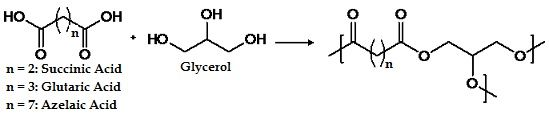
Degree of Branching in Hyperbranched Poly(glycerol-co-diacid)s Synthesized in Toluene
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Polymerizations in Toluene
2.3. Neat Polymerizations
2.4. Nuclear Magnetic Resonance (NMR)
3. Results and Discussion
3.1. NMR Assignments and Integrations
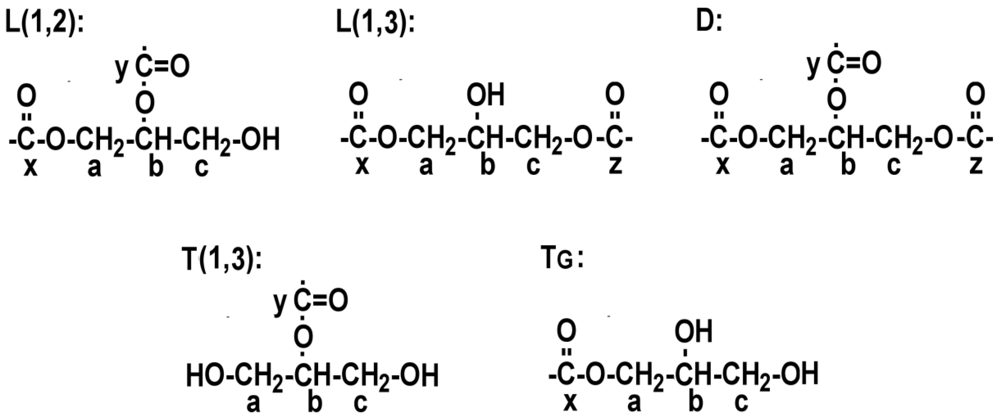

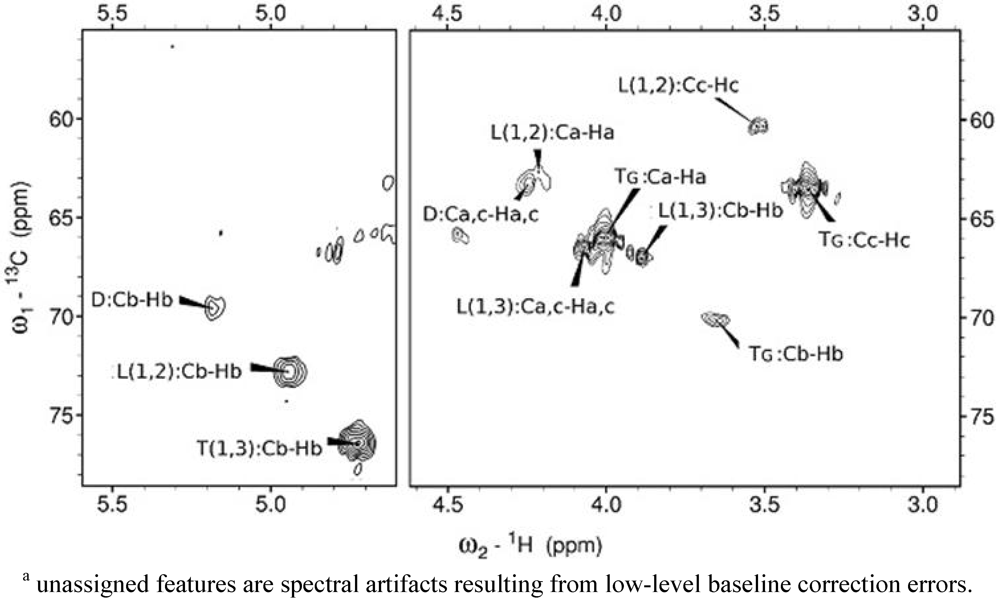

| Atom position in branching pattern | Glutaric Acid:Glycerol | Succinic Acid:Glycerol | Azelaic Acid:Glycerol | |||
|---|---|---|---|---|---|---|
| PPM | 1H | 13C | 1H | 13C | 1H | 13C |
| a | ||||||
| D | 4.29(±0.02) | 61.98(±0.29) | 4.24(±0.01) | 62.17(±0.07) | 4.27(±0.01) | 62.06(±0.26) |
| L(1,2) | 4.26(±0.01) | 62.57(±0.09) | 4.21(±0.01) | 62.68(±0.11) | 4.24(±0.02) | 62.62(±0.19) |
| T(1,3) | 3.44(±0.29) | 59.98(±0.05) | 3.47(±0.04) | 59.85(±0.09) | 3.46(±0.03) | 59.93(±0.05) |
| L(1,3) | 4.11(+0.08) | 65.05(+0.08) | 4.07(+0.01) | 65.32(+0.08) | 4.12(+0.03) | 64.88(±0.04) |
| TG | 4.08(±0.06) | 65.75(±0.11) | 4.04(±0.01) | 66.00(±0.07) | 4.08(±0.02) | 65.63(±0.03) |
| b | ||||||
| D | 5.22(±0.02) | 68.95(±0.26) | 5.19(±0.01) | 69.09(±0.10) | 5.22(±0.04) | 68.89(±0.03) |
| L(1,2) | 4.97(±0.01) | 72.30(±0.19) | 4.94(±0.01) | 72.51(±0.15) | 4.95(±0.08) | 72.29(±0.32) |
| T(1,3) | 4.77(±0.02) | 75.61(±0.40) | 4.73(±0.02) | 75.90(±0.20) | 4.75(±0.05) | 75.51(±0.03) |
| L(1,3) | 3.90(±0.02) | 66.34(±0.11) | 3.88(±0.02) | 66.26(±0.10) | 3.90(±0.04) | 66.30(±0.05) |
| TG | 3.65(±0.02) | 69.59(±0.31) | 3.65(±0.01) | 69.44(±0.11) | 3.66(±0.05) | 69.45(±0.04) |
| c | ||||||
| D | 4.29(±0.02) | 62.06(±0.36) | 4.24(±0.01) | 62.17(±0.07) | 4.27(±0.01) | 62.06(±0.26) |
| L(1,2) | 3.70(±0.38) | 60.48(±1.15) | 3.51(±0.02) | 59.56(±0.09) | 3.50(±0.02) | 59.68(±0.04) |
| T(1,3) | 3.45(±0.04) | 59.89(±0.16) | 3.47(±0.04) | 59.85(±0.09) | 3.46(±0.03) | 59.93(±0.05) |
| L(1,3) | 3.93(±0.33) | 63.63(±2.44) | 4.07(±0.01) | 65.33(±0.08) | 4.12(±0.03) | 64.88(±0.04) |
| TG | 3.52(±0.35) | 63.43(±0.95) | 3.39(±0.03) | 63.23(±0.07) | 3.39(±0.03) | 63.11(±0.22) |
