Pressure Retarded Osmosis and Forward Osmosis Membranes: Materials and Methods
Abstract
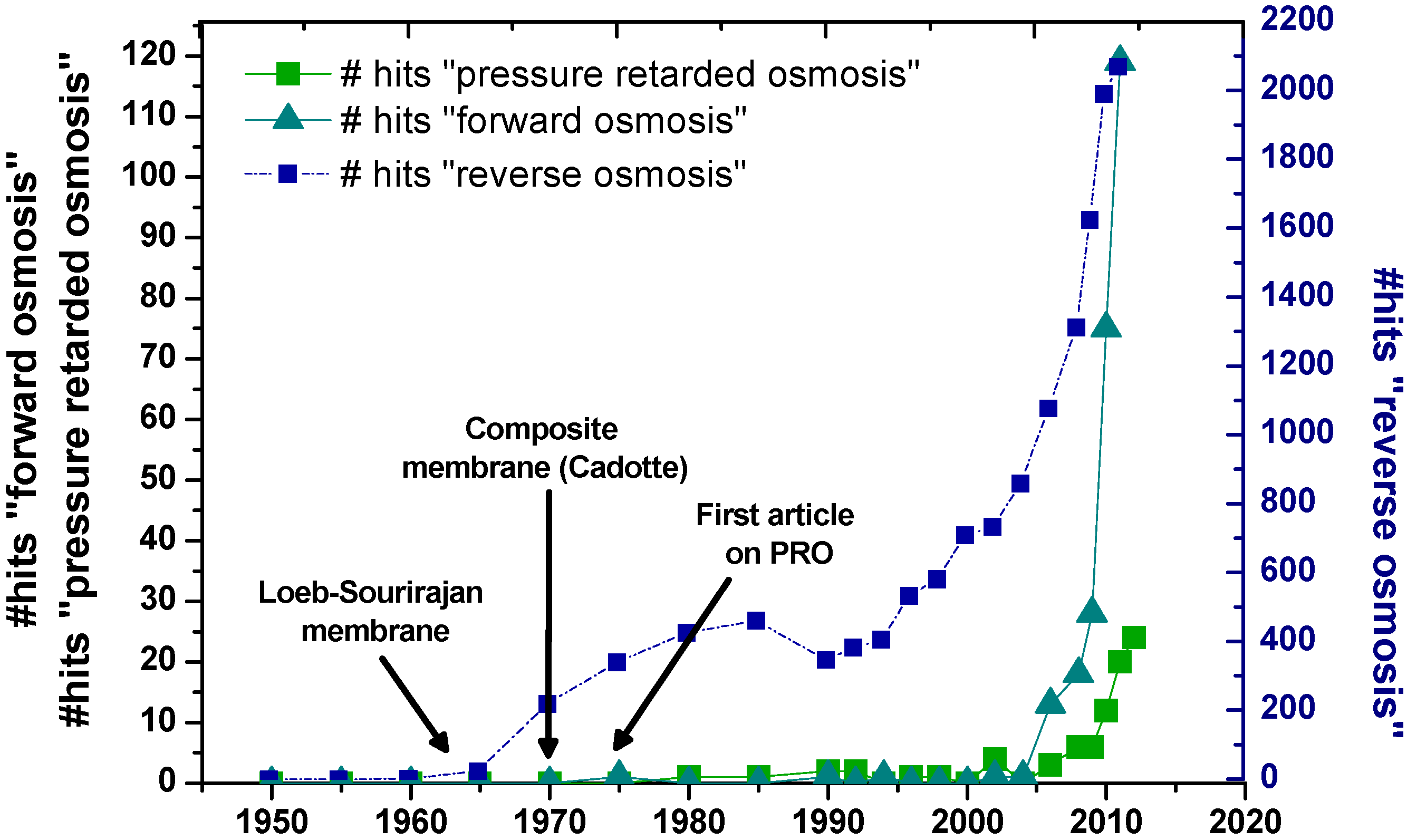
:1. Introduction
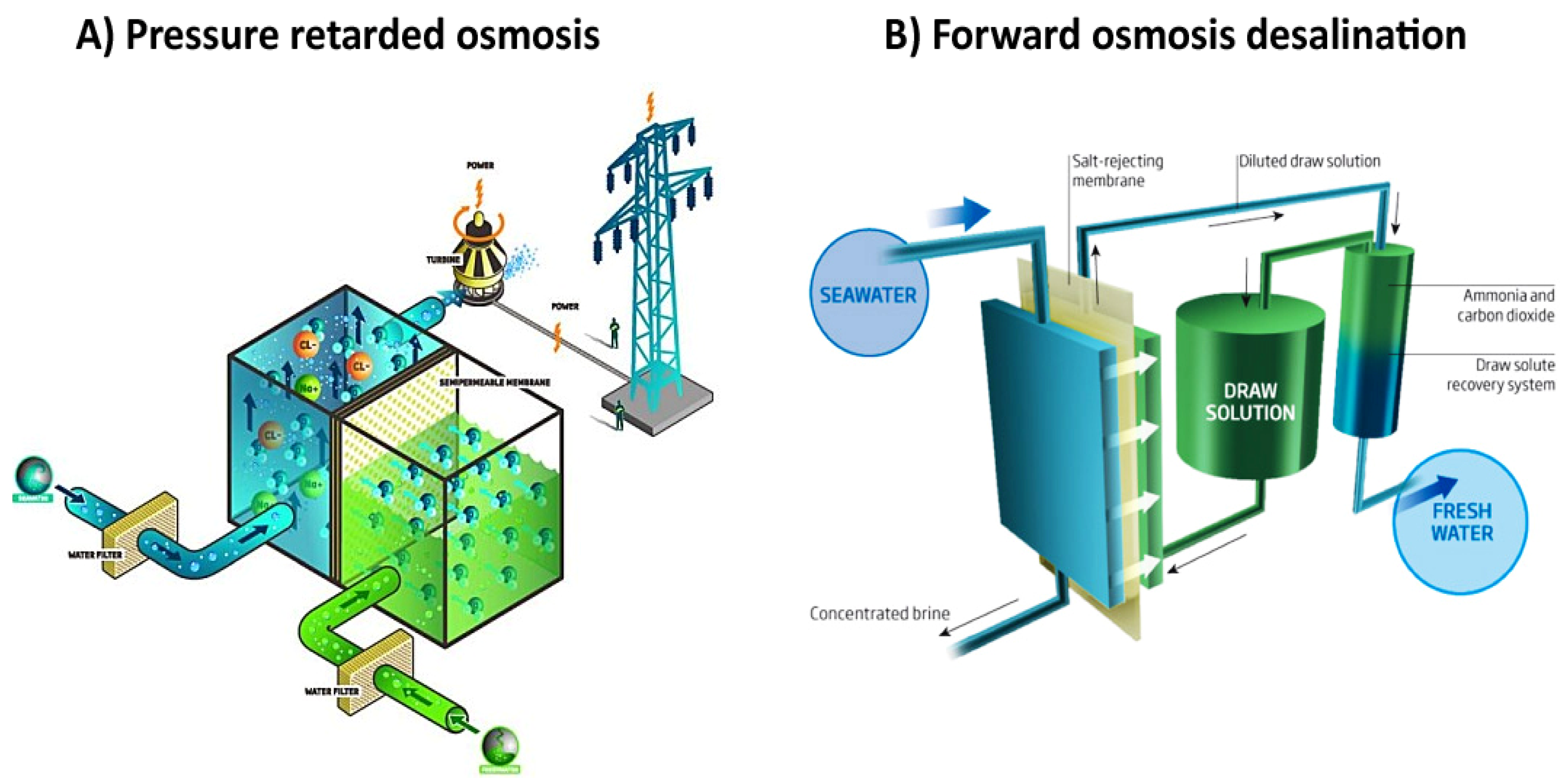
1.1. Osmotically Driven Membrane Processes
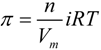
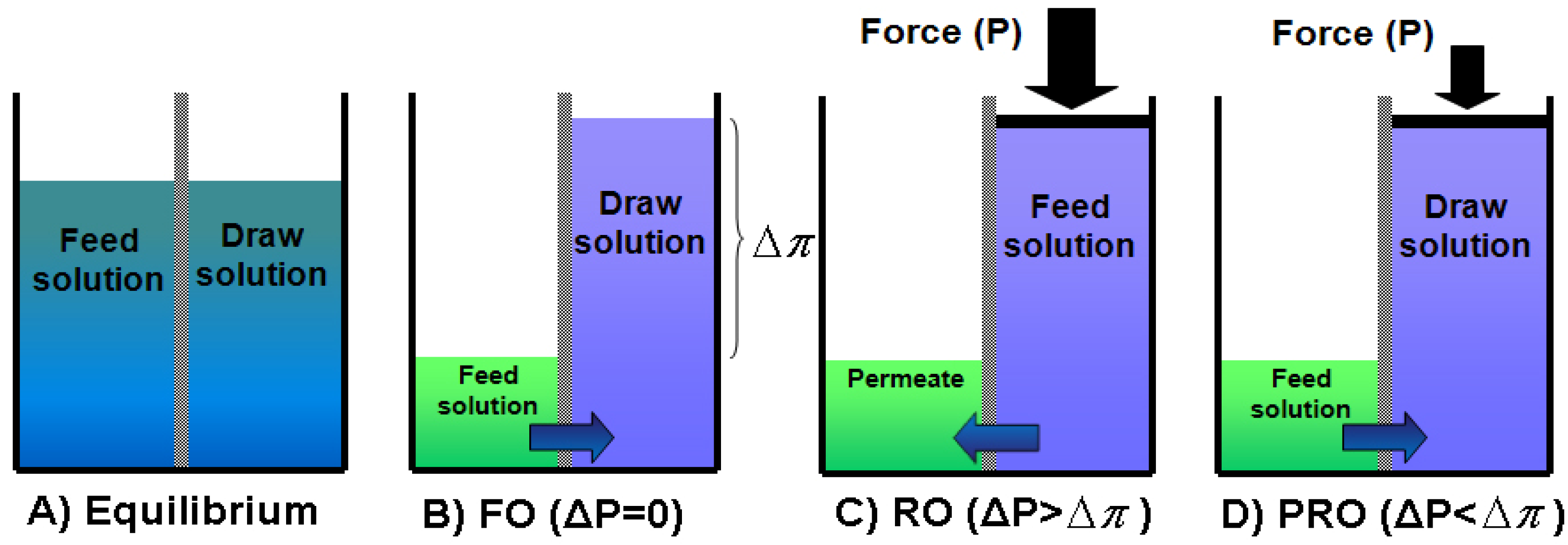
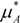 ) is equal to the chemical potential of the solvent in solution (μA) [40]:
) is equal to the chemical potential of the solvent in solution (μA) [40]:
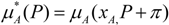
1.2. The A, B and S Parameters
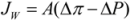

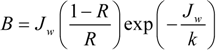
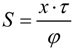
| Membrane Characteristics | S (μm) | Reference | ||
|---|---|---|---|---|
| FS * | Commercial FO membrane (CA) HTI | 481 | [41] | |
| 575 | [45] | |||
| FS * | TFC polysulfone-polyamide | 431 | [46] | |
| FS * | 670 | [47] | ||
| HF ** | TFC polyethersulfone-polyamide | 219 | [48] | |
| HF ** | 595 | [49] | ||
| FS * | Commercial RO membrane, DowFilmTec BW30 | With backing fabric | 37 500 | [47] |
| Without backing fabric | 14 000 | |||
1.3. Internal Concentration Polarization
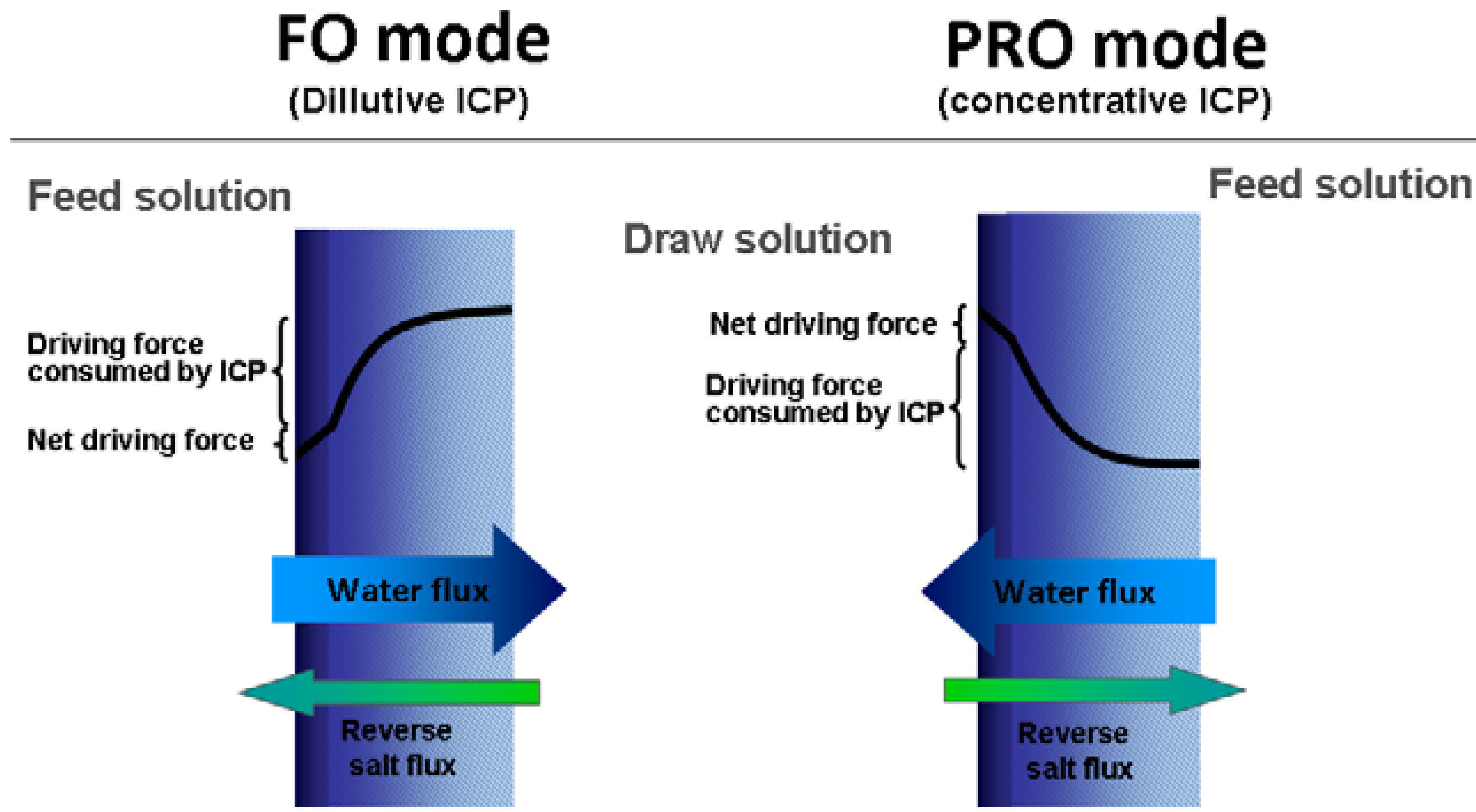
1.4. Reverse Draw Solute Permeation
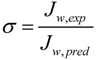
1.5. Fouling in Osmotically Driven Membrane Processes

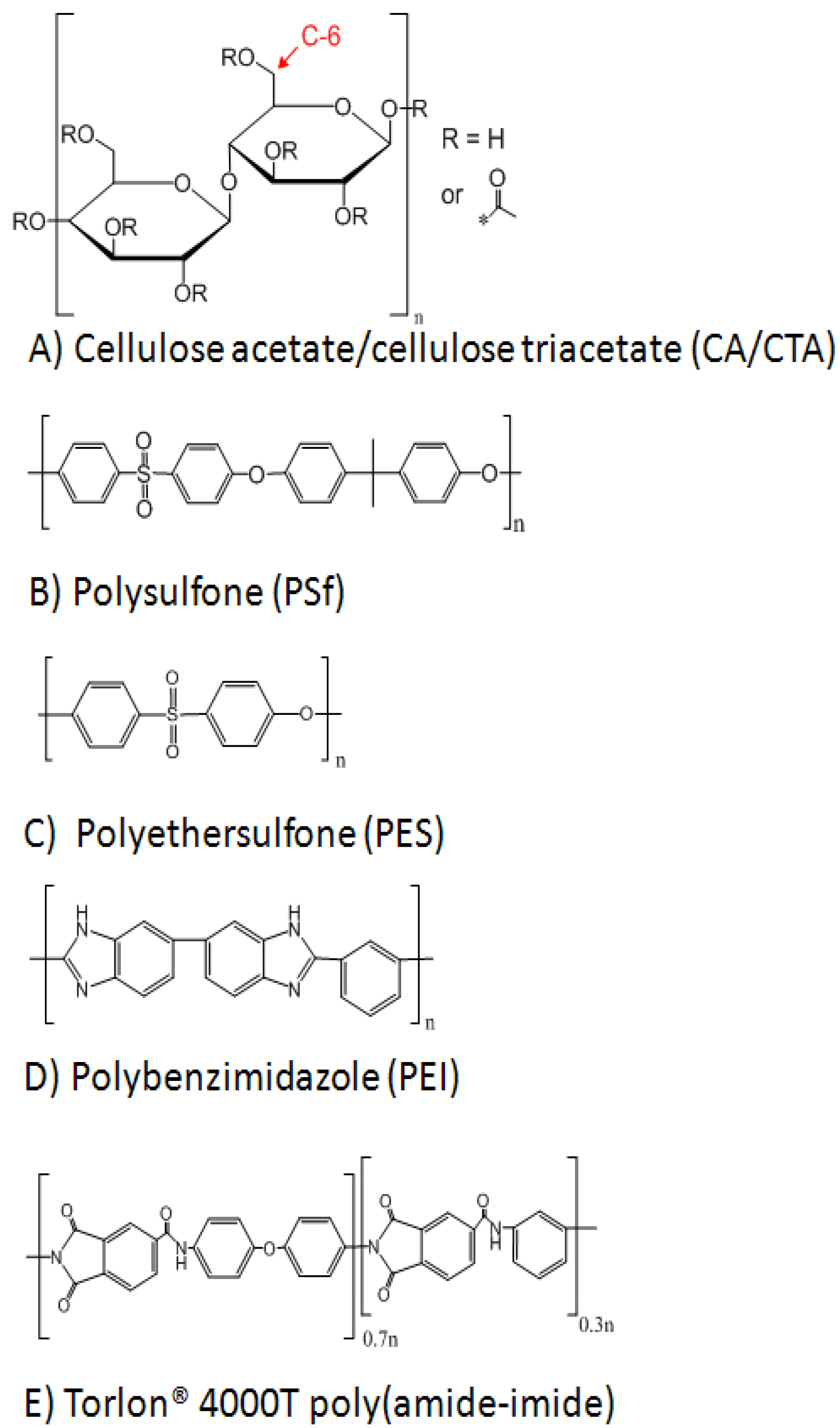
2. Membrane Materials
2.1. Cellulose Acetate
2.2. Polysulfone and Polyethersulfone
2.3. Polybenzimidazole
2.4. Poly(amide-imide)
