A Possibility for Construction of an Iodine Cleaning System Based on Doping for π-Conjugated Polymers
Abstract
: An iodine accumulation method using polyaniline (PANI) and a textile composite is proposed. PANI/pulp paper sheets prepared by a paper making technique are suitable for iodine adsorption, because of good processability. The PANI-based paper sheets can be applied for iodine cleanup as air filters, water filters, and floorcloth. This concept may lead to a development of an iodine cleaning machine or iodine shield cloth based on π-conjugated polymer composites. In-situ vapor phase doping of iodine, observation of surface images, and IR measurements are carried out to examine iodine doping function for the PANI/pulp paper sheets.1. Introduction
Since the discovery of halogen doping effect for polyacetylene which confirmed dramatic increase of electrical conductivity, the development of conducting polymers for practical applications has been rapid. These new technologies include solar cells, high-performance capacitors, field-effective transistors (FET), organic electroluminescence devices (O-EL), and electrochromic devices (EC), which represent a new approach of “plastic electronics” [1-5]. After the halogen doping effect was demonstrated for conducting polymers, iodine has been widely employed in both vapor phase and solution doping because iodine doping can prevent the occurrence of electrophilic addition to the π-conjugated system without disruption of π-conjugation of the main chain.
In the neutral state, conducting polymer precursors (π-conjugated polymers) are inherently insulators. However, doping by the addition of low concentrations of an electron donor yields a significant increase in conductivity, accompanied by generation of charge carriers. The π-conjugated system and dopants display very good mutual affinity due to donor-accepter interaction. This charge-transfer type interaction is applicable for adsorption of iodine in π-conjugated polymers.
In the family of conducting polymers, polyaniline (PANI) is one of the most promising electrically conducting polymers because of its chemical stability, relatively high conductivity, and interesting optical properties, such as electrochromism. Furthermore, production of PANI is low cost as compared to other conducting polymers, and polymerization of aniline can be performed in an environmentally friendly aqueous medium. Recently, polymerization of aniline in the presence of paper pulp was developed [6]. The PANI/pulp composite as a [synthetic polymer]/[natural polymer] composite has been processed with a traditional paper making technique to obtain a smooth conducting sheet. This technique largely eliminates the drawback of low processability of conducting polymers.
Treatment and accumulation of iodine in the air and water is difficult. Iodine has high vapor pressure at room temperature due to its low heat of sublimation. This vapor state further makes accumulating iodine difficult. Therefore, the donor-accepter interactions between π-conjugated polymers (polyacetylene, polythiophene, polypyrrole, polyphenylene, and their derivatives) and iodine can be considered as a useful method for accumulation of iodine. Iodine doping in PANI has been reported in previous studies [7-10]. In this study, a possibility of a PANI/pulp composite sheet for iodine absorption through vapor phase doping of iodine is investigated.
2. Results and Discussion
2.1. Preparation of Polyaniline/Pulp (PANI/Pulp) Paper Sheet
PANI/pulp is prepared by polymerization of aniline in the presence of pulp fiber (Scheme 1) [6]. After polymerization of aniline, the crude product is washed in ammonia/water to afford emeraldine base (EB, half-oxidized state) of PANI in the PANI/pulp composite. Further reduction treatment with hydrazine can produce leucoemeraldine base (LEB, fully reduced state). In this polymerization, the pulp fiber surface plays the role of a reaction site. The aniline (monomer) is absorbed on the pulp fiber surface by the capillary effect, and its polymerization is carried out at the surface. Therefore, effective polymerization and formation of a composite are performed. The PANI/pulp composite thus prepared is in cotton-like form, as shown in Figure 1(a).
