Optical and Physical Applications of Photocontrollable Materials: Azobenzene-Containing and Liquid Crystalline Polymers
Abstract
:1. Introduction
2. Photomechanical Effects
2.1. Photo-Actuator
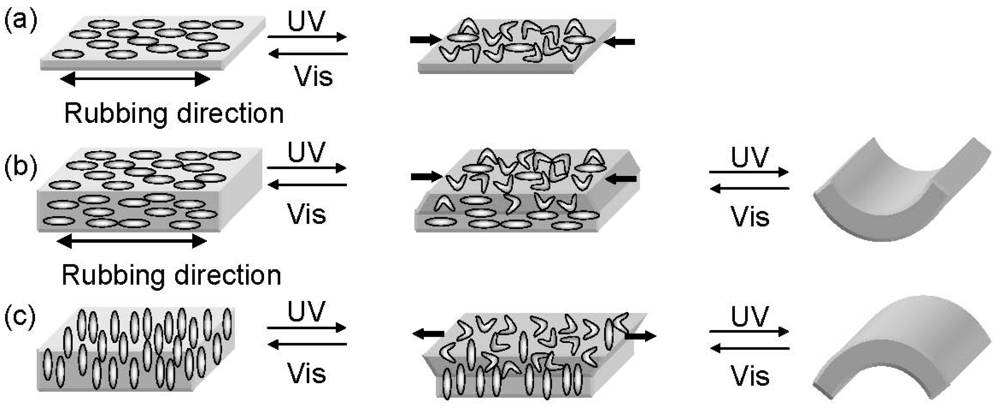
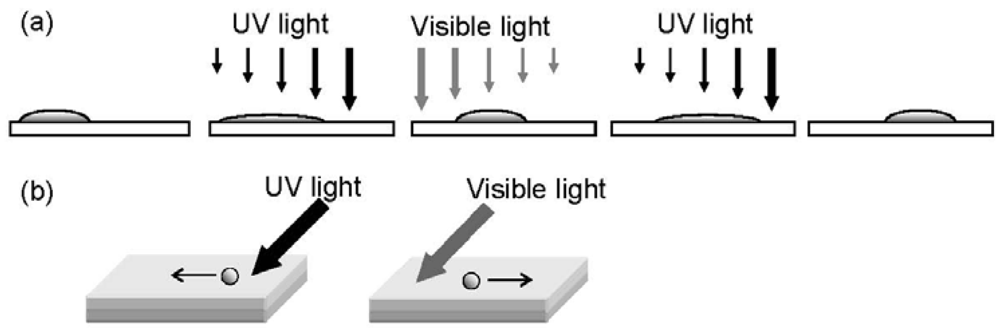
2.2. Photomanipulation
3. Photonic Crystals
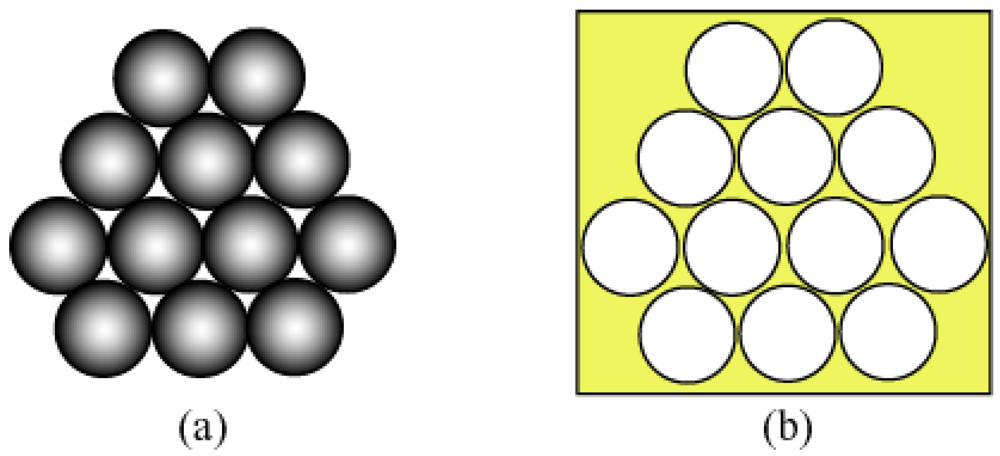
3.1. Colloidal Crystals

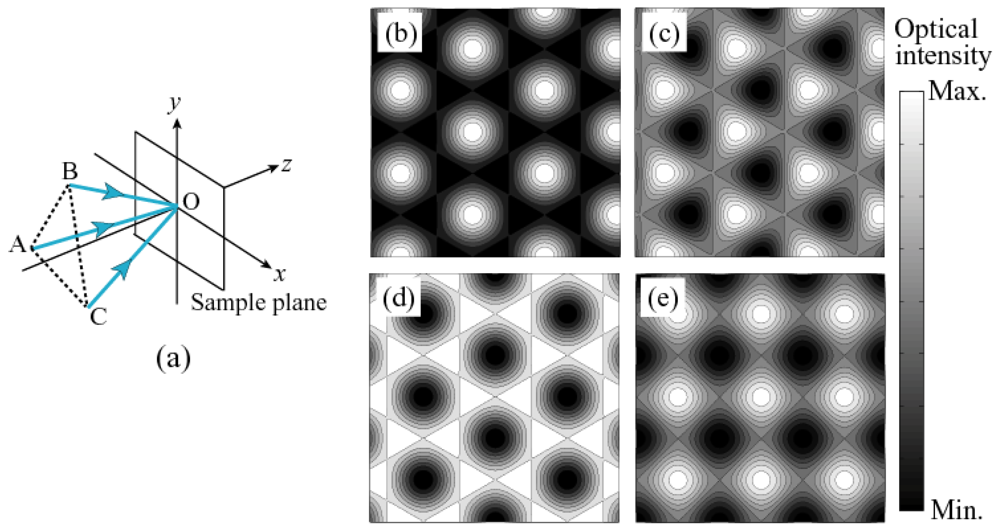
3.2. Holographic Fabrications

4. Diffractive Optical Elements
4.1. Anisotropic Gratings
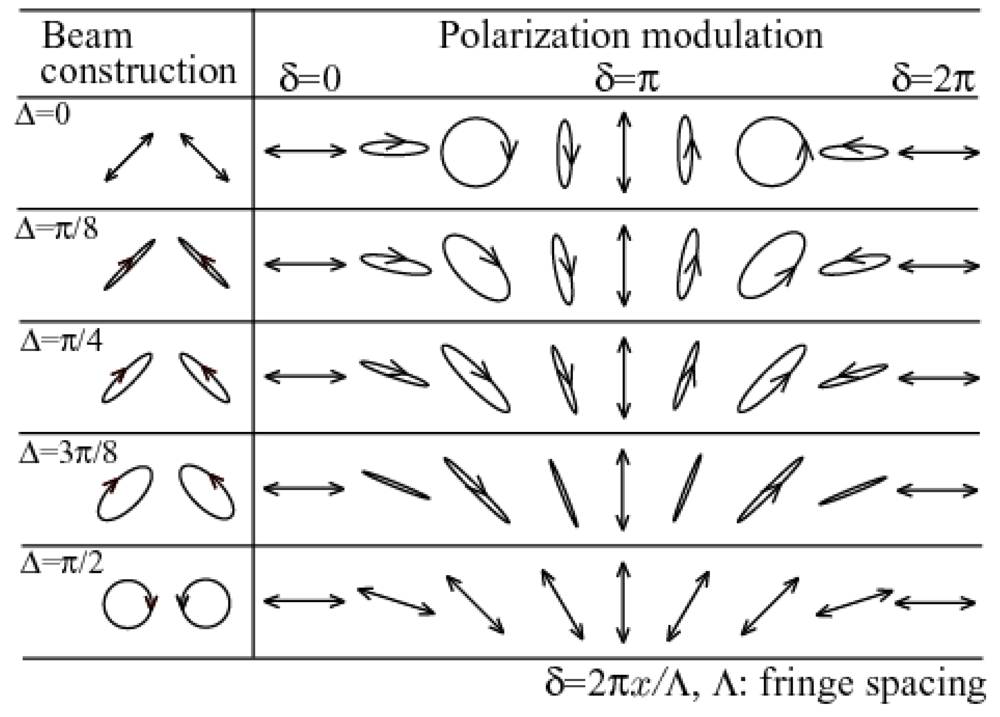


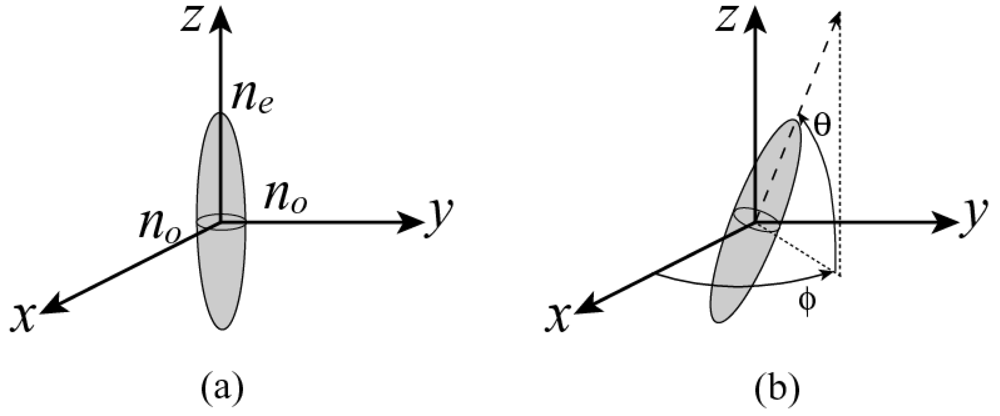
4.2. Applications
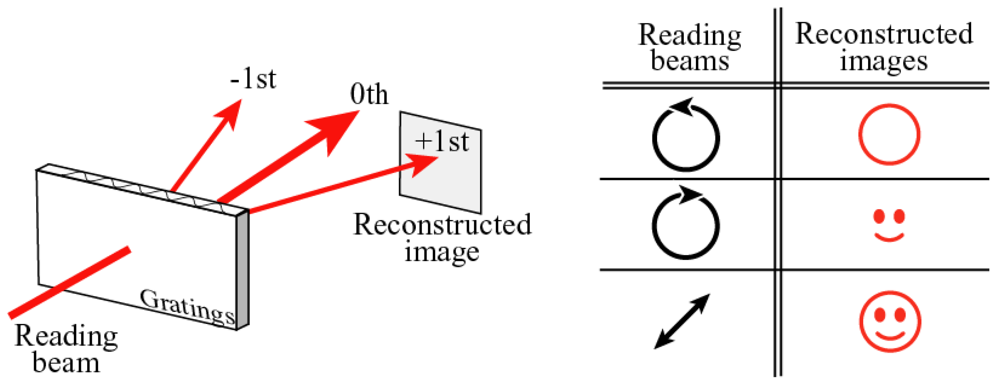


5. Relief Formation
5.1. Micro- and Nanoscale Formations

5.2. Applications
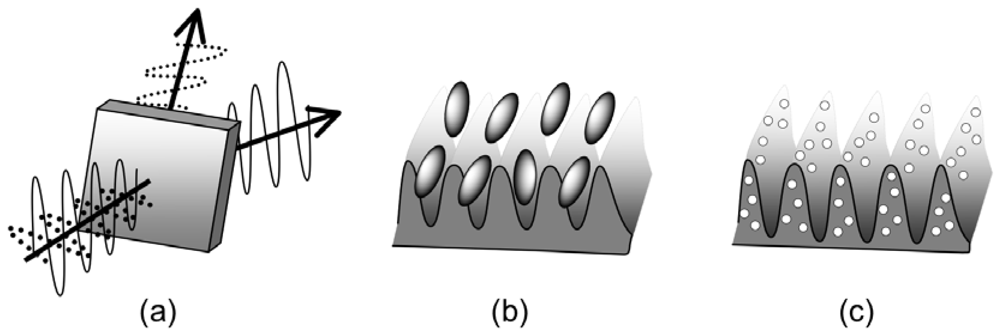
6. Self-Organized Structures
6.1. Mesoporous Structure

6.2. Nano Template

7. Photoinduced Chirality
7.1. Chirality Switching and Amplification

7.2. Supramolecular Chirality

8. Other Applications
9. Conclusions
Acknowledgments
References
- Ichimura, K. Photoalignment of liquid crystal systems. Chem. Rev. 2000, 100, 1847–1873. [Google Scholar] [CrossRef]
- Delaire, J.A.; Nakatani, K. Linear and nonlinear optical properties of photochromic molecules and materials. Chem. Rev. 2000, 100, 1817–1845. [Google Scholar] [CrossRef]
- Natansohn, A.; Rochon, P. Photoinduced motions in azo-containing polymers. Chem. Rev. 2002, 102, 4139–4175. [Google Scholar] [CrossRef]
- Ikeda, T.; Mamiya, J.; Yu, Y. Photomechanics of liquid-crystalline elastomers and other polymers. Angew. Chem. Int. Ed. 2007, 46, 506–528. [Google Scholar]
- Suzuki, A.; Tanaka, T. Phase transition in polymer gels induced by visible light. Nature 1990, 346, 345–347. [Google Scholar] [CrossRef]
- Tatsuma, T.; Takada, K.; Miyazaki, T. UV-light-induced swelling and visible-light-induced shrinking of a TiO2-containing redox gel. Adv. Mater. 2007, 19, 1249–1251. [Google Scholar] [CrossRef]
- Koshima, H.; Ojima, N.; Uchimoto, H. Mechanical motion of azobenzene crystals upon photoirradiation. J. Am. Chem. Soc. 2009, 131, 6890–6891. [Google Scholar] [CrossRef]
- Nakano, H. Direction control of photomechanical bending of a photochromic molecular fiber. J. Mater. Chem. 2010, 20, 2071–2074. [Google Scholar] [CrossRef]
- Bian, S.; Robinson, D.; Kuzyk, M.G. Optically activated cantilever using photomechanical effects in dye-doped polymer fibers. J. Opt. Soc. Am. B 2006, 23, 697–708. [Google Scholar] [CrossRef]
- Kobatake, S.; Takami, S.; Muto, H.; Ishikawa, T.; Irie, M. Rapid and reversible shape changes of molecular crystals on photoirradiation. Nature 2007, 446, 778–781. [Google Scholar] [CrossRef]
- Athanassiou, A.; Kalyva, M.; Lakiotaki, K.; Georgiou, S.; Fotakis, C. All-Optical reversible actuation of photochromic-polymer microsystems. Adv. Mater. 2005, 17, 988–992. [Google Scholar] [CrossRef]
- Lingyan, Z.; Al-Kaysi, R.O.; Bardeen, C.J. Reversible photoinduced twisting of molecular crystal microribbons. J. Am. Chem. Soc. 2011, 133, 12569–12575. [Google Scholar] [CrossRef]
- Al-Kaysi, R.O.; Bardeen, C.J. Reversible photoinduced shape changes of crystalline organic nanorods. Adv. Mater. 2007, 19, 1276–1280. [Google Scholar] [CrossRef]
- Eisenbach, C.D. Isomerization of aromatic azo chromophores in poly(ethyl acrylate) networks and photomechanical effect. Polymer 1980, 21, 1175–1179. [Google Scholar] [CrossRef]
- Wermter, H.; Finkelmann, H. Liquid crystalline elastomers as artificial muscles. e-Polymer 2001, 13, 1–13. [Google Scholar]
- Yu, H.; Ikeda, T. Photocontrollable liquid-crystalline actuators. Adv. Mater. 2011, 23, 2149–2180. [Google Scholar] [CrossRef]
- Ikeda, T.; Nakano, M.; Yu, Y.; Tsutsumi, O.; Kanazawa, A. Anisotropic bending and unbending behavior of azobenzene liquid-crystalline gels by light exposure. Adv. Mater. 2003, 15, 201–205. [Google Scholar] [CrossRef]
- Kondo, M.; Yu, Y.; Ikeda, T. How does the initial alignment of mesogens affect the photoinduced bending behavior of liquid-crystalline elastomers? Angew. Chem. Int. Ed. 2006, 45, 1378–1382. [Google Scholar]
- Van Oosten, C.L.; Harris, K.D.; Bastiaansen, C.W.M.; Broer, D.J. Glassy photomechanical liquid-crystal network actuators for microscale devices. Eur. Phys. J. E. 2007, 23, 329–336. [Google Scholar] [CrossRef]
- Yu, Y.; Nakano, M.; Ikeda, T. Miniaturizing a simple photomechanical system could expand its range of applications. Nature 2003, 425, 145. [Google Scholar] [CrossRef]
- Yoshino, T.; Mamiya, J.; Kinoshita, M.; Yu, Y.; Ikeda, T. Preparation and characterization of crosslinked azobenzene liquid-crystalline polymer fibers. Mol. Cryst. Liq. Cryst. 2007, 478, 989–999. [Google Scholar]
- Camacho-Lopez, M.; Finkelmann, H.; Palffy-Muhoray, P.; Shelley, M. Fast liquid-crystal elastomer swims into the dark. Nat. Mater. 2004, 3, 307–310. [Google Scholar] [CrossRef]
- Yamada, M.; Kondo, M.; Miyasato, R.; Naka, Y.; Mamiya, J.; Kinoshita, M.; Shishido, A.; Yu, Y.; Barrett, C.J.; Ikeda, T. Photomobile polymer materials—Various three-dimensional movements. J. Mater. Chem. 2009, 19, 60–62. [Google Scholar]
- Yamada, M.; Kondo, M.; Mamiya, J.; Yu, Y.; Kinoshita, M.; Barrett, C.J.; Ikeda, T. Photomobile polymer materials: Towards light-driven plastic motors. Angew. Chem. Int. Ed. 2008, 47, 4986–4988. [Google Scholar]
- Ichimura, K.; Oh, S.K.; Nakagawa, M. Light-driven motion of liquids on a photoresponsive surface. Science 2000, 288, 1624–1626. [Google Scholar] [CrossRef]
