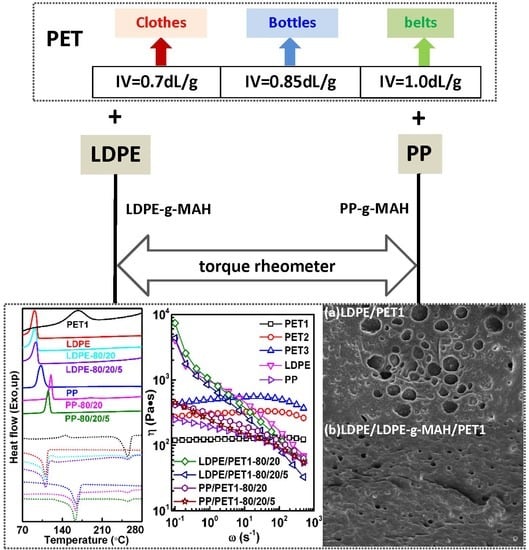
An Investigation and Comparison of the Blending of LDPE and PP with Different Intrinsic Viscosities of PET
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Measurements
3. Results and Discussion
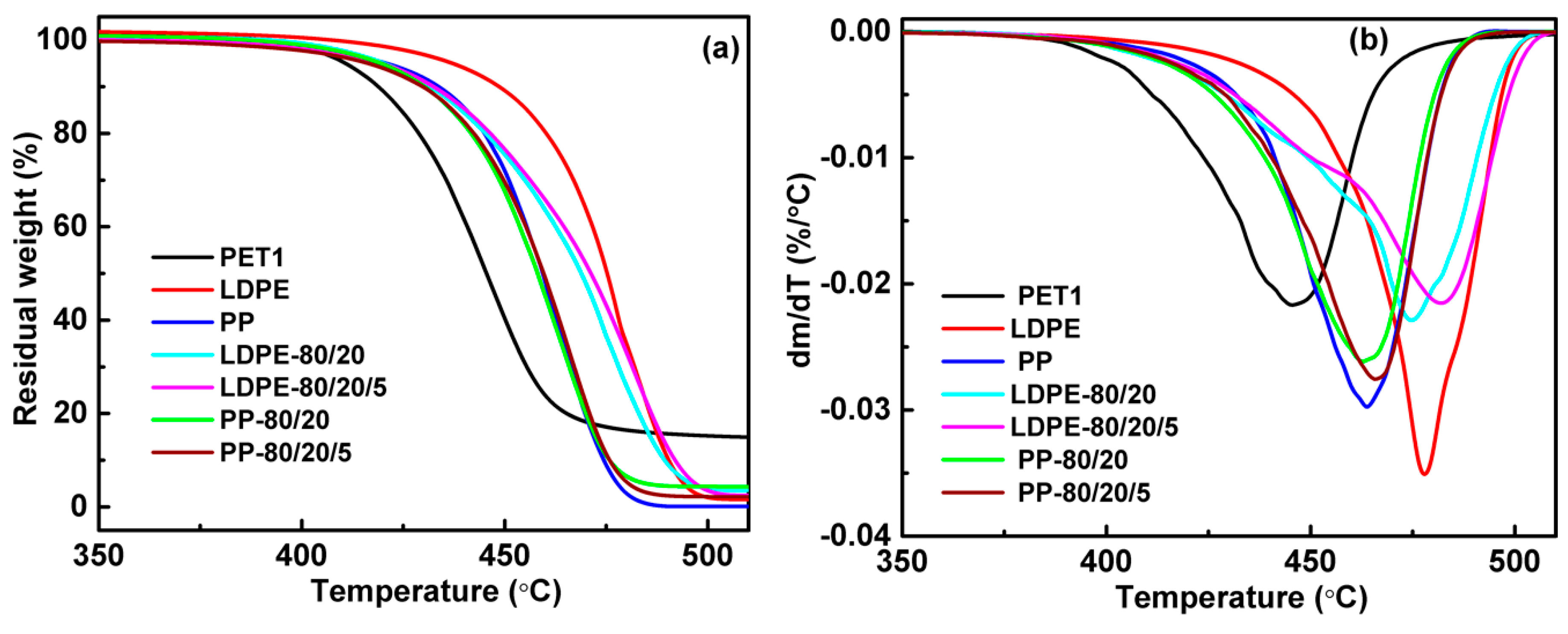
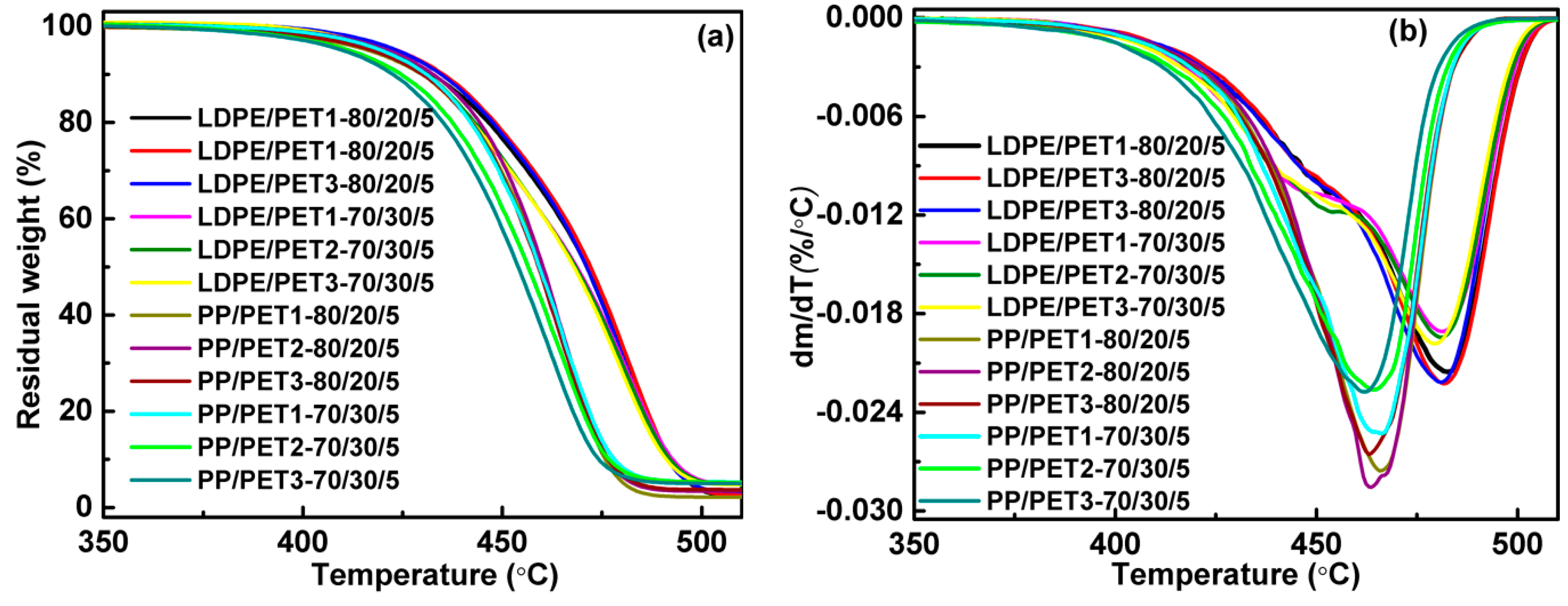
3.1. Thermal Properties
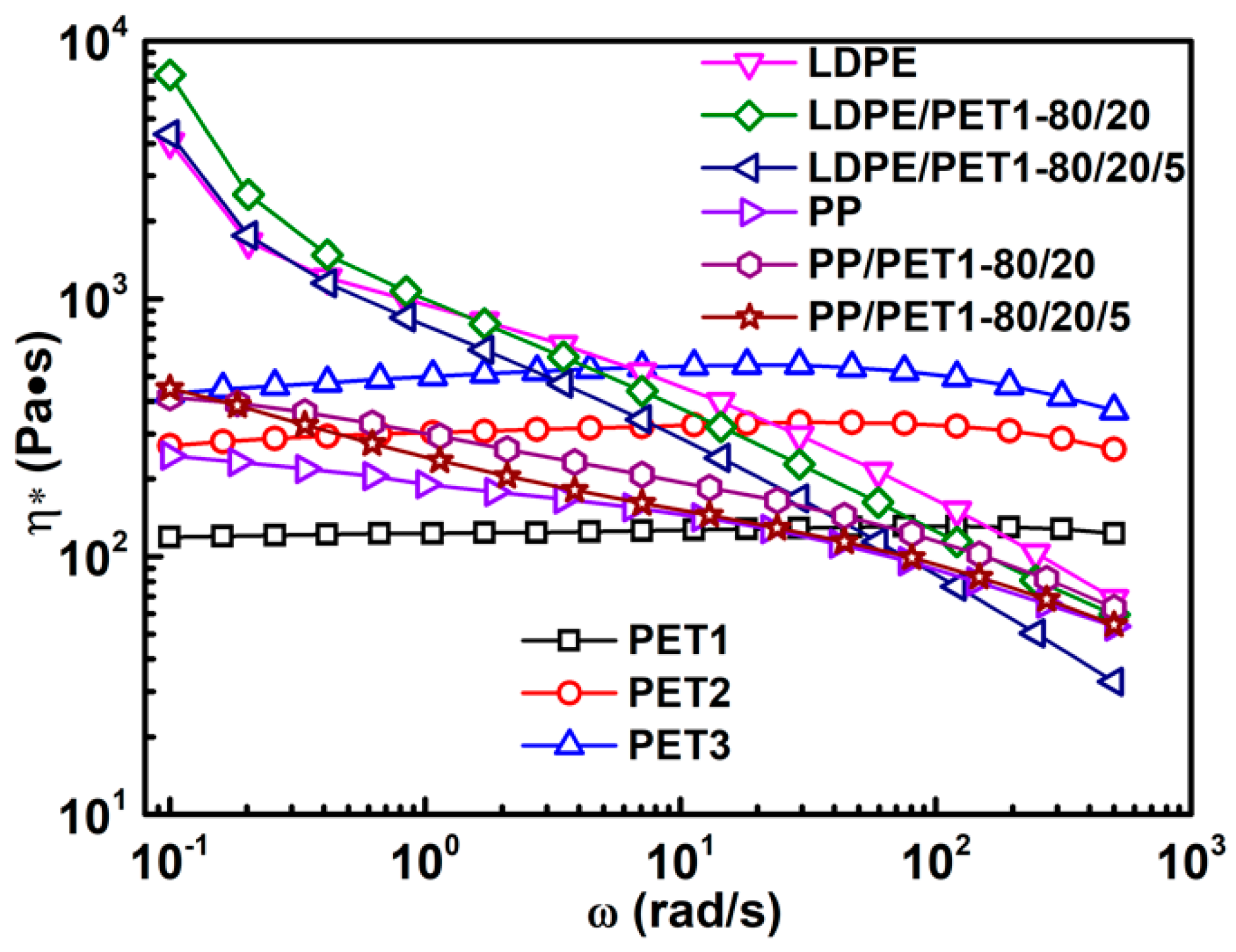
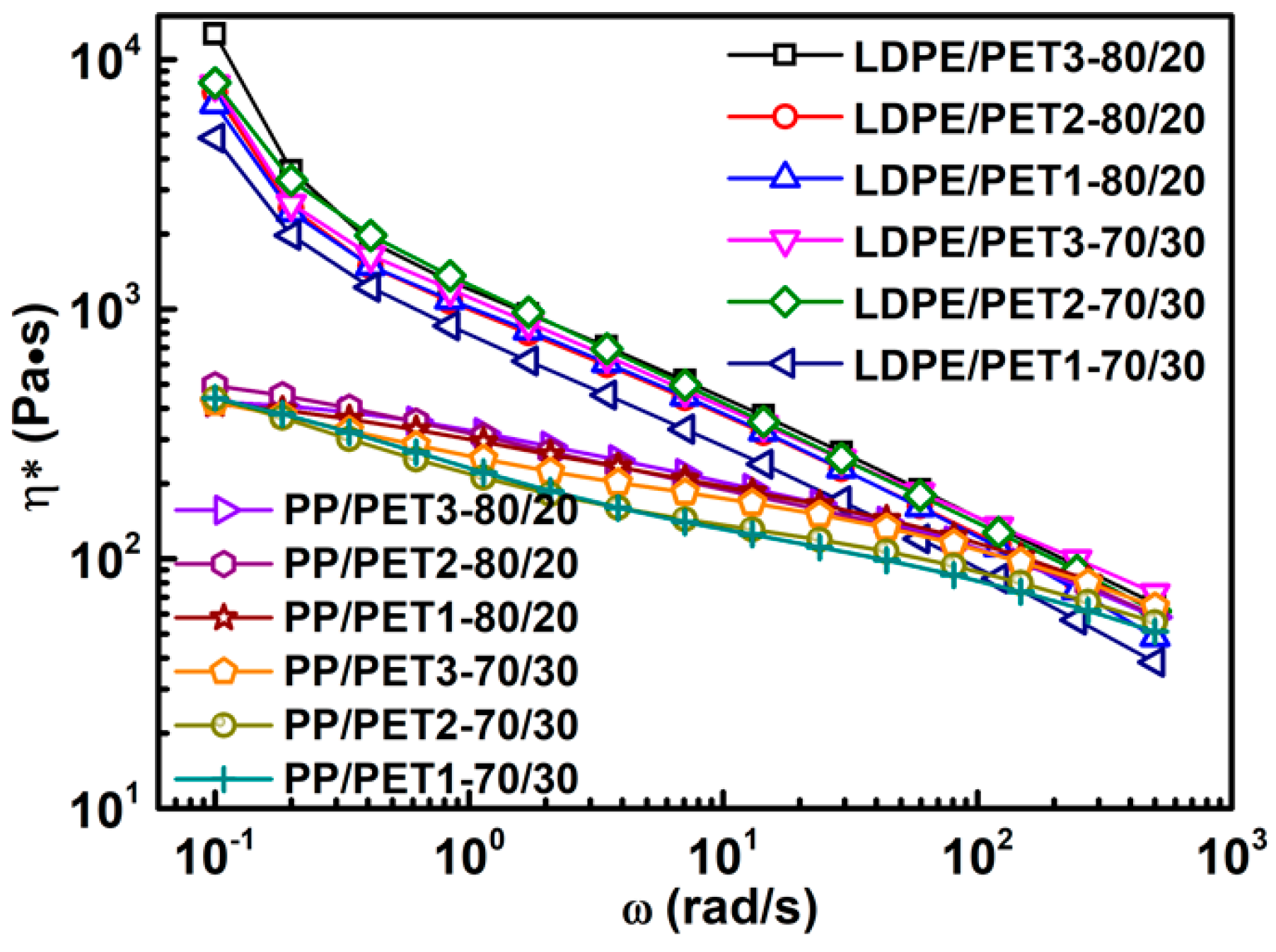
3.2. Analysis of Rheological Behavior
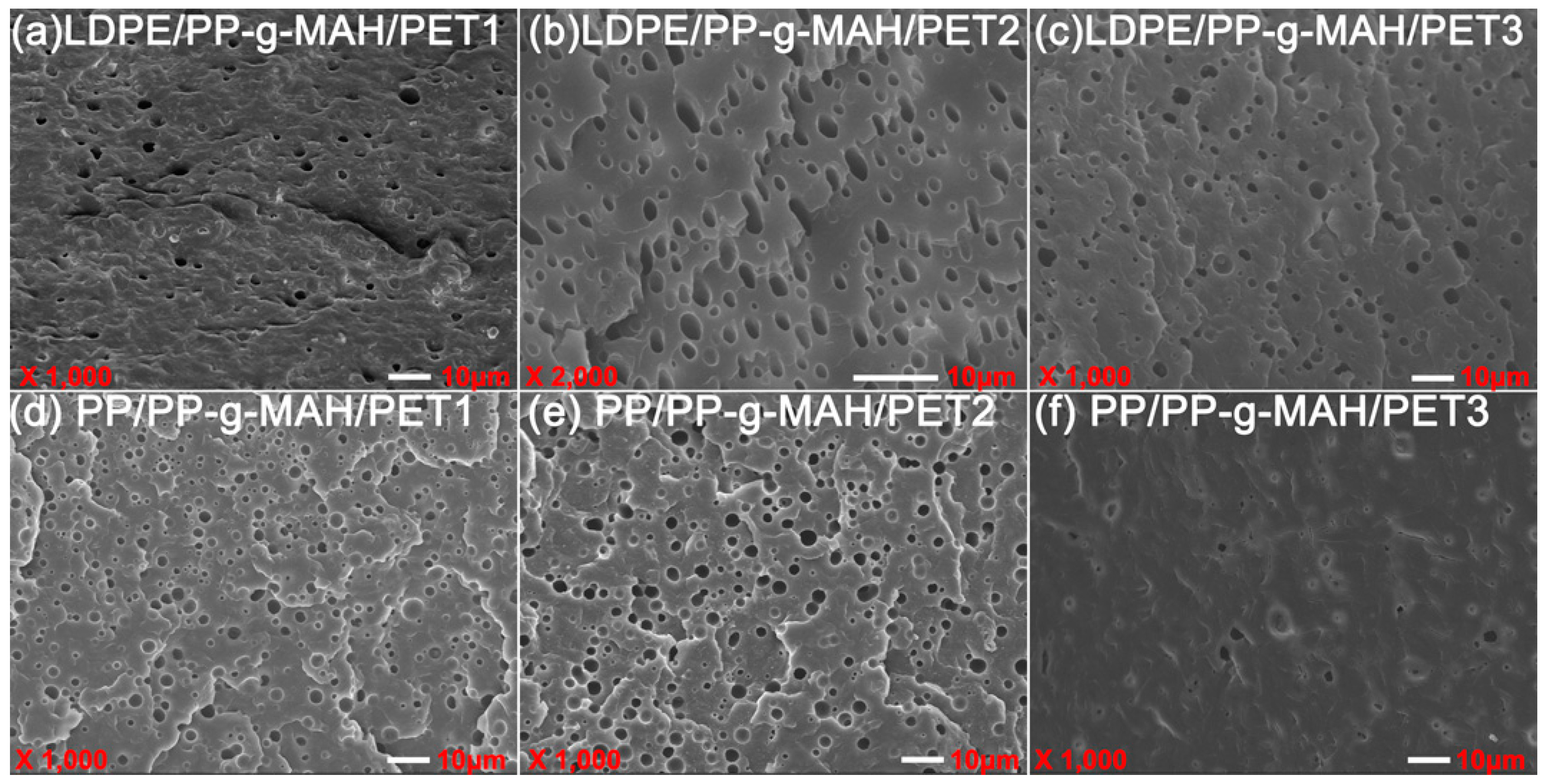
3.3. Morphological Characterization of Polymer Blends
3.4. Mechanical Properties of Polymers before and after Blending
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Chen, S.C.; Zhang, X.M.; Liu, M.; Ma, J.P.; Lu, W.Y.; Chen, W.X. Rheological characterization and thermal stability of different IV poly(ethylene terephthalate) in air and nitrogen. Int. Polym. Proc. 2016, 31, 292–300. [Google Scholar] [CrossRef]
- Raffa, P.; Coltelli, M.B.; Savi, S.; Bianchi, S.; Castelvetro, V. Chain extension and branching of poly(ethylene terephthalate) (PET) with di- and multifunctional epoxy or isocyanate additives: An experimental and modelling study. React. Funct. Polym. 2012, 72, 50–60. [Google Scholar] [CrossRef]
- Pracella, M.; Chionna, D.; Pawlak, A.; Galeski, A. Reactive mixing of PET and PET/PP blends with glycidyl methacrylate-modified styrene-b-(ethylene-co-olefin) block copolymers. J. Appl. Polym. Sci. 2005, 98, 2201–2211. [Google Scholar] [CrossRef]
- Khonakdar, H.A.; Jafari, S.H.; Mirzadeh, S.; Kalaee, M.R.; Zare, D.; Saeb, M.R. Rheology-morphology correlation in PET/PP blends: Influence of type of compatibilizer. J. Vinyl Addit. Technol. 2013, 19, 25–30. [Google Scholar] [CrossRef]
