Simvastatin Release from Poly(lactide-co-glycolide) Membrane Scaffolds
Abstract
:1. Introduction
2. Experimental Section
2.1. Materials

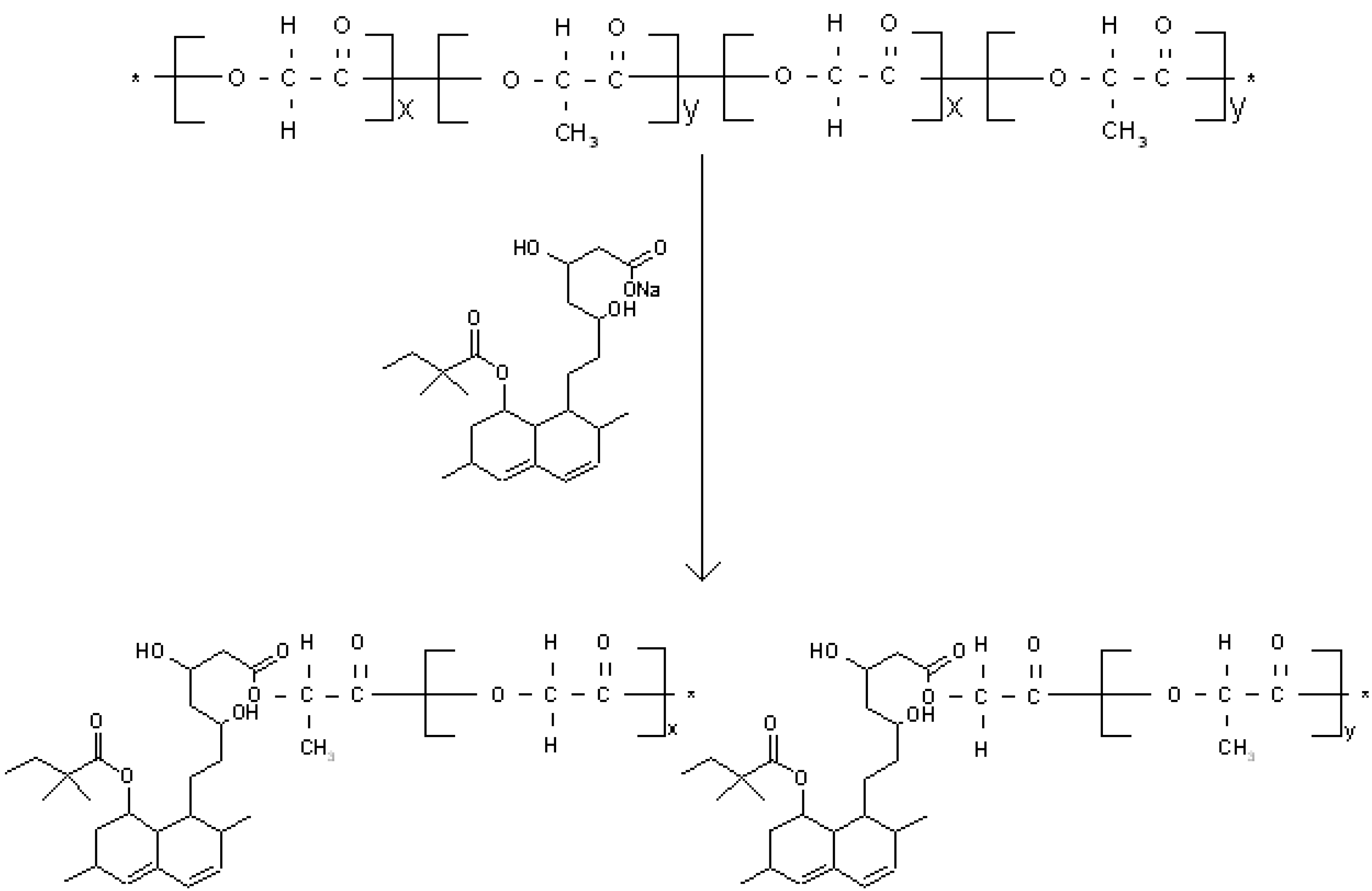
2.2. Extraction and Saponification of Simvastatin
2.3. Membrane Scaffold Preparation
2.4. Simvastatin Quantification by HPLC
2.5. Viscosity of the PLGA Membrane Casting Dopes
2.6. Morphology of PLGA Membrane Scaffolds
3. Results and Discussion
3.1. Viscosity of the Casting Dopes Was Reduced on Addition of Sap-Sim
| Dope Composition | PLGA | PLGA + Dis-Sim (1.0 mg sim/g PLGA) | PLGA + sap-sim (0.5 mg sim/g PLGA) | PLGA + sap-sim (1.0 mg sim/g PLGA) |
| Viscosity (Pa s) | 1.2 | 1.2 | 0.21 | 0.05 |
3.2. Covalent Bonding of Sap-Sim to PLGA Appears to Increase Porosity of Membranes

3.3. Simvastatin Release Mechanisms Are Different for Dis-Sim and Sap-Sim
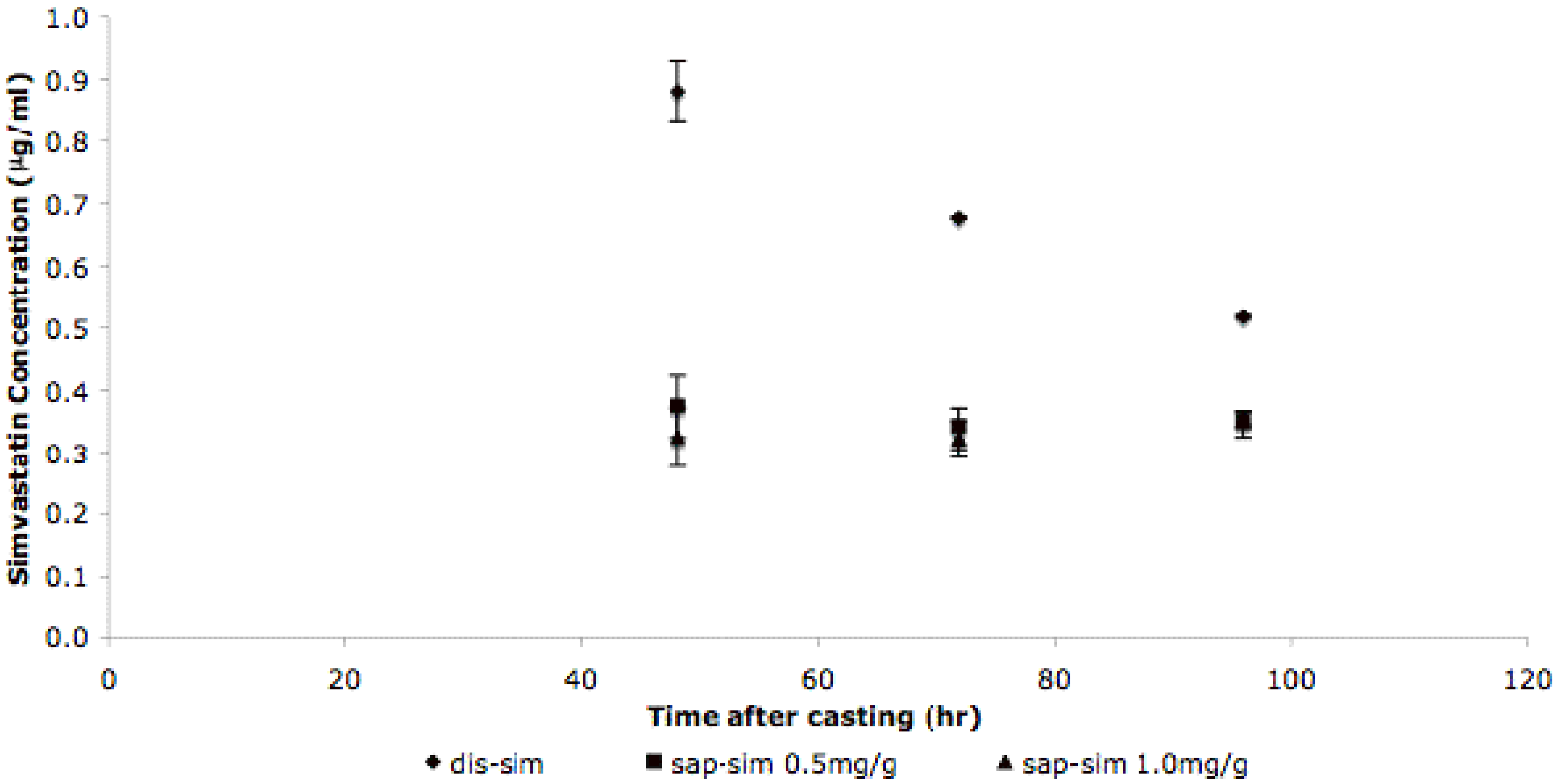
4. Conclusions
Acknowledgements
References
- Jadhav, S.B.; Jain, G.K. Statins and osteoporosis: New role for old drugs. J. Pharm. Pharmacol. 2006, 58, 3–18. [Google Scholar] [CrossRef] [PubMed]
- Canalis, E.; Hott, M.; Deloffre, P.; Tsouderos, Y.; Marie, P.J. The divalent strontium salt S12911 enhances bone cell replication and bone formation in vitro. Bone 1996, 18, 517–523. [Google Scholar] [CrossRef] [PubMed]
- Buehler, J.; Chappuis, P.; Saffar, J.L.; Tsouderos, Y.; Vignery, A. Strontium ranelate inhibits bone resorption while maintaining bone formation in alveolar bone in monkeys (Macaca fascicularis). Bone 2001, 29, 176–179. [Google Scholar] [CrossRef] [PubMed]
- Mundy, G.; Garrett, R.; Harris, S.; Chan, J.; Chen, D.; Rossini, G.; Boyce, B.; Zhao, M.; Gutierrez, G. Stimulation of bone formation in vitro and in rodents by statins. Science 1999, 286, 1946–1949. [Google Scholar] [CrossRef] [PubMed]
- Garrett, R.; Mundy, G. The role of statins as potential targets for bone formation. Arthritis Res. 2002, 4, 237–240. [Google Scholar] [CrossRef] [PubMed]
