Endocytosis Pathways of the Folate Tethered Star-Shaped PEG-PCL Micelles in Cancer Cell Lines
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
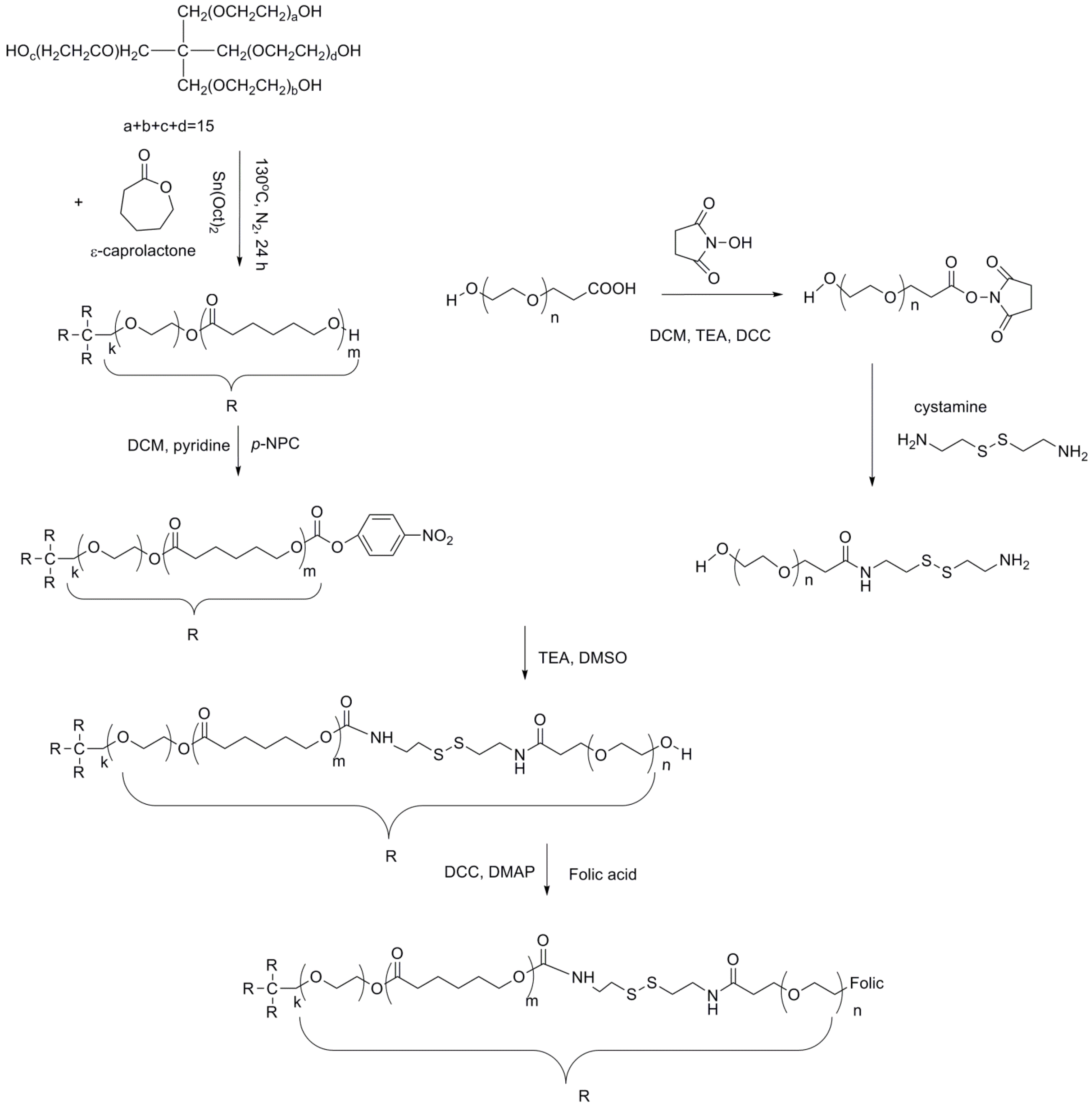
2.2. Synthesis of Folic-Tethered Star-Shaped Poly(ɛ-Caprolactone)-Poly(Ethylene Glycol) Block Copolymer 4SPCL-PEG-Folic
2.3. Preparation and Characteristic of 4SPCL-PEG-Folic Micelles
2.4. Experiment on in Vitro Release of DOX from 4SPCL-PEG-Folic Micelles
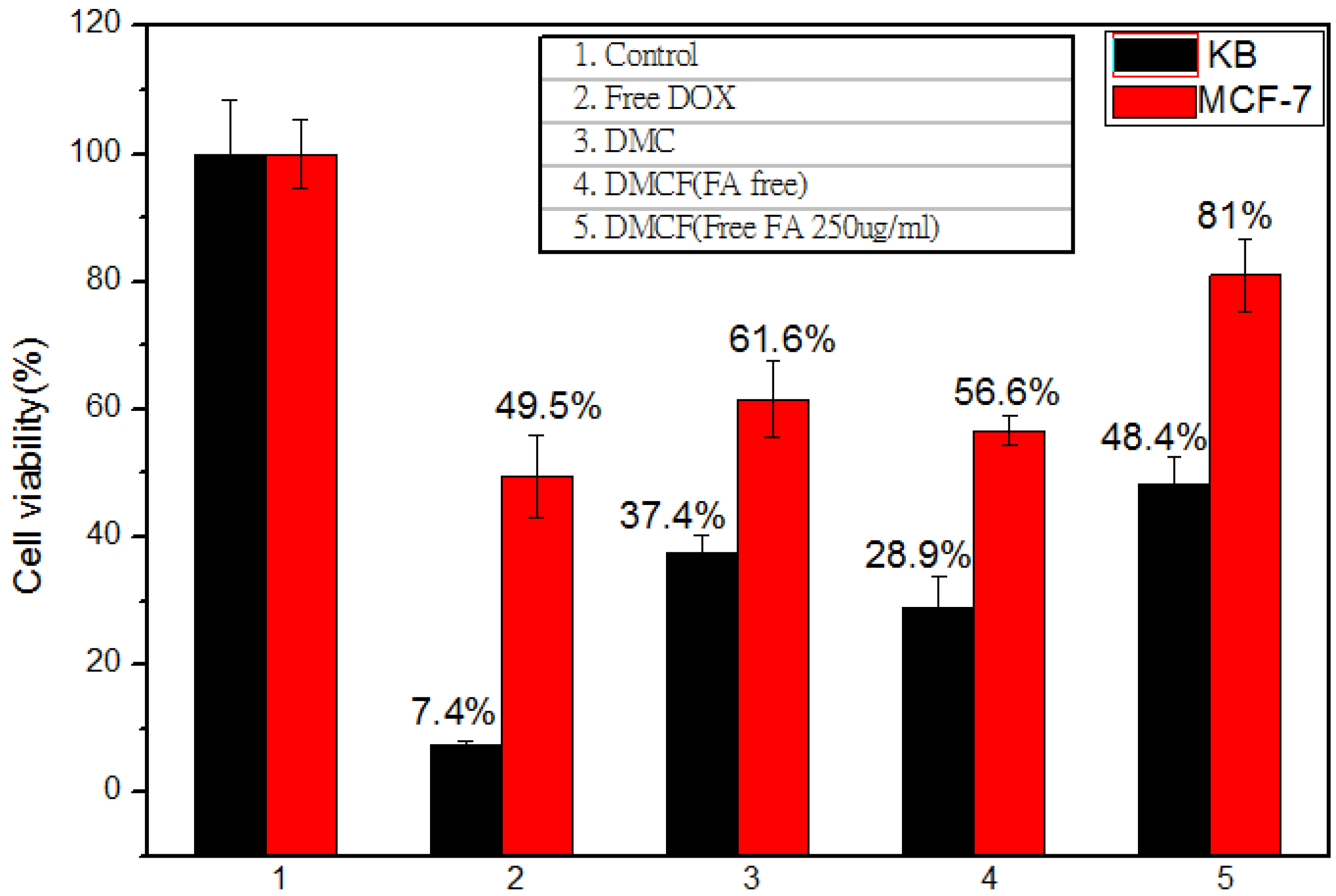
2.5. In Vitro Cytotoxicity of DOX-Loaded Micelle in KB and MCF-7 Cell Lines
2.6. Cellular Uptake of DOX-Loaded Micelles in the MCF-7 and KB Cell Lines
2.6.1. Flow Cytometry
2.6.2. Cellular Uptake of DOX
2.6.3. Pathways of Endocytosis
3. Results and Discussion
3.1. Preparation and Micellization Behavior of 4SPCL-PEG-Folic Copolymer
| Sample | Mw (theory) | Mn,NMR | Sample | Mw (theory) | Mn,NMR |
|---|---|---|---|---|---|
| 4SPCL50-PEG | 19,613 | 17,049 | 4SPCL50-PEG-Folic | 21,521 | 17,168 |
| 4SPCL100-PEG | 25,313 | 23,398 | 4SPCL100-PEG-Folic | 27,221 | 23,570 |
| 4SPCL150-PEG | 31,013 | 28,253 | 4SPCL150-PEG-Folic | 30,161 | 28,680 |
| Empty micelles | Drug loading micelles | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Sample | Particle size (nm) a | Zeta potential (mV) b | Poly Index | Sample | Particle size (nm) a | Zeta potential (mV) b | Poly Index | DLC c | DLE c (%) |