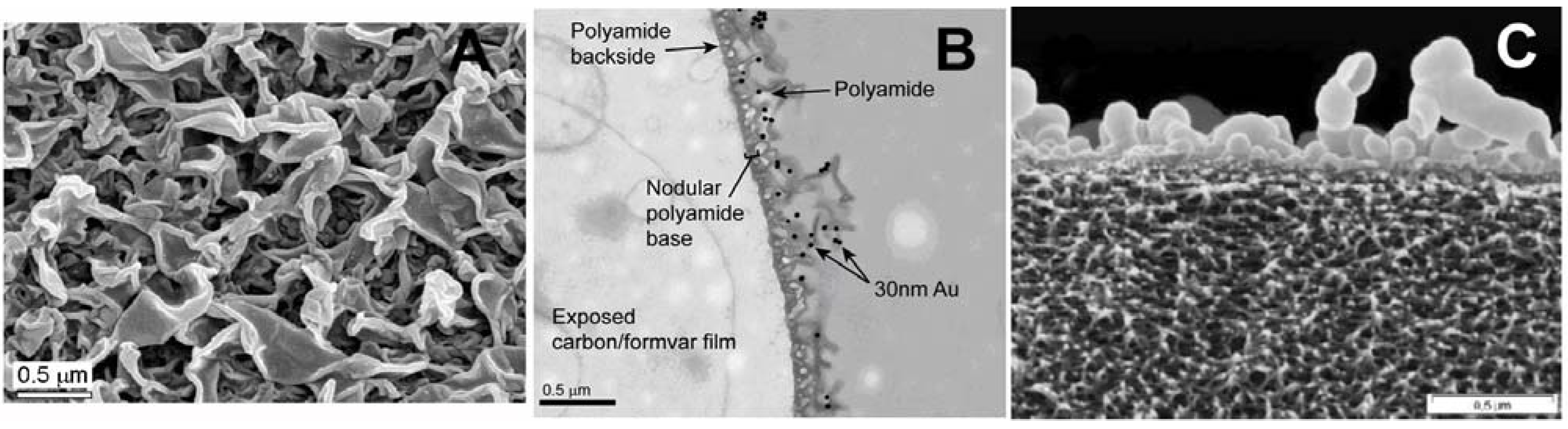
2.5. Polyamide

| Acid Chloride | Amine | Properties of Polyamide | Reference |
|---|---|---|---|
 Trimesoyl chloride (TMC) |  m-Phenylene diamine (MPD) | TMC-MPD is by far the most common combination of monomers in FO/PRO applications. This polyamide displays excellent flux and salt rejection properties. | [45,77,78,79] |
 p-Phenylene diamine (PPD) | Compared to the TMC-MPD polymer, the TMC-PPD membrane has lower molecular chain mobility. Resulting in a lower flux and higher salt rejection. | [80] | |
 2,2′-Benzidinedisulfonic acid (BDSA) | BDSA was used in addition to MPD and resulted in a more hydrophilic surface. Flux in RO increased as a function of BDSA concentration. Salt rejection increased with BDSA concentration (until 5% BDSA). | [81] | |
 2,6-Diaminetoluene(2,6-DAT) | Membranes synthesized from 2,6-DAT displayed a better chlorine tolerance than TMC-MPD membranes | [82] | |
 Methylated MPD | Methylated amino groups in MPD increases the average free volume of the polyamide. This resulted in decreased NaCl rejection and increase chlorine resistance. | [83] | |
 3,3,5,5-Biphenyl tetra-acyl chloride (BTEC) |  m-Phenylene diamine (MPD) | This combination gives a three-layer structure of polyamide. The dense middle layer consists of over 86% amide bonds. | [84] |
 Meta: Isophthaloyl chloride (IPC) Para: Terephthaloyl chloride (TPC) | The addition of diacyl chloride (IPC or TPC) in the organic solution resulted in enhanced flux in RO. | [85,86] |
3. Methods