The traditional paper making technique in the case of the PANI/pulp affords π-conjugated paper composite sheet. The paper sheet image is shown in Figure 1(b). The composite paper had a smooth surface and was sufficiently flexible to enable the preparation of paper-folding artwork. The traditional Japanese origami technique allows various processing of the PANI paper sheets [11]. Figure 1(c,d) (magnification image) shows an example of Miura folding of PANI/pulp sheet. Figure 1(e) displays thermograph of the Miura folding paper upon irradiation of infra-red (IR) light. The surface temperature increased with IR light because PANI absorbs UV and IR region lights. Processing of the PANI/pulp paper sheet with Miura folding having an apparently large surface area may be applied for IR light absorption sheet with good absorption efficiency.
2.2. Surface Image
Scanning electron microscopy (SEM) images of the PANI/pulp surface are shown in Figure 2(a,b). Fine fungal-like PANI structures located on the pulp fibers are observed.
2.3. Vapor Phase Doping of Iodine
The PANI component in this composite thus synthesized consists of π-conjugated main chains. The π-conjugated system can be doped by electron acceptors. The chemical doping with vapor phase iodine produces charge carriers along the main chain. In this section, in-situ iodine (electron acceptor) doping for this composite was examined.
The PANI/pulp (as prepared, emeraldine salt, ES) was treated with aqueous ammonia to yield EB state, as shown in Scheme 1. The EB state of the PANI/pulp paper shows a signal in electron spin resonance (ESR) because the EB is the half-oxidized state with an acid. Vapor phase doping of iodine in the air at the room temperature confirms increase of the sample weight (32.2 wt% increase after 30.5 h doping). While, sample weight of a pure paper sheet (as a blank sample, filter paper, pulp = 100%) resulted in 6.9 wt% increases