| 4SPCL50-PEG | 136.9 ± 7.7 | −1.8 ± 0.7 | 0.29 | DMC50 | 96.9 ± 9.7 | −2.8 ± 0.7 | 0.21 | 7.5 | 75 |
| 4SPCL100-PEG | 172.3 ± 1.5 | −1.4 ± 0.5 | 0.25 | DMC100 | 142.1 ± 14.9 | −2.1 ± 0.6 | 0.19 | 8.8 | 88 |
| 4SPCL150-PEG | 209.3 ± 8.8 | −1.3 ± 0.3 | 0.27 | DMC150 | 145.2 ± 14.1 | −1.9 ± 0.6 | 0.25 | 6.6 | 66 |
| 4SPCL50-PEG-Folic | 125.3 ± 2.6 | −1.9 ± 0.1 | 0.48 | DMC50F | 126.2 ± 1.1 | −3.4 ± 0.15 | 0.1 | 7.6 | 76 |
| 4SPCL100-PEG-Folic | 133.4 ± 7.4 | −2.3 ± 0.9 | 0.05 | DMC100F | 128.7 ± 8.9 | −3.9 ± 0.15 | 0.35 | 8.9 | 89 |
| 4SPCL150-PEG-Folic | 174.2 ± 1.9 | −2.0 ± 0.5 | 0.37 | DMC150F | 123.1 ± 7.9 | −2.4 ± 0.19 | 0.08 | 3.4 | 34 |
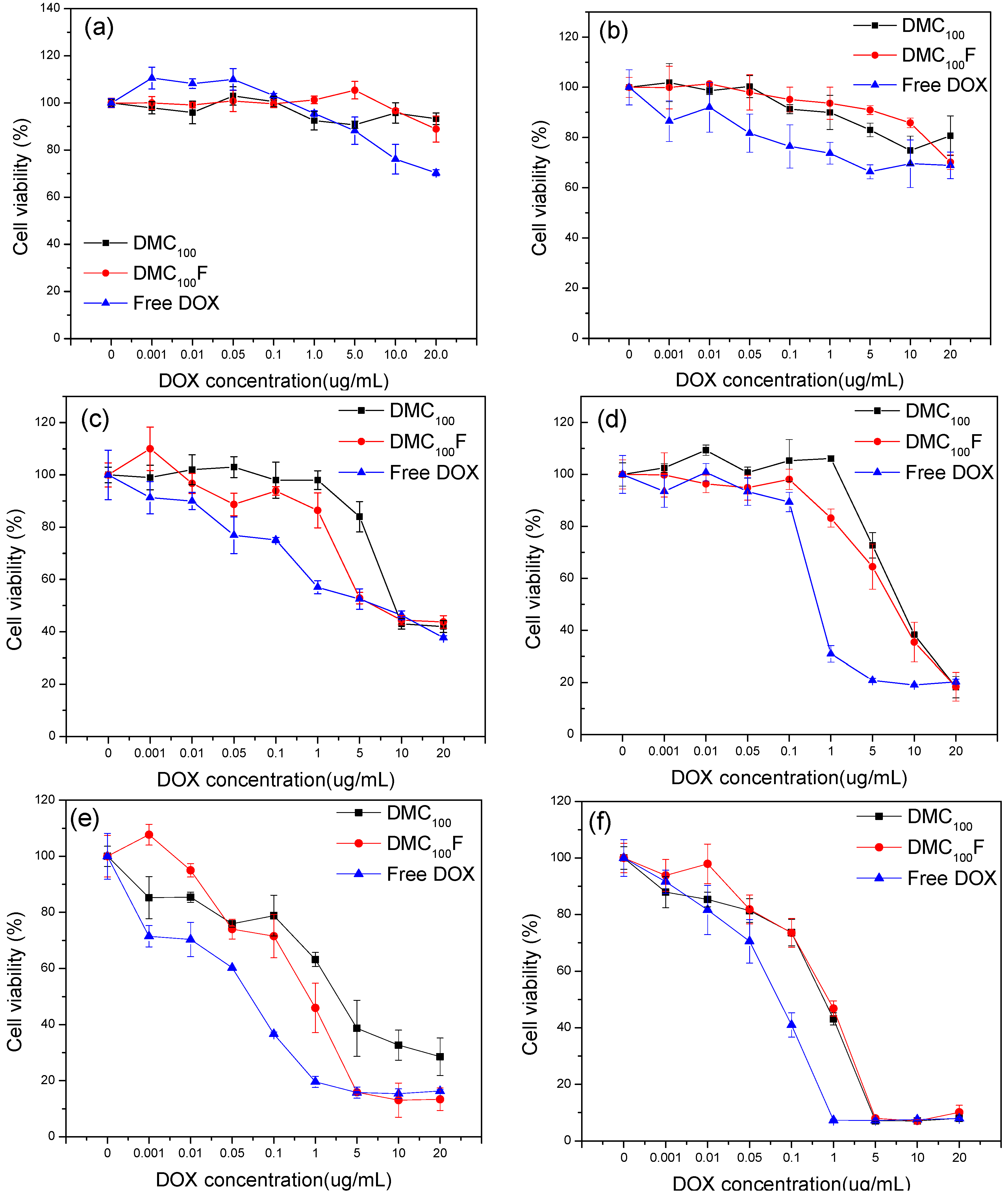
3.2. In Vitro Cytotoxicity of DOX-Loaded Micelles
3.3. Cellular Uptake of DOX-Loaded Micelles
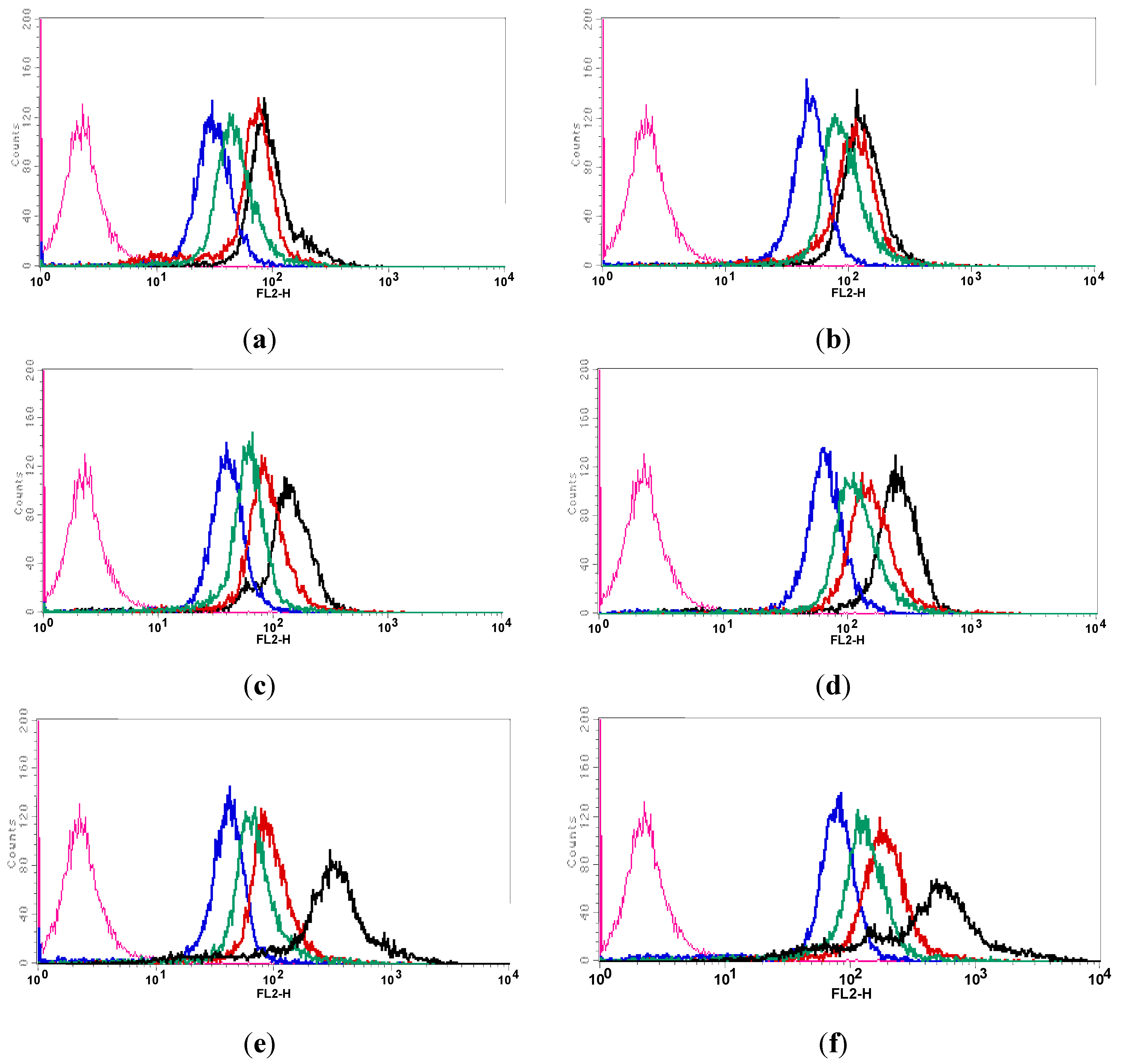
3.3.1. Flow Cytometry

3.3.2. Confocal Image of DOX-Loaded Micelles in KB and MCF-7 Cell Lines
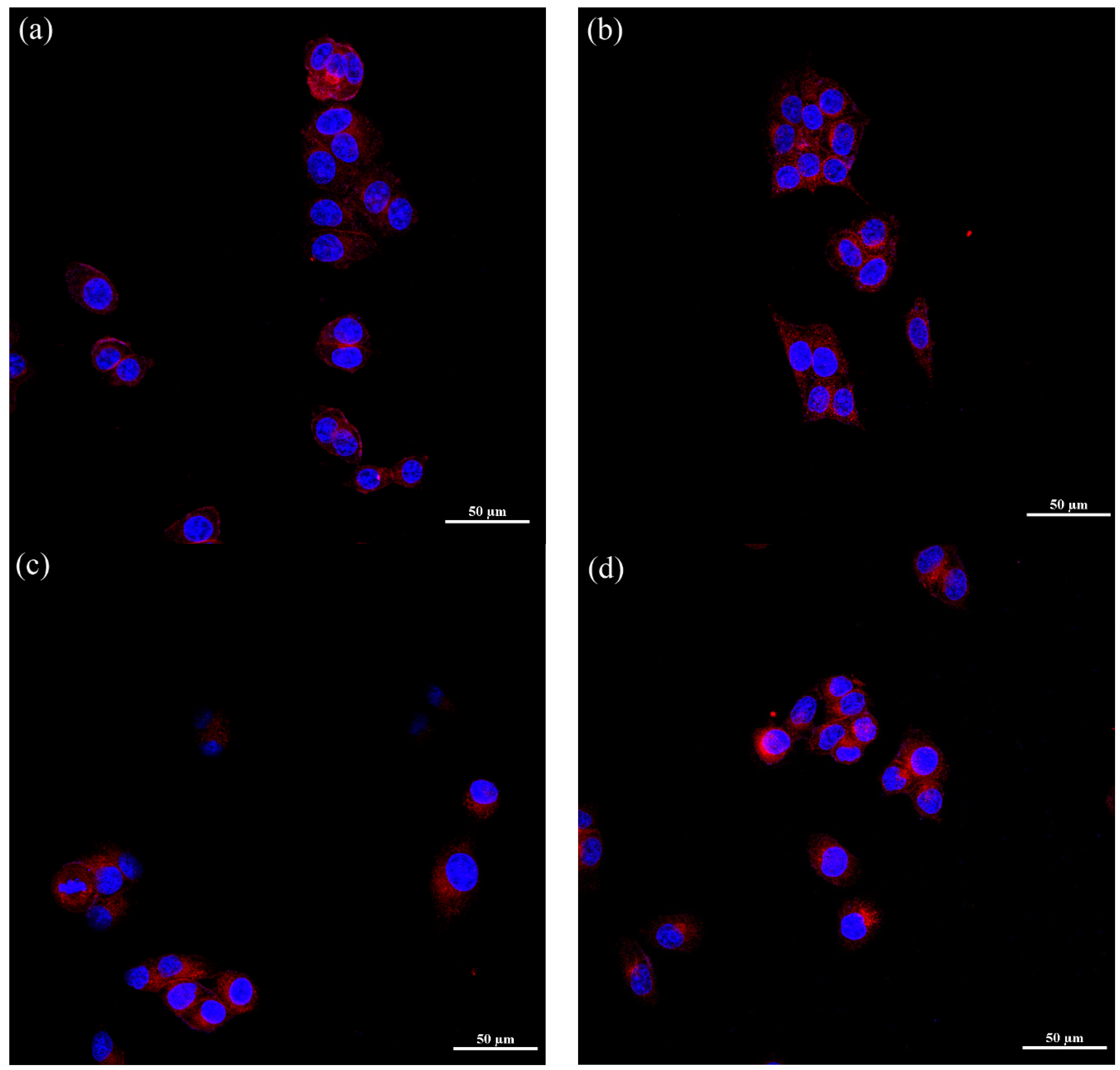
3.3.3. Endocytosis Pathway
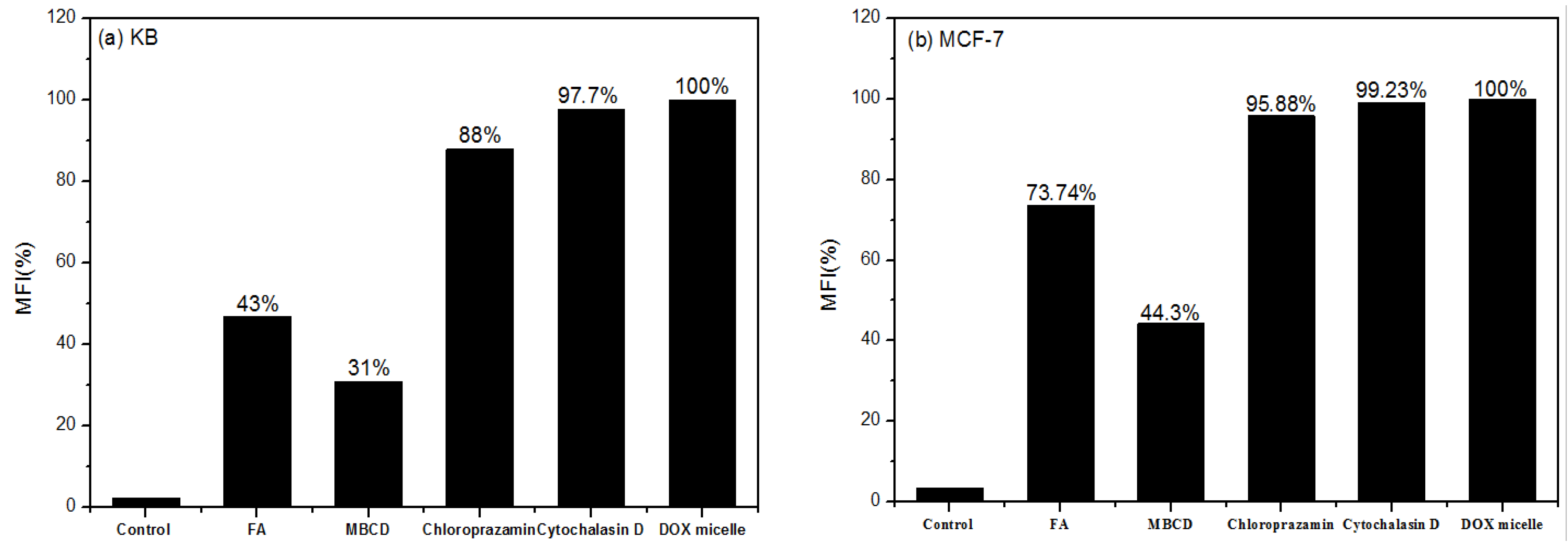
4. Conclusions
Acknowledgements
Author Contributions
Conflicts of Interest
References
- Byrne, J.D.; Betancourt, T.; Brannon-Peppas, L. Active targeting schemes for nanoparticle systems in cancer therapeutics. Adv. Drug Deliv. Rev. 2008, 60, 1615–1626. [Google Scholar] [CrossRef]
