1. Introduction
Air pollution is a serious environmental issue that is widely discussed around the globe, especially in the developing countries. The article published by the Lancet Respiratory Medicine in 2012 reported that 12.6 million deaths recorded worldwide were related to environmental pollution and, out of that number, 8.2 million were caused by air pollution [
1]. The study had also shown that indoor air pollution was the major factor contributing to the adverse health problems of humans [
1]. A poor level of indoor air quality is likely to cause sick building syndrome that affects both productivity and personal health. One of the effective ways to improve indoor air quality is to supply oxygen-enriched air to enhance air freshness [
2]. Additionally, oxygen-enriched air is highly sought after in medical applications for hypoxemic patients and internal combustion engines to reduce the environment pollution that arises from unburned hydrocarbons [
3,
4].
Currently, two conventional techniques that are commonly used in oxygen gas production are pressure swing absorption (PSA) and cryogenic distillation. These two techniques are commercially utilized in industrial applications to produce large volumes of high purity gases. Membrane technology, on other hand, is an emerging technology which is able to produce gases in relatively good purity at a moderate production volume. The main advantages of membrane technology compared to conventional methods are that membrane technology requires less setting up space and is less energy intensive [
5].
Generally, membranes act as a barrier for the gas separation whereby its characteristics and properties play an important role in the separation performance [
6]. Nevertheless, the gas separation performance is always limited by the selectivity and permeability trade-off as shown in the Robeson upper bound [
7,
8]. Permeability describes the ability of membrane to allow gas molecules to diffuse through the matrix and is governed by the Fick’s diffusion law [
9]. Selectivity meanwhile describes the ratio of the permeance of species A with respect to species B. To date, most of the works carried out are focused on the development of advanced polymeric-based membranes that demonstrate superior permeability and selectivity in lab-scale applications [
10].
One of the easiest and simplest ways to improve the existing performance of polymeric-based membranes is via surface coating. Polydimethylsiloxane (PDMS) and poly(ether block amide) (PEBAX) are widely used as coating materials for membrane to minimize not only its surface defects but also improve its separation performance. Studies showed that PDMS coating is able to repair membrane surface defects by eliminating the non-selective membrane pores and to enhance gas pair selectivity [
11,
12]. Wahab et al. [
13] and Moaddeb and Koros [
14] reported the potential use of PDMS-coated membranes to improve the gas separation process, especially in CO
2 separation for combustion. Unlike PDMS, PEBAX contains multiblock copolymers which consist of rigid polyamide chains separated by flexible polyether fragments. Previous work showed that the good species separation performance of PEBAX-coated membrane is attributed to its soft block of polyether fragments that allows gas molecules to permeate easily [
15,
16]. The hard polyamide block meanwhile provides good mechanical strength for the membrane [
16]. Because of this, PEBAX was applied as a coating material on the membrane to evaluate its performance in gas separation. Kim et al. [
15] reported that PEBAX-coated polyacrylonitrile (PAN) membrane was able to improve the sour gases (SO
2 and CO
2) separation performance from nitrogen. Liu et al. [
16] on the other hand found that polysulfone (PSF) flat sheet membranes coated with PEBAX via dip coating method were able to improve the permselectivity of CO
2/N
2.
In this study, two different coating materials i.e., PDMS and PEBAX were used to improve the separation properties of PSF hollow fiber membranes particularly for oxygen/nitrogen separation. The outer surfaces of membranes were coated with polymers at different concentrations followed by instrumental characterization before proceeding to gas separation performance evaluation.
2. Materials and Methods
2.1. Materials
The main material used for hollow fiber membrane fabrication was commercial PSF with the trade name UDEL-3500 (Amocco Chemicals, GA, USA). The solvent (N,N-dimethylacetamide, DMAc) and co-solvent (ethanol, EtOH) used to dissolve PSF pellets were obtained from Merck (Darmstadt, Germany), Tetrahydrofuran (THF) with purity >99% purchased from QReC (Selangor, Malaysia) was also used as co-solvent during dope solution preparation. The PDMS and PEBAX coating materials were purchased from Sigma-Aldrich, St. Louis, MO, USA and Arkema, PA, USA, respectively. They were used to form a selective layer on the outer surface of hollow fiber membrane. N-hexane obtained from Merck (Darmstadt, Germany) was used to prepare PDMS coating solution.
2.2. Fabrication of Hollow Fiber Membrane
The dope solution employed for the hollow fiber membrane fabrication was composed of 30 wt % PSF, 30 wt % DMAc, 30 wt % EtOH, and 10 wt % THF. Before dissolving in the solvent, the PSF pellets were dried in vacuum oven for one day at 70 °C to completely remove the moisture. The PSF pellets were then slowly added into the solution containing DMAc, EtOH, and THF under mechanical stirring. After the PSF pellets were completely dissolved, the mixture was continuously stirred for another 24 h in order to obtain a homogenous solution. The dope solution was then degassed in ultrasonic bath for 4 h to remove the air bubbles trapped in the solution.
