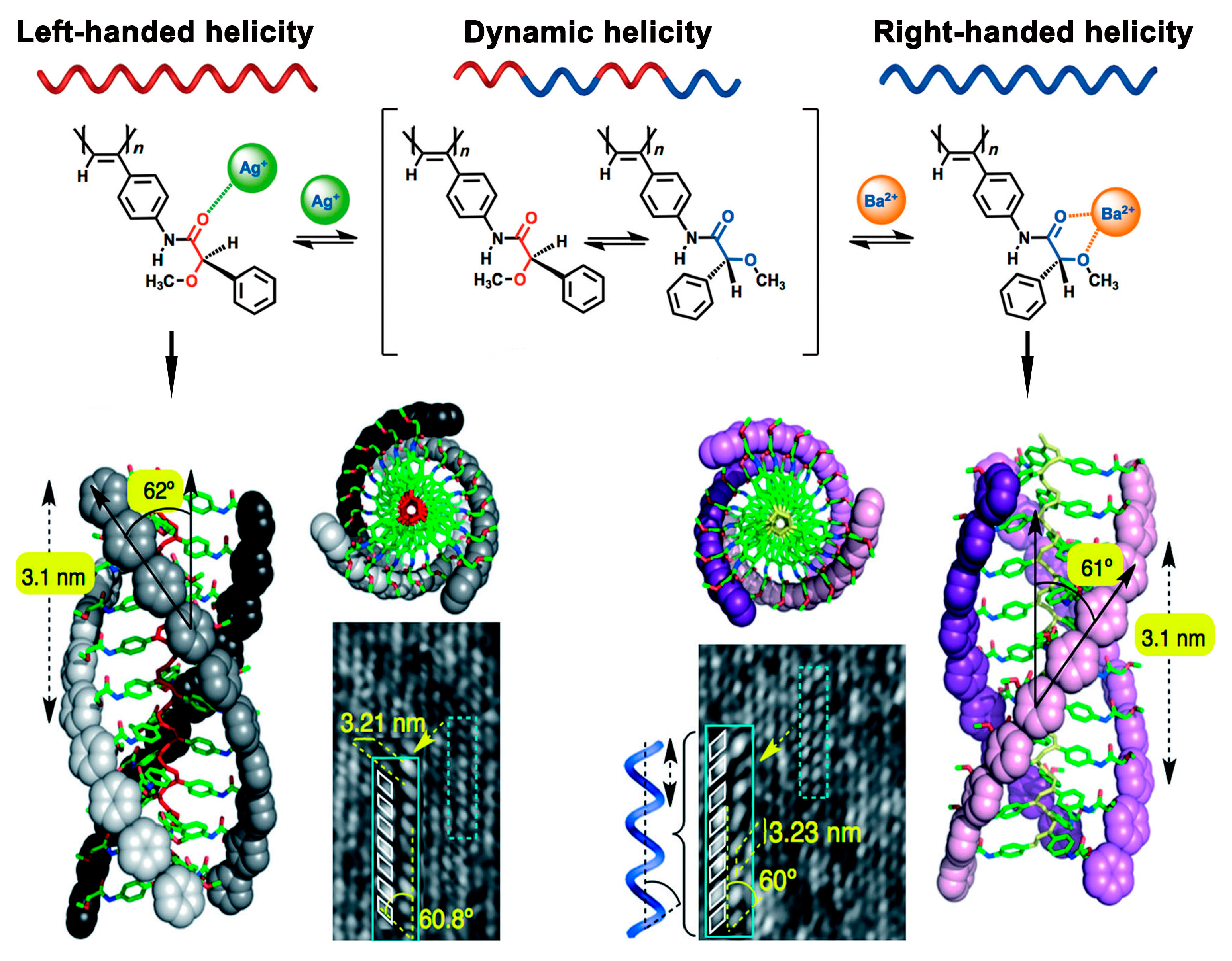
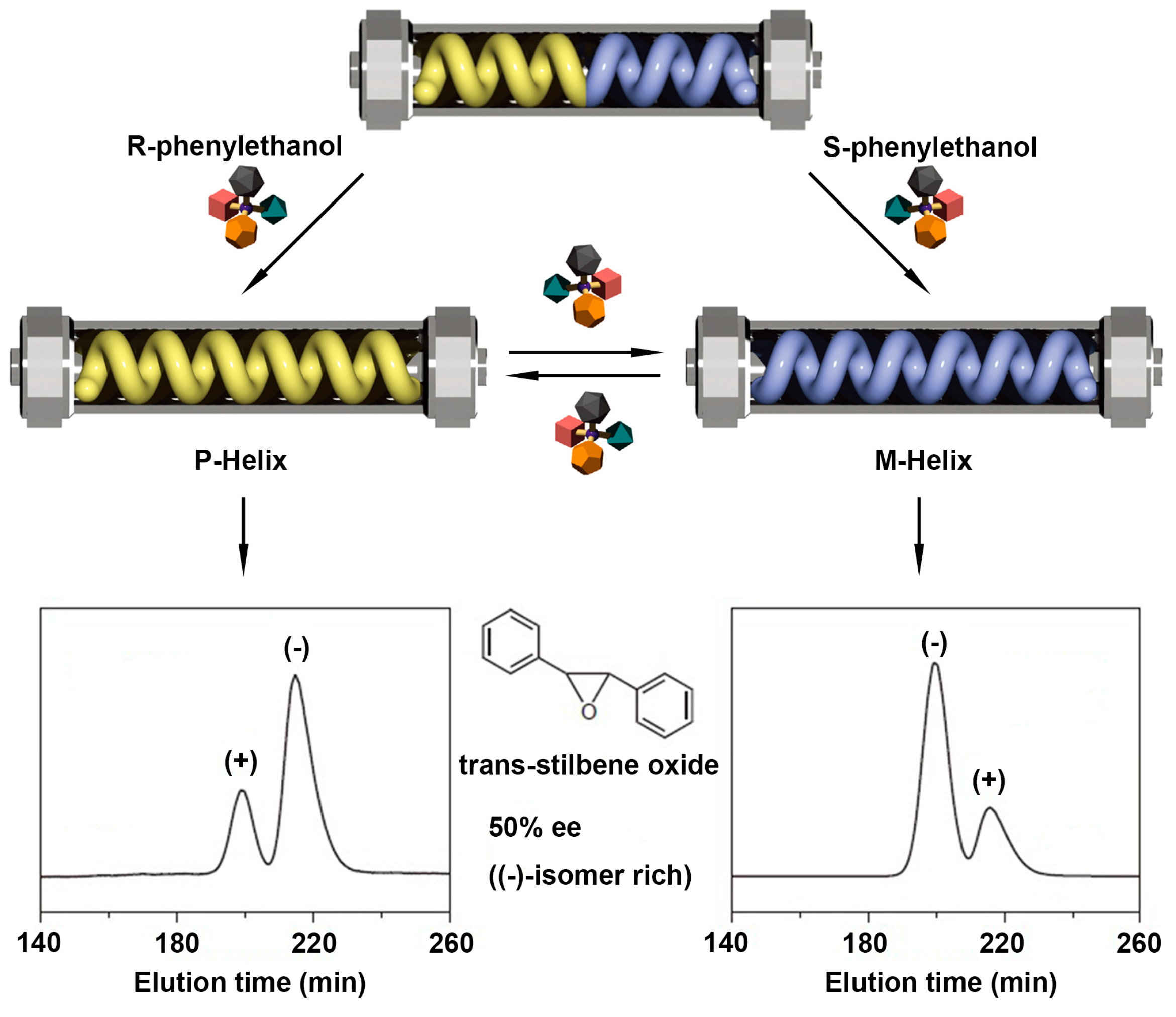
2.3. Ion-Directed Helical Chirality Inversion
2.4. pH-Directed Helical Chirality Inversion
2.5. Thermo-Directed Helical Chirality Inversion
3. Bio-Application of Helicity-Controllable Polymer
4. Conclusions and Outlook
Acknowledgments
Conflicts of Interest
References
- Barron, L.D. Chirality and life. Space Sci. Rev. 2008, 135, 187–201. [Google Scholar] [CrossRef]
- Pauling, L.; Corey, R.B.; Branson, H.R. The structure of proteins: Two hydrogen-bonded helical configurations of the polypeptide chain. Proc. Natl. Acad. Sci. USA 1951, 37, 205–211. [Google Scholar] [CrossRef] [PubMed]
- Nakano, T.; Okamoto, Y. Synthetic helical polymers: Conformation and function. Chem. Rev. 2001, 101, 4013–4038. [Google Scholar] [CrossRef] [PubMed]
- Yashima, E.; Maeda, K.; Iida, H.; Furusho, Y.; Nagai, K. Helical Polymers: Synthesis, Structures, and Functions. Chem. Rev. 2009, 109, 6102–6211. [Google Scholar] [CrossRef] [PubMed]
- Jockhart, D.J.; Winzeler, E.A. Genomics, gene expression and DNA arrays. Nature 2000, 405, 827–836. [Google Scholar] [CrossRef] [PubMed]
- Zotti, M.D.; Formaggio, F.; Crisma, M.; Peggion, C.; Moretto, A.; Toniolo, C. Handedness preference and switching of peptide helices. Part I: Helices based on protein amino acids. J. Pept. Sci. 2014, 20, 307–322. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Christensen, L.A.; Vasquez, K.M. Z-DNA-forming sequences generate large-scale deletions in mammalian cells. Proc. Natl. Acad. Sci. USA 2006, 103, 2677–2682. [Google Scholar] [CrossRef] [PubMed]
- Song, G.; Ren, J. Recognition and regulation of unique nucleic acid structures by small molecules. Chem. Commun. 2010, 46, 7283–7294. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.-G.; Lowenhaupt, K.; Maas, S.; Herbert, A.; Schwartz, T.; Rich, A. The zab domain of the human RNA editing enzyme ADAR1 recognizes Z-DNA when surrounded by B-DNA. J. Biol. Chem. 2000, 275, 26828–26833. [Google Scholar] [PubMed]
- Rich, A.; Zhang, S. Z-DNA: The long road to biological function. Nat. Rev. Genet. 2003, 4, 566–572. [Google Scholar] [CrossRef] [PubMed]
- Crespo, L.; Sanclimens, G.; Montaner, B.; Pérez-Tomàs, R.; Royo, M.; Pons, M.; Albericio, F.; Giralt, E. Peptide dendrimers based on polyproline helices. J. Am. Chem. Soc. 2002, 124, 8876–8883. [Google Scholar] [CrossRef] [PubMed]
- Samatey, F.A.; Imada, K.; Nagashima, S.; Vonderviszt, F.; Kumasaka, T.; Yamamoto, M.; Namba, K. Structure of the bacterial flagellar protofilament and implications for a switch for supercoiling. Nature 2001, 410, 331–337. [Google Scholar] [CrossRef] [PubMed]