- Oh, S.-K.; Nakagawa, M.; Ichimura, K. Relationship between the ability to control liquid crystal alignment and wetting properties of calix[4]resorcinarene monolayers. J. Mater. Chem. 2001, 11, 1563–1569. [Google Scholar]
- Diguet, A.; Guillermic, R.-M.; Magome, N.; Saint-Jalmes, A.; Chen, Y.; Yoshikawa, K.; Baigl, D. Photomanipulation of a droplet by the chromocapillary effect. Angew. Chem. Int. Ed. 2009, 48, 9281–9284. [Google Scholar]
- Loudet, J.-C.; Barois, P.; Poulin, P. Colloidal ordering from phase separation in a liquidcrystalline continuous phase. Nature 2000, 407, 611–613. [Google Scholar] [CrossRef]
- Nazarenko, V.G.; Nych, A.B.; Lev, B.I. Crystal structure in nematic emulsion. Phys. Rev. Lett. 2001, 87, 075504. [Google Scholar] [CrossRef]
- Petrov, P.G.; Terentjev, E.M. Formation of cellular solid in liquid crystal colloids. Langmuir 2001, 17, 2942–2949. [Google Scholar] [CrossRef]
- Yamamoto, T.; Yamamoto, J.; Lev, B.I.; Yokoyama, H. Light-induced assembly of tailored droplet arrays in nematic emulsions. Appl. Phys. Lett. 2002, 81, 2187–2189. [Google Scholar] [CrossRef]
- Kausar, A.; Nagano, H.; Ogata, T.; Nonaka, T.; Kurihara, S. Photocontrolled translational motion of a microscale solid object on azobenzene-doped liquid-crystalline films. Angew. Chem. Int. Ed. 2009, 48, 2144–2147. [Google Scholar]
- Kausar, A.; Nagano, H.; Kuwahara, Y.; Ogata, T.; Kurihara, S. Photocontrolled manipulation of a microscale object: A rotational or translational mechanism. Chem. Eur. J. 2011, 17, 508–515. [Google Scholar] [CrossRef]
- Yablonovitch, E. Inhibited spontaneous emission in solid-state physics and electronics. Phys. Rev. Lett. 1987, 58, 2059–2062. [Google Scholar] [CrossRef]
- Reynolds, A.; López-Tejeira, F.; Cassagne, D.; García-Vidal, F.J.; Jouanin, C.; Sánchez-Dehesa, J. Spectral properties of opal-based photonic crystals having a SiO2 matrix. Phys. Rev. B 1999, 60, 11422–11426. [Google Scholar]
- Li, H.-L.; Marlow, F. Controlled arrangement of colloidal crystal strips. Chem. Mater. 2005, 17, 3809–3811. [Google Scholar] [CrossRef]
- Busch, K.; John, S. Photonic band gap formation in certain self-organizing systems. Phys. Rev. E 1998, 58, 3896–3908. [Google Scholar] [CrossRef]
- Moritsugu, M.; Kim, S.N.; Ogata, T.; Nonaka, T.; Kurihara, S.; Kubo, S.; Segawa, H.; Sato, O. Photochemical switching behavior of azofunctionalized polymer liquid crystal/SiO2 composite photonic crystal. Appl. Phys. Lett. 2006, 89, 153131:1–153131:3. [Google Scholar]
- Moritsugu, M.; Kim, S.-N.; Kubo, S.; Ogata, T.; Nonaka, T.; Sato, O.; Kurihara, S. Photoswitching properties of photonic crystals infiltrated with polymer liquid crystals having azobenzene side chain groups with different methylene spacers. React. Funct. Polym. 2011, 71, 30–35. [Google Scholar] [CrossRef]
- Kurihara, S.; Moritsugu, M.; Kubo, S.; Kim, S.-N.; Ogata, T.; Nonaka, T.; Sato, O. Photoswitching properties of photonic band gap material containing azo-polymer liquid crystal. Eur. Polym. J. 2007, 43, 4951–4960. [Google Scholar] [CrossRef]
- Moritsugu, M.; Shirota, T.; Kubo, S.; Ogata, T.; Sato, O.; Kurihara, S. Enhanced photochemical-shift of reflection band from an inverse opal film based on larger birefringent polymer liquid crystals: Effect of tolane group on the photochemical shift behavior. J. Polym. Sci. Part B: Polym. Phys. 2009, 47, 1981–1990. [Google Scholar] [CrossRef]
- Hong, J.-C.; Park, J.-H.; Chun, C.; Kim, D.-Y. Photoinduced tuning of optical stop bands in azopolymer based inverse opal photonic crystals. Adv. Funct. Mater. 2007, 17, 2462–2469. [Google Scholar] [CrossRef]
- Li, Y.; Deng, Y.; Tong, X.; Wang, X. Formation of photoresponsive uniform colloidal spheres from an amphiphilic azobenzene- ontaining random copolymer. Macromolecules 2006, 39, 1108–1115. [Google Scholar] [CrossRef]
- Li, Y.; Deng, Y.; He, Y.; Tong, X.; Wang, X. Amphiphilic azo polymer spheres, colloidal monolayers, and photoinduced chromophore orientation. Langmuir 2005, 21, 6567–6571. [Google Scholar] [CrossRef]
- Liu, J.; He, Y.; Wang, X. Azo polymer colloidal spheres containing different amounts of functional groups and their photoinduced deformation behavior. Langmuir 2008, 24, 678–682. [Google Scholar] [CrossRef]
- Deng, Y.; Li, N.; He, Y.; Wang, X. Hybrid colloids composed of two amphiphilic azo polymers: Fabrication, characterization, and photoresponsive properties. Macromolecules 2007, 40, 6669–6678. [Google Scholar] [CrossRef]
- Li, Y.; He, Y.; Tong, X.; Wang, X. Stretching effect of linearly polarized Ar+ laser single-beam on azo polymer colloidal spheres. Langmuir 2006, 22, 2288–2291. [Google Scholar] [CrossRef]
- Kamenjicki, M.; Lednev, I.K.; Asher, S.A. Photoresponsive azobenzene photonic crystals. J. Phys. Chem. B 2004, 108, 12637–12639. [Google Scholar] [CrossRef]
- Kamenjicki, M.; Lednev, I.K.; Mikhonin, A.; Kesavamoorthy, R.; Asher, S.A. Photochemically controlled photonic crystals. Adv. Funct. Mater. 2003, 13, 774–780. [Google Scholar] [CrossRef]
- Gu, Z.-Z.; Hayami, S.; Meng, Q.-B.; Iyoda, T.; Fujishima, A.; Sato, O. Control of photonic band structure by molecular aggregates. J. Am. Chem. Soc. 2000, 122, 10730–10731. [Google Scholar]
- Kubo, S.; Gu, Z.-Z.; Takahashi, K.; Ohko, Y.; Sato, O.; Fujishima, A. Control of the optical band structure of liquid crystal infiltrated inverse opal by a photoinduced nematic-isotropic phase transition. J. Am. Chem. Soc. 2002, 124, 10950–10951. [Google Scholar]
- Gu, Z.-Z.; Iyoda, T.; Fujishima, A.; Sato, O. Photo-reversible regulation of optical stop bands. Adv. Mater. 2001, 13, 1295–1298. [Google Scholar] [CrossRef]