- Bettini, S.H.P.; De Mello, L.C.; Munoz, P.A.R.; Ruvolo-Filho, A. Grafting of maleic anhydride onto polypropylene, in the presence and absence of styrene, for compatibilization of poly(ethylene terephthalate)/(ethylene–propylene) blends. J. Appl. Polym. Sci. 2013, 127, 1001–1009. [Google Scholar] [CrossRef]
- Zhang, X.; Li, B.; Wang, K.; Zhang, Q.; Fu, Q. The effect of interfacial adhesion on the impact strength of immiscible PP/PETG blends compatibilized with triblock copolymers. Polymer 2009, 50, 4737–4744. [Google Scholar] [CrossRef]
- Li, H.; Zhang, X.M.; Zhu, S.Y.; Chen, W.X.; Feng, L.F. Preparation of polypropylene and polystyrene with -NCO and -NH2 functional groups and their applications in polypropylene/polystyrene blends. Polym. Eng. Sci. 2015, 55, 614–623. [Google Scholar] [CrossRef]
- Marcincin, A.; Kormendy, E.; Hricova, M.; Rusnak, A.; Aneja, A.P. Rheological behavior of polyester blend and mechanical properties of the polypropylene-polyester blend fibers. J. Appl. Polym. Sci. 2006, 102, 4222–4227. [Google Scholar] [CrossRef]
- Pracella, M.; Pazzagli, F.; Galeski, A. Reactive compatibilization and properties of recycled poly(ethylene terephthalate)/polyethylene blends. Polym. Bull. 2002, 48, 67–74. [Google Scholar] [CrossRef]
- Wang, C.; Dai, W.; Zhang, Z.; Mai, K. Effect of blending way on b-crystallization tendency of compatibilized b-nucleated isotactic polypropylene/poly(ethylene terephthalate) blends. J. Therm. Anal. Calorim. 2013, 111, 1585–1593. [Google Scholar] [CrossRef]
- Esmizadeh, E.; Moghri, M.; Saeb, M.R.; Mohsen, N.M.; Nobakht, N.; Bende, N.P. Application of Taguchi approach in describing the mechanical properties and thermal decomposition behavior of poly(vinyl chloride)/clay nanocomposites: Highlighting the role of organic modifier. J. Vinyl Addit. Technol. 2016, 22, 182–190. [Google Scholar] [CrossRef]
