Incorporation of Conductive Materials into Hydrogels for Tissue Engineering Applications
Abstract
:1. Introduction
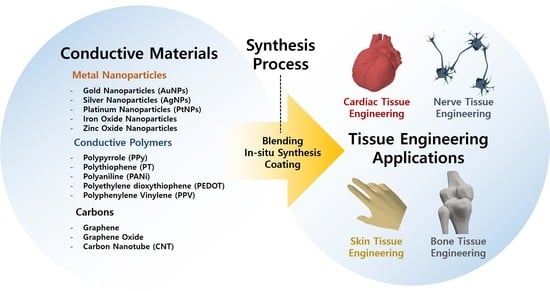
2. Types of Conductive Hydrogels
2.1. Materials
2.1.1. Metal Nanoparticles
2.1.2. Conductive Polymers
2.1.3. Carbons
2.1.4. Hybrid Materials
2.2. Synthesis Process
2.2.1. Blending Process
2.2.2. In Situ Process
2.2.3. Coating Process
3. Biomedical Applications for Tissue Engineering
3.1. Cardiac Tissue Engineering
3.2. Nerve Tissue Engineering
3.3. Bone Tissue Engineering
3.4. Skin Tissue Engineering
4. Conclusions and Future Perspectives
Funding
Conflicts of Interest
References
- Atala, A.; Kasper, F.K.; Mikos, A.G. Engineering Complex Tissues. Sci. Transl. Med. 2012, 4, 160rv112. [Google Scholar] [CrossRef] [PubMed]
- Gaharwar, A.K.; Peppas, N.A.; Khademhosseini, A. Nanocomposite hydrogels for biomedical applications. Biotechnol. Bioeng. 2014, 111, 441–453. [Google Scholar] [CrossRef] [PubMed]
- Harrison, B.S.; Atala, A. Carbon nanotube applications for tissue engineering. Biomaterials 2007, 28, 344–353. [Google Scholar] [CrossRef] [PubMed]
- Shevach, M.; Fleischer, S.; Shapira, A.; Dvir, T. Gold nanoparticle-decellularized matrix hybrids for cardiac tissue engineering. Nano Lett. 2014, 14, 5792–5796. [Google Scholar] [CrossRef] [PubMed]
- Balint, R.; Cassidy, N.J.; Cartmell, S.H. Conductive polymers: Towards a smart biomaterial for tissue engineering. Acta Biomater. 2014, 10, 2341–2353. [Google Scholar] [CrossRef] [PubMed]
- Kloxin, A.M.; Kloxin, C.J.; Bowman, C.N.; Anseth, K.S. Mechanical properties of cellularly responsive hydrogels and their experimental determination. Adv. Mater. 2010, 22, 3484–3494. [Google Scholar] [CrossRef] [PubMed]
- Mawad, D.; Stewart, E.; Officer, D.L.; Romeo, T.; Wagner, P.; Wagner, K.; Wallace, G.G. A Single Component Conducting Polymer Hydrogel as a Scaffold for Tissue Engineering. Adv. Funct. Mater. 2012, 22, 2692–2699. [Google Scholar] [CrossRef]
- Kaur, G.; Adhikari, R.; Cass, P.; Bown, M.; Gunatillake, P. Electrically conductive polymers and composites for biomedical applications. RSC Adv. 2015, 5, 37553–37567. [Google Scholar] [CrossRef] [Green Version]
- Yang, X.; Qiu, L.; Cheng, C.; Wu, Y.; Ma, Z.F.; Li, D. Ordered gelation of chemically converted graphene for next-generation electroconductive hydrogel films. Angew. Chem. Int. Ed. 2011, 50, 7325–7328. [Google Scholar] [CrossRef] [PubMed]
- Dhandayuthapani, B.; Yoshida, Y.; Maekawa, T.; Kumar, D.S. Polymeric Scaffolds in Tissue Engineering Application: A Review. Int. J. Polym. Sci. 2011, 2011, 290602. [Google Scholar] [CrossRef]
- Straley, K.S.; Foo, C.W.P.; Heilshorn, S.C. Biomaterial Design Strategies for the Treatment of Spinal Cord Injuries. J. Neurotrauma 2010, 27. [Google Scholar] [CrossRef] [PubMed]
- Mirkin, C.A. The beginning of a small revolution. Small 2005, 1, 14–16. [Google Scholar] [CrossRef] [PubMed]
- Campelo, J.M.; Luna, D.; Luque, R.; Marinas, J.M.; Romero, A.A. Sustainable preparation of supported metal nanoparticles and their applications in catalysis. ChemSusChem 2009, 2, 18–45. [Google Scholar] [CrossRef] [PubMed]
- Abdullah, M.; Khairurrijal, K.; Rajak, A.; Murniati, R.; Yuliza, E. Effect of Particle Size on the Electrical Conductivity of Metallic Particles. In Proceedings of the 2014 International Conference on Advances in Education Technology, Bandung, Indonesia, 16 October 2014. [Google Scholar]
- Otsuka, H.; Nagasaki, Y.; Kataoka, K. PEGylated nanoparticles for biological and pharmaceutical applications. Adv. Drug Deliv. Rev. 2012, 64, 246–255. [Google Scholar] [CrossRef] [Green Version]
- Ayush, V.; Francesco, S. Effect of Surface Properties on Nanoparticle–Cell Interactions. Small 2010, 6, 12–21. [Google Scholar]
- Skardal, A.; Zhang, J.; McCoard, L.; Xu, X.; Oottamasathien, S.; Prestwich, G.D. Photocrosslinkable hyaluronan-gelatin hydrogels for two-step bioprinting. Tissue Eng. Part A 2010, 16, 2675–2685. [Google Scholar] [CrossRef] [PubMed]
- Xing, R.; Liu, K.; Jiao, T.; Zhang, N.; Ma, K.; Zhang, R.; Zou, Q.; Ma, G.; Yan, X. An Injectable Self-Assembling Collagen–Gold Hybrid Hydrogel for Combinatorial Antitumor Photothermal/Photodynamic Therapy. Adv. Mater. 2016, 28, 3669–3676. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Li, X.; Takemura, T.; Hanagata, N.; Wu, G.; Chou, L.L. Genotoxicity and molecular response of silver nanoparticle (NP)-based hydrogel. J. Nanobiotechnol. 2012, 10, 16. [Google Scholar] [CrossRef] [PubMed]
- Paquet, C.; de Haan, H.W.; Leek, D.M.; Lin, H.-Y.; Xiang, B.; Tian, G.; Kell, A.; Simard, B. Clusters of Superparamagnetic Iron Oxide Nanoparticles Encapsulated in a Hydrogel: A Particle Architecture Generating a Synergistic Enhancement of the T2 Relaxation. ACS Nano 2011, 5, 3104–3112. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zare, M.; Ramezani, Z.; Rahbar, N. Development of zirconia nanoparticles-decorated calcium alginate hydrogel fibers for extraction of organophosphorous pesticides from water and juice samples: Facile synthesis and application with elimination of matrix effects. J. Chromatogr. A 2016, 1473, 28–37. [Google Scholar] [CrossRef] [PubMed]
- Shi, X.; Gu, W.; Li, B.; Chen, N.; Zhao, K.; Xian, Y. Enzymatic biosensors based on the use of metal oxide nanoparticles. Microchim. Acta 2014, 181, 1–22. [Google Scholar] [CrossRef]
- Gutiérrez-Sánchez, C.; Pita, M.; Vaz-Domínguez, C.; Shleev, S.; De Lacey, A.L. Gold Nanoparticles as Electronic Bridges for Laccase-Based Biocathodes. J. Am. Chem. Soc. 2012, 134, 17212–17220. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Arvizo, R.R.; Bhattacharyya, S.; Kudgus, R.A.; Giri, K.; Bhattacharya, R.; Mukherjee, P. Intrinsic therapeutic applications of noble metal nanoparticles: Past, present and future. Chem. Soc. Rev. 2012, 41, 2943–2970. [Google Scholar] [CrossRef] [PubMed]
- Prasanthkumar, S.; Gopal, A.; Ajayaghosh, A. Self-assembly of thienylenevinylene molecular wires to semiconducting gels with doped metallic conductivity. J. Am. Chem. Soc. 2010, 132, 13206–13207. [Google Scholar] [CrossRef] [PubMed]
- Shah, M.; Badwaik, V.; Kherde, Y.; Waghwani, H.K.; Modi, T.; Aguilar, Z.P.; Rodgers, H.; Hamilton, W.; Marutharaj, T.; Webb, C. Gold nanoparticles: Various methods of synthesis and antibacterial applications. Front. Biosci. (Landmark Ed.) 2014, 19, 1320–1344. [Google Scholar] [CrossRef] [PubMed]