- Yashima, E.; Maeda, K. Chirality-responsive helical polymers. Macromolecules 2008, 41, 3–12. [Google Scholar] [CrossRef]
- Qing, G.; Sun, T. The transformation of chiral signals into macroscopic properties of materials using chirality-responsive polymers. NPG Asia Mater. 2012, 4, e4. [Google Scholar] [CrossRef]
- Bisoyi, H.K.; Li, Q. Light-directed dynamic chirality inversion in functional self-organized helical superstructures. Angew. Chem. Int. Ed. 2016, 55, 2994–3010. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.; Qiu, M.; Deng, C.; Meng, F.; Zhang, J.; Cheng, R.; Zhong, Z. pH-Responsive chimaeric pepsomes based on asymmetric poly(ethylene glycol)-b-poly(l-leucine)-b-poly(l-glutamic acid) triblock copolymer for efficient loading and active intracellular delivery of doxorubicin hydrochloride. Biomacromolecules 2015, 16, 1322–1330. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, T.; Yamada, T.; Nagata, Y.; Suginome, M. High-molecular-weight polyquinoxaline-based helically chiral phosphine (PQXphos) as chirality-switchable, reusable, and highly enantioselective monodentate ligand in catalytic asymmetric hydrosilylation of styrenes. J. Am. Chem. Soc. 2010, 132, 7899–7901. [Google Scholar] [CrossRef] [PubMed]
- Pijper, D.; Jongejan, M.G.M.; Meetsma, A.; Feringa, B.L. Light-controlled supramolecular helicity of a liquid crystalline phase using a helical polymer functionalized with a single chiroptical molecular switch. J. Am. Chem. Soc. 2008, 130, 4541–4552. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kumar, M.; Brocorens, P.; Tonnelé, C.; Beljonne, D.; Surin, M.; George, S.J. A dynamic supramolecular polymer with stimuli-responsive handedness for in situ probing of enzymatic ATP hydrolysis. Nat. Commun. 2014, 5, 5793. [Google Scholar] [CrossRef] [PubMed]
- Miyake, H. Supramolecular chirality in dynamic coordination chemistry. Symmetry 2014, 6, 880–895. [Google Scholar] [CrossRef]
- Nagata, Y.; Yamada, T.; Adachi, T.; Akai, Y.; Yamamoto, T.; Suginome, M. Solvent-dependent switch of helical main-chain chirality in sergeants-and-soldiers-type poly(quinoxaline-2,3-diyl)s: Effect of the position and structures of the “sergeant” chiral units on the screw-sense induction. J. Am. Chem. Soc. 2013, 135, 10104–10113. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Dong, H.; Li, Y.; Liu, R.; Wang, Y.-F.; Bisoyi, H.K.; Sun, L.-D.; Yan, C.-H.; Li, Q. Luminescence-driven reversible handedness inversion of self-organized helical superstructures enabled by a novel near-infrared light nanotransducer. Adv. Mater. 2015, 27, 2065–2069. [Google Scholar] [CrossRef] [PubMed]