- Kubo, S.; Gu, Z.-Z.; Takahashi, K.; Fujishima, A.; Segawa, H.; Sato, O. Tunable photonic band gap crystals based on a liquid crystal-infiltrated inverse opal structure. J. Am. Chem. Soc. 2004, 126, 8314–8319. [Google Scholar]
- Kubo, S.; Gu, Z.-Z.; Takahashi, K.; Fujishima, A.; Segawa, H.; Sato, O. Control of the optical properties of liquid crystal-infiltrated inverse opal structures using photo irradiation and/or an electric field. Chem. Mater. 2005, 17, 2298–2309. [Google Scholar]
- Mao, W.; Zhong, Y.; Dong, J.; Wang, H. Crystallography of two-dimensional photonic lattices formed by holography of three noncoplanar beams. J. Opt. Soc. Am. B 2005, 22, 1085–1091. [Google Scholar] [CrossRef]
- Zhu, X.; Xu, Y.; Yang, S. Distortion of 3D su8 photonic structures fabricated by four-beam holographic lithography with umbrella configuration. Opt. Express 2007, 15, 16546–16560. [Google Scholar] [CrossRef]
- Liu, Y.J.; Dai, H.T.; Sun, X.W. Holographic fabrication of azo-dye-functionalized photonic structures. J. Mater. Chem. 2011, 21, 2982–2986. [Google Scholar]
- Ono, H.; Emoto, A.; Kawatsuki, N. Anisotropic photonic gratings formed in photocross-linkable polymer liquid crystals. J. Appl. Phys. 2006, 100, 013522:1–013522:7. [Google Scholar]
- Emoto, A.; Ono, H.; Kawatsuki, N.; Uchida, E. Two-dimensional anisotropic gratings formed in photocrosslinkable polymer liquid crystals by multiple interference. Jpn. J. Appl. Phys. 2006, 45, 1705–1709. [Google Scholar] [CrossRef]
- Kawatsuki, N.; Goto, K.; Kawakami, T.; Yamamoto, T. Reversion of alignment direction in the thermally enhanced photoorientation of photo-cross-linkable polymer liquid crystal films. Macromolecules 2002, 35, 706–713. [Google Scholar] [CrossRef]
- Kawatsuki, N.; Fujio, K.; Hasegawa, T.; Emoto, A.; Ono, H. Surface relief formation with molecular orientation in photoreactive liquid crystalline polymer film. J. Photopolym. Sci. Technol. 2006, 19, 151–156. [Google Scholar] [CrossRef]
- Yu, H.; Shishido, A.; Ikeda, T. Subwavelength modulation of surface relief and refractive index in preirradiated liquid-crystalline polymer films. Appl. Phys. Lett. 2008, 92, 103117:1–103117:3. [Google Scholar]
- Blaikie, R.J.; McNab, S.J. Evanescent interferometric lithography. Appl. Opt. 2001, 40, 1692–1698. [Google Scholar] [CrossRef]
- Martinez-Anton, J.C. Surface relief subwavelength gratings by means of total internal reflection evanescent wave interference lithography. J. Opt. A: Pure Appl. Opt. 2006, 8, S213–S218. [Google Scholar] [CrossRef]
- Ramanujam, P.S. Evanescent polarization holographic recording of sub-200-nm gratings in an azobenzene polyester. Opt. Lett. 2003, 28, 2375–2377. [Google Scholar] [CrossRef]
- Ohdaira, Y.; Hoshiyama, S.; Kawakami, T.; Shinbo, K.; Kato, K.; Kaneko, F. Fabrication of surface relief gratings on azo dye thin films utilizing an interference of evanescent waves. Appl. Phys. Lett. 2005, 86, 051102:1–051102:3. [Google Scholar]
- Goldenberg, L.M.; Gritsai, Y.; Kulikovska, O.; Stumpe, J. Three-dimensional planarized diffraction structures based on surface relief gratings in azobenzene materials. Opt. Lett. 2008, 33, 1309–1311. [Google Scholar] [CrossRef]
- Nikolova, L.; Ramanujam, P.S. Polarization Holography; Cambridge University Press: Cambridge, UK, 2009. [Google Scholar]
- Forcén, P.; Oriol, L.; Sánchez, C.; Alcalá, R.; Jankova, K.; Hvilsted, S. Pulsed recording of anisotropy and holographic polarization gratings in azo-polymethacrylates with different molecular architectures. J. Appl. Phys. 2008, 103, 123111. [Google Scholar]
- Baldus, O.; Leopold, A.; Hagen, R.; Bieringer, T.; Zilker, S.J. Surface relief gratings generated by pulsed holography: A simple way to polymer nanostructures without isomerizing side-chains. J. Chem. Phys. 2001, 114, 1344–1349. [Google Scholar]
- Rodríguez, F.J.; Sánchez, C.; Villacampa, B.; Alcalá, R.; Cases, R. Surface relief gratings induced by a nanosecond pulse in a liquid-crystalline azo-polymethacrylate. Appl. Phys. Lett. 2005, 87, 201914:1–201914:3. [Google Scholar]
- Sheng, C.X.; Norwood, R.A.; Wang, J.; Thomas, J.; Wu, Y.; Zheng, Z.; Tabirian, N.; Steeves, D.M.; Kimball, B.R.; Peyghambarian, N. Time-resolved studies of photoinduced birefringence in azobenzene dye-doped polymer films. Appl. Opt. 2008, 47, 5074–5077. [Google Scholar]
- Yager, K.G.; Barrett, C.J. Temperature modeling of laser-irradiated azo-polymer thin films. J. Chem. Phys. 2004, 120, 1089–1096. [Google Scholar] [CrossRef]
- Ono, H.; Nakamura, M.; Emoto, A.; Kawatsuki, N. Diffraction properties in polarization holography written by elliptical polarized light. Jpn. J. Appl. Phys. 2010, 49, 032502:1–032502:4. [Google Scholar]
- Fukuda, T. Rewritable high-density optical recording on azobenzene polymer thin film. Opt. Rev. 2005, 12, 126–129. [Google Scholar]
- Ono, H.; Emoto, A.; Kawatsuki, N.; Hasegawa, T. Multiplex diffraction from functionalized polymer liquid crystals and polarization conversion. Opt. Express 2003, 11, 2379–2384. [Google Scholar] [CrossRef]
- Ono, H.; Hatayama, A.; Emoto, A.; Kawatsuki, N.; Uchida, E. Two-dimensional crossed polarization gratings in photocrosslinkable polymer liquid crystals. Jpn. J. Appl. Phys. 2005, 44, L306–L309. [Google Scholar]
- Galstyan, A.V.; Zakharyan, G.G.; Hakobyan, R.S. The theory of light diffraction in thin anisotropic medium. Mol. Cryst. Liq.Cryst. 2006, 453, 203–213. [Google Scholar] [CrossRef]