3.1. Asymmetric Cellulose Acetate Membranes

3.2. Membrane Support Design

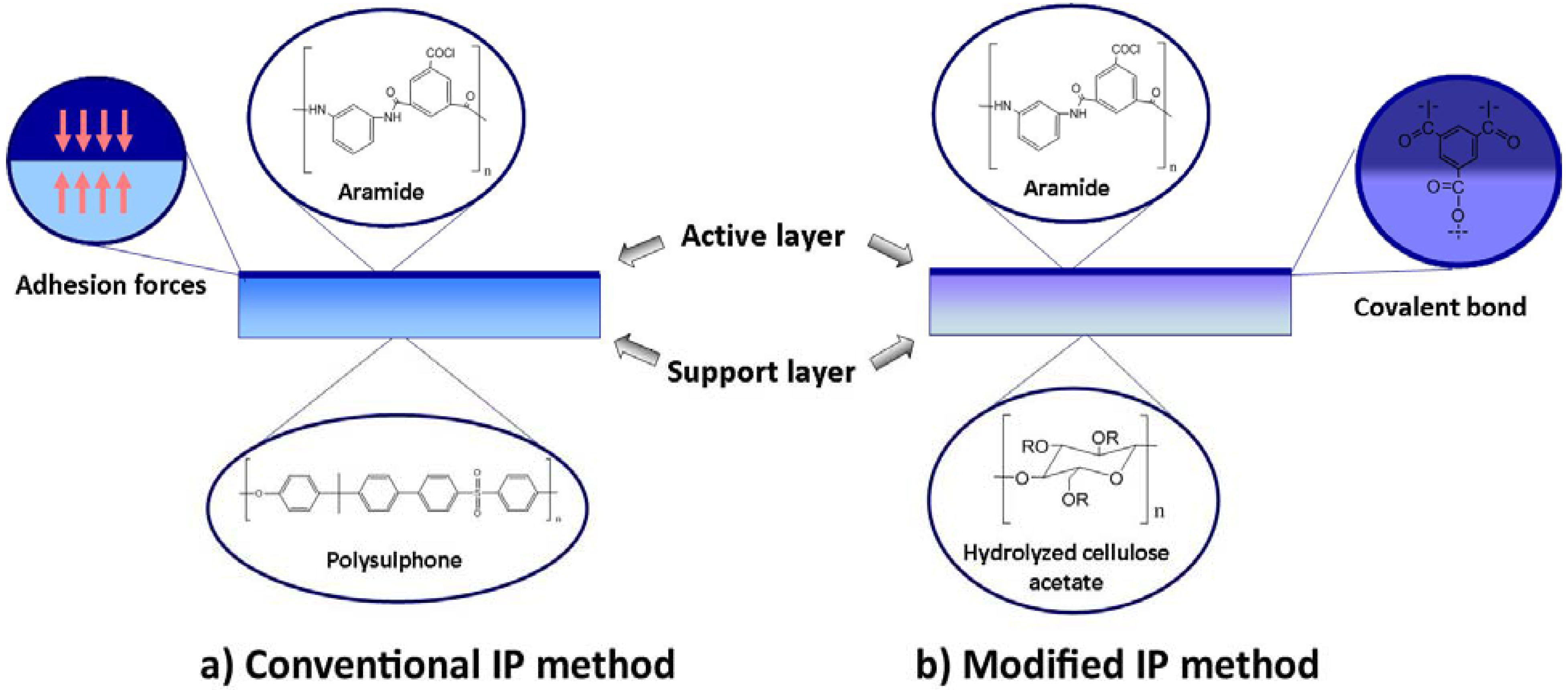
3.3. Interfacial Polymerization
3.4. Interfacial Polymerization on Hydrophilic Support
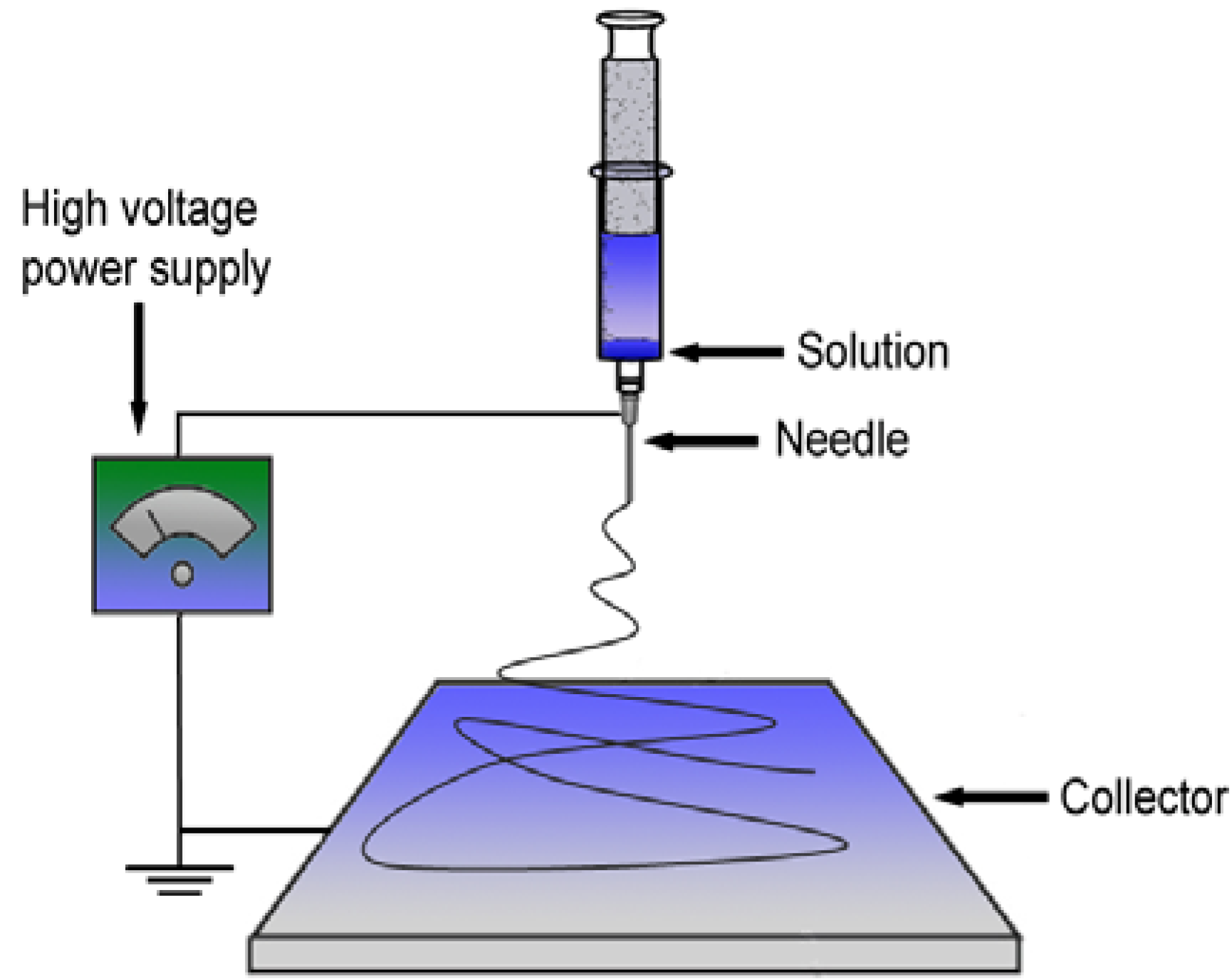
3.5. Electrospun Support
3.6. Hollow Fiber Membranes

3.7. Support Membrane Modification to Increase Hydrophilicity
3.8. New Trends in Composite Membrane Design
3.9. Draw Solutions (DS) an Important Variable to Consider along with the Material
4. Conclusions and Outlook
Acknowledgments
References and Notes
- Glater, J. The early history of reverse osmosis membrane development. Desalination 1998, 117, 297–309. [Google Scholar] [CrossRef]
- Nollet, A. Lecons de Physique Experimentale; Chez les Frères Guérin: Paris, France, 1748. [Google Scholar]
- Traube, M. Experimente zur Theorie der Zellenbildung und Endosmose. Archiv für Anatomie Physologie und wisserschlaftliche Med. 1867, 87–165. [Google Scholar]
- Loeb, S. The loeb-sourirajan membrane: How it came about. In Synthetic Membranes; American Chemical Society: Washington, DC, USA, 1981; Volume 153, pp. 1–9. [Google Scholar]
- Cadotte, J.E. Reverse Osmosis Membrane. U.S. Patent 4,039,440, 2 August 1977. [Google Scholar]
- Cadotte, J.E. Interfacially Synthesized Reverse Osmosis Membrane. U.S. Patent 4,277,344, 7 July 1981. [Google Scholar]
- Cadotte, J.E. Reverse Osmosis Membrane. U.S. Patent 4,259,183, 31 March 1981. [Google Scholar]
- Cadotte, J.E.; Petersen, R.J.; Larson, R.E.; Erickson, E.E. A new thin-film composite seawater reverse osmosis membrane. Desalination 1980, 32, 25–31. [Google Scholar] [CrossRef]