- Lu, Y.; Low, P.S. Folate-mediated delivery of macromolecular anticancer therapeutic agents. Adv. Drug Deliv. Rev. 2002, 54, 675–693. [Google Scholar] [CrossRef]
- Liu, S.Q.; Wiradharma, N.; Gao, S.J.; Tong, Y.W.; Yang, Y.Y. Bio-functional micelles self-assembled from a folate-conjugated block copolymer for targeted intracellular delivery of anticancer drugs. Biomaterials 2007, 28, 1423–1433. [Google Scholar] [CrossRef]
- Reddy, J.A.; Dorton, R.; Westrick, E.; Dawson, A.; Smith, T.; Xu, L.-C.; Vetzel, M.; Kleindl, P.; Vlahov, I.R.; Leamon, C.P. Preclinical evaluation of ec145, a folate-vinca alkaloid conjugate. Cancer Res. 2007, 67, 4434–4442. [Google Scholar] [CrossRef]
- Hilgenbrink, A.R.; Low, P.S. Folate receptor-mediated drug targeting: From therapeutics to diagnostics. J. Pharm. Sci. 2005, 94, 2135–2146. [Google Scholar] [CrossRef]
- Yang, S.J.; Lin, F.H.; Tsai, K.C.; Wei, M.F.; Tsai, H.M.; Wong, J.M.; Shieh, M.J. Folic acid-conjugated chitosan nanoparticles enhanced protoporphyrin ix accumulation in colorectal cancer cells. Bioconjugate Chem. 2010, 21, 679–689. [Google Scholar] [CrossRef]
- Wu, J.; Liu, Q.; Lee, R.J. A folate receptor-targeted liposomal formulation for paclitaxel. Int. J. Pharm. 2006, 316, 148–153. [Google Scholar] [CrossRef]