- Kuroda, K.; Matsuhashi, Y.; Fujimura, R.; Shimura, T. Theory of polarization holography. Opt. Rev. 2011, 18, 374–382. [Google Scholar] [CrossRef]
- Sasaki, T.; Miura, K.; Hanaizumi, O.; Emoto, A.; Ono, H. Coupled-wave analysis of vector holograms: Effects of modulation depth of anisotropic phase retardation. Appl. Opt. 2010, 49, 5205–5211. [Google Scholar] [CrossRef]
- Oh, C.; Escuti, M.J. Time-domain analysis of periodic anisotropic media at oblique incidence: An efficient FDTD implementation. Opt. Express 2006, 14, 11870–11884. [Google Scholar] [CrossRef]
- Oh, C.; Escuti, M.J. Numerical analysis of polarization gratings using the finite-difference time-domain method. Phys. Rev. A 2007, 76, 043815:1–043815:8. [Google Scholar]
- Ono, H.; Sekiguchi, T.; Emoto, A.; Kawatsuki, N. Light wave propagation in polarization holograms formed in photoreactive polymer liquid crystals. Jpn. J. Appl. Phys. 2008, 47, 3559–3563. [Google Scholar] [CrossRef]
- Emoto, A.; Wada, T.; Shioda, T.; Sasaki, T.; Manabe, S.; Kawatsuki, N.; Ono, H. Vector gartings fabricated by polarizer rotation exposure to hydrogen-bonded liquid crystalline polymers. Jpn. J. Appl. Phys. 2011, 50, 032502:1–032502:5. [Google Scholar]
- Todorov, T.; Nikolova, L.; Stoyanova, K.; Tomova, N. Polarization holography. 3: Some applications of polarization holographic recording. Appl. Opt. 1985, 24, 785–788. [Google Scholar] [CrossRef]
- Ilieva, D.; Nedelchev, L.; Petrova, T.; Tomova, N.; Dragostinova, V.; Nikolova, L. Holographic multiplexing using photoinduced anisotropy and surface relief in azopolymer films. J. Opt. A: Pure Appl. Opt. 2005, 7, 35–39. [Google Scholar] [CrossRef]
- Pan, X.; Wang, C.; Wang, C.; Zhang, X. Image storage based on circular-polarization holography in an azobenzene side-chain liquid-crystalline polymer. Appl. Opt. 2008, 47, 93–98. [Google Scholar] [CrossRef]
- Ono, H.; Suzuki, K.; Sasaki, T.; Iwato, T.; Emoto, A.; Shioda, T.; Kawatsuki, N. Reconstruction of polarized optical images in two- and three-dimensional vector holograms. J. Appl. Phys. 2009, 106, 083109:1–083109:7. [Google Scholar]
- Provenzano, C.; Cipparrone, G.; Mazzulla, A. Photopolarimeter based on two gratings recorded in thin organic films. Appl. Opt. 2006, 45, 3929–3934. [Google Scholar] [CrossRef]
- Todorov, T.; Nikolova, L.; Stoilov, G.; Hristov, B. Spectral stokesmeter. 1. Implementation of the device. Appl. Opt. 2007, 46, 6662–6668. [Google Scholar]
- Sasaki, T.; Hatayama, A.; Emoto, A.; Ono, H.; Kawatsuki, N. Simple detection of light polarization by using crossed polarization gratings. J. Appl. Phys. 2006, 100, 063502:1–063502:4. [Google Scholar]
- Provenzano, C.; Pagliusi, P.; Mazzulla, A.; Cipparrone, G. Method for artifact-free circular dichroism measurements based on polarization grating. Opt. Lett. 2010, 35, 1822–1824. [Google Scholar] [CrossRef]
- Zhou, J.; Shen, J.; Yang, J.; Ke, Y.; Wang, K.; Zhang, Q. Fabrication of a pure polarization grating in a cross-linked azopolymer by polarization-modulated holography. Opt. Lett. 2006, 31, 1370–1372. [Google Scholar]
- Ono, H.; Emoto, A.; Takahashi, F.; Kawatsuki, N.; Hasegawa, T. Highly stable polarization gratings in photocrosslinkable polymer liquid crystals. J. Appl. Phys. 2003, 94, 1298–1303. [Google Scholar] [CrossRef]
- Ono, H.; Nakamura, M.; Kawatsuki, N. Conversion of circularly polarized light into linearly polarized light in anisotropic phase gratings using photo-cross-linkable polymer liquid crystals. Appl. Phys. Lett. 2007, 90, 231107:1–231107:3. [Google Scholar]
- Emoto, A.; Fukuda, T.; Barada, D. Asymmetric polarization conversion in polarization holograms with surface relief. Jpn. J. Appl. Phys. 2008, 47, 3568–3571. [Google Scholar]
- Yang, X.; Zhang, C.; Qi, S.; Chen, K.; Tian, J.; Zhang, G. All-optical Boolean logic gate using azo-dye doped polymer film. Optik 2005, 116, 251–254. [Google Scholar] [CrossRef]
- Matharu, A.S.; Jeeva, S.; Ramanujam, P.S. Liquid crystals for holographic optical data strage. Chem. Soc. Rev. 2007, 36, 1868–1880. [Google Scholar] [CrossRef]
- Hvilsted, S.; Sánchez, C.; Alcalá, R. The volume holographic optical storage potential in azobenzene containing polymers. J. Mater. Chem. 2009, 19, 6641–6648. [Google Scholar] [CrossRef]
- Saishoji, A.; Sato, D.; Shishido, A.; Ikeda, T. Formation of bragg gratings with large angular multiplicity by means of the photoinduced reorientation of azobenzene copolymers. Langmuir 2007, 23, 320–326. [Google Scholar] [CrossRef]
- Ishiguro, M.; Sato, D.; Shishido, A.; Ikeda, T. Bragg-type polarization gratings formed in thick polymer films containing azobenzene and tolane moieties. Langmuir 2007, 23, 332–338. [Google Scholar]
- Minabe, J.; Maruyama, T.; Yasuda, S.; Kawano, K.; Hayashi, K.; Ogasawara, Y. Design of dye concentrations in azobenzene-containing polymer films for volume holographic storage. Jpn. J. Appl. Phys. 2004, 43, 4964–4967. [Google Scholar]
- Forcén, P.; Oriol, L.; Sánchez, C.; Rodríguez, F.J.; Alcalá, R.; Hvilsted, S.; Jankova, K. Methacrylic azopolymers for holographic storage: a comparison among different polymer types. Eur. Polym. J. 2007, 43, 3292–3300. [Google Scholar] [CrossRef]
- Häckel, M.; Kador, L.; Kropp, D.; Schmidt, H.-S. Polymer blends with azobenzene-containing block copolymers as stable rewritable volume holographic media. Adv. Mater. 2007, 19, 227–231. [Google Scholar] [CrossRef]
- Rochon, P.; Batalla, E.; Natansohn, A. Optically induced surface gratings on azoaromatic polymer films. Appl. Phys. Lett. 1995, 66, 136–138. [Google Scholar] [CrossRef]