| Diacid | D | L(1,2) | T(1,3) | L(1,3) | TG | D.B.% | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1:1 molar ratio to glycerol (no Dean Stark apparatus), 110 °C, 24 h | |||||||||||||
| 1 | Succinic | 3.5 | 9.7 | 7.3 | 33.7 | 45.9 | 13.7 | ||||||
| 2 | Glutaric | 5.2 | 11.1 | 4.4 | 44.8 | 34.5 | 15.7 | ||||||
| 3 | Azelaic | 4.1 | 12.6 | 4.3 | 42.6 | 36.4 | 13.0 | ||||||
| 1:1 molar ratio to glycerol, 135 °C, 24 h | |||||||||||||
| 4 | Succinic | 13.74 | 22.7 | 8.51 | 35.8 | 19.3 | 31.8 | ||||||
| 5 | Glutaric | (Peaks too broad to interpret–see text) | |||||||||||
| 6 | Azelaic | 2.6 | 12 | 4.8 | 33.3 | 47.4 | 10.2 | ||||||
| 2:1 molar ratio to glycerol, 135 °C, 24 h | |||||||||||||
| 7 | Succinic | 22.0 | 25.6 | 8.88 | 34.4 | 9.08 | 42.4 | ||||||
| 8 | Glutaric | 61.3 | 8.3 | 1.2 | 18.2 | 11 | 82.2 | ||||||
| 9 | Azelaic | 3.2 | 17.9 | 4.78 | 21.0 | 53.1 | 14.1 | ||||||
| 1:1 molar ratio to glycerol, 155 °C, 24 h | |||||||||||||
| 10 | Glutaric | (Peaks too broad to interpret–see text) | |||||||||||
| 2:1 molar ratio with glycerol, 135oC, 72 h | |||||||||||||
| 11 | Azelaic | 24.7 | 40.7 | 1.2 | 28.4 | 5.1 | 41.7 | ||||||
| 2:1 molar ratio with glycerol, 135 °C, 10 h | |||||||||||||
| 12 | Glutaric | 47.5 | 18.6 | .5 | 29.1 | 4.3 | 66.6 | ||||||
| 2:1 molar ratio with glycerol, 155 °C, neat, 10 h | |||||||||||||
| 13 | Glutaric | 52.6 | 15.0 | 3.6 | 24.3 | 4.4 | 72.9 | ||||||
| 2:1 molar ratio to glycerol, 155 °C, 24 h | |||||||||||||
| 14 | Glutaric | (Peaks too broad to interpret – see text) | |||||||||||
| 2:1 molar ratio with glycerol, 155 °C, neat, 24 h | |||||||||||||
| 15 | Glutaric | (Peaks too broad to interpret–see text) | |||||||||||
| Diacid | D | L(1,2) | T(1,3) | L(1,3) | TG | D.B.% | |||||||
| 1:1 molar ratio to glycerol (no Dean Stark apparatus), 110 °C, 24 h | |||||||||||||
| 1 | Succinic | 3.5 | 30.7 | 3.5 | 22.9 | 43.0 | 11.5 | ||||||
| 2 | Glutaric | 4.1 | 29.7 | 4.6 | 24.6 | 37.0 | 13.2 | ||||||
| 3 | Azelaic | 4.4 | 28.4 | 5.1 | 21.8 | 40.3 | 15.0 | ||||||
| 1:1 molar ratio to glycerol, 135 °C, 24 h | |||||||||||||
| 4 | Succinic | 7.8 | 23.9 | 5.1 | 30.2 | 33.0 | 39.4 | ||||||