- Li, S.C.; Lu, L.N.; Zeng, W. Thermostimulative shape-memory effect of reactive compatibilized high-density polyethylene/poly(ethylene terephthalate) blends by an ethylene-butyl acrylate-glycidyl methacrylate terpolymer. J. Appl. Polym. Sci. 2009, 112, 3341–3346. [Google Scholar] [CrossRef]
- Nonato, R.C.; Bonse, B.C. A study of PP/PET composites: Factorial design, mechanical and thermal properties. Polym. Test. 2016, 56, 167–173. [Google Scholar] [CrossRef]
- Li, H.; Xie, X.M. Morphology development and superior mechanical properties of PP/PA6/SEBS ternary blends compatibilized by using a highly efficient multi-phase compatibilizer. Polymer 2017, 108, 1–10. [Google Scholar] [CrossRef]
- Jazani, O.M.; Rastin, H.; Formela, K.; Hejna, A.; Shahbazi, M.; Farkiani, B.; Saeb, M.R. An investigation on the role of GMA grafting degree on the efficiency of PET/PP-g-GMA reactive blending: Morphology and mechanical properties. Polym. Bull. 2017, 74, 4483–4497. [Google Scholar] [CrossRef]
- Mantia, F.P.L.; Ceraulo, M.; Giacchi, G.; Mistretta, M.C.; Botta, L. Effect of a compatibilizer on the morphology and properties of polypropylene/poly(ethylene therephthalate) spun fibers. Polymers 2017, 9, 47. [Google Scholar] [CrossRef]
- Si, X.; Guo, L.; Wang, Y.; Lau, K. Preparation and study of polypropylene/poly(ethylene terephthalate) composite fibers. Compos. Sci. Technol. 2008, 68, 2943–2947. [Google Scholar] [CrossRef]
- Lei, Y.; Wu, Q.; Zhang, Q. Morphology and properties of microfibrillar composites based on recycled poly(ethylene terephthalate) and high density polyethylene. Compos. Part A Appl. Sci. Manuf. 2009, 40, 904–912. [Google Scholar] [CrossRef]