- Cho, I.-H.; Bhunia, A.; Irudayaraj, J. Rapid pathogen detection by lateral-flow immunochromatographic assay with gold nanoparticle-assisted enzyme signal amplification. Int. J. Food Microbiol. 2015, 206, 60–66. [Google Scholar] [CrossRef] [PubMed]
- Chung, U.S.; Kim, J.-H.; Kim, B.; Kim, E.; Jang, W.-D.; Koh, W.-G. Dendrimer porphyrin-coated gold nanoshells for the synergistic combination of photodynamic and photothermal therapy. Chem. Commun. 2016, 52, 1258–1261. [Google Scholar] [CrossRef] [PubMed]
- Rengan, A.K.; Bukhari, A.B.; Pradhan, A.; Malhotra, R.; Banerjee, R.; Srivastava, R.; De, A. In vivo analysis of biodegradable liposome gold nanoparticles as efficient agents for photothermal therapy of cancer. Nano Lett. 2015, 15, 842–848. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, L.C.; Bickford, L.R.; Lewinski, N.A.; Coughlin, A.J.; Hu, Y.; Day, E.S.; West, J.L.; Drezek, R.A. A new era for cancer treatment: Gold-nanoparticle-mediated thermal therapies. Small 2011, 7, 169–183. [Google Scholar] [CrossRef] [PubMed]
- Kang, Z.; Yan, X.; Zhao, L.; Liao, Q.; Zhao, K.; Du, H.; Zhang, X.; Zhang, X.; Zhang, Y. Gold nanoparticle/ZnO nanorod hybrids for enhanced reactive oxygen species generation and photodynamic therapy. Nano Res. 2015, 8, 2004–2014. [Google Scholar] [CrossRef]
- Nossier, A.I.; Eissa, S.; Ismail, M.F.; Hamdy, M.A.; Azzazy, H.M.E.-S. Direct detection of hyaluronidase in urine using cationic gold nanoparticles: A potential diagnostic test for bladder cancer. Biosens. Bioelectron. 2014, 54, 7–14. [Google Scholar] [CrossRef] [PubMed]
- Sabella, S.; Galeone, A.; Vecchio, G.; Cingolani, R.; Pompa, P. AuNPs are toxic in vitro and in vivo: A review. J. Nanosci. Lett. 2011, 1, 145–165. [Google Scholar]
- Baei, P.; Jalili-Firoozinezhad, S.; Rajabi-Zeleti, S.; Tafazzoli-Shadpour, M.; Baharvand, H.; Aghdami, N. Electrically conductive gold nanoparticle-chitosan thermosensitive hydrogels for cardiac tissue engineering. Mater. Sci. Eng. C 2016, 63, 131–141. [Google Scholar] [CrossRef] [PubMed]
- Iravani, S.; Korbekandi, H.; Mirmohammadi, S.V.; Zolfaghari, B. Synthesis of silver nanoparticles: Chemical, physical and biological methods. Res. Pharm. Sci. 2014, 9, 385. [Google Scholar] [PubMed]
- Durán, N.; Nakazato, G.; Seabra, A.B. Antimicrobial activity of biogenic silver nanoparticles, and silver chloride nanoparticles: An overview and comments. Appl. Microbiol. Biotechnol. 2016, 100, 6555–6570. [Google Scholar] [CrossRef] [PubMed]
- Johnston, H.J.; Hutchison, G.; Christensen, F.M.; Peters, S.; Hankin, S.; Stone, V. A review of the in vivo and in vitro toxicity of silver and gold particulates: Particle attributes and biological mechanisms responsible for the observed toxicity. Crit. Rev. Toxicol. 2010, 40, 328–346. [Google Scholar] [CrossRef] [PubMed]
- Rai, M.; Deshmukh, S.; Ingle, A.; Gade, A. Silver nanoparticles: The powerful nanoweapon against multidrug-resistant bacteria. J. Appl. Microbiol. 2012, 112, 841–852. [Google Scholar] [CrossRef] [PubMed]
- Meng, M.; He, H.; Xiao, J.; Zhao, P.; Xie, J.; Lu, Z. Controllable in situ synthesis of silver nanoparticles on multilayered film-coated silk fibers for antibacterial application. J. Colloid Interface Sci. 2016, 461, 369–375. [Google Scholar] [CrossRef] [PubMed]
- Xiang, D.; Zheng, Y.; Duan, W.; Li, X.; Yin, J.; Shigdar, S.; O’Connor, M.L.; Marappan, M.; Zhao, X.; Miao, Y.; et al. Inhibition of A/Human/Hubei/3/2005 (H3N2) influenza virus infection by silver nanoparticles in vitro and in vivo. Int. J. Nanomed. 2013, 8, 4103–4114. [Google Scholar] [CrossRef] [PubMed]
- Austin, L.A.; Mackey, M.A.; Dreaden, E.C.; El-Sayed, M.A. The optical, photothermal, and facile surface chemical properties of gold and silver nanoparticles in biodiagnostics, therapy, and drug delivery. Arch. Toxicol. 2014, 88, 1391–1417. [Google Scholar] [CrossRef] [PubMed]
- Jeyaraj, M.; Sathishkumar, G.; Sivanandhan, G.; MubarakAli, D.; Rajesh, M.; Arun, R.; Kapildev, G.; Manickavasagam, M.; Thajuddin, N.; Premkumar, K. Biogenic silver nanoparticles for cancer treatment: An experimental report. Colloids Surf. B Biointerfaces 2013, 106, 86–92. [Google Scholar] [CrossRef] [PubMed]
- Stepanov, A.; Golubev, A.; Nikitin, S.; Osin, Y. A review on the fabrication and properties of platinum nanoparticles. Rev. Adv. Mater. Sci. 2014, 38, e175. [Google Scholar]
- Lee, S.; Kwon, D.; Yim, C.; Jeon, S. Facile detection of Troponin I using dendritic platinum nanoparticles and capillary tube indicators. Anal. Chem. 2015, 87, 5004–5008. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Bo, X.; Mu, Z.; Zhang, Y.; Guo, L. Electrodeposition of nickel oxide and platinum nanoparticles on electrochemically reduced graphene oxide film as a nonenzymatic glucose sensor. Sens. Actuators B Chem. 2014, 192, 261–268. [Google Scholar] [CrossRef]
- Hikosaka, K.; Kim, J.; Kajita, M.; Kanayama, A.; Miyamoto, Y. Platinum nanoparticles have an activity similar to mitochondrial NADH: Ubiquinone oxidoreductase. Colloids Surf. B Biointerfaces 2008, 66, 195–200. [Google Scholar] [CrossRef] [PubMed]
- Xie, J.; Liu, G.; Eden, H.S.; Ai, H.; Chen, X. Surface-Engineered Magnetic Nanoparticle Platforms for Cancer Imaging and Therapy. Acc. Chem. Res. 2011, 44, 883–892. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xu, Q.; Li, J.; Li, S.; Pan, H. A highly sensitive electrochemiluminescence immunosensor based on magnetic nanoparticles and its application in CA125 determination. J. Solid State Electrochem. 2012, 16, 2891–2898. [Google Scholar] [CrossRef]
- Li, M.; Zhu, L.; Lin, D. Toxicity of ZnO nanoparticles to Escherichia coli: Mechanism and the influence of medium components. Environ. Sci. Technol. 2011, 45, 1977–1983. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Chen, B.; Jiang, H.; Wang, C.; Wang, H.; Wang, X. A strategy for ZnO nanorod mediated multi-mode cancer treatment. Biomaterials 2011, 32, 1906–1914. [Google Scholar] [CrossRef] [PubMed]
- Fan, Z.; Lu, J.G. Zinc oxide nanostructures: Synthesis and properties. J. Nanosci. Nanotechnol. 2005, 5, 1561–1573. [Google Scholar] [CrossRef] [PubMed]
- Genchi, G.G.; Nuhn, H.; Liakos, I.; Marino, A.; Marras, S.; Athanassiou, A.; Mattoli, V.; Desai, T.A. Titanium dioxide nanotube arrays coated with laminin enhance C2C12 skeletal myoblast adhesion and differentiation. RSC Adv. 2016, 6, 18502–18514. [Google Scholar] [CrossRef]