- Larson, R.E.; Cadotte, J.E.; Petersen, R.J. The FT-30 seawater reverse osmosis membrane—Element test results. Desalination 1981, 38, 473–483. [Google Scholar] [CrossRef]
- ACS Scifinder Home Page. Available online: http://www.cas.org/products/scifinder (accessed on 11 March 2013).
- Sidney, L. Production of energy from concentrated brines by pressure-retarded osmosis: I. Preliminary technical and economic correlations. J. Membr. Sci. 1976, 1, 49–63. [Google Scholar] [CrossRef]
- Loeb, S.; van Hessen, F.; Shahaf, D. Production of energy from concentrated brines by pressure-retarded osmosis: II. Experimental results and projected energy costs. J. Membr. Sci. 1976, 1, 249–269. [Google Scholar] [CrossRef]
- Statkraft Home Page. Available online: http://www.statkraft.no/jobb-og-karriere/ (accessed on 11 March 2013).
- Skilhagen, S.E.; Dugstad, J.E.; Aaberg, R.J. Osmotic power—Power production based on the osmotic pressure difference between waters with varying salt gradients. Desalination 2008, 220, 476–482. [Google Scholar] [CrossRef]
- Batchelder, G.W. Process for the Demineralization of Water. U.S. Patent 3,171,799, 3 February 1965. [Google Scholar]
- Glew, D.N. Process for Liquid Recovery and Solution Concentration. U.S. Patent 3,216,930, 11 September 1965. [Google Scholar]
- Frank, B.S. Desalination of Sea Water. U.S. Patent 3,670,897, 20 June 1972. [Google Scholar]