by the vapor phase doping of iodine for 30.5 h. This experiment was carried out with the PANI/pulp sheet (in the same condition as the doping for PANI/pulp sheet). The result thus obtained indicates that the PANI/pulp sheet exactly captured iodine vapor with good efficiency. Visual inspection with a combination of readout optical microscope and quartz microbalance confirmed increase of the sample weight after the iodine doping. The experimental equipments are shown in Figure S1 (Electronic Supplementary Information, ESI).
The ESR spectra of the PANI/pulp paper (EB) show asymmetric Dysonian line shape, as shown in Figure 3. The intensity is gradually increased upon in-situ vapor phase doping of iodine. The line shape broadens with progress of the iodine doping.
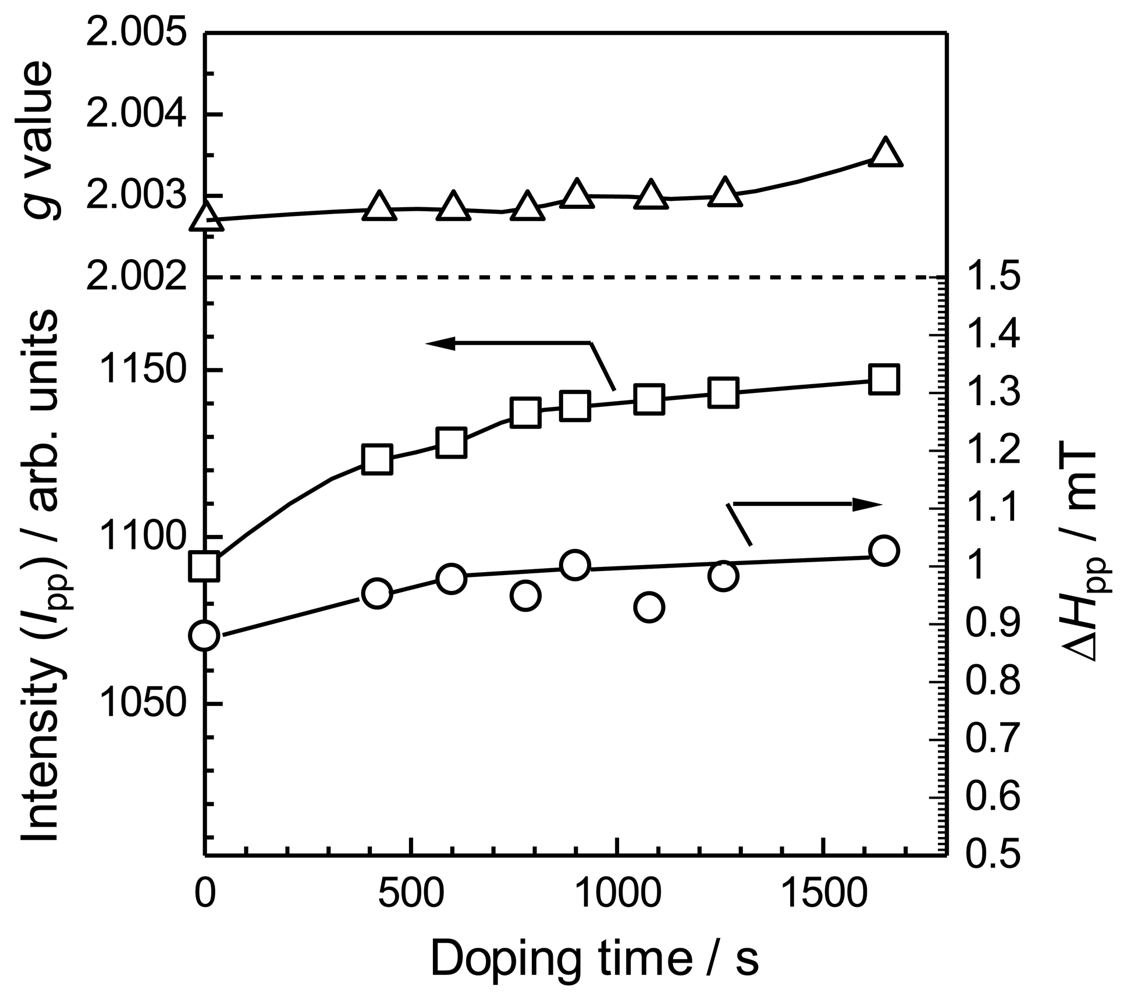
Figure 4 summarizes the changes in g-value, ESR intensity (Ipp), and peak-to-peak linewidth (ΔHpp) during the vapor phase doping of iodine in the PANI/pulp. The g-values gradually increase with doping time, suggesting the generation of polarons (radical cations) and bipolarons (dications) along the main chain of the PANI layer.
The ESR intensity decreases with heavy doping (green line for 171 min of doping). The narrow Δ Hpp value for a light doping level suggests delocalization of the charge carriers on the main chain. Subsequent heavy doping produces broad ESR line shapes. This is a typical behavior of the doping process with generation of bipolarons in conducting polymers.
The pristine PANI (EB) is slightly doped with sulfuric acid, and further doping with electron acceptor is possible. Therefore, the PANI (EB) is further doped with iodine vapor. LEB as a fully reduced state prepared by hydrazine treatment can have good doping susceptibility. However, the treatment of PANI with ammonia is convenient and safe as compared to employment of hydrazine for the preparation of the reduced state.