- Freire, F.; Seco, J.M.; Quiñoá, E.; Riguera, R. Chiral amplification and helical-sense tuning by mono- and divalent metals on dynamic helical polymers. Angew. Chem. Int. Ed. 2011, 50, 11692–11696. [Google Scholar] [CrossRef] [PubMed]
- Janssen, P.G.A.; Ruiz-Carretero, A.; González-Rodríguez, D.; Meijer, E.W.; Schenning, A.P.H.J. pH-switchable helicity of DNA-templated assemblies. Angew. Chem. Int. Ed. 2009, 48, 8103–8106. [Google Scholar] [CrossRef] [PubMed]
- Huang, Z.; Kang, S.-K.; Banno, M.; Yamaguchi, T.; Lee, D.; Seok, C.; Yashima, E.; Lee, M. Pulsating tubules from noncovalent macrocycles. Science 2012, 337, 1521–1526. [Google Scholar] [CrossRef] [PubMed]
- Martinez, A.; Guy, L.; Dutasta, J.-P. Reversible, solvent-induced chirality switch in atrane structure: Control of the unidirectional motion of the molecular propeller. J. Am. Chem. Soc. 2010, 132, 16733–16734. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.; Jin, Y.-J.; Kim, H.; Suzuki, N.; Fujiki, M.; Sakaguchi, T.; Kim, S.K.; Lee, W.-E.; Kwak, G. Solvent-to-polymer chirality transfer in intramolecular stack structure. Macromolecules 2012, 45, 5379–5386. [Google Scholar] [CrossRef]
- Johnson, R.S.; Yamazaki, T.; Kovalenko, A.; Fenniri, H. Molecular basis for water-promoted supramolecular chirality inversion in helical rosette nanotubes. J. Am. Chem. Soc. 2007, 129, 5735–5743. [Google Scholar] [CrossRef] [PubMed]
- Isare, B.; Linares, M.; Zargarian, L.; Fermandjian, S.; Miura, M.; Motohashi, S.; Vanthuyne, N.; Lazzaroni, R.; Bouteiller, L. Chirality in dynamic supramolecular nanotubes induced by a chiral solvent. Chem. Eur. J. 2010, 16, 173–177. [Google Scholar] [CrossRef] [PubMed]
- Ke, Y.-Z.; Nagata, Y.; Yamada, T.; Suginome, M. Majority-rules-type helical poly(quinoxaline-2,3-diyl)s as highly efficient chirality-amplification systems for asymmetric catalysis. Angew. Chem. Int. Ed. 2015, 54, 9333–9337. [Google Scholar] [CrossRef] [PubMed]
- Nagata, Y.; Nishikawa, T.; Suginome, M. Exerting control over the helical chirality in the main chain of sergeants-and-soldiers-type poly(quinoxaline-2,3-diyl)s by changing from random to block copolymerization protocols. J. Am. Chem. Soc. 2015, 137, 4070–4073. [Google Scholar] [CrossRef] [PubMed]