- Kravath, R.E.; Davis, J.A. Desalination of sea water by direct osmosis. Desalination 1975, 16, 151–155. [Google Scholar] [CrossRef]
- Stache, K. Apparatus for Transforming Sea Water, Brackish Water, Polluted Water or the Like into a Nutritious Drink by Means of Osmosis. U.S. Patent 4,879,030, 11 July 1989. [Google Scholar]
- Yaeli, J. Method and Apparatus for Processing Liquid Solutions of Suspensions Particularly Useful in the Desalination of Saline Water. U.S. Patent 5,098,575, 24 March 1992. [Google Scholar]
- McGinnis, R. Osmotic Desalination Process. U.S. Patent 7,560,029 B2, 14 July 2009. [Google Scholar]
- Ling, M.M.; Wang, K.Y.; Chung, T.-S. Highly water-soluble magnetic nanoparticles as novel draw solutes in forward osmosis for water reuse. Ind. Eng. Chem. Res. 2010, 49, 5869–5876. [Google Scholar] [CrossRef]
- McCutcheon, J.R.; McGinnis, R.L.; Elimelech, M. A novel ammonia-carbon dioxide forward (direct) osmosis desalination process. Desalination 2005, 174, 1–11. [Google Scholar] [CrossRef]
- Cath, T.Y.; Childress, A.E.; Elimelech, M. Forward osmosis: Principles, applications, and recent developments. J. Membr. Sci. 2006, 281, 70–87. [Google Scholar] [CrossRef]
- Cath, T.Y.; Gormly, S.; Beaudry, E.G.; Flynn, M.T.; Adams, V.D.; Childress, A.E. Membrane contactor processes for wastewater reclamation in space: Part I. Direct osmotic concentration as pretreatment for reverse osmosis. J. Membr. Sci. 2005, 257, 85–98. [Google Scholar] [CrossRef]
- Hydration Technology Innovations Home Page. Available online: http://www.htiwater.com/ (accessed on 11 March 2013).