- Zheng, H.; Mathe, M. Enhanced conductivity and stability of composite membranes based on poly(2,5-benzimidazole) and zirconium oxide nanoparticles for fuel cells. J. Power Sources 2011, 196, 894–898. [Google Scholar] [CrossRef]
- Gerard, M.; Chaubey, A.; Malhotra, B.D. Application of conducting polymers to biosensors. Biosens. Bioelectron. 2002, 17, 345–359. [Google Scholar] [CrossRef]
- Mozafari, M.; Mehraien, M.; Vashaee, D.; Tayebi, L. Electroconductive nanocomposite scaffolds: A new strategy into tissue engineering and regenerative medicine. In Nanocomposites-New Trends and Developments; InTech: Rijeka, Croatia, 2012. [Google Scholar]
- Rylie, A.G.; Sungchul, B.; Laura, A.P.-W.; Penny, J.M. Conducting polymer-hydrogels for medical electrode applications. Sci. Technol. Adv. Mater. 2010, 11, 014107. [Google Scholar] [Green Version]
- Sajesh, K.M.; Jayakumar, R.; Nair, S.V.; Chennazhi, K.P. Biocompatible conducting chitosan/polypyrrole–alginate composite scaffold for bone tissue engineering. Int. J. Biol. Macromol. 2013, 62, 465–471. [Google Scholar] [CrossRef] [PubMed]
- Kai, D.; Prabhakaran, M.P.; Jin, G.; Ramakrishna, S. Polypyrrole-contained electrospun conductive nanofibrous membranes for cardiac tissue engineering. J. Biomed. Mater. Res. Part A 2011, 99, 376–385. [Google Scholar] [CrossRef] [PubMed]
- Ghasemi-Mobarakeh, L.; Prabhakaran, M.P.; Morshed, M.; Nasr-Esfahani, M.H.; Baharvand, H.; Kiani, S.; Al-Deyab, S.S.; Ramakrishna, S. Application of conductive polymers, scaffolds and electrical stimulation for nerve tissue engineering. J. Tissue Eng. Regen. Med. 2011, 5, e17–e35. [Google Scholar] [CrossRef] [PubMed]
- Chougule, M.A.; Pawar, S.G.; Godse, P.R.; Mulik, R.N.; Sen, S.; Patil, V.B. Synthesis and characterization of polypyrrole (PPy) thin films. Soft Nanosci. Lett. 2011, 1, 6–10. [Google Scholar] [CrossRef]
- Brezoi, D.V. Polypyrrole films prepared by chemical oxidation of pyrrole in aqueous FeCl3 solution. J. Sci. Arts 2010, 1, 53–58. [Google Scholar]
- Song, Y.; Liu, T.Y.; Xu, X.X.; Feng, D.Y.; Li, Y.; Liu, X.X. Pushing the Cycling Stability Limit of Polypyrrole for Supercapacitors. Adv. Funct. Mater. 2015, 25, 4626–4632. [Google Scholar] [CrossRef]
- Huang, Z.-B.; Yin, G.-F.; Liao, X.-M.; Gu, J.-W. Conducting polypyrrole in tissue engineering applications. Front. Mater. Sci. 2014, 8, 39–45. [Google Scholar] [CrossRef]
- Stewart, E.M.; Liu, X.; Clark, G.M.; Kapsa, R.M.I.; Wallace, G.G. Inhibition of smooth muscle cell adhesion and proliferation on heparin-doped polypyrrole. Acta Biomater. 2012, 8, 194–200. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Lu, L.; Zhang, J.; Hu, X.; Zhang, Y.; Liang, W.; Wu, S.; Luo, Z. Electrical stimulation to conductive scaffold promotes axonal regeneration and remyelination in a rat model of large nerve defect. PLoS ONE 2012, 7, e39526. [Google Scholar] [CrossRef] [PubMed]
- Abidian, M.R.; Daneshvar, E.D.; Egeland, B.M.; Kipke, D.R.; Cederna, P.S.; Urbanchek, M.G. Hybrid Conducting Polymer–Hydrogel Conduits for Axonal Growth and Neural Tissue Engineering. Adv. Healthc. Mater. 2012, 1, 762–767. [Google Scholar] [CrossRef] [PubMed]
- Runge, M.B.; Dadsetan, M.; Baltrusaitis, J.; Knight, A.M.; Ruesink, T.; Lazcano, E.A.; Lu, L.; Windebank, A.J.; Yaszemski, M.J. The development of electrically conductive polycaprolactone fumarate-polypyrrole composite materials for nerve regeneration. Biomaterials 2010, 31, 5916–5926. [Google Scholar] [CrossRef] [PubMed]
- Zou, Y.; Qin, J.; Huang, Z.; Yin, G.; Pu, X.; He, D. Fabrication of Aligned Conducting PPy-PLLA Fiber Films and Their Electrically Controlled Guidance and Orientation for Neurites. ACS Appl. Mater. Interfaces 2016, 8, 12576–12582. [Google Scholar] [CrossRef] [PubMed]
- Tian, L.; Prabhakaran, M.P.; Hu, J.; Chen, M.; Besenbacher, F.; Ramakrishna, S. Synergistic effect of topography, surface chemistry and conductivity of the electrospun nanofibrous scaffold on cellular response of PC12 cells. Colloids Surf. B Biointerfaces 2016, 145, 420–429. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Wang, K.; Xing, Y.; Yu, Q. Lysine-doped polypyrrole/spider silk protein/poly(l-lactic) acid containing nerve growth factor composite fibers for neural application. Mater. Sci. Eng. C 2015, 56, 564–573. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Choe, G.; Yang, S.; Jo, H.; Lee, J.Y. Polypyrrole-incorporated conductive hyaluronic acid hydrogels. Biomater. Res. 2016, 20, 31. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Jayatissa, A.H. Comparison study of graphene based conductive nanocomposites using poly(methyl methacrylate) and polypyrrole as matrix materials. J. Mater. Sci. Mater. Electron. 2015, 26, 7780–7783. [Google Scholar] [CrossRef]
- Björninen, M.; Siljander, A.; Pelto, J.; Hyttinen, J.; Kellomäki, M.; Miettinen, S.; Seppänen, R.; Haimi, S. Comparison of Chondroitin Sulfate and Hyaluronic Acid Doped Conductive Polypyrrole Films for Adipose Stem Cells. Ann. Biomed. Eng. 2014, 42, 1889–1900. [Google Scholar] [CrossRef] [PubMed]
- Bendrea, A.-D.; Cianga, L.; Cianga, I. Review paper: Progress in the Field of Conducting Polymers for Tissue Engineering Applications. J. Biomater. Appl. 2011, 26, 3–84. [Google Scholar] [CrossRef] [PubMed]
- Thomas, C.; Zong, K.; Schottland, P.; Reynolds, J. Poly(3,4-alkylenedioxypyrrole)s as highly stable aqueous-compatible conducting polymers with biomedical implications. Adv. Mater. 2000, 12, 222–225. [Google Scholar] [CrossRef]
- Thompson, B.C.; Moulton, S.E.; Richardson, R.T.; Wallace, G.G. Effect of the dopant anion in polypyrrole on nerve growth and release of a neurotrophic protein. Biomaterials 2011, 32, 3822–3831. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.-C.; Sun, Y.-C.; Chen, Y.-H. Electrically conductive nanofibers with highly oriented structures and their potential application in skeletal muscle tissue engineering. Acta Biomater. 2013, 9, 5562–5572. [Google Scholar] [CrossRef] [PubMed]
- Humpolicek, P.; Kasparkova, V.; Saha, P.; Stejskal, J. Biocompatibility of polyaniline. Synth. Met. 2012, 162, 722–727. [Google Scholar] [CrossRef]
- Borriello, A.; Guarino, V.; Schiavo, L.; Alvarez-Perez, M.; Ambrosio, L. Optimizing PANi doped electroactive substrates as patches for the regeneration of cardiac muscle. J. Mater. Sci. Mater. Med. 2011, 22, 1053–1062. [Google Scholar] [CrossRef] [PubMed]