- Yamada, T.; Nagata, Y.; Suginome, M. Non-hydrogen-bonding-based, solvent-dependent helix inversion between pure P-helix and pure M-helix in poly(quinoxaline-2,3-diyl)s bearing chiral side chains. Chem. Commun. 2010, 46, 4914–4916. [Google Scholar] [CrossRef] [PubMed]
- Nagata, Y.; Takagi, K.; Suginome, M. Solid polymer films exhibiting handedness-switchable, full-color-tunable selective reflection of circularly polarized light. J. Am. Chem. Soc. 2014, 136, 9858–9861. [Google Scholar] [CrossRef] [PubMed]
- Louzao, I.; Seco, J.M.; Quiñoá, E.; Riguera, R. Control of the helicity of poly(phenylacetylene)s: From the conformation of the pendant to the chirality of the backbone. Angew. Chem. Int. Ed. 2010, 49, 1430–1433. [Google Scholar] [CrossRef] [PubMed]
- Leiras, S.; Freire, F.; Seco, J.M.; Quiñoá, E.; Riguera, R. Controlled modulation of the helical sense and the elongation of poly(phenylacetylene)s by polar and donor effects. Chem. Sci. 2013, 4, 2735–2743. [Google Scholar] [CrossRef]
- Sakurai, S.; Okoshi, K.; Kumaki, J.; Yashima, E. Two-dimensional surface chirality control by solvent induced helicity inversion of a helical polyacetylene on graphite. J. Am. Chem. Soc. 2006, 128, 5650–5651. [Google Scholar] [CrossRef] [PubMed]
- Destoop, I.; Ghijsens, E.; Katayama, K.; Tahara, K.; Mali, K.S.; Tobe, Y.; Feyter, S.D. Solvent-induced homochirality in surface-confined low-density nanoporous molecular networks. J. Am. Chem. Soc. 2012, 134, 19568–19571. [Google Scholar] [CrossRef] [PubMed]
- Russew, M.-M.; Hecht, S. Photoswitches: From molecules to materials. Adv. Mater. 2010, 22, 3348–3360. [Google Scholar] [CrossRef] [PubMed]
- Van Delden, R.A.; ter Wiel, M.K.J.; Vicario, J.; Koumura, N.; Feringa, B.L. Unidirectional molecular motor on a gold surface. Nature 2005, 437, 1337–1340. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Feringa, B.L. Dynamic control of chiral space in a catalytic asymmetric reaction using a molecular motor. Science 2011, 331, 1429–1432. [Google Scholar] [CrossRef] [PubMed]
- Pijper, D.; Feringa, B.L. Molecular transmission: Controlling the twist sense of a helical polymer with a single light-driven molecular motor. Angew. Chem. Int. Ed. 2007, 46, 3693–3696. [Google Scholar] [CrossRef] [PubMed]