- Xu, B.; Yuan, J.; Ding, T.; Gao, Q. Amphiphilic biodegradable poly(ε-caprolactone)-poly(ethylene glycol)-poly(ε-caprolactone) triblock copolymers: Synthesis, characterization and their use as drug carriers for folic acid. Polym. Bull. 2010, 64, 537–551. [Google Scholar] [CrossRef]
- Lu, T.; Sun, J.; Chen, X.; Zhang, P.; Jing, X. Folate-conjugated micelles and their folate-receptor-mediated endocytosis. Macromol. Biosci. 2009, 9, 1059–1068. [Google Scholar] [CrossRef]
- Kim, D.; Gao, Z.G.; Lee, E.S.; Bae, Y.H. In vivo evaluation of doxorubicin-loaded polymeric micelles targeting folate receptors and early endosomal pH in drug-resistant ovarian cancer. Mol. Pharm. 2009, 6, 1353–1362. [Google Scholar] [CrossRef]
- Zhang, Z.; Yao, J. Preparation of irinotecan-loaded folate-targeted liposome for tumor targeting delivery and its antitumor activity. AAPS PharmSciTech 2012, 13, 802–810. [Google Scholar] [CrossRef]
- Zhao, F.; Zhao, Y.; Liu, Y.; Chang, X.; Chen, C.; Zhao, Y. Cellular uptake, intracellular trafficking, and cytotoxicity of nanomaterials. Small 2011, 7, 1322–1337. [Google Scholar] [CrossRef]
- Sahay, G.; Alakhova, D.Y.; Kabanov, A.V. Endocytosis of nanomedicines. J. Control. Release 2010, 145, 182–195. [Google Scholar] [CrossRef]
- Bareford, L.M.; Swaan, P.W. Endocytic mechanisms for targeted drug delivery. Adv. Drug Deliv. Rev. 2007, 59, 748–758. [Google Scholar] [CrossRef]