2.4. IR
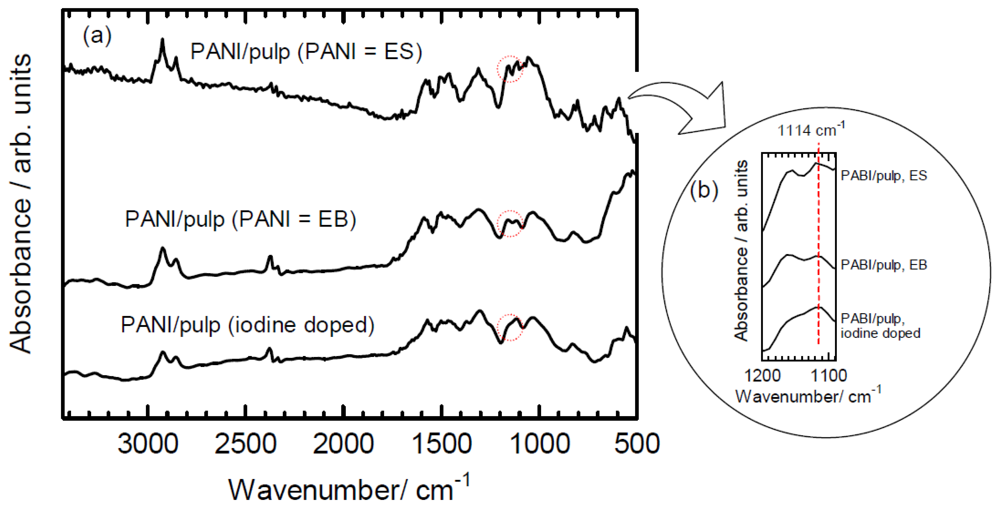
Infrared absorption (IR) spectra were collected for PANI/pulp (as polymerized, emeraldine salt (ES)), PANI/pulp (after treatment of ammonia, emeraldine base (EB)), and iodine-doped PANI/pulp (Figure 5). The absorption bands at 2,850–2,930 cm−1 are assignable to νCH2, CH3 of the methylene group derived from cellulose (pulp). The absorption band at 1,590 cm−1 is ascribed to benzene (C=C) structure of PANI. The samples show both cellulose and PANI characteristics. The PANI/pulp (as polymerized, ES) and iodine-doped samples show characteristic absorption at 1,114 cm−1 (Figure 5(b)), suggesting this band is related to doping. Therefore, the IR results confirm the chemical structure of PANI/pulp and doping effect.
2.5. Solution Doping
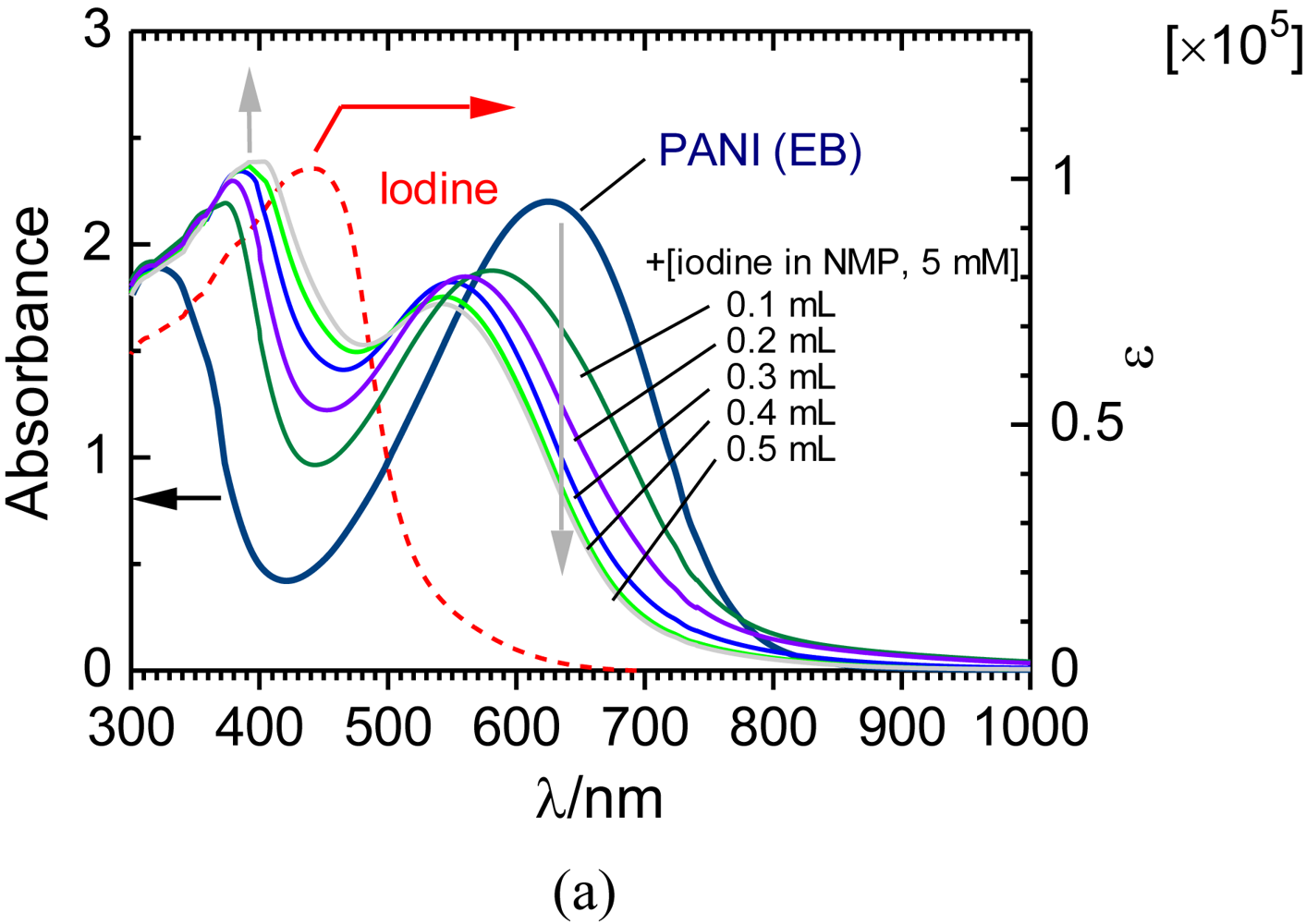
Solution doping of iodine for PANI (EB) [12] in N-methyl-2-pyrrolidone (NMP) was carried out to confirm doping effect of PANI in the solution. Firstly, a PANI (EB) in NMP solution (PANI (EB)/NMP, 0.0048 wt%) was prepared. Next, solution doping of iodine to approximately 3 mL of the PANI (EB)/NMP was conducted by addition of certain amount (0.1–0.5 mL) of iodine in NMP solution (5 mM) for the UV-Vis optical absorption spectroscopy measurements. Figure 5 shows the absorption spectra of PANI (EB) in NMP, iodine in NMP solution, and PANI (EB) with iodine doping in NMP. PANI (EB) displays absorption maxima at 320 nm (π–π* transition of benzene ring) and at 626 nm in the spectrum. The absorption band at 626 nm is assignable to the transition of electron from the HOMO (highest occupied molecular orbital) of the benzenoid part to the LUMO (lowest unoccupied molecular orbital) of