Dry-jet wet phase inversion method was employed in this study to fabricate the PSF hollow fiber membrane and detailed description of the method can be found elsewhere [
17,
18]. The spinning machine is schematically shown in
Figure 1 and the detailed spinning parameters are shown in
Table 1. The dope solution was first transferred to a stainless-steel dope reservoir prior to the fabrication process. A gear pump was used to deliver the dope solution from the reservoir to the spinneret, passing through the annular spinneret. Bore fluid composed of distilled water would flow through the center of the spinneret to instigate the solvent/non-solvent exchange between the dope solution and bore fluid, forming lumen as shown in the insertion in
Figure 1.
Nascent hollow fiber membrane was formed when it was contacted with large amount of water in the coagulation bath. The hollow fiber was then guided manually to the washing bath before being collected by wind-up drum. The hollow fibers collected from the drum were subjected to two-day immersion in water bath in order to completely remove the solvent residual. Lastly, the hollow fiber membranes were dried in the vacuum oven at 70 °C for one day in order to remove the moisture/residual solvent content. As the solvents from the dope solution are considered hazardous and will eventually be discharged into the drain, post-treatment using solvent absorbents is recommended. However, as our work only dealt with lab-scale membrane fabrication, there is no such treatment process implemented for the time being [
19]. For large scale membrane fabrication process by phase inversion method, wastewater treatment process is required in order to treat the effluent containing solvents before being discharged to environment [
20].
2.3. Coating of Membrane Surface
In this study, the coating solutions containing either PDMS or PEBAX were prepared at three different concentrations, i.e., 1, 3, and 5 wt % and were used to form a thin coating layer on the outer surface of hollow fiber membrane. To prepare the PDMS coating solution, the PDMS base was first mixed with the predetermined volume of n-hexane solution to obtain the desired weight percentage. The solution was continuously stirred for 2 h followed by 4 h sonication to remove microbubbles trapped in the solution. The dried PSF membranes were then dipped in the PDMS coating solution for 2 min before subjecting to heat treatment (curing step) in an oven at 70 °C for 4 h. The procedures were repeated five times in order to obtain good PDMS coating layer.
To prepare the PEBAX coating solution, PEBAX with specific quantity was first dissolved in a solution composed of distilled water and ethanol at volume ratio of 30:70. The mixture was then stirred at room temperature for 2 h prior before proceeding to the sonication process for 4 h. Similar to PDMS coating technique, the hollow fiber membranes were dipped into the PEBAX solution for 2 min followed by drying at 70 °C for 4 h. Similar to the PDMS coating protocol, the procedures were repeated five times in order to obtain good PEBAX coating layer. It must be noted that during the curing process, the PEBAX-coated fibers had to be kept separately to avoid them from sticking.
Table 2 shows the types of the hollow fiber membranes coated with different materials at different concentrations.
2.4. Membrane Characterization
The cross sectional and morphology of the hollow fiber membrane were carefully examined by scanning electron microscope (Hitachi, Tokyo, Japan, S3400N). Prior to analysis, the hollow fiber membranes were cryogenically cracked using liquid nitrogen in order to obtain a clear morphology of the membrane without defect. The samples were then coated with a layer of gold using sputter coating machine (Emitech, East Sussex, UK, SC7620) to improve the surface conductivity [
21]. Energy dispersive X-ray (EDX) spectrometer was used to analyze the elements present on the membrane outer surface. The element that can be used to confirm the existence of the coating layer were silicon (Si) for PDMS coating and nitrogen (N) for PEBAX coating. For each membrane sample, the membrane characterization was repeated five times to yield the average results with standard deviation.
2.5. Gas Permeation Study
The gas permeation setup is schematically illustrated in
Figure 2. In this study, the gases used for experiment were pure oxygen and nitrogen gas supplied by MegaMount Industrial Gas, Johor, Malaysia with purity >99.99%. Five units of hollow fiber membranes with the length of 23 cm were bundled and placed within the stainless steel membrane housing. The hollow fiber membranes were sealed with dead end manner using epoxy resin (Loctite, Düsseldorf, Germany). The flow arrangement in this study was shell-side feed where the feed gas from the cylinder would diffuse through the outer surface of the hollow fiber membranes and the permeated gas will pass through the lumen of fibers. The permeated gas is connected to a soap bubble flow meter in which the gas permeance could be determined as
where
PA/
l is the gas permeance (GPU) (Note: 1 GPU = 10
−6 cm
3 (STP)/cm
2 cm Hg),
Q is the volumetric flowrate of gas diffuse across the membrane (cm
3/s, STP),
A is the effective membrane area (cm
2), Δ
P is the transmembrane pressure (cm Hg), and
T is the temperature. The experiments were carried out at room temperature (28 °C) with constant feed pressure of 5 bar. For each membrane sample, the experiments were repeated five times to yield the average results with standard deviation. The membrane selectivity,
αA/B describing the ratio of gas pair permeability can be calculated by the pressure normalized flux ratio of oxygen over nitrogen gas.