- Guarino, V.; Alvarez-Perez, M.A.; Borriello, A.; Napolitano, T.; Ambrosio, L. Conductive PANi/PEGDA macroporous hydrogels for nerve regeneration. Adv. Healthc. Mater. 2013, 2, 218–227. [Google Scholar] [CrossRef] [PubMed]
- Prabhakaran, M.P.; Ghasemi-Mobarakeh, L.; Jin, G.; Ramakrishna, S. Electrospun conducting polymer nanofibers and electrical stimulation of nerve stem cells. J. Biosci. Bioeng. 2011, 112, 501–507. [Google Scholar] [CrossRef] [PubMed]
- Dong, R.; Zhao, X.; Guo, B.; Ma, P.X. Self-healing conductive injectable hydrogels with antibacterial activity as cell delivery carrier for cardiac cell therapy. ACS Appl. Mater. Interfaces 2016, 8, 17138–17150. [Google Scholar] [CrossRef] [PubMed]
- Sista, P.; Ghosh, K.; Martinez, J.S.; Rocha, R.C. Polythiophenes in biological applications. J. Nanosci. Nanotechnol. 2014, 14, 250–272. [Google Scholar] [CrossRef] [PubMed]
- Rad, A.T.; Ali, N.; Kotturi, H.S.R.; Yazdimamaghani, M.; Smay, J.; Vashaee, D.; Tayebi, L. Conducting scaffolds for liver tissue engineering. J. Biomed. Mater. Res. Part A 2014, 102, 4169–4181. [Google Scholar]
- Karagkiozaki, V.; Karagiannidis, P.; Gioti, M.; Kavatzikidou, P.; Georgiou, D.; Georgaraki, E.; Logothetidis, S. Bioelectronics meets nanomedicine for cardiovascular implants: PEDOT-based nanocoatings for tissue regeneration. Biochim. Biophys. Acta (BBA)-Gen. Subj. 2013, 1830, 4294–4304. [Google Scholar] [CrossRef] [PubMed]
- Luo, S.-C.; Mohamed Ali, E.; Tansil, N.C.; Yu, H.-H.; Gao, S.; Kantchev, E.A.; Ying, J.Y. Poly(3,4-ethylenedioxythiophene)(PEDOT) nanobiointerfaces: Thin, ultrasmooth, and functionalized PEDOT films with in vitro and in vivo biocompatibility. Langmuir 2008, 24, 8071–8077. [Google Scholar] [CrossRef] [PubMed]
- Strakosas, X.; Wei, B.; Martin, D.C.; Owens, R.M. Biofunctionalization of polydioxythiophene derivatives for biomedical applications. J. Mater. Chem. B 2016, 4, 4952–4968. [Google Scholar] [CrossRef] [Green Version]
- Wang, G.-X.; Qian, Y.; Cao, X.-X.; Xia, X.-H. Direct electrochemistry of cytochrome c on a graphene/poly(3,4-ethylenedioxythiophene) nanocomposite modified electrode. Electrochem. Commun. 2012, 20, 1–3. [Google Scholar] [CrossRef]
- Groenendaal, L.; Jonas, F.; Freitag, D.; Pielartzik, H.; Reynolds, J.R. Poly(3,4-ethylenedioxythiophene) and its derivatives: Past, present, and future. Adv. Mater. 2000, 12, 481–494. [Google Scholar] [CrossRef]
- Spencer, A.R.; Primbetova, A.; Koppes, A.N.; Koppes, R.A.; Fenniri, H.; Annabi, N. Electroconductive Gelatin Methacryloyl-PEDOT:PSS Composite Hydrogels: Design, Synthesis, and Properties. ACS Biomater. Sci. Eng. 2018, 4, 1558–1567. [Google Scholar] [CrossRef]
- Schweizer, T.M. Electrical Characterization and Investigation of the Piezoresistive Effect of PEDOT: PSS Thin Films. Master’s Thesis, Georgia Institute of Technology, Atlanta, GA, USA, 2005. [Google Scholar]
- Courté, M.; Alaaeddine, M.; Barth, V.; Tortech, L.; Fichou, D. Structural and electronic properties of 2,2′,6,6′-tetraphenyl-dipyranylidene and its use as a hole-collecting interfacial layer in organic solar cells. Dyes Pigments 2017, 141, 487–492. [Google Scholar] [CrossRef]
- Niamlang, S.; Buranut, T.; Niansiri, A.; Sirivat, A. Electrically controlled Aloin from poly(p-phenylene vinylene)/polyacrylamide hydrogel system. In Proceedings of the 9th Eco-Energy and Materials Science and Engineering Symposium, Chiang-Rai, Thailand, 25–28 May 2011; Intellectual Repository of Rajamangala University of Technology Thanyaburi: Pathum Thani, Thailand, 2011. [Google Scholar]
- Niamlang, S.; Buranut, T.; Niansiri, A.; Sirivat, A. Controlled aloin release from crosslinked polyacrylamide hydrogels: Effects of mesh size, electric field strength and a conductive polymer. Materials 2013, 6, 4787–4800. [Google Scholar] [CrossRef] [PubMed]
- Lee, G.-H.; Cooper, R.C.; An, S.J.; Lee, S.; van der Zande, A.; Petrone, N.; Hammerberg, A.G.; Lee, C.; Crawford, B.; Oliver, W.; et al. High-Strength Chemical-Vapor–Deposited Graphene and Grain Boundaries. Science 2013, 340, 1073–1076. [Google Scholar] [CrossRef] [PubMed]
- Novoselov, K.S.; Fal′ko, V.I.; Colombo, L.; Gellert, P.R.; Schwab, M.G.; Kim, K. A roadmap for graphene. Nature 2012, 490, 192. [Google Scholar] [CrossRef] [PubMed]
- Mayorov, A.S.; Gorbachev, R.V.; Morozov, S.V.; Britnell, L.; Jalil, R.; Ponomarenko, L.A.; Blake, P.; Novoselov, K.S.; Watanabe, K.; Taniguchi, T.; et al. Micrometer-Scale Ballistic Transport in Encapsulated Graphene at Room Temperature. Nano Lett. 2011, 11, 2396–2399. [Google Scholar] [CrossRef] [PubMed]
- Shao, Y.; Wang, J.; Wu, H.; Liu, J.; Aksay, I.A.; Lin, Y. Graphene based electrochemical sensors and biosensors: A review. Electroanalysis 2010, 22, 1027–1036. [Google Scholar] [CrossRef]
- Xu, Y.; Lin, Z.; Huang, X.; Wang, Y.; Huang, Y.; Duan, X. Functionalized Graphene Hydrogel-Based High-Performance Supercapacitors. Adv. Mater. 2013, 25, 5779–5784. [Google Scholar] [CrossRef] [PubMed]
- MacHado, B.F.; Serp, P. Graphene-based materials for catalysis. Catal. Sci. Technol. 2012, 2, 54–75. [Google Scholar] [CrossRef]
- Hoa, L.T.; Chung, J.S.; Hur, S.H. A highly sensitive enzyme-free glucose sensor based on Co3O4 nanoflowers and 3D graphene oxide hydrogel fabricated via hydrothermal synthesis. Sens. Actuators B 2016, 223, 76–82. [Google Scholar] [CrossRef]
- Lee, W.C.; Lim, C.H.Y.X.; Shi, H.; Tang, L.A.L.; Wang, Y.; Lim, C.T.; Loh, K.P. Origin of Enhanced Stem Cell Growth and Differentiation on Graphene and Graphene Oxide. ACS Nano 2011, 5, 7334–7341. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Zhu, J.; Qiu, L.; Li, D. Bioinspired effective prevention of restacking in multilayered graphene films: Towards the next generation of high-performance supercapacitors. Adv. Mater. 2011, 23, 2833–2838. [Google Scholar] [CrossRef] [PubMed]
- Loh, K.P.; Bao, Q.; Eda, G.; Chhowalla, M. Graphene oxide as a chemically tunable platform for optical applications. Nat. Chem. 2010, 2, 1015–1024. [Google Scholar] [CrossRef] [PubMed]
- Gao, W.; Alemany, L.B.; Ci, L.; Ajayan, P.M. New insights into the structure and reduction of graphite oxide. Nat. Chem. 2009, 1, 403–408. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Yu, D.; Zeng, C.; Miao, Z.; Dai, L. Biocompatible graphene oxide-based glucose biosensors. Langmuir 2010, 26, 6158–6160. [Google Scholar] [CrossRef] [PubMed]