- Schreiber, R.; Luong, N.; Fan, Z.; Kuzyk, A.; Nickels, P.C.; Zhang, T.; Smith, D.M.; Yurke, B.; Kuang, W.; Govorov, A.X.; et al. Chiral plasmonic DNA nanostructures with switchable circular dichroism. Nat. Commun. 2013, 4, 2948. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Lee, J.; Kim, W.Y.; Kim, H.; Lee, S.; Lee, H.C.; Lee, Y.S.; Seo, M.; Kim, S.Y. Induction and control of supramolecular chirality by light in self-assembled helical nanostructures. Nat. Commun. 2015, 6, 6959. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.-T.; Chen, C.-H.; Ong, T.-G. Complementary helicity interchange of optically switchable supramolecular-enantiomeric helicenes with (−)-gel-sol-(+)-gel transition ternary logic. J. Am. Chem. Soc. 2013, 135, 5294–5297. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.-J.; Shin, B.-G.; Kim, J.-J.; Kim, D.-Y. Photoinduced supramolecular chirality in amorphous azobenzene polymer films. J. Am. Chem. Soc. 2002, 124, 3504–3505. [Google Scholar] [CrossRef] [PubMed]
- Mathews, M.; Zola, R.S.; Hurley, S.; Yang, D.-K.; White, T.J.; Bunning, T.J.; Li, Q. Light-driven reversible handedness inversion in self-organized helical superstructures. J. Am. Chem. Soc. 2010, 132, 18361–18366. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Urbas, A.; Li, Q. Reversible visible-light tuning of self-organized helical superstructures enabled by unprecedented light-driven axially chiral molecular switches. J. Am. Chem. Soc. 2012, 134, 3342–3345. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Z.-G.; Li, Y.; Bisoyi, H.K.; Wang, L.; Bunning, T.J.; Li, Q. Three-dimensional control of the helical axis of a chiral nematic liquid crystal by light. Nature 2016, 531, 352–356. [Google Scholar] [CrossRef] [PubMed]
- Jose, B.A.S.; Matsushita, S.; Akagi, K. Lyotropic chiral nematic liquid crystalline aliphatic conjugated polymers based on disubstituted polyacetylene derivatives that exhibit high dissymmetry factors in circularly polarized luminescence. J. Am. Chem. Soc. 2012, 134, 19795–19807. [Google Scholar] [CrossRef] [PubMed]
- Iamsaard, S.; Aßhoff, S.J.; Matt, B.; Kudernac, T.; Cornelissen, J.J.L.M.; Fletcher, S.P.; Katsonis, N. Conversion of light into macroscopic helical motion. Nat. Chem. 2014, 6, 229–235. [Google Scholar] [CrossRef] [PubMed]
- Toyoda, K.; Miyamoto, K.; Aoki, N.; Morita, R.; Omatsu, T. Using optical vortex to control the chirality of twisted metal nanostructures. Nano Lett. 2012, 12, 3645–3649. [Google Scholar] [CrossRef] [PubMed]
- Knipe, P.C.; Thompson, S.; Hamilton, A.D. Ion-mediated conformational switches. Chem. Sci. 2015, 6, 1630–1639. [Google Scholar] [CrossRef]
- Bergueiro, J.; Freire, F.; Wendler, E.P.; Seco, J.M.; Quiñoá, E.; Riguera, R. The on/off switching by metal ions of the “sergeants and soldiers” chiral amplification effect on helical poly(phenylacetylene)s. Chem. Sci. 2014, 5, 2170–2176. [Google Scholar] [CrossRef]
