The following equations were used to calculate the crystallinity (
χ) of the polymers in the blends:
where
ΔHfm is the melting enthalpy of PP,
ΔHim is the melting enthalpy of LDPE,
is the percentage crystallinity of PP,
is the percentage crystallinity of LDPE,
ΔH0m(
LDPE) is the equilibrium melting enthalpy of LDPE = 288 J·g
−1 [
20],
ΔH0m(
PP) is the equilibrium melting enthalpy of PP = 207 J·g
−1 [
21],
ϕPP is the weight fraction of PP, and
ϕLDPE is the weight fraction of LDPE. In
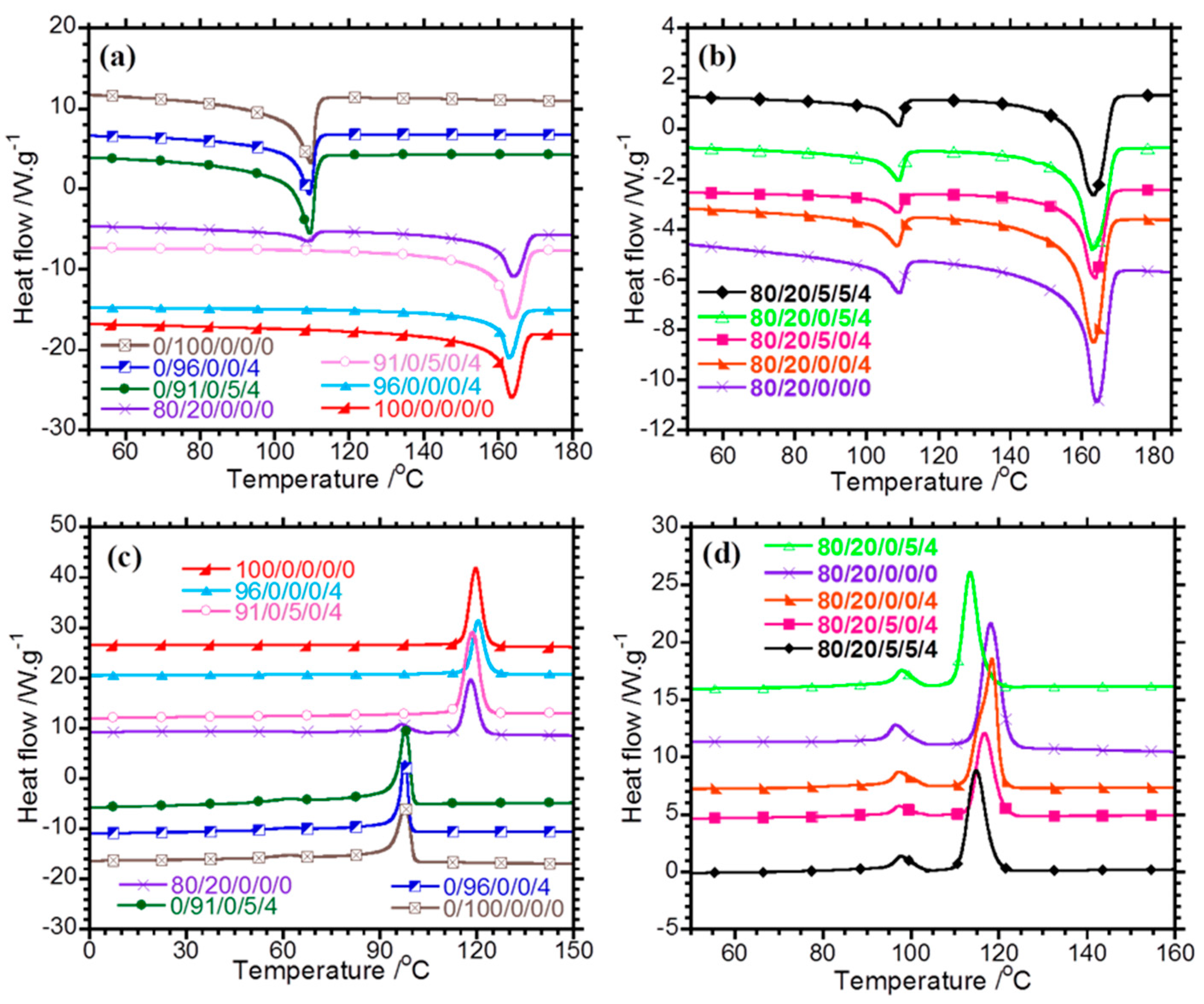
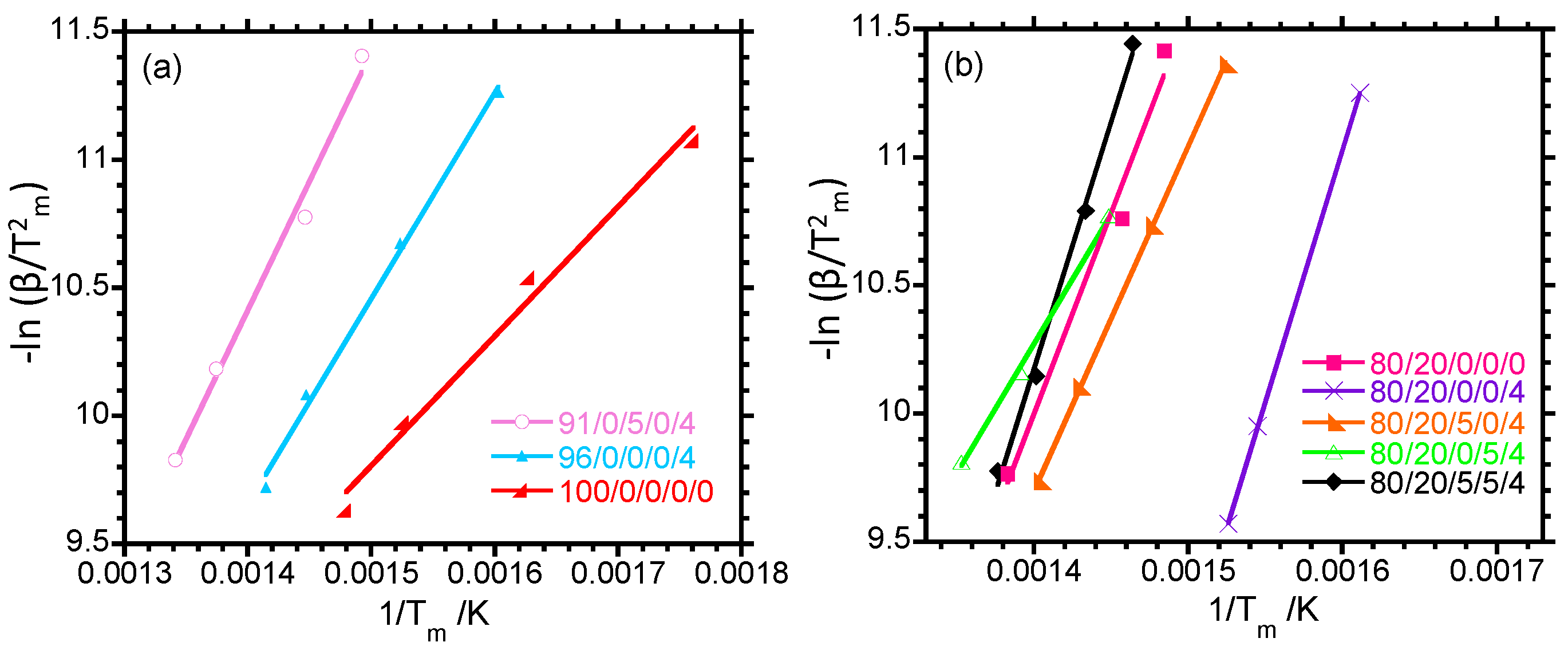
Figure 1a, neat PP has melting temperature (
Tfm) of 163 °C, whereas the melting temperature of LDPE (
Tim) is 109 °C. The 80/20/0/0/0 blend has two melting peaks associated with the melting of the individual polymers. The melting temperatures of PP and LDPE in the binary (
Figure 1a) and ternary (
Figure 1b) composites are the same as those of neat PP and LDPE, suggesting that the thickness of the polymer crystals is not affected by the incorporation of clay and its spatial distribution in different phases of the polymeric part of the composites. The clay particles in the composites are confined to the amorphous phase and do not affect the development of crystals in the polymer matrix. The clay particles can also decrease or increase the crystallisation rate by acting as nucleating agents. In
Figure 1c and
Table 2, PP has a crystallisation temperature (
Tfc) of 119 °C, whereas neat LDPE has two crystallisation peaks at 97 and 61 °C. The larger crystallisation peak of LDPE at 97 °C is attributed to primary crystallisation into thick lamellae (
Tic), whereas the smaller peak at 61 °C is attributed to secondary crystallisation into thin lamellae. The incorporation of clay into PP and LDPE has no effect on
Tfc and
Tic, and this can be attributed to the poor interactions between the modifier of clay and the polymers. PP and LDPE are non-polar in nature and form poor or no interactions with the polar silicate layers of clay in PP/clay and LDPE/clay composites.