the quinoid structure of PANI [8,13]. This band decreased in intensity and shifted toward short wavelengths (from 626 nm to 541 nm) accompanied by the progress of the doping. Furthermore, a new absorption band at 372–398 nm with isosbestic point at around 580 nm appeared. This result strongly indicates electronic interaction between PANI and iodine. This absorption band is not due to optical absorption of iodine because the absorption band of iodine is observed at 440 nm, as shown in Figure 6(a) (red dashed line). Figure 6(b) shows CIE color space chromaticity diagram of the PANI (EB) with solution doping of iodine. The solution color of PANI (EB) shifted toward the red region accompanied by progress of the iodine doping in the solution. This result confirms color change upon the iodine doping. Absorption spectra of the PANI/pulp paper sheet cannot be obtained because of insolubility in solvents. However, the result of the solution doping for PANI (EB) suggests that the iodine in the solution can be adsorbed onto the PANI/pulp sheet via the electronic interaction.
2.6. Possible Mechanism
The PANI/pulp paper sheet is capable of iodine vapor absorption through the conducting polymer doping process. Iodine doping occurs in the form of triiodide anion (I3ˉ) by the oxidation of the benzenoid diamine units [8].
The iodine can attach to the surface of the PANI due to physical adsorption and electronic interactions (charge-transfer type interactions) based on the doping mechanism observed for conducting polymers. The doping can be carried out, not only in the vapor phase, but also in solution. The PANI/pulp can absorb very fine, molecular iodine species by the electronic interaction.