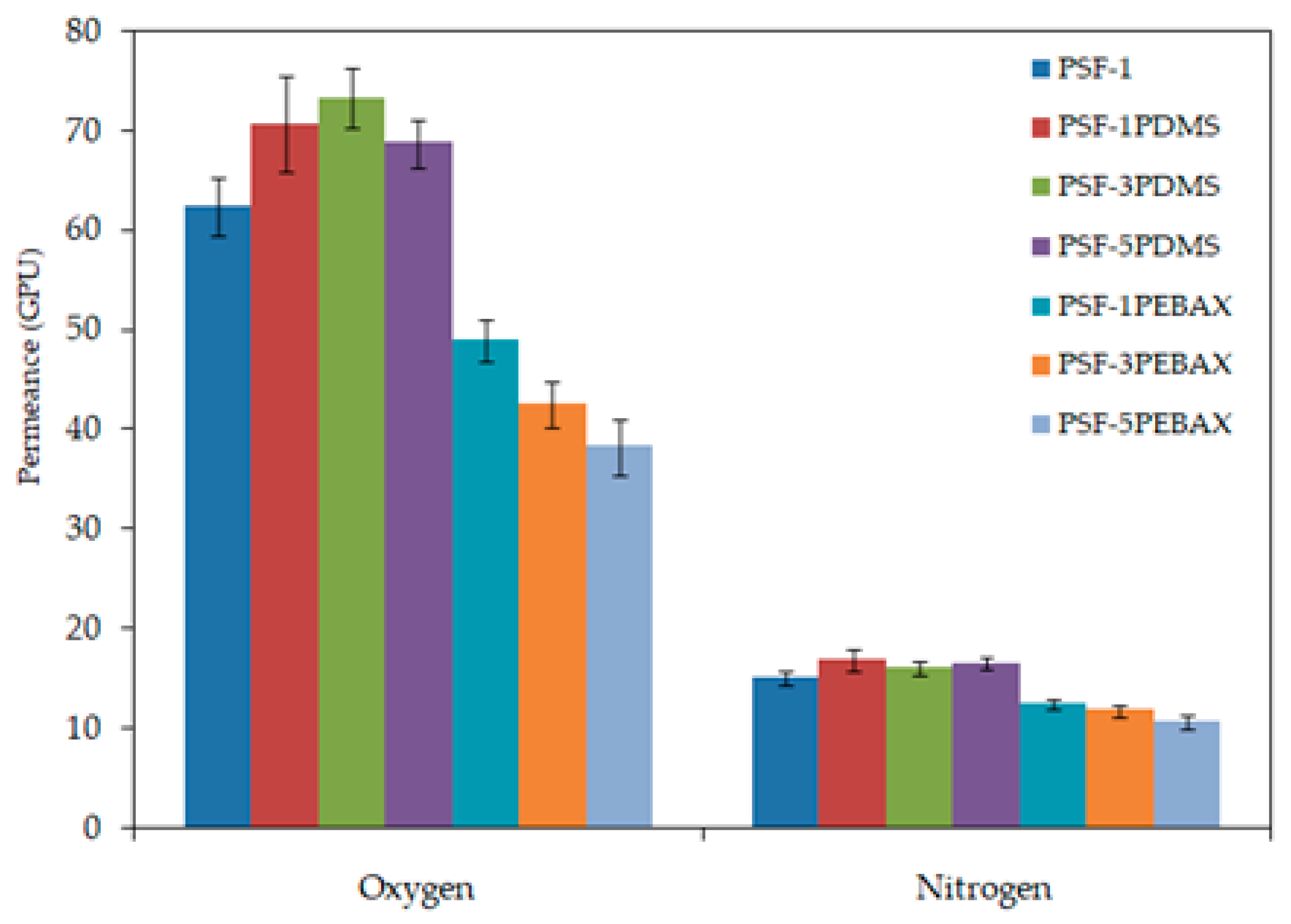
4. Conclusions
In this study, the surface properties of the PSF hollow fiber membranes were modified by subjecting the membranes to dip-coating process using either PDMS or PEBAX at different concentrations. Results showed that the membranes coated with PDMS exhibited better permeance and selectivity in oxygen/nitrogen separation process in comparison to the membranes coated with PEBAX. Upon coating with 3 wt % PDMS, the membrane exhibited the best performance, showing permeance of 18.31 and 4.01 GPU for oxygen and nitrogen gas, respectively and recording oxygen/nitrogen selectivity of 4.56. Meanwhile, the best performing PEBAX-coated membrane (1 wt % PEBAX) only showed 12.23 and 3.11 GPU for oxygen and nitrogen gas, respectively and selectivity of 3.94. Although previous research works have shown that the PEBAX coating layer is more selective compared to the PDMS coating layer, contradictory results were obtained in this work. This is likely because the previous works only investigated the effects of PEBAX coating on the flat sheet membranes and when the same conditions were applied to the circular surface of hollow fiber membrane, it led to different results. As a conclusion, PDMS coating is more promising to improve the performance of PSF hollow fiber membrane for oxygen enrichment process compared to the PEBAX coating.