Tfc of PP in the 80/20/0/0/0 blend is the same as that of neat PP, suggesting that an LDPE content lower than 20 wt % is not sufficient to induce a change in the peak position of
Tfc. In the previous studies [
6,
22], it was reported that the incorporation of LDPE into PP reduces the crystallisation temperature of PP. The 80/20/0/5/4 nanocomposite has the lowest
Tfc of the ternary nanocomposites, and this might be due to its higher total mass fraction of LDPE, as observed in
Table S1 taking into consideration the presence of PE in PE-
g-MA. From
Figure 1d and
Table 2, it is observed that
Tic of LDPE in the ternary composites remains the same, whereas the
Tfc of PP in the ternary composites changes. In fact,
Tfc of PP for the 80/20/0/0/4 nanocomposite is equal to that in the neat blend, whereas the values of
Tfc of the PP-
g-MA- or PE-
g-MA-containing blend composites are lower than that of the neat 80/20/0/0/0 blend. These results suggest that there is a link between the microstructure of the blend composites and the crystallisability of PP therein. The crystallisabilty of PP in the blend composites is influenced by intervention of nanoclay and dispersion of the LDPE phase. Briefly, the TEM images in
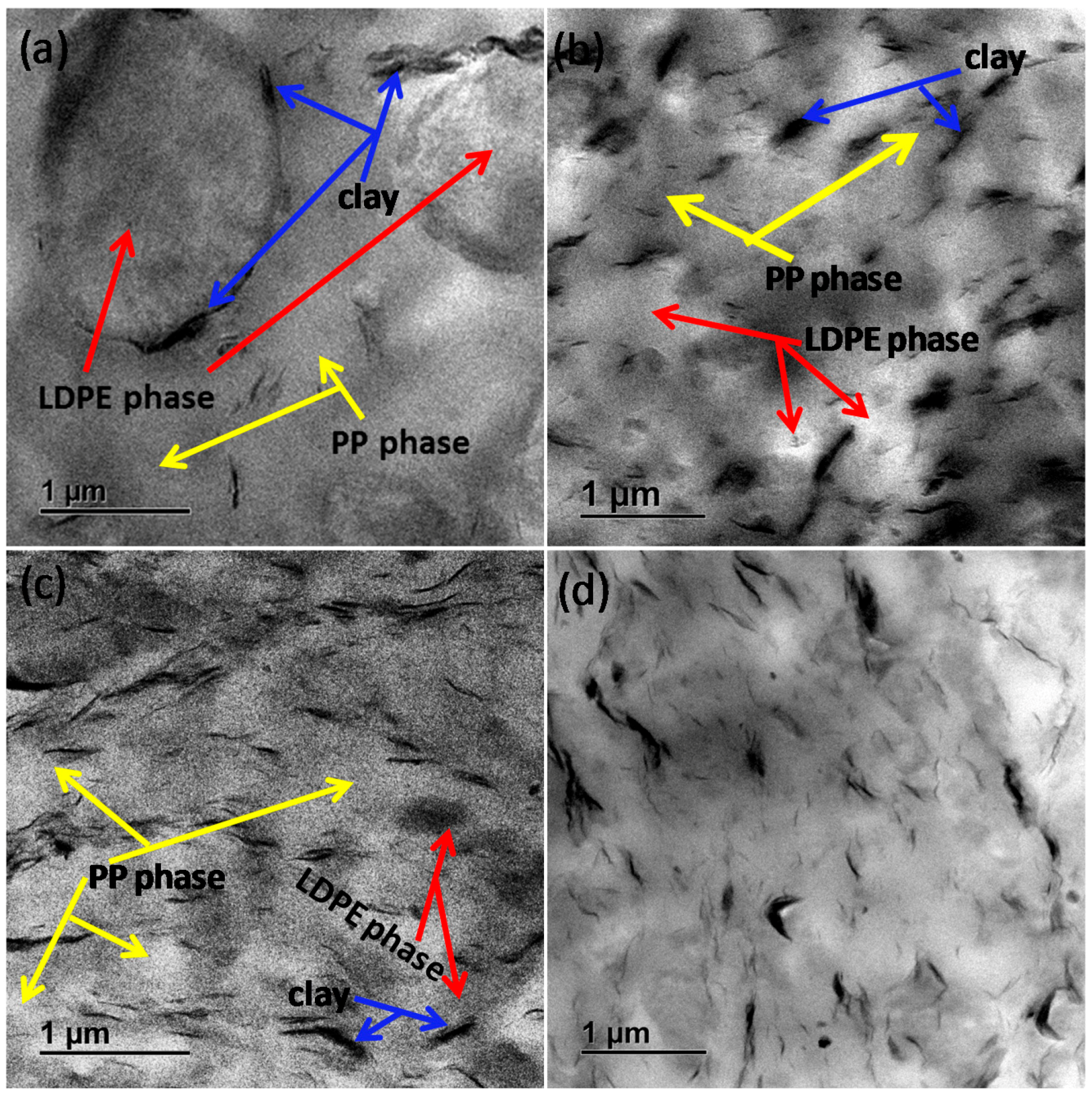
Figure 2 of the PP-
g-MA and PE-
g-MA-containing blend composites show well-distributed and dispersed clay particles in the polymeric phases, and more details can be found in our previous study [
18]. In addition, the LDPE phase is better dispersed in the PP-
g-MA and PE-
g-MA-containing blend composites due to combined interventions of clay and maleated compatibilisers. Therefore, the clay particles in the PP-
g-MA and PE-
g-MA containing blend composites serve as anti-nucleating agents, which provide enhanced hindrance of the chain mobility of PP; hence,
Tfc is lowered. Moreover, the LDPE phase in the PP-
g-MA and PE-
g-MA containing blend composites also contribute to disrupting the chain mobility of PP due to their high surface area to volume ratio. However, incorporating clay alone into the PP/LDPE blend allowed localisation of clay tactoids at the interphase [