- Yang, K.; Gong, H.; Shi, X.; Wan, J.; Zhang, Y.; Liu, Z. In vivo biodistribution and toxicology of functionalized nano-graphene oxide in mice after oral and intraperitoneal administration. Biomaterials 2013, 34, 2787–2795. [Google Scholar] [CrossRef] [PubMed]
- Zhou, M.; Zhai, Y.; Dong, S. Electrochemical Sensing and Biosensing Platform Based on Chemically Reduced Graphene Oxide. Anal. Chem. 2009, 81, 5603–5613. [Google Scholar] [CrossRef] [PubMed]
- Han, L.; Liu, K.; Wang, M.; Wang, K.; Fang, L.; Chen, H.; Zhou, J.; Lu, X. Mussel-Inspired Adhesive and Conductive Hydrogel with Long-Lasting Moisture and Extreme Temperature Tolerance. Adv. Funct. Mater. 2018, 28, 1704195. [Google Scholar] [CrossRef]
- Joung, Y.S.; Ramirez, R.B.; Bailey, E.; Adenekan, R.; Buie, C.R. Conductive hydrogel films produced by freestanding electrophoretic deposition and polymerization at the interface of immiscible liquids. Compos. Sci. Technol. 2017, 153, 128–135. [Google Scholar] [CrossRef]
- Samanta, S.K.; Pal, A.; Bhattacharya, S.; Rao, C. Carbon nanotube reinforced supramolecular gels with electrically conducting, viscoelastic and near-infrared sensitive properties. J. Mater. Chem. 2010, 20, 6881–6890. [Google Scholar] [CrossRef]
- Esawi, A.; Morsi, K.; Sayed, A.; Taher, M.; Lanka, S. Effect of carbon nanotube (CNT) content on the mechanical properties of CNT-reinforced aluminium composites. Compos. Sci. Technol. 2010, 70, 2237–2241. [Google Scholar] [CrossRef]
- Wong, K.K.H.; Zinke-Allmang, M.; Hutter, J.L.; Hrapovic, S.; Luong, J.H.; Wan, W. The effect of carbon nanotube aspect ratio and loading on the elastic modulus of electrospun poly(vinyl alcohol)-carbon nanotube hybrid fibers. Carbon 2009, 47, 2571–2578. [Google Scholar] [CrossRef] [Green Version]
- Cellot, G.; Toma, F.M.; Kasap Varley, Z.; Laishram, J.; Villari, A.; Quintana, M.; Cipollone, S.; Prato, M.; Ballerini, L. Carbon Nanotube Scaffolds Tune Synaptic Strength in Cultured Neural Circuits: Novel Frontiers in Nanomaterial–Tissue Interactions. J. Neurosci. 2011, 31, 12945–12953. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, F.; Weidmann, A.; Nebe, J.B.; Burkel, E. Osteoblast cell response to surface-modified carbon nanotubes. Mater. Sci. Eng. C 2012, 32, 1057–1061. [Google Scholar] [CrossRef]
- Shvedova, A.A.; Castranova, V.; Kisin, E.R.; Schwegler-Berry, D.; Murray, A.R.; Gandelsman, V.Z.; Maynard, A.; Baron, P. Exposure to carbon nanotube material: Assessment of nanotube cytotoxicity using human keratinocyte cells. J. Toxicol. Environ. Health Part A 2003, 66, 1909–1926. [Google Scholar] [CrossRef] [PubMed]
- Yan, L.; Zhao, F.; Li, S.; Hu, Z.; Zhao, Y. Low-toxic and safe nanomaterials by surface-chemical design, carbon nanotubes, fullerenes, metallofullerenes, and graphenes. Nanoscale 2011, 3, 362–382. [Google Scholar] [CrossRef] [PubMed]
- Davide, P.; Ravi, S.; David, M.; Mathieu, E.; Jean-Paul, B.; Maurizio, P.; Kostas, K.; Alberto, B. Functionalized Carbon Nanotubes for Plasmid DNA Gene Delivery. Angew. Chem. Int. Ed. 2004, 43, 5242–5246. [Google Scholar]
- Shim, W.; Kwon, Y.; Jeon, S.Y.; Yu, W.R. Optimally conductive networks in randomly dispersed CNT:graphene hybrids. Sci. Rep. 2015, 5, 16568. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, H.; Ma, Z. A cascade reaction signal-amplified amperometric immunosensor platform for ultrasensitive detection of tumour marker. Sens. Actuators B Chem. 2018, 254, 642–647. [Google Scholar] [CrossRef]
- Li, J.; Liu, C.-Y.; Liu, Y. Au/graphene hydrogel: Synthesis, characterization and its use for catalytic reduction of 4-nitrophenol. J. Mater. Chem. 2012, 22, 8426–8430. [Google Scholar] [CrossRef]
- Vaitkuviene, A.; Ratautaite, V.; Mikoliunaite, L.; Kaseta, V.; Ramanauskaite, G.; Biziuleviciene, G.; Ramanaviciene, A.; Ramanavicius, A. Some biocompatibility aspects of conducting polymer polypyrrole evaluated with bone marrow-derived stem cells. Colloids Surf. A Physicochem. Eng. Asp. 2014, 442, 152–156. [Google Scholar] [CrossRef]
- Liu, X.; Miller Ii, A.L.; Park, S.; Waletzki, B.E.; Terzic, A.; Yaszemski, M.J.; Lu, L. Covalent crosslinking of graphene oxide and carbon nanotube into hydrogels enhances nerve cell responses. J. Mater. Chem. B 2016, 4, 6930–6941. [Google Scholar] [CrossRef]
- Huyen, D. Carbon Nanotubes and Semiconducting Polymer Nanocomposites. In Carbon Nanotubes-Synthesis, Characterization, Applications; InTech: Rijeka, Croatia, 2011. [Google Scholar] [Green Version]
- Gupta, N.D.; Maity, S.; Chattopadhyay, K.K. Field emission enhancement of polypyrrole due to band bending induced tunnelling in polypyrrole-carbon nanotubes nanocomposite. J. Ind. Eng. Chem. 2014, 20, 3208–3213. [Google Scholar] [CrossRef]
- Patton, A.J.; Poole-Warren, L.A.; Green, R.A. Mechanisms for Imparting Conductivity to Nonconductive Polymeric Biomaterials. Macromol. Biosci. 2016, 16, 1103–1121. [Google Scholar] [CrossRef] [PubMed]
- Mietta, J.L.; Tamborenea, P.I.; Negri, R.M. Anisotropic reversible piezoresistivity in magnetic–metallic/polymer structured elastomeric composites: Modelling and experiments. Soft Matter 2016, 12, 422–431. [Google Scholar] [CrossRef] [PubMed]
- García, M.; Batalla, P.; Escarpa, A. Metallic and polymeric nanowires for electrochemical sensing and biosensing. TrAC Trends Anal. Chem. 2014, 57, 6–22. [Google Scholar] [CrossRef]
- Gong, S.; Schwalb, W.; Wang, Y.; Chen, Y.; Tang, Y.; Si, J.; Shirinzadeh, B.; Cheng, W. A wearable and highly sensitive pressure sensor with ultrathin gold nanowires. Nat. Commun. 2014, 5, 3132. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hsu, P.-C.; Kong, D.; Wang, S.; Wang, H.; Welch, A.J.; Wu, H.; Cui, Y. Electrolessly deposited electrospun metal nanowire transparent electrodes. J. Am. Chem. Soc. 2014, 136, 10593–10596. [Google Scholar] [CrossRef] [PubMed]
- Kim, A.; Won, Y.; Woo, K.; Kim, C.-H.; Moon, J. Highly transparent low resistance ZnO/Ag nanowire/ZnO composite electrode for thin film solar cells. ACS Nano 2013, 7, 1081–1091. [Google Scholar] [CrossRef] [PubMed]