| 5 | Glutaric | 13.7 | 19.6 | 3.0 | 40.7 | 23.1 | 31.2 | ||||||
| 6 | Azelaic | 2.8 | 32.5 | 5.5 | 18.8 | 40.4 | 9.9 | ||||||
| 2:1 molar ratio to glycerol, 135 °C, 24 h | |||||||||||||
| 7 | Succinic | 9.1 | 19.4 | 3.0 | 37.6 | 31.0 | 41.9 | ||||||
| 8 | Glutaric | 63.9 | 5.6 | 0 | 15.9 | 14.5 | 85.6 | ||||||
| 9 | Azelaic | 4.0 | 27.0 | 4.9 | 22.1 | 42.1 | 13.9 | ||||||
| 1:1 molar ratio to glycerol, 155 °C, 24 h | |||||||||||||
| 10 | Glutaric | 15.6 | 19.5 | 0.9 | 43.2 | 20.8 | 33.3 | ||||||
| 2:1 molar ratio with glycerol, 135 °C, 72 h | |||||||||||||
| 11 | Azelaic | 27.4 | 16.2 | 1.2 | 41.9 | 13.4 | 48.5 | ||||||
| 2:1 molar ratio with glycerol, 135 °C, 10 h | |||||||||||||
| 12 | Glutaric | 54.7 | 5.0 | 0.0 | 39.8 | 0.5 | 70.9 | ||||||
| 2:1 molar ratio with glycerol, 155 °C, neat, 10 h | |||||||||||||
| 13 | Glutaric | 50.8 | 11.8 | 0.0 | 30.7 | 6.7 | 70.5 | ||||||
| 2:1 molar ratio with glycerol, 155 °C, 24 h | |||||||||||||
| 14 | Glutaric | 62.2 | 11.5 | 0.0 | 26.3 | 0.0 | 76.7 | ||||||
| 2:1 molar ratio with glycerol, 155 °C, neat, 24 h | |||||||||||||
| 15 | Glutaric | 78.2 | 3.9 | 0 | 17.9 | 0.0 | 87.8 | ||||||
3.2. Degree of Branching (D.B.%)
4. Conclusions
Acknowledgments
Disclaimer
References
- Lin, Q.; Long, T.E. Polymerization of A2 with B3 monomers: A facile approach to hyperbranched poly(aryl ester)s. Macromolecules 2003, 36, 9809–9816. [Google Scholar]
- Stumbe, J.-F.; Bruchmann, B. Hyperbranched polyesters based on adipic acid and glycerol. Macromol. Rapid Commun. 2004, 25, 921–924. [Google Scholar] [CrossRef]
- Kulshrestha, A.S.; Gao, W.; Gross, R.A. Glycerol copolyesters: Control of branching and molecular weight using lipase catalyst. Macromolecules 2005, 38, 3193–3204. [Google Scholar]
- Jikei, M.; Kakimoto, M.A. Hyperbranched aromatic polyamides prepared by direct polycondensation. High Perform. Polym. 2001, 13, 33–43. [Google Scholar]
- Fang, J.; Kita, H.; Okamoto, K. Hyperbranched polyimide for gas separation applications. 1. Synthesis and characterization. Macromolecules 2000, 33, 4639–4646. [Google Scholar]