- Dauty, E.; Remy, J.-S.; Zuber, G.; Behr, J.-P. Intracellular delivery of nanometric DNA particles via the folate receptor. Bioconjugate Chem. 2002, 13, 831–839. [Google Scholar] [CrossRef]
- Hillaireau, H.; Couvreur, P. Nanocarriers’ entry into the cell: Relevance to drug delivery. Cell. Mol. Life Sci. 2009, 66, 2873–2896. [Google Scholar] [CrossRef]
- Cuong, N.-V.; Li, Y.-L.; Hsieh, M.-F. Targeted delivery of doxorubicin to human breast cancers by folate-decorated star-shaped PEG-PCL micelle. J. Mater. Chem. 2012, 22, 1006–1020. [Google Scholar] [CrossRef]
- Cuong, N.V.; Hsieh, M.F.; Chen, Y.T.; Liau, I. Doxorubicin-loaded nanosized micelles of a star-shaped poly(ε-caprolactone)-polyphosphoester block co-polymer for treatment of human breast cancer. J. Biomater. Sci. Polym. Ed. 2011, 22, 1409–1426. [Google Scholar] [CrossRef]
- Cuong, N.V.; Hsieh, M.F.; Chen, Y.T.; Liau, I. Synthesis and characterization of PEG-PCL-PEG triblock copolymers as carriers of doxorubicin for the treatment of breast cancer. J. Appl. Polym. Sci. 2010, 117, 3694–3703. [Google Scholar]
- Cuong, N.V.; Jiang, J.L.; Li, Y.L.; Chen, J.R.; Jwo, S.C.; Hsieh, M.F. Doxorubicin-loaded PEG-PCL-PEG micelle using xenograft model of nude mice: Effect of multiple administration of micelle on the suppression of human breast cancer. Cancers 2011, 3, 61–78. [Google Scholar]