- Langley, D.; Giusti, G.; Mayousse, C.; Celle, C.; Bellet, D.; Simonato, J.-P. Flexible transparent conductive materials based on silver nanowire networks: A review. Nanotechnology 2013, 24, 452001. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Wang, R.; Wen, M.; Weng, D.; Cui, X.; Sun, J.; Li, H.; Lu, Y. Synthesis of ultralong copper nanowires for high-performance transparent electrodes. J. Am. Chem. Soc. 2012, 134, 14283–14286. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Lee, P.; Lee, H.; Lee, D.; Lee, S.S.; Ko, S.H. Very long Ag nanowire synthesis and its application in a highly transparent, conductive and flexible metal electrode touch panel. Nanoscale 2012, 4, 6408–6414. [Google Scholar] [CrossRef] [PubMed]
- Yuan, T.; Zhang, L.; Li, K.; Fan, H.; Fan, Y.; Liang, J.; Zhang, X. Collagen hydrogel as an immunomodulatory scaffold in cartilage tissue engineering. J. Biomed. Mater. Res. Part B Appl. Biomater. 2014, 102, 337–344. [Google Scholar] [CrossRef] [PubMed]
- Lu, X.; Zhang, W.; Wang, C.; Wen, T.-C.; Wei, Y. One-dimensional conducting polymer nanocomposites: Synthesis, properties and applications. Prog. Polym. Sci. 2011, 36, 671–712. [Google Scholar] [CrossRef]
- Gouma, P.; Kalyanasundaram, K.; Bishop, A. Electrospun single-crystal MoO3 nanowires for biochemistry sensing probes. J. Mater. Res. 2006, 21, 2904–2910. [Google Scholar] [CrossRef]
- Souier, T.; Stefancich, M.; Chiesa, M. Characterization of multi-walled carbon nanotube–polymer nanocomposites by scanning spreading resistance microscopy. Nanotechnology 2012, 23, 405704. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Jiang, D. Influence of geometries of multi-walled carbon nanotubes on the pore structures of Buckypaper. Compos. Part A Appl. Sci. Manuf. 2012, 43, 469–474. [Google Scholar] [CrossRef]
- Yang, J.; Wang, X.; Wang, X.; Jia, R.; Huang, J. Preparation of highly conductive CNTs/polyaniline composites through plasma pretreating and in-situ polymerization. J. Phys. Chem. Solids 2010, 71, 448–452. [Google Scholar] [CrossRef]
- Xiao, X.; Wu, G.; Zhou, H.; Qian, K.; Hu, J. Preparation and Property Evaluation of Conductive Hydrogel Using Poly(Vinyl Alcohol)/Polyethylene Glycol/Graphene Oxide for Human Electrocardiogram Acquisition. Polymers 2017, 9, 259. [Google Scholar] [CrossRef]
- Lai, J.; Zhang, L.; Niu, W.; Qi, W.; Zhao, J.; Liu, Z.; Zhang, W.; Xu, G. One-pot synthesis of gold nanorods using binary surfactant systems with improved monodispersity, dimensional tunability and plasmon resonance scattering properties. Nanotechnology 2014, 25, 125601. [Google Scholar] [CrossRef] [PubMed]
- Harada, M.; Tamura, N.; Takenaka, M. Nucleation and growth of metal nanoparticles during photoreduction using in situ time-resolved SAXS analysis. J. Phys. Chem. C 2011, 115, 14081–14092. [Google Scholar] [CrossRef]
- Dang, Z.-M.; Yuan, J.-K.; Zha, J.-W.; Zhou, T.; Li, S.-T.; Hu, G.-H. Fundamentals, processes and applications of high-permittivity polymer–matrix composites. Prog. Mater. Sci. 2012, 57, 660–723. [Google Scholar] [CrossRef]
- Zhao, W.; Odelius, K.; Edlund, U.; Zhao, C.; Albertsson, A.-C. In situ synthesis of magnetic field-responsive hemicellulose hydrogels for drug delivery. Biomacromolecules 2015, 16, 2522–2528. [Google Scholar] [CrossRef] [PubMed]
- Ma, P.-C.; Siddiqui, N.A.; Marom, G.; Kim, J.-K. Dispersion and functionalization of carbon nanotubes for polymer-based nanocomposites: A review. Compos. Part A Appl. Sci. Manuf. 2010, 41, 1345–1367. [Google Scholar] [CrossRef]
- Siddiqui, N.A.; Li, E.L.; Sham, M.-L.; Tang, B.Z.; Gao, S.L.; Mäder, E.; Kim, J.-K. Tensile strength of glass fibres with carbon nanotube–epoxy nanocomposite coating: Effects of CNT morphology and dispersion state. Compos. Part A Appl. Sci. Manuf. 2010, 41, 539–548. [Google Scholar] [CrossRef]
- Kim, Y.S.; Cho, K.; Lee, H.J.; Chang, S.; Lee, H.; Kim, J.H.; Koh, W.-G. Highly conductive and hydrated PEG-based hydrogels for the potential application of a tissue engineering scaffold. React. Funct. Polym. 2016, 109, 15–22. [Google Scholar] [CrossRef]
- Luo, X.; Cui, X.T. Sponge-like nanostructured conducting polymers for electrically controlled drug release. Electrochem. Commun. 2009, 11, 1956–1959. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, H.; Han, H.; Ma, Z. Conductive hydrogel composed of 1,3,5-benzenetricarboxylic acid and Fe3+ used as enhanced electrochemical immunosensing substrate for tumor biomarker. Bioelectrochemistry 2017, 114, 48–53. [Google Scholar] [CrossRef] [PubMed]
- Kurniawan, D.; Nor, F.; Lee, H.; Lim, J. Elastic properties of polycaprolactone at small strains are significantly affected by strain rate and temperature. Proc. Inst. Mech. Eng. Part H J. Eng. Med. 2011, 225, 1015–1020. [Google Scholar] [CrossRef] [PubMed]
- Xie, J.; Macewan, M.R.; Willerth, S.M.; Li, X.; Moran, D.W.; Sakiyama-Elbert, S.E.; Xia, Y. Conductive Core-Sheath Nanofibers and Their Potential Application in Neural Tissue Engineering. Adv. Funct. Mater. 2009, 19, 2312–2318. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.-J.; Yi, H.-G.; Kim, S.-W.; Cho, D.-W. 3D Cell Printed Tissue Analogues: A New Platform for Theranostics. Theranostics 2017, 7, 3118–3137. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pati, F.; Jang, J.; Ha, D.-H.; Kim, S.W.; Rhie, J.-W.; Shim, J.-H.; Kim, D.-H.; Cho, D.-W. Printing three-dimensional tissue analogues with decellularized extracellular matrix bioink. Nat. Commun. 2014, 5, 3935. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, B.; Yao, F.; Hao, T.; Fang, W.; Ye, L.; Zhang, Y.; Wang, Y.; Li, J.; Wang, C. Development of Electrically Conductive Double-Network Hydrogels via One-Step Facile Strategy for Cardiac Tissue Engineering. Adv. Healthc. Mater. 2016, 5, 474–488. [Google Scholar] [CrossRef] [PubMed]
- Jing, X.; Mi, H.-Y.; Napiwocki, B.N.; Peng, X.-F.; Turng, L.-S. Mussel-inspired electroactive chitosan/graphene oxide composite hydrogel with rapid self-healing and recovery behavior for tissue engineering. Carbon 2017, 125, 557–570. [Google Scholar] [CrossRef]
- Sun, H.; Zhou, J.; Huang, Z.; Qu, L.; Lin, N.; Liang, C.; Dai, R.; Tang, L.; Tian, F. Carbon nanotube-incorporated collagen hydrogels improve cell alignment and the performance of cardiac constructs. Int. J. Nanomed. 2017, 12, 3109–3120. [Google Scholar] [CrossRef] [PubMed]
- Gajendiran, M.; Choi, J.; Kim, S.-J.; Kim, K.; Shin, H.; Koo, H.-J.; Kim, K. Conductive biomaterials for tissue engineering applications. J. Ind. Eng. Chem. 2017, 51, 12–26. [Google Scholar] [CrossRef]
- Hosseinzadeh, S.; Rezayat, S.M.; Vashegani-Farahani, E.; Mahmoudifard, M.; Zamanlui, S.; Soleimani, M. Nanofibrous hydrogel with stable electrical conductivity for biological applications. Polymer 2016, 97, 205–216. [Google Scholar] [CrossRef]