3. Experiment Section
3.1. Materials
Aniline (Tokyo Kasei, TCI) and water were distilled prior to use. Ammonium persulfate (APS, (NH4)2S2O8) (Kanto Chemicals, Japan) was used as received. Needle bleached Kraft pulp (NBKP) was employed.
3.2. Techniques
ESR spectroscopy measurements were carried out using a JEOL JES TE-200 spectrometer with 100 kHz modulation. SEM observations were carried out with JEOL JSM-521. IR spectra were obtained with a JASCO FT/IR 550 spectrometer. Gravimetric measurement was conducted with a readout optical microscopy system (PRM-2, PIKA industry, Tokyo) combined with a quartz microbalance. A thermographic image of the PANI/pulp paper was obtained with FLIR i5 (FLIR Systems, USA).
3.3. Typical Polymerization Method of Aniline in the Presence of Pulp
A solution of NBKP (100 g), aniline (75 g), H2SO4 (60 g) in 3 L water was stirred for 1 h at room temperature. The solution was cooled to ca. 5 °C, and ammonium persulfate (APS, 60 g) was gradually added. The color of the solution turned from brown to dark green. After 24 h, the solution was filtered, and the resultant was washed with 3 L of distilled water for 24 h. After filtration, the dark green material was further washed with 1 L of methanol for 24 h, and dried to afford PANI/pulp fibers as product.
3.4. Papermaking
Papermaking was performed by dispersion of the PANI/pulp in water, filtration with net, removal of the water in the sheet by press, and drying.
The PANI/pulp paper sheet was submerged in aqueous ammonia solution to afford the EB. After 1 h, the sheet was washed with acetone, and dried to yield PANI/pulp paper sheet in the EB state.
Also, as prepared PANI/pulp fibers (ES) can be reduced before papermaking. Subsequently, papermaking technique for the PANI/pulp fibers (ES) allows production of PANI/pulp paper sheet in the EB state.
3.5. Doping
In-situ vapor phase doping was carried out in the 5 mm quartz tube under the presence of iodine for the ESR measurements. The sublimed iodine vapor is adsorbed onto the PANI/pulp sample. The vapor phase doping of iodine was carried out in the air at room temperature.
4. Conclusion and Perspectives
The vapor phase doping of PANI/pulp with iodine was confirmed by the ESR measurements. The iodine absorption behavior of PANI/pulp is similar to that observed upon doping PANI with acid iodides, such as hydrogen iodide. On the other hand, application of alkaline iodides doping for PANI might be restricted. The PANI based paper sheets can be applied to clean up radioactive iodine (e.g., 126I, 131I) [14] as air filters (mixed with a charcoal filter), water filters, and floorcloth. A scheme of radioactive decay of 131I (β-decay) is shown in Scheme S1 (Electronic Supplementary Information, ESI). Half life time of 131I is 8.02 days, and 131I subsequently transforms into radioactive 131Xe in the form of gas, and it may be released from the PANI [15]. Therefore, PANI as a 131I absorber can be valid in short term application for the protection from the vapor.
Although the specific surface area is small compared to the powder form of PANI, a combination of π-conjugated polymers and textiles allows good processability and utility as materials to clean iodine decontamination in the air. Preparation of other composites of π-conjugated polymers and textiles, such as cotton, silk, and synthetic textiles is possible [16]. Furthermore, a combination of π-conjugated polymers and starch may provide good iodine absorption properties. This concept leads to the possibility of designing an iodine cleaner or iodine shield cloth based on π-conjugated systems.
Supplementary Material
polymers-03-00875-s001.pdf




Acknowledgments
This research was supported by ILC project of University of Tsukuba.