- Kim, D.Y.; Tripathy, S.K.; Li, L.; Kumar, J. Laser-induced holographic surface relief gratings on nonlinear optical polymer films. Appl. Phys. Lett. 1995, 66, 1166–1168. [Google Scholar] [CrossRef]
- Viswanathan, N.K.; Balasubramanian, S.; Li, L.; Tripathy, S.K.; Films, A.P. A detailed investigation of the polarization-dependent surface-relief-grating formation process on azo polymer films. Jpn. J. Appl. Phys. 1999, 38, 5928–5937. [Google Scholar] [CrossRef]
- Barrett, C.J.; Natansohn, A.L.; Rochon, P.L. Mechanism of optically inscribed high-efficiency diffraction gratings in azo polymer films. J. Phys. Chem. 1996, 100, 8836–8842. [Google Scholar]
- Pedersen, T.G.; Johansen, P.M. Mean-field theory of photoinduced molecular reorientation in azobenzene liquid crystalline side-chain polymers. Phys. Rev. Lett. 1997, 79, 2470–2473. [Google Scholar] [CrossRef]
- Bian, S.; Li, L.; Kumar, J.; Kim, D.Y.; Williams, J.; Tripathy, S.K. Single laser beam-induced surface deformation on azobenzene polymer films. Appl. Phys. Lett. 1998, 73, 1817–1819. [Google Scholar] [CrossRef]
- Lefin, P.; Fiorini, C.; Nunzi, J.M. Anisotropy of the photo-induced translation diffusion of azobenzene dyes in polymer matrices. Pure Appl. Opt. 1998, 7, 71–82. [Google Scholar] [CrossRef]
- Barada, D.; Fukuda, T.; Itoh, M.; Yatagai, T. Numerical analysis of photoinduced surface relief formed by particle method. Opt. Rev. 2005, 12, 271–273. [Google Scholar] [CrossRef]
- Bellini, B.; Ackermann, J.; Klein, H.; Grave, Ch.; Dumas, Ph.; Safarov, V. Light-induced molecular motion of azobenzenecontaining molecules: A random-walk model. J. Phys.: Condens. Matter 2006, 18, S1817–S1835. [Google Scholar]
- Juan, M.L.; Plain, J.; Bachelot, R.; Royer, P.; Gray, S.K.; Wiederrecht, G.P. Multiscale model for photoinduced molecular motion in azo polymers. ACSNano 2009, 3, 1573–1579. [Google Scholar]
- Viswanathan, N.K.; Kim, D.Y.; Bian, S.; Williams, J.; Liu, W.; Li, L.; Samuelson, L.; Kumar, J.; Tripathy, S.K. Surface relief structures on azo polymer films. J. Mater. Chem. 1999, 9, 1941–1955. [Google Scholar] [CrossRef]
- Fukuda, T.; Matsuda, H.; Shiraga, T.; Kimura, T.; Kato, M.; Viswanathan, N.K.; Kumar, J.; Tripathy, S.K. Photofabrication of surface relief grating on films of azobenzene polymer with different dye functionalization. Macromolecules 2000, 33, 4220–4225. [Google Scholar]
- Fukuda, T.; Sumaru, K.; Yamanaka, T.; Matsuda, H. Photo-induced formation of the surface relief grating on azobenzene polymers: Analysis based on the fluid mechanics. Mol. Cryst. Liq. Cryst. 2000, 345, 263–268. [Google Scholar] [CrossRef]
- Kumar, J.; Li, L.; Jiang, X.L.; Kim, D.Y.; Lee, T.S.; Tripathy, S. Gradient force: The mechanism for surface relief grating formation in azobenzene functionalized polymers. Appl. Phys. Lett. 1998, 72, 2096–2098. [Google Scholar]
- Bian, S.; Williams, J.M.; Kim, D.Y.; Li, L.; Balasubramanian, S.; Kumar, J.; Tripathy, S. Photoinduced surface deformations on azobenzene polymer films. J. Appl. Phys. 1999, 86, 4498–4508. [Google Scholar] [CrossRef]
- Holme, N.C.R.; Nikolova, L.; Hvilsted, S.; Rasmussen, P.H.; Berg, R.H.; Ramanujam, P.S. Optically induced surface relief phenomena in azobenzene polymers. Appl. Phys. Lett. 1999, 74, 519–521. [Google Scholar]
- Fiorini, C.; Prudhomme, N.; De Veyrac, G.; Maurin, I.; Raimond, P.; Nunzi, J.M. Molecular migration mechanism for laser induced surface relief grating formation. Synth. Met. 2000, 115, 121–125. [Google Scholar] [CrossRef]
- Ikawa, T.; Mitsuoka, T.; Hasegawa, M.; Tsuchimori, M.; Watanabe, O. Optical near field induced change in viscoelasticity on an azobenzene-containing polymer surface. J. Phys. Chem. B 2000, 104, 9055–9058. [Google Scholar]
- Landraud, N.; Peretti, J.; Chaput, F.; Lampel, G.; Boilot, J.P.; Lahlil, K.; Safarov, V.I. Near-field optical patterning on azo-hybrid sol-gel films. Appl. Phys. Lett. 2001, 79, 4562–4564. [Google Scholar]
- Fukuda, T.; Sumaru, K.; Kimura, T.; Matsuda, H.; Narita, Y. Observation of optical near-field as photo-induced surface relief formation. Jpn. J. Appl. Phys. 2001, 40, L900–L902. [Google Scholar]
- Fukuda, T.; Sumaru, K.; Matsuda, H.; Narita, Y.; Inoue, T.; Sato, F. Simple and effective technique for the evaluation of optical field emitted from a SNOM probe tip. Proc. SPIE 4642, 138–147. [Google Scholar]
- Ishitobi, H.; Tanabe, M.; Sekkat, Z.; Kawata, S. Nanomovement of azo polymers induced by metal tip enhanced nearfield irradiation. Appl. Phys. Lett. 2007, 91, 091911. [Google Scholar] [CrossRef]
- Karageorgiev, P.; Neher, D.; Schulz, B.; Stiller, B.; Pietsch, U.; Giersig, M.; Brehmer, L. From anisotropic photo-fluidity towards nanomanipulation in the optical near-field. Nat. Mater. 2005, 4, 699–703. [Google Scholar] [CrossRef]
- Ubukata, T.; Seki, T.; Ichimura, T. Surface relief gratings in host-guest supramolecular materials. Adv. Mater. 2000, 12, 1675–1678. [Google Scholar] [CrossRef]
- Ubukata, T.; Takahashi, K.; Yokoyama, Y. Photoinduced surface relief structures formed on polymer films doped with photochromic spiropyrans. J. Phys. Org. Chem. 2007, 20, 981–984. [Google Scholar] [CrossRef]
- Kikuchi, A.; Harada, A.; Yagi, M.; Ubukata, T.; Yokoyamaa, Y.; Abeb, J. Photoinduced diffusive mass transfer in o-Cl-HABI amorphous thin films. Chem. Commun. 2010, 46, 2262–2264. [Google Scholar]
- Natansohn, A.; Rochon, P. Photoinduced motions in azobenzene-based amorphous polymers: Possible photonic devices. Adv. Mater. 1999, 11, 1387–1391. [Google Scholar] [CrossRef]