- Dalla Rosa, M.; Giroux, F. Osmotic treatments (OT) and problems related to the solution management. J. Food Eng. 2001, 49, 223–236. [Google Scholar] [CrossRef]
- Wright, J.C.; Johnson, R.M.; Yum, S.I. Duros® osmotic pharmaceutical systems for parenteral & site-directed therapy. Drug Dev. Deliv. 2003, 3, 64–73. [Google Scholar]
- Holloway, R.W.; Childress, A.E.; Dennett, K.E.; Cath, T.Y. Forward osmosis for concentration of anaerobic digester centrate. Water Res. 2007, 41, 4005–4014. [Google Scholar] [CrossRef]
- Elimelech, M.; Phillip, W.A. The future of seawater desalination: Energy, technology, and the environment. Science 2011, 333, 712–717. [Google Scholar] [CrossRef]
- Zhao, S.; Zou, L.; Tang, C.Y.; Mulcahy, D. Recent developments in forward osmosis: Opportunities and challenges. J. Membr. Sci. 2012, 396, 1–21. [Google Scholar] [CrossRef]
- Chung, T.-S.; Zhang, S.; Wang, K.Y.; Su, J.; Ling, M.M. Forward osmosis processes: Yesterday, today and tomorrow. Desalination 2012, 287, 78–81. [Google Scholar] [CrossRef]
- Hoover, L.A.; Phillip, W.A.; Tiraferri, A.; Yip, N.Y.; Elimelech, M. Forward with osmosis: Emerging applications for greater sustainability. Environ. Sci. Technol. 2011, 45, 9824–9830. [Google Scholar]
- Achilli, A.; Cath, T.Y.; Childress, A.E. Power generation with pressure retarded osmosis: An experimental and theoretical investigation. J. Membr. Sci. 2009, 343, 42–52. [Google Scholar] [CrossRef]
- Achilli, A.; Cath, T.Y.; Marchand, E.A.; Childress, A.E. The forward osmosis membrane bioreactor: A low fouling alternative to mbr processes. Desalination 2009, 239, 10–21. [Google Scholar] [CrossRef]
- Chung, T.-S.; Li, X.; Ong, R.C.; Ge, Q.; Wang, H.; Han, G. Emerging forward osmosis (FO) technologies and challenges ahead for clean water and clean energy applications. Curr. Opin. Chem. Eng. 2012, 1, 246–257. [Google Scholar] [CrossRef]
- Achilli, A.; Childress, A.E. Pressure retarded osmosis: From the vision of sidney loeb to the first prototype installation—Review. Desalination 2010, 261, 205–211. [Google Scholar] [CrossRef]
- Zhang, S.; Fu, F.; Chung, T.-S. Substrate modifications and alcohol treatment on thin film composite membranes for osmotic power. Chem. Eng. Sci. 2013, 87, 40–50. [Google Scholar] [CrossRef]
- Greentech Media Home Page. Available online: http://www.greentechmedia.com/ (accessed on 11 March 2013).
- Mulder, M. Basic Principles of Membrane Technology; Kluver Academic Publishers: Dordrecht, The Netherlands, 1996. [Google Scholar]
- Phillip, W.A.; Yong, J.S.; Elimelech, M. Reverse draw solute permeation in forward osmosis: Modeling and experiments. Environ. Sci. Technol. 2010, 44, 5170–5176. [Google Scholar] [CrossRef]
- Loeb, S.; Titelman, L.; Korngold, E.; Freiman, J. Effect of porous support fabric on osmosis through a loeb-sourirajan type asymmetric membrane. J. Membr. Sci. 1997, 129, 243–249. [Google Scholar]
- Tiraferri, A.; Yip, N.Y.; Phillip, W.A.; Schiffman, J.D.; Elimelech, M. Relating performance of thin-film composite forward osmosis membranes to support layer formation and structure. J. Membr. Sci. 2011, 367, 340–352. [Google Scholar] [CrossRef]
- Gerstandt, K.; Peinemann, K.V.; Skilhagen, S.E.; Thorsen, T.; Holt, T. Membrane processes in energy supply for an osmotic power plant. Desalination 2008, 224, 64–70. [Google Scholar] [CrossRef]
- Chou, S.; Shi, L.; Wang, R.; Tang, C.Y.; Qiu, C.; Fane, A.G. Characteristics and potential applications of a novel forward osmosis hollow fiber membrane. Desalination 2010, 261, 365–372. [Google Scholar] [CrossRef]
- Yip, N.Y.; Tiraferri, A.; Phillip, W.A.; Schiffman, J.D.; Elimelech, M. High performance thin-film composite forward osmosis membrane. Environ. Sci. Technol. 2010, 44, 3812–3818. [Google Scholar]
- Wei, J.; Qiu, C.; Tang, C.Y.; Wang, R.; Fane, A.G. Synthesis and characterization of flat-sheet thin film composite forward osmosis membranes. J. Membr. Sci. 2011, 372, 292–302. [Google Scholar] [CrossRef]
- Sukitpaneenit, P.; Chung, T.-S. High performance thin-film composite forward osmosis hollow fiber membranes with macrovoid-free and highly porous structure for sustainable water production. Environ. Sci. Technol. 2012, 46, 7358–7365. [Google Scholar] [CrossRef]
- Wang, R.; Shi, L.; Tang, C.Y.; Chou, S.; Qiu, C.; Fane, A.G. Characterization of novel forward osmosis hollow fiber membranes. J. Membr. Sci. 2010, 355, 158–167. [Google Scholar] [CrossRef]
- Gray, G.T.; McCutcheon, J.R.; Elimelech, M. Internal concentration polarization in forward osmosis: Role of membrane orientation. Desalination 2006, 197, 1–8. [Google Scholar] [CrossRef]
- McCutcheon, J.R.; Elimelech, M. Influence of concentrative and dilutive internal concentration polarization on flux behavior in forward osmosis. J. Membr. Sci. 2006, 284, 237–247. [Google Scholar] [CrossRef]
- Lee, K.L.; Baker, R.W.; Lonsdale, H.K. Membranes for power generation by pressure-retarded osmosis. J. Membr. Sci. 1981, 8, 141–171. [Google Scholar] [CrossRef]
- Hancock, N.T.; Cath, T.Y. Solute coupled diffusion in osmotically driven membrane processes. Environ. Sci. Technol. 2009, 43, 6769–6775. [Google Scholar] [CrossRef]
- Hancock, N.T.; Phillip, W.A.; Elimelech, M.; Cath, T.Y. Bidirectional permeation of electrolytes in osmotically driven membrane processes. Environ. Sci. Technol. 2011, 45, 10642–10651. [Google Scholar] [CrossRef]
- Yong, J.S.; Phillip, W.A.; Elimelech, M. Coupled reverse draw solute permeation and water flux in forward osmosis with neutral draw solutes. J. Membr. Sci. 2012, 392–393, 9–17. [Google Scholar]
- Zelman, A. Membrane permeability: Generalization of the reflection coefficient method of describing volume and solute flows. Biophys. J. 1972, 12, 414–419. [Google Scholar] [CrossRef]
- Amy, G. Fundamental understanding of organic matter fouling of membranes. Desalination 2008, 231, 44–51. [Google Scholar] [CrossRef]