- Arias, S.; Freire, F.; Quiñoá, E.; Riguera, R. The leading role of cation–π interactions in polymer chemistry: The control of the helical sense in solution. Polym. Chem. 2015, 6, 4725–4733. [Google Scholar] [CrossRef]
- Arias, S.; Freire, F.; Quiñoá, E.; Riguera, R. Nanospheres, nanotubes, toroids, and gels with controlled macroscopic chirality. Angew. Chem. Int. Ed. 2014, 53, 13720–13724. [Google Scholar] [CrossRef] [PubMed]
- Arias, S.; Bergueiro, J.; Freire, F.; Quiñoá, E.; Riguera, R. Chiral nanostructures from helical copolymer–metal complexes: Tunable cation–π interactions and sergeants and soldiers effect. Small 2016, 12, 238–244. [Google Scholar] [CrossRef] [PubMed]
- Akine, S.; Hotate, S.; Nabeshima, T. A molecular leverage for helicity control and helix inversion. J. Am. Chem. Soc. 2011, 133, 13868–13871. [Google Scholar] [CrossRef] [PubMed]
- Park, S.H.; Jung, S.H.; Ahn, J.; Lee, J.H.; Kwon, K.-Y.; Jeon, J.; Kim, H.; Jaworski, J.; Jung, J.H. Reversibly tunable helix inversion in supramolecular gels trigged by Co2+. Chem. Commun. 2014, 50, 13495–13498. [Google Scholar] [CrossRef] [PubMed]
- Haberhauer, G. A metal-ion-driven supramolecular chirality pendulum. Angew. Chem. Int. Ed. 2010, 49, 9286–9289. [Google Scholar] [CrossRef] [PubMed]
- Miyake, H.; Kamon, H.; Miyahara, I.; Sugimoto, H.; Tsukube, H. Time-programmed peptide helix inversion of a synthetic metal complex triggered by an achiral NO3− anion. J. Am. Chem. Soc. 2008, 130, 792–793. [Google Scholar] [CrossRef] [PubMed]
- Suk, J.-M.; Naidu, V.R.; Liu, X.; Lah, M.S.; Jeong, K.-S. A foldamer-based chiroptical molecular switch that displays complete inversion of the helical sense upon anion binding. J. Am. Chem. Soc. 2011, 133, 13938–13941. [Google Scholar] [CrossRef] [PubMed]
- Li, B.S.; Cheuk, K.K.L.; Salhi, F.; Law, J.W.Y.; Cha, J.A.K.; Xiao, X.; Bai, C.; Tang, B.Z. Tuning the chain helicity and organizational morphology of an l-valine-containing polyacetylene by pH change. Nano Lett. 2001, 1, 323–328. [Google Scholar] [CrossRef]
- Cho, J.-I.; Tanaka, M.; Sato, S.; Kinbara, K.; Aida, T. Oligo(4-aminopiperidine-4-carboxylic acid): An unusual basic oligopeptide with an acid-induced helical conformation. J. Am. Chem. Soc. 2010, 132, 13176–13178. [Google Scholar] [CrossRef] [PubMed]
- Sanji, T.; Kato, N.; Tanaka, M. Switching of optical activity in oligosilane through pH-responsive chiral wrapping with amylose. Macromolecules 2006, 39, 7508–7512. [Google Scholar] [CrossRef]