18], and the LDPE phase of the 80/20/0/0/4 composite has a relatively lower surface area to volume ratio. Therefore, the poor dispersion of clay tactoids and LDPE phase in the 80/20/0/0/4 composite allow chain mobility of PP; hence
Tfc is equal to that in the neat blend.
In the binary composites, clay has a greater effect on
, whereas
is unchanged when clay is introduced.
decreases from 52.8 for PP to 48 for the 96/0/0/0/4 nanocomposite, suggesting that the extent of crystallisation of PP is reduced by the presence of clay. The clay particles in the binary composite form a barrier that retards the development and growth of PP crystals. Regarding the ternary composites containing the maleated compatibilisers in comparison with the neat blend,
and
of the ternary composites are lower than those of the neat blend. The simultaneous incorporation of maleated compatibilisers and clay in the ternary composites has promoted better interaction between them and PP/LDPE chains. This is evidenced in
Figure 2 by the better distribution and dispersion of clay in the 80/20/5/0/4, 80/20/0/5/4, and 80/20/5/5/4 nanocomposites. As a consequence, this disrupts the chain-folding process and crystallisability of PP and LDPE; hence,
and
are lower for the ternary composites containing maleated compatibilisers. The 80/20/0/0/4 nanocomposite has a higher
than the neat blend and other ternary nanocomposites, suggesting increased crystallisability. This is attributed to the observed localisation of clay tactoids at the PP/LDPE interface in
Figure 2a promoted by the poor interaction of the clay with either PP or LDPE [
18]. Therefore, the clay tactoids at the interface for the 80/20/0/0/4 nanocomposite, which do not interact well with the blended polymers, make it easy for the PP matrix to undergo the chain-folding process. It can be assumed that the localisation of clay at the PP/LDPE interface allows for the easier crystallisation of PP.