- Rana, D.; Matsuura, T. Surface modifications for antifouling membranes. Chem. Rev. 2010, 110, 2448–2471. [Google Scholar] [CrossRef]
- Seidel, A.; Elimelech, M. Coupling between chemical and physical interactions in natural organic matter (NOM) fouling of nanofiltration membranes: Implications for fouling control. J. Membr. Sci. 2002, 203, 245–255. [Google Scholar] [CrossRef]
- Thorsen, T. Concentration polarisation by natural organic matter (NOM) in NF and UF. J. Membr. Sci. 2004, 233, 79–91. [Google Scholar] [CrossRef]
- Mi, B.; Elimelech, M. Gypsum scaling and cleaning in forward osmosis: Measurements and mechanisms. Environ. Sci. Technol. 2010, 44, 2022–2028. [Google Scholar] [CrossRef]
- Lee, S.; Boo, C.; Elimelech, M.; Hong, S. Comparison of fouling behavior in forward osmosis (FO) and reverse osmosis (RO). J. Membr. Sci. 2010, 365, 34–39. [Google Scholar] [CrossRef]
- Boo, C.; Lee, S.; Elimelech, M.; Meng, Z.; Hong, S. Colloidal fouling in forward osmosis: Role of reverse salt diffusion. J. Membr. Sci. 2012, 390–391, 277–284. [Google Scholar]
- Mi, B.; Elimelech, M. Chemical and physical aspects of organic fouling of forward osmosis membranes. J. Membr. Sci. 2008, 320, 292–302. [Google Scholar] [CrossRef]
- Liu, Y.; Mi, B. Combined fouling of forward osmosis membranes: Synergistic foulant interaction and direct observation of fouling layer formation. J. Membr. Sci. 2012, 407–408, 136–144. [Google Scholar] [CrossRef]
- Mi, B.; Elimelech, M. Organic fouling of forward osmosis membranes: Fouling reversibility and cleaning without chemical reagents. J. Membr. Sci. 2010, 348, 337–345. [Google Scholar] [CrossRef]
- Carraher, C.E. Polymer Chemistry, 6th ed; Marcel Dekker: New York, NY, USA, 2003. [Google Scholar]
- McCutcheon, J.R.; Elimelech, M. Influence of membrane support layer hydrophobicity on water flux in osmotically driven membrane processes. J. Membr. Sci. 2008, 318, 458–466. [Google Scholar] [CrossRef]
- Saunders, K.J. Organic Polymer Chemistry, 2nd ed; Chapman Hall: New York, NY, USA, 1988. [Google Scholar]
- Wang, K.Y.; Chung, T.-S.; Qin, J.-J. Polybenzimidazole (pbi) nanofiltration hollow fiber membranes applied in forward osmosis process. J. Membr. Sci. 2007, 300, 6–12. [Google Scholar] [CrossRef]
- Sawyer, L.C.; Jones, R.S. Observations on the structure of first generation polybenzimidazole reverse osmosis membranes. J. Membr. Sci. 1984, 20, 147–166. [Google Scholar] [CrossRef]
- Solvay Specicality Polymers. Technical Data Sheet, Torlon®4000t. Available online: http://catalog.ides.com/datasheet.aspx?I=42041&FMT=PDF&E=135275 (accessed on 11 March 2013).
- Robertson, G.P.; Guiver, M.D.; Yoshikawa, M.; Brownstein, S. Structural determination of torlon® 4000t polyamide–imide by nmr spectroscopy. Polymer 2004, 45, 1111–1117. [Google Scholar] [CrossRef]
- Setiawan, L.; Wang, R.; Li, K.; Fane, A.G. Fabrication of novel poly(amide–imide) forward osmosis hollow fiber membranes with a positively charged nanofiltration-like selective layer. J. Membr. Sci. 2011, 369, 196–205. [Google Scholar] [CrossRef]
- Wittbecker, E.L.; Morgan, P.W. Interfacial polycondensation. I. J. Polym. Sci. 1959, 40, 289–297. [Google Scholar] [CrossRef]
- Morgan, P.W.; Kwolek, S.L. Interfacial polycondensation. II. Fundamentals of polymer formation at liquid interfaces. J. Polym. Sci. 1959, 40, 299–327. [Google Scholar] [CrossRef]
- Alsvik, I.L.; Hägg, M.B. Preparation of thin film composite membranes with polyamide film on hydrophilic supports. J. Membr. Sci. 2013, 428, 225–231. [Google Scholar] [CrossRef]
- Nilsen, T.-N.; Alsvik, I.L. Thin Film Composites. WO Patent 2011/152735, 8 December 2011. [Google Scholar]
- Shi, L.; Chou, S.R.; Wang, R.; Fang, W.X.; Tang, C.Y.; Fane, A.G. Effect of substrate structure on the performance of thin-film composite forward osmosis hollow fiber membranes. J. Membr. Sci. 2011, 382, 116–123. [Google Scholar] [CrossRef]
- Juhn Roh, I. Effect of the physicochemical properties on the permeation performance in fully aromatic crosslinked polyamide thin films. J. Appl. Polym. Sci. 2003, 87, 569–576. [Google Scholar] [CrossRef]
- Baroña, G.N.B.; Lim, J.; Jung, B. High performance thin film composite polyamide reverse osmosis membrane prepared via m-phenylenediamine and 2,2′-benzidinedisulfonic acid. Desalination 2012, 291, 69–77. [Google Scholar] [CrossRef]
- Son, S.H.; Jegal, J. Preparation and characterization of polyamide reverse-osmosis membranes with good chlorine tolerance. J. Appl. Polym. Sci. 2011, 120, 1245–1252. [Google Scholar] [CrossRef]
- Shintani, T.; Shimazu, A.; Yahagi, S.; Matsuyama, H. Characterization of methyl-substituted polyamides used for reverse osmosis membranes by positron annihilation lifetime spectroscopy and md simulation. J. Appl. Polym. Sci. 2009, 113, 1757–1762. [Google Scholar] [CrossRef]
- Liu, Y.; He, B.; Li, J.; Sanderson, R.D.; Li, L.; Zhang, S. Formation and structural evolution of biphenyl polyamide thin film on hollow fiber membrane during interfacial polymerization. J. Membr. Sci. 2011, 373, 98–106. [Google Scholar] [CrossRef]
- Yu, S.; Liu, M.; Liu, X.; Gao, C. Performance enhancement in interfacially synthesized thin-film composite polyamide-urethane reverse osmosis membrane for seawater desalination. J. Membr. Sci. 2009, 342, 313–320. [Google Scholar]
- Zhou, Y.; Yu, S.; Liu, M.; Chen, H.; Gao, C. Effect of mixed crosslinking agents on performance of thin-film-composite membranes. Desalination 2006, 192, 182–189. [Google Scholar]
- Herron, J. Asymmetric Forward Osmosis Membranes. U.S. Patent 7,445,712, 4 November 2008. [Google Scholar]
- Herron, J. Two-Layer Membrane. U.S. Patent 0,175,300 A1, 12 July 2012. [Google Scholar]
- Smoke, J. HTI’s New Thin Film Forward Osmosis Membrane in Production. Available online: http://www.htiwater.com/news/press-room/content/2012/press-HTI-HTIThinFilmMembrane042512.pdf (accessed on 11 March 2013).