- Komber, H.; Voit, B.; Monticelli, O.; Russo, S. 1H and 13C Spectra of a hyperbranched aromatic polyamide from p-phenylenediamine and trimesic acid. Macromolecules 2001, 34, 5487–5493. [Google Scholar]
- Wyatt, V.T.; Haas, M.J.; Strahan, G.D.; Nunez, A. Characterization of thermal and mechanical properties of hyperbranched oligo(glycerol-glutaric acid)s. J. Biobased Mater. Bioenergy 2011, 5, 92–101. [Google Scholar]
- Voit, B.I. Dendritic polymers: From aesthetic macromolecules to commercially interesting materials. Acta Polym. 1995, 46, 87–99. [Google Scholar]
- Tomalia, D.A.; Frechet, J.M.J. Discovery of dendrimers and dendritic polymers: A brief historical perspective. J. Polym. Sci. Part A: Polym. Chem. 2002, 40, 2719–2728. [Google Scholar] [CrossRef]
- Kim, Y.H. Hyperbranched polymers 10 years after. J. Polym. Sci. Part A: Polym. Chem. 1998, 36, 1685–1698. [Google Scholar] [CrossRef]
- Haag, R.; Sunder, A.; Stumbe, J.-F. An approach to glycerol dendrimers and pseudo-dendritic polyglycerols. J. Am. Chem. Soc. 2000, 122, 2954–2955. [Google Scholar]
- Sunder, A.; Hanselmann, R.; Frey, H.; Mulhaupt, R. Controlled synthesis of hyperbranched polyglycerols by ring-opening multibranching polymerization. Macromolecules 1999, 32, 4240–4246. [Google Scholar]
- Holter, D.; Burgath, A.; Frey, H. Degree of branching in hyperbranched polymers. Acta Polym. 1997, 48, 30–35. [Google Scholar]
- Holter, D.; Frey, H. Degree of branching in hyperbranched polymers. 2. Enhancement of DB: Scope and limitations. Acta Polym. 1997, 48, 298–309. [Google Scholar] [CrossRef]
- Flory, P.J. Molecular size distribution in three dimensional polymers. I. Gelation. J. Am. Chem. Soc. 1941, 63, 3083–3096. [Google Scholar] [CrossRef]
- Wyatt, V.T. The Lewis-Acid catalyzed synthesis of hyperbranched polymers based on glycerol and diacids in toluene. J. Am. Oil Chem. Soc. 2012, 89, 313–319. [Google Scholar]
- Wyatt, V.T.; Nuñez, A.; Strahan, G.D. The Lewis acid-catalyzed synthesis of hyperbranched oligo(glycerol-diacid)s in aprotic polar media. J. Am. Oil Chem. Soc. 2010, 87, 1539–1369. [Google Scholar]
- Frazza, E.J.; Schmitt, E.E. A new absorbable suture. J. Biomed. Mater. Res. Symp. 1971, 1, 43–58. [Google Scholar]