- Raffa, P.; Coltelli, M.; Castelvetro, V. Expanding the application field of post-consumer poly(ethylene terephthalate) through structural modification by reactive blending. J. Appl. Polym. Sci. 2014, 131, 5829–5836. [Google Scholar] [CrossRef]
- Yousfi, M.; Soulestin, J.; Vergnes, B.; Lacrampe, M.F.; Krawczak, P. Morphology and mechanical properties of PET/PE blends compatibilized by nanoclays: Effect of thermal stability of nanofiller organic modifier. J. Appl. Polym. Sci. 2013, 128, 2766–2778. [Google Scholar] [CrossRef]
- Zhu, S.Y.; Li, H.; Zhang, X.M.; Chen, W.X.; Feng, L.F. Synthesis, copolymer composition, and rheological behavior of functionalized polystyrene with isocyanate and amine side groups. Des. Monomers Polym. 2015, 132, 232–241. [Google Scholar] [CrossRef]
- Sun, M.H.; Zhang, X.M.; Chen, W.X.; Feng, L.F. Surface functionalization of polypropylene-bearing isocyanate groups in solid state and their cyclotrimerization with diisocyanates. J. Appl. Polym. Sci. 2015, 132, 42186. [Google Scholar] [CrossRef]
- Sun, M.H.; Zhang, X.M.; Chen, W.X.; Feng, L.F. The effect of tunable modification parameters on the structure and properties of polypropylene with cyclotrimerization surface of isocyanates. J. Appl. Polym. Sci. 2016, 133, 43327. [Google Scholar] [CrossRef]
- Zhang, X.M.; Sun, M.H.; Chen, W.X. Synergistic effects of silica nanoparticles and reactive compatibilizer on the compatibilization of polystyrene/polyamide 6 blends. Polym. Eng. Sci. 2017, 57, 24511. [Google Scholar] [CrossRef]
- Chiu, F.C.; Yen, H.Z.; Lee, C.E. Characterization of PP/HDPE blend-based nanocomposites using different maleated polyolefins as compatibilizers. Polym. Test. 2010, 29, 397–406. [Google Scholar] [CrossRef]