- Ubukata, T.; Isoshima, T.; Hara, M. Wavelength-programmable organic distributed-feedback laser based on a photoassisted polymer-migration system. Adv. Mater. 2005, 17, 1630–1633. [Google Scholar] [CrossRef]
- Lia, X.T.; Natansohn, A.; Rochon, P. Photoinduced liquid crystal alignment based on a surface relief grating in an assembled cell. Appl. Phys. Lett. 1999, 74, 3791–3793. [Google Scholar] [CrossRef]
- Kim, M.H.; Kim, J.D.; Fukuda, T.; Matsuda, H. Alignment control of liquid crystals on surface relief gratings. Liquid. Cryst. 2000, 27, 1633–1640. [Google Scholar] [CrossRef]
- Parfenov, A.; Tamaoki, N.; Ohnishi, S. Photoinduced alignment of nematic liquid crystal on the polymer surface microrelief. J. Appl. Phys. 2000, 87, 2043–2045. [Google Scholar] [CrossRef]
- Chung, D.H.; Fukuda, T.; Takanishi, Y.; Ishikawa, K.; Matsuda, H.; Takezoe, H.; Osipov, M.A. Competitive effects of grooves and photoalignment on nematic liquid-crystal alignment using azobenzene polymer. J. Appl. Phys. 2002, 92, 1841–1844. [Google Scholar]
- Ye, Y.H.; Badilescu, S.; Truong, V.V.; Rochon, P.; Natansohn, A. Self-assembly of colloidal spheres on patterned substrates. Appl. Phys. Lett. 2001, 79, 872–874. [Google Scholar]
- Yi, D.K.; Seo, E.M.; Kim, D.Y. Surface-modulation-controlled three-dimensional colloidal crystals. Appl. Phys. Lett. 2002, 80, 225–227. [Google Scholar]
- Yi, D.K.; Kim, M.J.; Kim, D.Y. Surface relief grating induced colloidal crystal structures. Langmuir 2002, 18, 2019–2023. [Google Scholar] [CrossRef]
- Watanabe, O.; Ikawa, T.; Kato, T.; Tawata, M.; Shimoyama, H. Area-selective photoimmobilization of a two-dimensional array of colloidal spheres on a photodeformed template formed in photoresponsive azopolymer film. Appl. Phys. Lett. 2006, 88, 204107:1–204107:3. [Google Scholar]
- Fukuda, T.; Keum, C.D.; Matsuda, H.; Yase, K.; Tamada, K. Photo-induced surface relief on Azo polymer for optical component fabrication. Proc. SPIE 5183, 155–162. [Google Scholar]
- Ubukata, T.; Hara, M.; Ichimura, K.; Seki, T. Phototactic mass transport in polymer films for micropatterning and alignment of functional materials. Adv. Mater. 2004, 16, 220–223. [Google Scholar] [CrossRef]
- Zettsu, N.; Ubukata, T.; Seki, T. Two-dimensional manipulation of poly(3-dodecylthiophene) using light-driven instant mass migration as a molecular conveyer. Jpn. J. Appl. Phys. 2004, 43, L1169–L1171. [Google Scholar] [CrossRef]
- Beck, J.S.; Vartuli, J.C. Recent advances in the synthesis, characterization and applications of mesoporous molecular sieves. Curr. Opin. Solid State Mater. Sci. 1996, 1, 76–87. [Google Scholar] [CrossRef]
- Beck, J.S.; VartUli, J.C.; Roth, W.J.; Leonowicz, M.E.; Kresge, C.T.; Schmitt, K.D.; Chu, C.T.-W.; Olson, D.H.; Sheppard, E.W.; McCullen, S.B.; et al. A new family of mesoporous molecular sieves prepared with liquid crystal templates. J. Am. Chem. Soc. 1992, 114, 10834–10843. [Google Scholar]
- Wan, Y.; Zhao, D. On the controllable soft-templating approach to mesoporous silicates. Chem. Rev. 2007, 107, 2821–2860. [Google Scholar] [CrossRef]
- Huo, Q.; Zhao, D.; Feng, J.; Weston, K.; Buratto, S.K.; Stucky, G.D.; Schacht, S.; Schüth, F. Room temperature growth of mesoporous silica fibers: A new high-surface-area optical waveguide. Adv. Mater. 1997, 9, 974–978. [Google Scholar] [CrossRef]
- Marlow, F.; McGehee, M.D.; Zhao, D.; Chmelka, B.F.; Stucky, G.D. Doped mesoporous silica fibers: A new laser material. Adv. Mater. 1999, 11, 632–636. [Google Scholar] [CrossRef]
- Schomburg, C.; Wark, M.; Rohlfing, Y.; Schulz-Ekloff, G.; Wohrle, D. Photochromism of spiropyran in molecular sieve voids: effects of host-guest interaction on isomer status, switching stability and reversibility. J. Mater. Chem. 2001, 11, 2014–2021. [Google Scholar] [CrossRef]
- Doshi, D.A.; Huesing, N.K.; Lu, M.; Fan, H.; Lu, Y.; Simmons-Potter, K.; Potter, B.G., Jr; Hurd, A.J.; Brinker, C.J. Optically defined multifunctional patterning of photosensitive thin-film silica mesophases. Science 2000, 290, 107–111. [Google Scholar]
- Yang, C.M.; Cho, A.T.; Pan, F.M.; Tsai, T.G.; Chao, K.J. Spin-on mesoporous silica films with ultralow dielectric constants, ordered pore structures, and hydrophobic surfaces. Adv. Mater. 2001, 13, 1099–1102. [Google Scholar] [CrossRef]
- Yanagisawa, T.; Shimizu, T.; Kuroda, K.; Kato, C. Trimethylsily derivatives of alkyltrimethylammonium-kanemite complexes and their conversion to microporous SiO2 materials. Bull. Chem. Soc. Jpn. 1990, 63, 1535–1537. [Google Scholar] [CrossRef]
- Kresge, C.T.; Leonowicz, M.E.; Roth, W.J.; Vartuli, J.C.; Beck, J.S. Ordered mesoporous molecular sieves synthesized by a liquid-crystal template mechanism. Nature 1992, 359, 710–712. [Google Scholar] [CrossRef]
- Tolbert, S.H.; Firouzi, A.; Stucky, G.D.; Chmelka, B.F. Magnetic field alignment of ordered silicate-surfactant composites and mesoporous silica. Science 1997, 278, 264–268. [Google Scholar]
- Miyata, H.; Kuroda, K. Formation of a continuous mesoporous silica film with fully aligned mesochannels on a glass substrate. Chem. Mater. 2000, 12, 49–54. [Google Scholar] [CrossRef]
- Ichimura, K.; Fujiwara, T.; Momose, M.; Matsunaga, D. Surface-assisted photoalignment control of lyotropic liquid crystals. Part 1. Characterisation and photoalignment of aqueous solutions of a water-soluble dye as lyotropic liquid crystals. J. Mater. Chem. 2002, 12, 3380–3386. [Google Scholar] [CrossRef]