- Vert, M.; Li, S.M. Bioresorbability and biocompatibility of aliphatic polyesters. J. Mater. Sci. Mater. Med. 1992, 3, 432–446. [Google Scholar] [CrossRef]
- Shalaby, S.W.; Johnson, R.A. Biomedical Polymers; Shalaby, S.W., Ed.; Hanser Publishing: New York, NY, USA, 1994; pp. 2–34, Chapter 1. [Google Scholar]
- Seiler, M.; Kohler, D.; Arlt, W. Hyperbranched polymers: New selective solvents for extractive distillation and solvent extraction. Sep. Purif. Technol. 2003, 30, 179–197. [Google Scholar]
- Rabiller, C.; Maze, F. Quantitative analysis and determination of the enantiomeric purity of glycerides by 13C NMR Spectroscopy. Application to the lipase-catalysed transesterification of triacylglycerides. Magn. Reson. Chem. 1989, 27, 582–584. [Google Scholar]
- Halldorsson, A.; Magnusson, C.D.; Haraldsson, G.G. Chemoenzymatic synthesis of structured triacylglycerols by highly regioselective acylation. Tetrahedron 2003, 59, 9101–9109. [Google Scholar]
- Magnusson, H.; Malmstrum, E.; Hult, A. Influence of reaction conditions on degree of branching in byperbranced aliphatic polyethers from 3-ethyl-3 (hydroxymethyl)oxetane. Macromolecules 2001, 34, 5786–5791. [Google Scholar]
- Hawker, C.J.; Lee, R.; Frechet, J.M.J. One-step synthesis of hyperbranched dendritic polyesters. J. Am. Chem. Soc. 1991, 113, 4583–4588. [Google Scholar]
- Kim, Y.H. Highly branched aromatic polymers prepared by single step syntheses. Macromol. Symp. 1994, 77, 21–33. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Wyatt, V.T.; Strahan, G.D. Degree of Branching in Hyperbranched Poly(glycerol-co-diacid)s Synthesized in Toluene. Polymers 2012, 4, 396-407. https://doi.org/10.3390/polym4010396
Wyatt VT, Strahan GD. Degree of Branching in Hyperbranched Poly(glycerol-co-diacid)s Synthesized in Toluene. Polymers. 2012; 4(1):396-407. https://doi.org/10.3390/polym4010396
Chicago/Turabian StyleWyatt, Victor T., and Gary D. Strahan. 2012. "Degree of Branching in Hyperbranched Poly(glycerol-co-diacid)s Synthesized in Toluene" Polymers 4, no. 1: 396-407. https://doi.org/10.3390/polym4010396