- Phillips, B.; Belmonte, N.; Vernochet, C.; Ailhaud, G.; Dani, C. Compactin enhances osteogenesis in murine embryonic stem cells. Biochem. Biophys. Res. Commun. 2001, 284, 478–484. [Google Scholar] [CrossRef] [PubMed]
- Garrett, I.R.; Esparza, J.; Chen, D.; Zhao, M.; Gutierrez, G.; Escobedo, A.; Horn, D.; Mundy, G.R. Statins mediate their effects on osteoblasts by inhibition of HMG-CoA reductase and ultimately BMP-2. J. Bone Miner. Res. 2000, 15, S225–S225. [Google Scholar]
- Maeda, T.; Kawane, T.; Horiuchi, N. Statins augment vascular endothelial growth factor expression in osteoblastic cells vial inhibition of protein prenylation. Endocrinology 2003, 144, 681–692. [Google Scholar] [CrossRef] [PubMed]
- Sugiyama, M.; Kodama, T.; Konishi, K.; Abe, K.; Asami, S.; Oikawa, S. Compactin and simvastatin, but not pravastatin, induce bone morphogenetic protein-2 in human osteosarcoma cells. Biochem. Biophys. Res. Commun. 2000, 271, 688–692. [Google Scholar] [CrossRef] [PubMed]
- Garrett, I.R.; Escobedo, A.; Esparza, J.; Mundy, G.R. Cerivastatin increases BMP-2 expression in vivo and bone formation in concentrations of two orders of magnitude lower than other statins. J. Bone Miner. Res. 1999, 14, S180–S180. [Google Scholar]
- Maeda, T.; Matsunuma, A.; Kawane, T.; Horiuchi, N. Simvastatin promotes osteoblast differentiation and mineralization in MC3T3-E1 cells. Biochem. Biophys. Res. Commun. 2001, 280, 874–877. [Google Scholar] [CrossRef] [PubMed]
- Woo, J.T.; Kasai, S.; Stern, P.H.; Nagai, K. Compactin suppresses bone resorption by inhibiting the fusion of prefusion osteoclasts and disrupting the actin ring in osteoclasts. J. Bone Miner. Res. 2000, 15, 650–662. [Google Scholar] [CrossRef] [PubMed]
- Von Stechow, D.; Fish, S.; Yahalom, D.; Bab, I.; Chorev, M.; Müller, R.; Alexander, J.M. Does simvastatin stimulate bone formation in vivo? BMC Musculoskel. Disord. 2003. [Google Scholar] [CrossRef]
- Bauer, D.C. HMG CoA reductase inhibitors and the skeleton: A comprehensive review. Osteoporos. Int. 2003, 14, 273–282. [Google Scholar] [CrossRef] [PubMed]