- Aliabadi, H.M.; Mahmud, A.; Sharifabadi, A.D.; Lavasanifar, A. Micelles of methoxy poly(ethylene oxide)-b-poly(ε-caprolactone) as vehicles for the solubilization and controlled delivery of cyclosporine A. J. Control. Release 2005, 104, 301–311. [Google Scholar] [CrossRef]
- Cui, J.; Li, C.; Guo, W.; Li, Y.; Wang, C.; Zhang, L.; Zhang, L.; Hao, Y.; Wang, Y. Direct comparison of two pegylated liposomal doxorubicin formulations: Is auc predictive for toxicity and efficacy? J. Control. Release 2007, 118, 204–215. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, Y.; Xiang, J.; Yao, K. Target-specific cellular uptake of taxol-loaded heparin-peg-folate nanoparticles. Biomacromolecules 2010, 11, 3531–3538. [Google Scholar] [CrossRef]
- Shuai, X.; Ai, H.; Nasongkla, N.; Kim, S.; Gao, J. Micellar carriers based on block copolymers of poly(ε-caprolactone) and poly(ethylene glycol) for doxorubicin delivery. J. Control. Release 2004, 98, 415–426. [Google Scholar] [CrossRef]
- Allen, C.; Maysinger, D.; Eisenberg, A. Nano-engineering block copolymer aggregates for drug delivery. Colloid Surf. B Biointerfaces 1999, 16, 3–27. [Google Scholar] [CrossRef]
- Minotti, G.; Menna, P.; Salvatorelli, E.; Cairo, G.; Gianni, L. Anthracyclines: Molecular advances and pharmacologic developments in antitumor activity and cardiotoxicity. Pharmacol. Rev. 2004, 56, 185–229. [Google Scholar] [CrossRef]