References and Notes
- Shirakawa, H. The discovery of polyacetylene film: The dawning of an era of conducting polymers. Angew. Chem. Int. Ed. 2001, 40, 2574–2580. [Google Scholar]
- MacDiarmid, A.G. “Synthetic metals”: A novel role for organic polymers. Angew. Chem. Int. Ed. 2001, 40, 2581–2590. [Google Scholar]
- Heeger, A.J. Semiconducting and metallic polymers: The fourth generation of polymeric materials. Angew. Chem. Int. Ed. 2001, 40, 2591–2611. [Google Scholar]
- Advances in Synthetic Metals, Twenty Years of Progress in Science and Technology; Bernier, P., Lefrant, S., Bidan, G., Eds.; Elsevier Science S. A.: Lausanne, Switzerland, 1999.
- Goto, H.; Yoneyama, H.; Togashi, F.; Ohta, R.; Tsujimoto, A.; Kita, E.; Ohshima, K. Preparation of conducting polymers by electrochemical methods and demonstration of a polymer battery. J. Chem. Edu. 2008, 85, 1067–1070. [Google Scholar]
- Goto, H. Electrically conducting paper from a polyaniline/pulp composite and paper folding art work for a 3D object. Text. Res. J. 2011, 81, 122–127. [Google Scholar]
- Wang, L.; Jing, X.; Wang, F. On the iodine-doping of polyaniline and poly-ortho-methylaniline. Synth. Met. 1991, 41, 739–744. [Google Scholar]
- Gizdavic-Nikolaidis, M.; Graham, A.; Bowmaker, G. Iodine vapour doped polyaniline. Polymer 2008, 49, 3070–3075. [Google Scholar]
- Sarkar, A.; Ghosh, P.; Meikap, A.K.; Chattopadhyay, S.K.; Chatterjee, S.K.; Chowdhury, P.; Roy, K.; Saha, B. Electrical-transport properties of iodine-doped conducting polyaniline. J. Appl. Sci. 2008, 108, 2312–2320. [Google Scholar]
- Sajeev, U.S.; Joseph Mathai, C.; Saravanan, S.; Ashokan, R.R.; Venkatachalam, S.; Ananthraman, M.R. On the optical and electrical properties of rf and a.c. plasma polymerized aniline thin films. Bull. Mater. Sci. 2006, 29, 159–163. [Google Scholar]
- Nishiyama, Y. Miura folding: Applying origami to space exploration. Available online: http://www.osaka-ue.ac.jp/zemi/nishiyama/math2010/miura.pdf (accessed on 30 March 2011).
- PANI (ES) was prepared by basically the same method as preparation of the PANI/pulp except addition of pulp in the polymerization reaction. PANI (EB) was prepared by treatment of PANI(ES) with a large amount of aqueous ammonia.
- Huang, W.S.; MacDiarmid, A.G. Optical properties of polyaniline. Polymer 1993, 34, 1833–1845. [Google Scholar]
- Wu, C.Y.; Satteson, M.; Wang, K.; Cline, D.; Gove, H.E.; Hanna, S.S.; Hass, M.; Ibbotson, R.; Ramayya, A.V. Isomers in iodine nuclei. Phys. Rev. C 1990, 41, 1600–1605. [Google Scholar]
- Xenon is heavier than air.
- Jwa, J.-M.; Kawashima, H.; Goto, H. Polyaniline/cotton composite. Fiber. Prep. J. 2011. in press. [Google Scholar]
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Goto, H. A Possibility for Construction of an Iodine Cleaning System Based on Doping for π-Conjugated Polymers. Polymers 2011, 3, 875-885. https://doi.org/10.3390/polym3020875
Goto H. A Possibility for Construction of an Iodine Cleaning System Based on Doping for π-Conjugated Polymers. Polymers. 2011; 3(2):875-885. https://doi.org/10.3390/polym3020875
Chicago/Turabian StyleGoto, Hiromasa. 2011. "A Possibility for Construction of an Iodine Cleaning System Based on Doping for π-Conjugated Polymers" Polymers 3, no. 2: 875-885. https://doi.org/10.3390/polym3020875