- Tao, Y.; Mai, K. Non-isothermal crystallization and melting behavior of compatibilized polypropylene/recycled poly(ethylene terephthalate) blends. Eur. Polym. J. 2007, 43, 3538–3549. [Google Scholar] [CrossRef]
- Yee, M.; Souza, A.M.C.; Valera, T.S.; Demarquette, N.R. Stress relaxation behavior of PMMA/PS polymer blends. Rheol. Acta 2009, 48, 527–541. [Google Scholar] [CrossRef]
- Souza, A.M.C.; Calvao, P.S.; Demarquette, N.R. Linear viscoelastic behavior of compatibilized PMMA/PP blends. J. Appl. Polym. Sci. 2013, 129, 1280–1289. [Google Scholar] [CrossRef]
- De Souza, A.M.C.; Caldeira, C.B. An investigation on recycled PET/PP and recycled PET/PP-EP compatibilized blends: Rheological, morphological, and mechanical properties. J. Appl. Polym. Sci. 2015, 132, 41892. [Google Scholar] [CrossRef]
- Hale, W.; Keskkula, H.; Paul, D.R. Compatibilization of PBT/ABS blends by methyl methacrylate-glycidyl methacrylate-ethyl acrylate terpolymers. Polymer 1999, 40, 365–377. [Google Scholar] [CrossRef]
- Sanchez, S.A.; Calderas, F.; Manero, O. Influence of maleic anhydride grafting on the rheological properties of polyethylene terephthalate–styrene butadiene blends. Polymer 2001, 42, 7335–7342. [Google Scholar] [CrossRef]
- Akbari, M.; Zadhoush, A.; Haghighat, M. PET/PP blending by using PP-g-MA synthesized by solid phase. J. Appl. Polym. Sci. 2007, 104, 3986–3993. [Google Scholar] [CrossRef]
- Yousfi, M.; Lepretre, S.; Soulestin, J.; Vergnes, B.; Lacrampe, M.F.; Krawczak, P. Processing-induced degradation of nanoclay organic modifier in melt-mixed PET/PE blends during twin screw extrusion at industrial scale: Effect on morphology and mechanical behavior. J. Appl. Polym. Sci. 2013, 131, 1001–1007. [Google Scholar] [CrossRef]