- Upadhyay, K.K.; Bhatt, A.N.; Mishra, A.K.; Dwarakanath, B.S.; Jain, S.; Schatz, C.; le Meins, J.F.; Farooque, A.; Chandraiah, G.; Jain, A.K.; et al. The intracellular drug delivery and anti tumor activity of doxorubicin loaded poly(gamma-benzyl l-glutamate)-b-hyaluronan polymersomes. Biomaterials 2010, 31, 2882–2892. [Google Scholar] [CrossRef]
- Ostacolo, L.; Marra, M.; Ungaro, F.; Zappavigna, S.; Maglio, G.; Quaglia, F.; Abbruzzese, A.; Caraglia, M. In vitro anticancer activity of docetaxel-loaded micelles based on poly(ethylene oxide)-poly(ε-caprolactone) block copolymers: Do nanocarrier properties have a role. J. Control. Release 2010, 148, 255–263. [Google Scholar] [CrossRef]
- Zhao, H.; Duong, H.H.P.; Yung, L.Y.L. Folate-conjugated polymer micelles with pH-triggered drug release properties. Macromol. Rapid Commun. 2010, 31, 1163–1169. [Google Scholar] [CrossRef]
- Mohan, P.; Rapoport, N. Doxorubicin as a molecular nanotheranostic agent: Effect of doxorubicin encapsulation in micelles or nanoemulsions on the ultrasound-mediated intracellular delivery and nuclear trafficking. Mol. Pharm. 2010, 7, 1959–1973. [Google Scholar] [CrossRef]
- Tsukioka, Y.; Matsumura, Y.; Hamaguchi, T.; Koike, H.; Moriyasu, F.; Kakizoe, T. Pharmaceutical and biomedical differences between micellar doxorubicin (NK911) and liposomal doxorubicin (Doxil). Jpn. J. Cancer Res. 2002, 93, 1145–1153. [Google Scholar] [CrossRef]