- Kawashima, Y.; Nakagawa, M.; Ichimura, K.; Seki, T. Photo-orientation of mesoporous silica materials via transfer from an azobenzene-containing polymer monolayer. J. Mater. Chem. 2004, 14, 328–335. [Google Scholar] [CrossRef]
- Kawashima, Y.; Nakagawa, M.; Seki, T.; Ichimura, K. Photo-orientation of mesostructured silica via hierarchical multiple transfer. Chem. Mater. 2002, 14, 2842–2844. [Google Scholar] [CrossRef]
- Yang, H.; Kuperman, A.; Coombs, N.; Mamiche-Afara, S.; Ozin, G.A. Synthesis of oriented films of mesoporous silica on mica. Nature 1996, 379, 703–705. [Google Scholar] [CrossRef]
- Seki, T.; Fukuda, K.; Ichimura, K. Photocontrol of polymer chain organization using a photochromic monolayer. Langmuir 1999, 15, 5098–5101. [Google Scholar] [CrossRef]
- Fukumoto, H.; Nagano, S.; Seki, T. In situ polymerization of liquid crystalline monomers within photoaligned mesoporous silica thin film. Chem. Lett. 2006, 35, 180–181. [Google Scholar] [CrossRef]
- Kawatsuki, N.; Kawakami, T.; Yamamoto, T. A photoinduced birefringent film with a high orientational order obtained from a novel polymer liquid crystal. Adv. Mater. 2001, 13, 1337–1339. [Google Scholar] [CrossRef]
- Fukumoto, H.; Nagano, S.; Kawatsuki, N.; Seki, T. Photo-orientation of mesoporous silica thin films on photocrosslinkable polymer films. Adv. Mater. 2005, 17, 1035–1039. [Google Scholar] [CrossRef]
- Park, M.; Harrison, C.; Chaikin, P.M.; Register, R.A.; Adamson, D.H. Block copolymer lithography: periodic arrays of ~1011 holes in 1 square centimeter. Science 1997, 276, 1401–1404. [Google Scholar] [CrossRef]
- Sidorenko, A.; Tokarev, I.; Minko, S.; Stamm, M. Ordered reactive nanomembranes/nanotemplates from thin films of block copolymer supramolecular assembly. J. Am. Chem. Soc. 2003, 125, 12211–12216. [Google Scholar]
- Osuji, C.; Ferreira, P.J.; Mao, G.; Ober, C.K.; Vander Sande, J.B.; Thomas, E.L. Alignment of self-assembled hierarchical microstructure in liquid crystalline diblock copolymers using high magnetic fields. Macromolecules 2004, 37, 9903–9908. [Google Scholar] [CrossRef]
- Morkved, T.L.; Lu, M.; Urbas, A.M.; Ehrichs, E.E.; Jaeger, H.M.; Mansky, P.; Russell, T.P. Local control of microdomain orientation in diblock copolymer thin films with electric fields. Science 1996, 273, 931–933. [Google Scholar]
- Tomikawa, N.; Lu, Z.; Itoh, T.; Imrie, C.T.; Adachi, M.; Tokita, M.; Watanabe, J. Orientation of microphase-segregated cylinders in liquid crystalline diblock copolymer by magnetic field. Jpn. J. Appl. Phys. 2005, 44, L711–L714. [Google Scholar]
- Hamley, I.W.; Castelletto, V.; Lu, Z.B.; Imrie, C.T.; Itoh, T.; Al-hussein, A. Interplay between smectic ordering and microphase separation in a series of side-group liquid-crystal block copolymers. Macromolecules 2004, 37, 4798–4807. [Google Scholar]
- Tokita, M.; Adachi, M.; Takazawa, F.; Watanabe, J. Shear flow orientation of cylindrical microdomain in liquid crystalline diblock copolymer and its potentiality as anchoring substrate for nematic mesogens. Jpn. J. Appl. Phys. 2006, 45, 9152–9156. [Google Scholar] [CrossRef]
- Yu, H.; Iyoda, T.; Ikeda, T. Photoinduced alignment of nanocylinders by supramolecular cooperative motions. J. Am. Chem. Soc. 2006, 128, 11010–11011. [Google Scholar]
- Morikawa, Y.; Nagano, S.; Watanabe, K.; Kamata, K.; Iyoda, T.; Seki, T. Optical alignment and patterning of nanoscale microdomains in a block copolymer thin film. Adv. Mater. 2006, 18, 883–886. [Google Scholar] [CrossRef]
- Tian, Y.; Watanabe, K.; Kong, X.; Abe, J.; Iyoda, T. Synthesis, nanostructures, and functionality of amphiphilic liquid crystalline block copolymers with azobenzene moieties. Macromolecules 2002, 35, 3739–3747. [Google Scholar] [CrossRef]
- Chen, A.; Komura, M.; Kamata, K.; Iyoda, T. Highly ordered arrays of mesoporous silica nanorods with tunable aspect ratios from block copolymer thin films. Adv. Mater. 2008, 20, 763–767. [Google Scholar] [CrossRef]
- Li, J.; Kamata, K.; Watanabe, S.; Iyoda, T. Template- and vacuum-ultraviolet-assisted fabrication of a Ag-nanoparticle array on flexible and rigid substrates. Adv. Mater. 2007, 19, 1267–1271. [Google Scholar] [CrossRef]
- Watanabe, S.; Fujiwara, R.; Hada, M.; Okazaki, Y.; Iyoda, T. Site-specific recognition of nanophase-separated surfaces of amphiphilic block copolymers by hydrophilic and hydrophobic gold nanoparticles. Angew. Chem. Int. Ed. 2007, 46, 1120–1123. [Google Scholar]
- Watanabe, R.; Kamata, K.; Iyoda, T. Nanodimple arrays fabricated on SiO2 surfaces by wet etching through block copolymer thin films. Jpn. J. Appl. Phys. 2008, 47, 5039–5041. [Google Scholar] [CrossRef]
- Li, J.; Kamata, K.; Komura, M.; Yamada, T.; Yoshida, H.; Iyoda, T. Anisotropic ion conductivity in liquid crystalline diblock copolymer membranes with perpendicularly oriented PEO cylindrical domains. Macromolecules 2007, 40, 8125–8128. [Google Scholar] [CrossRef]
- Huck, N.P.M.; Jager, W.F.; Lange, B.D.; Feringa, B.L. Dynamic control and amplification of molecular chirality by circular polarized light. Science 1996, 273, 1686–1688. [Google Scholar] [CrossRef]
- Suarez, M.; Schuster, G.B. Photoresolution of an axially chiral bicyclo[3.3.0]octan-3-one: Phototriggers for a liquid-crystal-based optical switch. J. Am. Chem. Soc. 1995, 117, 6732–6738. [Google Scholar] [CrossRef]
- Choi, S.W.; Ha, N.Y.; Shiromo, K.; Rao, N.V.S.; Paul, M.K.; Toyooka, T.; Nishimura, S.; Wu, J.W.; Park, B.; Takanishi, Y.;et al. Photoinduced circular anisotropy in a photochromic w-shaped-molecule-doped polymeric liquid crystal film. Phys. Rev. E 2006, 73, 021702:1–021702:6. [Google Scholar]