- Tavanaie, M.A.; Shoushtari, A.M.; Goharpey, F.; Mojtahedi, M.R. Matrix-fibril morphology development of polypropylene/poly(butylenesterephthalate) blend fibers at different zones of melt spinning process and its relation to mechanical properties. Fiber Polym. 2013, 14, 396–404. [Google Scholar] [CrossRef]





| PET | [η] (dL/g) | [COOH] (mol/t) | H2O (wt %) |
|---|---|---|---|
| PET1 | 0.70 | 22 | 0.35 |
| PET2 | 0.85 | 18 | 0.26 |
| PET3 | 1.00 | 15 | 0.10 |
| No. | PET1 | PET2 | PET3 | PP | PP-g-MAH | LDPE | LDPE-g-MAH |
|---|---|---|---|---|---|---|---|
| 1 | 20 | 80 | |||||
| 2 | 20 | 80 | |||||
| 3 | 20 | 80 | |||||
| 4 | 30 | 70 | |||||
| 5 | 30 | 70 | |||||
| 6 | 30 | 70 | |||||
| 7 | 20 | 80 | 5 | ||||
| 8 | 20 | 80 | 5 | ||||
| 9 | 20 | 80 | 5 | ||||
| 10 | 30 | 70 | 5 | ||||
| 11 | 30 | 70 | 5 | ||||
| 12 | 30 | 70 | 5 | ||||
| 13 | 20 | 80 | |||||
| 14 | 20 | 80 | |||||
| 15 | 20 | 80 | |||||
| 16 | 30 | 70 | |||||
| 17 | 30 | 70 | |||||
| 18 | 30 | 70 | |||||
| 19 | 20 | 80 | 5 | ||||
| 20 | 20 | 80 | 5 | ||||
| 21 | 20 | 80 | 5 | ||||
| 22 | 30 | 70 | 5 | ||||
| 23 | 30 | 70 | 5 | ||||
| 24 | 30 | 70 | 5 |
| Samples | Tc1 (°C) | Tm1 (°C) | Tm2 (°C) |
|---|---|---|---|
| LDPE | 91.09 | 110.12 | — |
| PP | 102.51 | 165.33 | — |
| PET1 | 167.69 | — | 254.33 |
| LDPE/PET1-80/20 | 91.85 | 110.68 | 255.67 |
| LDPE/PET2-80/20 | 92.96 | 110.58 | 254.81 |
| LDPE/PET3-80/20 | 93.43 | 110.23 | 253.32 |
| LDPE/PET1-70/30 | 92.49 | 110.77 | 2557.5 |
| LDPE/PET2-70/30 | 93.44 | 109.68 | 254.34 |
| LDPE/PET3-70/30 | 94.44 | 109.86 | 252.88 |
| PP/PET1-80/20 | 119.61 | 163.31 | 255.28 |
| PP/PET2-80/20 | 119.14 | 162.96 | 255.25 |
| PP/PET3-80/20 | 119.21 | 163.33 | 254.87 |
| PP/PET1-70/30 | 119.13 | 163.70 | 255.75 |
| PP/PET2-70/30 | 118.67 | 162.96 | 254.83 |