- Zhao, H.; Yung, L.Y.L. Selectivity of folate conjugated polymer micelles against different tumor cells. Int. J. Pharm. 2008, 349, 256–268. [Google Scholar] [CrossRef]
- Chiu, Y.L.; Ho, Y.C.; Chen, Y.M.; Peng, S.F.; Ke, C.J.; Chen, K.J.; Mi, F.L.; Sung, H.W. The characteristics, cellular uptake and intracellular trafficking of nanoparticles made of hydrophobically-modified chitosan. J. Control. Release 2010, 146, 152–159. [Google Scholar] [CrossRef]
- Liu, Y.; Sun, J.; Cao, W.; Yang, J.; Lian, H.; Li, X.; Sun, Y.; Wang, Y.; Wang, S.; He, Z. Dual-targeting folate-conjugated hyaluonic acid polymeric micelles for paclitaxel delivery. Int. J. Pharm. 2011, 421, 160–169. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Li, Y.-L.; Van Cuong, N.; Hsieh, M.-F. Endocytosis Pathways of the Folate Tethered Star-Shaped PEG-PCL Micelles in Cancer Cell Lines. Polymers 2014, 6, 634-650. https://doi.org/10.3390/polym6030634
Li Y-L, Van Cuong N, Hsieh M-F. Endocytosis Pathways of the Folate Tethered Star-Shaped PEG-PCL Micelles in Cancer Cell Lines. Polymers. 2014; 6(3):634-650. https://doi.org/10.3390/polym6030634
Chicago/Turabian StyleLi, Yu-Lun, Nguyen Van Cuong, and Ming-Fa Hsieh. 2014. "Endocytosis Pathways of the Folate Tethered Star-Shaped PEG-PCL Micelles in Cancer Cell Lines" Polymers 6, no. 3: 634-650. https://doi.org/10.3390/polym6030634