- Jo, H.; Sim, M.; Kim, S.; Yang, S.; Yoo, Y.; Park, J.H.; Yoon, T.H.; Kim, M.G.; Lee, J.Y. Electrically conductive graphene/polyacrylamide hydrogels produced by mild chemical reduction for enhanced myoblast growth and differentiation. Acta Biomater. 2017, 48, 100–109. [Google Scholar] [CrossRef] [PubMed]
- Annabi, N.; Shin, S.R.; Tamayol, A.; Miscuglio, M.; Bakooshli, M.A.; Assmann, A.; Mostafalu, P.; Sun, J.-Y.; Mithieux, S.; Cheung, L.; et al. Highly Elastic and Conductive Human-Based Protein Hybrid Hydrogels. Adv. Mater. 2016, 28, 40–49. [Google Scholar] [CrossRef] [PubMed]
- Ahadian, S.; Yamada, S.; Ramon-Azcon, J.; Estili, M.; Liang, X.; Nakajima, K.; Shiku, H.; Khademhosseini, A.; Matsue, T. Hybrid hydrogel-aligned carbon nanotube scaffolds to enhance cardiac differentiation of embryoid bodies. Acta Biomater. 2016, 31, 134–143. [Google Scholar] [CrossRef] [PubMed]
- Navaei, A.; Saini, H.; Christenson, W.; Sullivan, R.T.; Ros, R.; Nikkhah, M. Gold nanorod-incorporated gelatin-based conductive hydrogels for engineering cardiac tissue constructs. Acta Biomater. 2016, 41, 133–146. [Google Scholar] [CrossRef] [PubMed]
- Hosoyama, K.; Ahumada, M.; McTiernan, C.; Bejjani, J.; Variola, F.; Ruel, M.; Xu, B.; Liang, W.; Suuronen, E.; Alarcon, E. Multi-functional thermo-crosslinkable collagen-metal nanoparticle composites for tissue regeneration: Nanosilver vs. nanogold. RSC Adv. 2017, 7, 47704–47708. [Google Scholar] [CrossRef]
- Liu, N.; Chen, J.; Zhuang, J.; Zhu, P. Fabrication of engineered nanoparticles on biological macromolecular (PEGylated chitosan) composite for bio-active hydrogel system in cardiac repair applications. Int. J. Biol. Macromol. 2018, 117, 553–558. [Google Scholar] [CrossRef] [PubMed]
- Kharaziha, M.; Shin, S.R.; Nikkhah, M.; Topkaya, S.N.; Masoumi, N.; Annabi, N.; Dokmeci, M.R.; Khademhosseini, A. Tough and flexible CNT–polymeric hybrid scaffolds for engineering cardiac constructs. Biomaterials 2014, 35, 7346–7354. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shin, S.R.; Jung, S.M.; Zalabany, M.; Kim, K.; Zorlutuna, P.; Kim, S.B.; Nikkhah, M.; Khabiry, M.; Azize, M.; Kong, J. Carbon-nanotube-embedded hydrogel sheets for engineering cardiac constructs and bioactuators. ACS Nano 2013, 7, 2369–2380. [Google Scholar] [CrossRef] [PubMed]
- Shi, Z.; Gao, H.; Feng, J.; Ding, B.; Cao, X.; Kuga, S.; Wang, Y.; Zhang, L.; Cai, J. In situ synthesis of robust conductive cellulose/polypyrrole composite aerogels and their potential application in nerve regeneration. Angew. Chem. Int. Ed. Engl. 2014, 53, 5380–5384. [Google Scholar] [CrossRef] [PubMed]
- Bu, Y.; Xu, H.-X.; Li, X.; Xu, W.-J.; Yin, Y.-X.; Dai, H.-L.; Wang, X.-B.; Huang, Z.-J.; Xu, P.-H. A conductive sodium alginate and carboxymethyl chitosan hydrogel doped with polypyrrole for peripheral nerve regeneration. RSC Adv. 2018, 8, 10806–10817. [Google Scholar] [CrossRef] [Green Version]
- Yang, S.; Jang, L.; Kim, S.; Yang, J.; Yang, K.; Cho, S.W.; Lee, J.Y. Polypyrrole/alginate hybrid hydrogels: Electrically conductive and soft biomaterials for human mesenchymal stem cell culture and potential neural tissue engineering applications. Macromol. Biosci. 2016, 16, 1653–1661. [Google Scholar] [CrossRef] [PubMed]
- Imaninezhad, M.; Pemberton, K.; Xu, F.; Kalinowski, K.; Bera, R.; Zustiak, S. Directed and enhanced neurite outgrowth following exogenous electrical stimulation on carbon nanotube-hydrogel composites. J. Neural Eng. 2018, 15, 056034. [Google Scholar] [CrossRef] [PubMed]
- Jafarkhani, M.; Salehi, Z.; Nematian, T. Preparation and characterization of chitosan/graphene oxide composite hydrogels for nerve tissue Engineering. Mater. Today Proc. 2018, 5, 15620–15628. [Google Scholar] [CrossRef]
- Zhao, Y.; Wang, Y.; Niu, C.; Zhang, L.; Li, G.; Yang, Y. Construction of polyacrylamide/graphene oxide/gelatin/sodium alginate composite hydrogel with bioactivity for promoting Schwann cells growth. J. Biomed. Mater. Res. Part A 2018, 106, 1951–1964. [Google Scholar] [CrossRef] [PubMed]
- Rose, J.C.; Cámara-Torres, M.; Rahimi, K.; Köhler, J.; Möller, M.; De Laporte, L. Nerve cells decide to orient inside an injectable hydrogel with minimal structural guidance. Nano Lett. 2017, 17, 3782–3791. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Holzwarth, J.M.; Yan, Y.; Xu, P.; Zheng, H.; Yin, Y.; Li, S.; Ma, P.X. Conductive PPY/PDLLA conduit for peripheral nerve regeneration. Biomaterials 2014, 35, 225–235. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, X.; Miller, A.L.; Park, S.; Waletzki, B.E.; Zhou, Z.; Terzic, A.; Lu, L. Functionalized Carbon Nanotube and Graphene Oxide Embedded Electrically Conductive Hydrogel Synergistically Stimulates Nerve Cell Differentiation. ACS Appl. Mater. Interfaces 2017, 9, 14677–14690. [Google Scholar] [CrossRef] [PubMed]
- Javadi, M.; Gu, Q.; Naficy, S.; Farajikhah, S.; Crook, J.M.; Wallace, G.G.; Beirne, S.; Moulton, S.E. Conductive Tough Hydrogel for Bioapplications. Macromol. Biosci. 2018, 18. [Google Scholar] [CrossRef] [PubMed]
- Bose, S.; Roy, M.; Bandyopadhyay, A. Recent advances in bone tissue engineering scaffolds. Trends Biotechnol. 2012, 30, 546–554. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Demirtaş, T.T.; Irmak, G.; Gümüşderelioğlu, M. A bioprintable form of chitosan hydrogel for bone tissue engineering. Biofabrication 2017, 9, 035003. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; Zhang, J.; Yi, C.; Yang, M. The effects of gold nanoparticles on the proliferation, differentiation, and mineralization function of MC3T3-E1 cells in vitro. Chin. Sci. Bull. 2010, 55, 1013–1019. [Google Scholar] [CrossRef]
- Heo, D.N.; Ko, W.-K.; Bae, M.S.; Lee, J.B.; Lee, D.-W.; Byun, W.; Lee, C.H.; Kim, E.-C.; Jung, B.-Y.; Kwon, I.K. Enhanced bone regeneration with a gold nanoparticle–hydrogel complex. J. Mater. Chem. B 2014, 2, 1584–1593. [Google Scholar] [CrossRef]
- Khorshidi, S.; Karkhaneh, A. Hydrogel/fiber conductive scaffold for bone tissue engineering. J. Biomed. Mater. Res. A 2018, 106, 718–724. [Google Scholar] [CrossRef] [PubMed]