| PP/PET3-70/30 | 118.65 | 164.42 | 254.81 |
| LDPE/PET1-80/20/5 | 93.61 | 110.63 | 255.18 |
| LDPE/PET2-80/20/5 | 91.39 | 111.66 | 255.96 |
| LDPE/PET3-80/20/5 | 92.13 | 111.13 | 253.88 |
| LDPE/PET1-70/30/5 | 91.78 | 111.40 | 256.22 |
| LDPE/PET2-70/30/5 | 92.32 | 111.63 | 254.93 |
| LDPE/PET3-70/30/5 | 91.23 | 111.66 | 254.66 |
| PP/PET1-80/20/5 | 114.91 | 162.62 | 255.96 |
| PP/PET2-80/20/5 | 115.61 | 162.86 | 254.93 |
| PP/PET3-80/20/5 | 115.07 | 163.39 | 254.66 |
| PP/PET1-70/30/5 | 114.36 | 162.09 | 255.44 |
| PP/PET2-70/30/5 | 115.45 | 162.87 | 255.71 |
| PP/PET3-70/30/5 | 116.00 | 162.09 | 253.88 |
| Blend | 0/100 | 80/20 | 70/30 | 80/20/5 | 70/30/5 | 100/0 | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| T5% (°C) | Tcmax (°C) | T5% (°C) | Tcmax (°C) | T5% (°C) | Tcmax (°C) | T5% (°C) | Tcmax (°C) | T5% (°C) | Tcmax (°C) | T5% (°C) | Tcmax (°C) | |
| LDPE/PET1 | 410.8 | 444.3 | 419.7 | 473.4 | 420.5 | 478.7 | 419.6 | 482.8 | 419.4 | 481.7 | 437.5 | 477.7 |
| LDPE/PET2 | 407.5 | 445.7 | 421.3 | 475.9 | 416.3 | 476.0 | 423.2 | 481.7 | 420.3 | 480.9 | - | - |
| LDPE/PET3 | 407.2 | 447.7 | 422.5 | 480.3 | 416.4 | 475.8 | 423.6 | 480.8 | 419.5 | 479.2 | - | - |
| PP/PET1 | 410.8 | 444.3 | 419.5 | 463.5 | 420.3 | 463.3 | 416.3 | 465.9 | 419.7 | 465.7 | 420.8 | 461.3 |
| PP/PET2 | 407.5 | 445.7 | 417.1 | 463.4 | 414.3 | 461.4 | 421.1 | 463.3 | 411.5 | 465.0 | - | - |
| PP/PET3 | 407.2 | 447.7 | 420.7 | 463.2 | 412.1 | 460.5 | 417.5 | 462.9 | 409.9 | 461.8 | - | - |
| Blend | 0/100 (%) | 80/20 (%) | 70/30 (%) | 80/20/5 (%) | 70/30/5 (%) | 100/0 (%) |
|---|---|---|---|---|---|---|
| LDPE/PET1 | 8.55 | 51.13 | 25.51 | 67.45 | 37.52 | 223.45 |
| LDPE/PET2 | 8.45 | 44.67 | 23.50 | 78.25 | 41.47 | - |