- Kurouski, D.; Lombardi, R.A.; Dukor, R.K.; Lednev, I.K.; Nafie, L.A. Direct observation and pH control of reversed supramolecular chirality in insulin fibrils by vibrational circular dichroism. Chem. Commun. 2010, 46, 7154–7156. [Google Scholar] [CrossRef] [PubMed]
- Komori, H.; Inai, Y. Control of peptide helix sense by temperature tuning of noncovalent chiral domino effect. J. Org. Chem. 2007, 72, 4012–4022. [Google Scholar] [CrossRef] [PubMed]
- George, S.J.; de Bruijn, R.; Tomović, Ž.; Averbeke, B.V.; Beljonne, D.; Lazzaroni, R.; Schenning, A.P.H.J.; Meijer, E.W. Asymmetric noncovalent synthesis of self-assembled one-dimensional stacks by a chiral supramolecular auxiliary approach. J. Am. Chem. Soc. 2012, 134, 17789–17796. [Google Scholar] [CrossRef] [PubMed]
- Gopal, A.; Hifsudheen, M.; Furumi, S.; Takeuchi, M.; Ajayaghosh, A. Thermally assisted photonic inversion of supramolecular handedness. Angew. Chem. Int. Ed. 2012, 51, 10505–10509. [Google Scholar] [CrossRef] [PubMed]
- Shigeno, M.; Kushida, Y.; Yamaguchi, M. Heating/cooling stimulus induces three-state molecular switching of pseudoenantiomeric aminomethylenehelicene oligomers: Reversible nonequilibrium thermodynamic processes. J. Am. Chem. Soc. 2014, 136, 7972–7980. [Google Scholar] [CrossRef] [PubMed]
- Miyagawa, T.; Furuko, A.; Maeda, K.; Katagiri, H.; Furusho, Y.; Yashima, E. Dual memory of enantiomeric helices in a polyacetylene induced by a single enantiomer. J. Am. Chem. Soc. 2005, 127, 5018–5019. [Google Scholar] [CrossRef] [PubMed]
- Maeda, K.; Mochizuki, H.; Watanabe, M.; Yashima, E. Switching of macromolecular helicity of optically active poly(phenylacetylene)s bearing cyclodextrin pendants induced by various external stimuli. J. Am. Chem. Soc. 2006, 128, 7639–7650. [Google Scholar] [CrossRef] [PubMed]
- Mochida, Y.; Cabral, H.; Miura, Y.; Albertini, F.; Fukushima, S.; Osada, K.; Nishiyama, N.; Kataoka, K. Bundled assembly of helical nanostructures in polymeric micelles loaded with platinum drugs enhancing therapeutic efficiency against pancreatic tumor. ACS Nano 2014, 8, 6724–6738. [Google Scholar] [CrossRef] [PubMed]
- Bellomo, E.G.; Wyrsta, M.D.; Pakstis, L.; Pochan, D.J.; Deming, T.J. Stimuli-responsive polypeptide vesicles by conformation-specific assembly. Nat. Mater. 2004, 3, 244–248. [Google Scholar] [CrossRef] [PubMed]
- Shi, S.-Y.; He, Y.-G.; Chen, W.-W.; Liu, N.; Zhu, Y.-Y.; Ding, Y.-S.; Yin, J.; Wu, Z.-Q. Polypeptide-b-poly(phenyl isocyanide) hybrid rod-rod copolymers: One-pot synthesis, self-assembly, and cell imaging. Macromol. Rapid. Commun. 2015, 36, 1511–1520. [Google Scholar] [CrossRef] [PubMed]