- Vera, F.; Tejedor, R.M.; Romero, P.; Barberá, J.; Ros, M.B.; Serrano, J.L.; Sierra, T. Light-driven supramolecular chirality in propeller-like hydrogen-bonded complexs that show columnar mesomorphism. Angew. Chem. Int. Ed. 2007, 46, 1873–1877. [Google Scholar]
- Tejedor, R.M.; Millaruelo, M.; Oriol, L.; Serrano, J.L.; Alcalá, R.; Rodríguez, F.J.; Villacampa, B. Photoinduced supramolecular chirality in side-chain liquid crystalline azopolymers. J. Mater. Chem. 2006, 16, 1674–1680. [Google Scholar]
- Maxein, G.; Zentel, R. Photochemical inversion of the helical twist sense in chiral polyisocyanates. Macromolecules 1995, 28, 8438–8440. [Google Scholar] [CrossRef]
- Mayer, S.; Zentel, R. Switching of the helical polymer conformation in a solid polymer film. Macromol. Rapid Commun. 2000, 21, 927–930. [Google Scholar] [CrossRef]
- Angiolini, L.; Bozio, R.; Giorgini, L.; Pedron, D.; Turco, G.; Daurú, A. Photomodulation of the chiroptical properties of new chiral methacrylic polymers with side chain azobenzene moieties. Chem. Eur. J. 2002, 8, 4241–4247. [Google Scholar]
- Nikilova, L.; Todorov, T.; Ivanov, M.; Andruzzi, F.; Hvilsted, S.; Ramanujam, P.S. Photoinduced circular anisotropy in side-chain azobenzene polyesters. Opt. Mater. 1997, 8, 255–258. [Google Scholar] [CrossRef]
- Nikolova, L.; Nedelchev, L.; Todorov, T.; Petrova, Tz.; Tomova, N.; Dragostinova, V.; Ramanujam, P.S.; Hvilsted, S. Self-induced light polarization rotation in azobenzene-containing polymers. Appl. Phys. Lett. 2000, 77, 657–659. [Google Scholar]
- Iftime, G.; Lagugné-Labarthet, F.; Natansohn, A.; Rochon, P. Control of chirality of an azobenzene liquid crystalline polymer with circularly polarized light. J. Am. Chem. Soc. 2000, 122, 126646–12650. [Google Scholar]
- Kim, M.J.; Shin, B.G.; Kim, J.J.; Kim, D.Y. Photoinduced supramolecular chirality in amorphous azobenzene polymer films. J. Am. Chem. Soc. 2002, 124, 3504–3505. [Google Scholar]
- Sumimura, H.; Fukuda, T.; Kim, J.Y.; Barada, D.; Itoh, M.; Yatagai, T. Photoinduced chirality in azobenzene amorphous copolymer bearing large birefringent moiety. Jpn. J. Appl. Phys. 2006, 45, 451–455. [Google Scholar]
- Fukuda, T.; Barada, D.; Sumimura, H.; Kim, J.Y.; Itoh, M.; Yatagai, T. Numerical analysis of photoinduced chirality in azobenzene polymer and its application as photoaddressable polarization altering elements. Jpn. J. Appl. Phys. 2008, 47, 1196–1202. [Google Scholar]
- Pagés, S.; Lagugné-Labarthet, F.; Buffeteau, T.; Sourisseau, C. Photoinduced linear andor circular birefringences from light propergation through amorphous or semectic azopolymer films. Appl. Phys. B 2002, 75, 541–548. [Google Scholar] [CrossRef]
- Barada, D.; Fukuda, T.; Sumimura, H.; Kim, J.Y.; Itoh, M.; Yatagai, T. Polarization recording in photoinduced chiral material for optical storage. Jpn. J. Appl. Phys. 2007, 46, 3928–3932. [Google Scholar]
- Yaroshchuk, O.V.; Kiselev, A.D.; Zakrevskyy, Y.; Bidna, T.; Kelly, J.; Chien, L.-C.; Lindau, J. Photoinduced three-dimensional orientational order in side chain liquid crystalline azopolymers. Phys. Rev. E 2003, 68, 011803:1–011803:15. [Google Scholar]
- Yaroshchuk, O.V.; Dumont, M.; Zakrevskyy, Y.A.; Bidna, T.V.; Lindau, J. Molecular structure of azopolymers and photoinduced 3D orientational order. 1. Azobenzene polyesters. J. Phys. Chem. B 2004, 108, 4647–4658. [Google Scholar]
- Yager, K.G.; Tanchak, O.M.; Godbout, C.; Fritzsche, H.; Barrett, C.J. Photomechanical effects in azo-polymers studied by neutron reflectometry. Macromolecules 2006, 39, 9311–9319. [Google Scholar]
- Karimi-Alavijeh, H.; Parsanasab, G.-M.; Baghban, M.-A.; Sarailou, E.; Gharavi, A.; Javadpour, S.; Shkunov, V. Fabrication of graded index waveguides in azo polymers using a direct writing technique. Appl. Phys. Lett. 2008, 92, 041105:1–041105:3. [Google Scholar]
- Bang, C.-U.; Shishido, A.; Ikeda, T. Azobenzene liquid-crystalline polymer for optical switching of grating waveguide couplers with a flat surface. Macromol. Rapid Commun. 2007, 28, 1040–1044. [Google Scholar] [CrossRef]
- Liu, J.; Wang, M.; Li, Y.; Zhong, H. Photo-triggered assembly/disassembly of macroscopically ordered monodomain lamellar structure in side-chain liquid crystalline polymer containing strong polar azobenzene mesogens. Liquid. Cryst. 2011, 38, 105–113. [Google Scholar] [CrossRef]
- Kawatsuki, N.; Hasegawa, T.; Ono, H.; Yamamoto, T. Scattering linear polarizer based on a polymer blend of photo-cross-linkable polymer liquid crystal and photoinactive polymer. Chem. Lett. 2002, 31, 1256–1257. [Google Scholar]
- Gomy, C.; Schmitzer, A.R. Synthesis and photoresponsive properties of a molecularly imprinted polymer. Org. Lett. 2007, 9, 3865–3868. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Emoto, A.; Uchida, E.; Fukuda, T. Optical and Physical Applications of Photocontrollable Materials: Azobenzene-Containing and Liquid Crystalline Polymers. Polymers 2012, 4, 150-186. https://doi.org/10.3390/polym4010150
Emoto A, Uchida E, Fukuda T. Optical and Physical Applications of Photocontrollable Materials: Azobenzene-Containing and Liquid Crystalline Polymers. Polymers. 2012; 4(1):150-186. https://doi.org/10.3390/polym4010150
Chicago/Turabian StyleEmoto, Akira, Emi Uchida, and Takashi Fukuda. 2012. "Optical and Physical Applications of Photocontrollable Materials: Azobenzene-Containing and Liquid Crystalline Polymers" Polymers 4, no. 1: 150-186. https://doi.org/10.3390/polym4010150