- Freger, V. Nanoscale heterogeneity of polyamide membranes formed by interfacial polymerization. Langmuir 2003, 19, 4791–4797. [Google Scholar] [CrossRef]
- Freger, V.; Srebnik, S. Mathematical model of charge and density distributions in interfacial polymerization of thin films. J. Appl. Polym. Sci. 2003, 88, 1162–1169. [Google Scholar] [CrossRef]
- Freger, V. Kinetics of film formation by interfacial polycondensation. Langmuir 2005, 21, 1884–1894. [Google Scholar] [CrossRef]
- Schaep, J.; Vandecasteele, C. Evaluating the charge of nanofiltration membranes. J. Membr. Sci. 2001, 188, 129–136. [Google Scholar] [CrossRef]
- Pacheco, F.A.; Pinnau, I.; Reinhard, M.; Leckie, J.O. Characterization of isolated polyamide thin films of ro and nf membranes using novel tem techniques. J. Membr. Sci. 2010, 358, 51–59. [Google Scholar] [CrossRef]
- Ghosh, A.K.; Hoek, E.M.V. Impacts of support membrane structure and chemistry on polyamide–polysulfone interfacial composite membranes. J. Membr. Sci. 2009, 336, 140–148. [Google Scholar] [CrossRef]
- Ghosh, A.K.; Jeong, B.-H.; Huang, X.; Hoek, E.M.V. Impacts of reaction and curing conditions on polyamide composite reverse osmosis membrane properties. J. Membr. Sci. 2008, 311, 34–45. [Google Scholar] [CrossRef]
- Alsvik, I.L.; Katherine, Z.; Elimelech, M.; Hägg, M.-B. Polyamide formation on a cellulose triacetate support for osmotic membranes: Effect of linking molecules on membrane performance. Desalination 2013, 312, 2–9. [Google Scholar] [CrossRef]
- Ramakrishna, S. Introduction to Electrospinning and Nanofibers; World Scientific Publishing Co.: River Edge, NJ, USA, 2005. [Google Scholar]
- Reneker, D.H.; Chun, I. Nanometre diameter fibres of polymer, produced by electrospinning. Nanotechnology 1996, 7, 216. [Google Scholar] [CrossRef]
- He, J.-H. Electrospun Nanofibers and Their Applications; Smithers Rapra: Shrewsbury, UK, 2008. [Google Scholar]
- Stranger, J.; Tucker, N.; Staiger, M. Electrospinning; Smithers Rapra: Shrevsbury, UK, 2009. [Google Scholar]
- Bui, N.-N.; Lind, M.L.; Hoek, E.M.V.; McCutcheon, J.R. Electrospun nanofiber supported thin film composite membranes for engineered osmosis. J. Membr. Sci. 2011, 385–386, 10–19. [Google Scholar]
- Clausi, D.T.; Koros, W.J. Formation of defect-free polyimide hollow fiber membranes for gas separations. J. Membr. Sci. 2000, 167, 79–89. [Google Scholar] [CrossRef]
- Wang, K.Y.; Yang, Q.; Chung, T.-S.; Rajagopalan, R. Enhanced forward osmosis from chemically modified polybenzimidazole (pbi) nanofiltration hollow fiber membranes with a thin wall. Chem. Eng. Sci. 2009, 64, 1577–1584. [Google Scholar] [CrossRef]
- Fang, W.; Wang, R.; Chou, S.; Setiawan, L.; Fane, A.G. Composite forward osmosis hollow fiber membranes: Integration of RO- and NF-like selective layers to enhance membrane properties of anti-scaling and anti-internal concentration polarization. J. Membr. Sci. 2012, 394–395, 140–150. [Google Scholar]
- Arena, J.T.; McCloskey, B.; Freeman, B.D.; McCutcheon, J.R. Surface modification of thin film composite membrane support layers with polydopamine: Enabling use of reverse osmosis membranes in pressure retarded osmosis. J. Membr. Sci. 2011, 375, 55–62. [Google Scholar] [CrossRef]
- Tiraferri, A.; Kang, Y.; Gianellis, E.P.; Elimelech, M. Highly hydrophilic thin-film composite forward osmosis membranes functionalized with surface-tailored nanoparticles. ACS Appl. Mater. Interf. 2012, 4, 5044–5053. [Google Scholar] [CrossRef]
- Hoover, L.A.; Schiffman, J.D.; Elimelech, M. Nanofibers in thin-film composite membrane support layers: Enabling expanded application of forward and pressure retarded osmosis. Desalination 2013, 308, 73–81. [Google Scholar] [CrossRef]
- Phuntsho, S.; Shon, H.K.; Hong, S.; Lee, S.; Vigneswaran, S. A novel low energy fertilizer driven forward osmosis desalination for direct fertigation: Evaluation the performance of fertilizer draw solutions. J. Membr. Sci. 2011, 375, 172–181. [Google Scholar] [CrossRef]
- Achili, A.; Cath, T.Y.; Childress, A.E. Selection of inorganic-based draw solutions for forward osmosis applications. J. Membr. Sci. 2010, 364, 233–241. [Google Scholar] [CrossRef]
- Ngu, H.Y.; Tang, W. Forward (direct) osmosis: A novel and prospective process for brine control. Water Environ. Found. 2006, 4345–4352. [Google Scholar]
- Ling, M.M.; Chung, T.-S. Novel dual–stage FO system for sustainable protein enrichment using nanoparticles as intermediate draw solutes. J. Membr. Sci. 2011, 372, 201–209. [Google Scholar] [CrossRef]
- Checkli, L.; Phuntsho, S.; Shon, H.K.; Vigneswaran, S.; Kandasamy, J.; Chahan, A. A review of draw solutes in forward osmosis process and their use in modern applications. Desalin. Water Treat. 2012, 43, 167–184. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Alsvik, I.L.; Hägg, M.-B. Pressure Retarded Osmosis and Forward Osmosis Membranes: Materials and Methods. Polymers 2013, 5, 303-327. https://doi.org/10.3390/polym5010303
Alsvik IL, Hägg M-B. Pressure Retarded Osmosis and Forward Osmosis Membranes: Materials and Methods. Polymers. 2013; 5(1):303-327. https://doi.org/10.3390/polym5010303
Chicago/Turabian StyleAlsvik, Inger Lise, and May-Britt Hägg. 2013. "Pressure Retarded Osmosis and Forward Osmosis Membranes: Materials and Methods" Polymers 5, no. 1: 303-327. https://doi.org/10.3390/polym5010303