- Gasser, J.A. Fluvastatin and cerivastatin are not anabolic for bone after local or systemic administration of non-toxic doses in mice and rats. J. Bone Miner. Res. 2001, 16, S295–S295. [Google Scholar]
- Yao, W.; Li, C.Y.; Farmer, R.W.; Chen, J.L.; Mo, A.; Cooper, R.; Chmielewski, P.; Setterberg, R.B.; Lee, W.S.S.; Lundy, M.W. Simvastatin did not prevent bone loss in ovariectomized rats. J. Bone Miner. Res. 2001, 16, S294. [Google Scholar]
- Maritz, F.J.; Conradie, M.M.; Hulley, P.A.; Gopal, R.; Hough, S. Effect of statins on bone mineral density and bone histomorphometry in rodents. Arterioscler. Thromb. Vasc. Biol. 2001, 21, 1636–1641. [Google Scholar] [CrossRef] [PubMed]
- Mulder, M. Basic Principles of Membrane Technology; Kluwer Academic Publishers: Dordrecht, The Netherlands, 1992. [Google Scholar] [Green Version]
- Whang, K.M.; Grageda, E.; Khan, A.; Mcdonald, J.; Lawton, M.; Satsangi, N. A novel osteotropic biomaterial OG-PLG: In vitro efficacy. J. Biomed. Mater. Res. A 2005, 74A, 247–253. [Google Scholar] [CrossRef] [PubMed]
- Crawford, D.T.; Qi, H.; Chidsey-Frink, K.L.; Simmons, H.A.; Thompson, D.D.; Ke, H.Z. Statin increases cortical bone in young male rats by single, local administration but fails to restore bone in ovariectomized (OVX) rats by daily systemic administration. J. Bone Miner. Res. 2001, 16, S295–S295. [Google Scholar]
- Whang, K.; Zhao, M.; Qiao, M.; Rossini, G.; Horn, D.; Garrett, I.R.; Mundy, G.R.; Chen, D. Administration of lovastatin locally in low doses in a novel delivery system induces prolonged bone formation. J. Bone Miner. Res. 2000, 15, F380. [Google Scholar]
- Ellis, M.J.; Chaudhuri, J.B. Poly(lactic-co-glycolic acid) hollow fibre membranes for use as a tissue engineering scaffold. Biotech. Bioeng. 2007, 96, 177–187. [Google Scholar] [CrossRef]
- Whang, K.M.; McDonald, J.; Khan, A.; Satsangi, N. A novel osteotropic biomaterial OG-PLG: Synthesis and in vitro release. J. Biomed. Mater. Res. A 2005, 74A, 237–246. [Google Scholar] [CrossRef] [PubMed]