| LDPE/PET3 | 6.41 | 44.16 | 22.21 | 148.54 | 58.97 | - |
| PP/PET1 | 8.55 | 15.1 | 11.7 | 38.5 | 30.5 | 428 |
| PP/PET2 | 8.45 | 14.7 | 11.5 | 34.8 | 28.0 | - |
| PP/PET3 | 6.41 | 13.7 | 10.2 | 31.3 | 24.3 | - |
| Blend | 0/100 MPa | 80/20 MPa | 70/30 MPa | 80/20/5 MPa | 70/30/5 MPa | 100/0 MPa |
|---|---|---|---|---|---|---|
| LDPE/PET1 | 26.0 | 16.71 | 23.52 | 14.32 | 18.25 | 12.31 |
| LDPE/PET2 | 28.8 | 15.62 | 17.25 | 13.88 | 12.74 | - |
| LDPE/PET3 | 37.5 | 14.45 | 17.01 | 12.90 | 12.24 | - |
| PP/PET1 | 26.0 | 17.1 | 24.2 | 11.3 | 12.5 | 35.1 |
| PP/PET2 | 28.8 | 20.6 | 25.0 | 11.9 | 12.41 | - |
| PP/PET3 | 37.5 | 24.4 | 25.2 | 12.0 | 12.2 | - |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, S.-C.; Zhang, L.-H.; Zhang, G.; Zhong, G.-C.; Li, J.; Zhang, X.-M.; Chen, W.-X. An Investigation and Comparison of the Blending of LDPE and PP with Different Intrinsic Viscosities of PET. Polymers 2018, 10, 147. https://doi.org/10.3390/polym10020147
Chen S-C, Zhang L-H, Zhang G, Zhong G-C, Li J, Zhang X-M, Chen W-X. An Investigation and Comparison of the Blending of LDPE and PP with Different Intrinsic Viscosities of PET. Polymers. 2018; 10(2):147. https://doi.org/10.3390/polym10020147
Chicago/Turabian StyleChen, Shi-Chang, Li-Hao Zhang, Guo Zhang, Guo-Cai Zhong, Jian Li, Xian-Ming Zhang, and Wen-Xing Chen. 2018. "An Investigation and Comparison of the Blending of LDPE and PP with Different Intrinsic Viscosities of PET" Polymers 10, no. 2: 147. https://doi.org/10.3390/polym10020147
APA StyleChen, S.-C., Zhang, L.-H., Zhang, G., Zhong, G.-C., Li, J., Zhang, X.-M., & Chen, W.-X. (2018). An Investigation and Comparison of the Blending of LDPE and PP with Different Intrinsic Viscosities of PET. Polymers, 10(2), 147. https://doi.org/10.3390/polym10020147