- Shimomura, K.; Ikai, T.; Kanoh, S.; Yashima, E.; Maeda, K. Switchable enantioseparation based on macromolecular memory of a helical polyacetylene in the solid state. Nat. Chem. 2014, 6, 429–434. [Google Scholar] [CrossRef] [PubMed]
- Wen, A.M.; Steinmetz, N.F. Design of virus-based nanomaterials for medicine, biotechnology, and energy. Chem. Soc. Rev. 2016, 45, 4074–4126. [Google Scholar] [CrossRef] [PubMed]
- Barry, E.; Beller, D.; Dogic, Z. A model liquid crystalline system based on rodlike viruses with variable chirality and persistence length. Soft Matter 2009, 5, 2563–2570. [Google Scholar] [CrossRef]
- Chung, W.-J.; Oh, J.-W.; Kwak, K.; Lee, B.Y.; Meyer, J.; Wang, E.; Hexemer, A.; Lee, S.-W. Biomimetic self-templating supramolecular structures. Nature 2011, 478, 364–368. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.; Zhang, G.; Liu, S. Enzyme-responsive polymeric assemblies, nanoparticles and hydrogels. Chem. Soc. Rev. 2012, 41, 5933–5949. [Google Scholar] [CrossRef] [PubMed]
- Lv, Z.; Li, X.; Chen, Z.; Chen, J.; Chen, C.; Xiong, P.; Sun, T.; Qing, G. Surface stiffness-a parameter for sensing the chirality of saccharides. ACS Appl. Mater. Interfaces 2015, 7, 27223–27233. [Google Scholar] [CrossRef] [PubMed]


| Polymers | Stimulation | Regulating mechanism | Ref |
|---|---|---|---|
| Poly(phenylacetylene) | Solvent | The effect of solvent polarity | [34,35,36] |
| Ion | Cation–π Interactions | [56] | |
| pH | Electrical repulsion | [62] | |
| Temperature | Noncovalent helix induction | [70,71] | |
| Poly(peptide) | Ion | Chirality transfer | [60] |
| - | Cis-trans isomerization | [11] | |
| Temperature | Chiral domino effect | [66] | |
| Poly(quinoxaline) | Solvent | The effect of solvent polarity | [21,30,31,32,33] |
| Poly(diphenylacetylene) | Solvent | Chirality transfer | [27] |
| Poly(hexylisocyanate) | Light | Photoisomerization | [41] |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lv, Z.; Chen, Z.; Shao, K.; Qing, G.; Sun, T. Stimuli-Directed Helical Chirality Inversion and Bio-Applications. Polymers 2016, 8, 310. https://doi.org/10.3390/polym8080310
Lv Z, Chen Z, Shao K, Qing G, Sun T. Stimuli-Directed Helical Chirality Inversion and Bio-Applications. Polymers. 2016; 8(8):310. https://doi.org/10.3390/polym8080310
Chicago/Turabian StyleLv, Ziyu, Zhonghui Chen, Kenan Shao, Guangyan Qing, and Taolei Sun. 2016. "Stimuli-Directed Helical Chirality Inversion and Bio-Applications" Polymers 8, no. 8: 310. https://doi.org/10.3390/polym8080310