- Piskin, E.; Isoglu, E.; Bölgen, N.; Vargel, I.; Griffiths, S.; Çavusoglu, T.; Korkusuz, P.; Güzel, E.; Cartmell, S. In vivo Performance of simvastatin loaded electrospun spiral-wounded polycaprolactone scaffolds in reconstruction of cranial bone defects in the rat model. J. Biomed. Mat. Res. A 2009, 15, 1137–1151. [Google Scholar] [CrossRef]
- Yang, S.F.; Leong, K.F.; Du, Z.H.; Chua, C.K. The design of scaffolds for use in tissue engineering. Part 1. Traditional factors. Tissue Eng. 2001, 7, 679–689. [Google Scholar] [CrossRef] [PubMed]
- Benoit, D.S.W.; Nuttelman, C.R.; Collins, S.D.; Anseth, K.S. Synthesis and characterization of a fluvastatin-releasing hydrogel delivery system to modulate hMSC differentiation and function for bone regeneration. Biomaterials 2006, 27, 6102–6110. [Google Scholar] [CrossRef] [PubMed]
- Shearer, H.; Ellis, M.J.; Perera, S.P.; Chaudhuri, J.B. Effects of common sterilization methods on the structure and properties of poly(D,L lactic-co-glycolic acid) scaffolds. Tissue Eng. 2006, 12, 2717–2727. [Google Scholar] [CrossRef] [PubMed]
© 2010 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Rashidi, H.; Ellis, M.J.; Cartmell, S.H.; Chaudhuri, J.B. Simvastatin Release from Poly(lactide-co-glycolide) Membrane Scaffolds. Polymers 2010, 2, 709-718. https://doi.org/10.3390/polym2040709
Rashidi H, Ellis MJ, Cartmell SH, Chaudhuri JB. Simvastatin Release from Poly(lactide-co-glycolide) Membrane Scaffolds. Polymers. 2010; 2(4):709-718. https://doi.org/10.3390/polym2040709
Chicago/Turabian StyleRashidi, Hassan, Marianne J. Ellis, Sarah H. Cartmell, and Julian B. Chaudhuri. 2010. "Simvastatin Release from Poly(lactide-co-glycolide) Membrane Scaffolds" Polymers 2, no. 4: 709-718. https://doi.org/10.3390/polym2040709
APA StyleRashidi, H., Ellis, M. J., Cartmell, S. H., & Chaudhuri, J. B. (2010). Simvastatin Release from Poly(lactide-co-glycolide) Membrane Scaffolds. Polymers, 2(4), 709-718. https://doi.org/10.3390/polym2040709