- Zanjanizadeh Ezazi, N.; Shahbazi, M.A.; Shatalin, Y.V.; Nadal, E.; Makila, E.; Salonen, J.; Kemell, M.; Correia, A.; Hirvonen, J.; Santos, H.A. Conductive vancomycin-loaded mesoporous silica polypyrrole-based scaffolds for bone regeneration. Int. J. Pharm. 2018, 536, 241–250. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, M.; Ferraz, M.P.; Monteiro, F.J.; Fernandes, M.H.; Beppu, M.M.; Mantione, D.; Sardon, H. Antibacterial silk fibroin/nanohydroxyapatite hydrogels with silver and gold nanoparticles for bone regeneration. Nanomed. Nanotechnol. Biol. Med. 2017, 13, 231–239. [Google Scholar] [CrossRef] [PubMed]
- Pelto, J.; Bjorninen, M.; Palli, A.; Talvitie, E.; Hyttinen, J.; Mannerstrom, B.; Suuronen Seppanen, R.; Kellomaki, M.; Miettinen, S.; Haimi, S. Novel polypyrrole-coated polylactide scaffolds enhance adipose stem cell proliferation and early osteogenic differentiation. Tissue Eng. Part. A 2013, 19, 882–892. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.; Cheng, C.; He, Y.S.; Lyu, C.; Wang, Y.; Yu, J.; Qiu, L.; Zou, D.; Li, D. Multilayered Graphene Hydrogel Membranes for Guided Bone Regeneration. Adv. Mater. 2016, 28, 4025–4031. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Yu, M.; Guo, B.; Ma, P.X.; Yin, Z. Conductive nanofibrous composite scaffolds based on in-situ formed polyaniline nanoparticle and polylactide for bone regeneration. J. Colloid Interface Sci. 2018, 514, 517–527. [Google Scholar] [CrossRef] [PubMed]
- Tandon, B.; Magaz, A.; Balint, R.; Blaker, J.J.; Cartmell, S.H. Electroactive biomaterials: Vehicles for controlled delivery of therapeutic agents for drug delivery and tissue regeneration. Adv. Drug Deliv. Rev. 2018, 129, 148–168. [Google Scholar] [CrossRef] [PubMed]
- Zare, E.N.; Lakouraj, M.M.; Mohseni, M. Biodegradable polypyrrole/dextrin conductive nanocomposite: Synthesis, characterization, antioxidant and antibacterial activity. Synth. Met. 2014, 187, 9–16. [Google Scholar] [CrossRef]
- Zhao, X.; Li, P.; Guo, B.; Ma, P.X. Antibacterial and conductive injectable hydrogels based on quaternized chitosan-graft-polyaniline/oxidized dextran for tissue engineering. Acta Biomater. 2015, 26, 236–248. [Google Scholar] [CrossRef] [PubMed]
- Darabi, M.A.; Khosrozadeh, A.; Mbeleck, R.; Liu, Y.; Chang, Q.; Jiang, J.; Cai, J.; Wang, Q.; Luo, G.; Xing, M. Skin-Inspired Multifunctional Autonomic-Intrinsic Conductive Self-Healing Hydrogels with Pressure Sensitivity, Stretchability, and 3D Printability. Adv. Mater. 2017, 29, 1700533. [Google Scholar] [CrossRef] [PubMed]
- Deng, Z.; Guo, Y.; Ma, P.X.; Guo, B. Rapid thermal responsive conductive hybrid cryogels with shape memory properties, photothermal properties and pressure dependent conductivity. J. Colloid Interface Sci. 2018, 526, 281–294. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Wu, H.; Guo, B.; Dong, R.; Qiu, Y.; Ma, P.X. Antibacterial anti-oxidant electroactive injectable hydrogel as self-healing wound dressing with hemostasis and adhesiveness for cutaneous wound healing. Biomaterials 2017, 122, 34–47. [Google Scholar] [CrossRef] [PubMed]



















| Kinds of Nanoparticles | Size (nm) | Shape | Advantages | Disadvantages | Application |
|---|---|---|---|---|---|
| Gold Nanoparticles (AuNPs) | 1–60 | Spherical Rod Polygonal Floral | High stability Low cytotoxicity in initial step Possibility of high scale production | Relatively weak optical signal Long-term cytotoxicity High price | Labelling and visualization, diagnostics, therapeutics, catalysis, cancer cell treatment |
| Silver Nanoparticles (AgNPs) | 4–120 | Spherical Wire Oval Polygonal Rod | Anti-microbacterial High optical signal | Cytotoxicity Low stability before surface treatment High price | Anti-microbial, gas/vapor sensing, water sterilization, cancer cell treatment |
| Platinum Nanoparticles (PtNPs) | 10–100 | Spherical Cuboidal Floral | Catalysis High optical-signal High stability | High price Cytotoxicity | Biosensing of molecules, enhancement of bone strength, detection of cancer cells |
| Iron Oxide Nanoparticles | 4–45 | Tube Spherical Cluster | Super-paramagnetic property Low cytotoxicity Economical | Weak strength Low stability Toxic solvent is needed | Gas sensing, magnetic resonance imaging |
| Zinc Oxide Nanoparticles | 20–600 | Flower Rod Wire Sheet | Piezo- and pyroelectric Wide range of UV absorption High optical signal Economical Anti-bacterial effect | Cytotoxicity Low stability Toxic solvent is needed | Photocatalyst, absorber of UV radiation, biosensors, gas sensing |
| Kinds of Conductive Polymers | Conductivity (mS·cm−1) | Advantages | Disadvantages | Application |
|---|---|---|---|---|
| Polypyrrole (PPy) | 103~5 × 104 | High conductivity High stability Biocompatibility High mechanical strength | Easy to Fragile Susceptible to irreversible oxidation Insoluble in water | Biosensors, antioxidants, drug delivery, neural prosthetics, tissue engineering |
| Polyaniline (PANi) | 102~108 | High conductivity High stability High conductivity Water solubility | Lack of plasticity Poor cell adhesion and growth Low solubility | Biosensors, antioxidants, drug delivery, bioactuators, food industry, tissue engineering |
| Polythiophene (PT) | 10−1~10−4 | Good optical property Biocompatibility Can obtain various functions according to the reactions | Low conductivity Low stability Low solubility | Biosensors, food industry, tissue engineering |
| Poly (3,4-ethylene dioxythiophene) (PEDOT) | 3 × 105~5 × 105 | High stability High conductivity Biocompatibility High mechanical strength Water solubility (doped with PSS) | Relatively low mechanical strength | Antioxidants, drug delivery, neural prosthetics electrode |
| Poly(p-phenylene vinylene) (PPV) | 1~1 × 105 | Its precursors can be manipulated in aqueous solution Good optical properties High stability | Insoluble in water Doping is essential to increase conductivity | Biosensors light-emitting diodes Photovoltaic devices |
| Kinds of Carbons | Conductivity (mS·cm−1) | Advantages | Disadvantages |
|---|---|---|---|
| Graphene | 108~109 | High mechanical strength High conductivity Easy synthesis | Oxidative stress Serious aggregation Toxicity Hydrophobicity |
| Graphene Oxide | Depend on oxidation and humidity (10−1~10−5) | Biocompatibility Hydrophilicity Interacting with various inorganic and organic materials Controllable electrical/optical properties | Low conductivity (or even insulator) Sensitive to humidity Weak mechanical strength |
| Carbon Nanotube (CNTs) | 107~108 | High mechanical strength High conductivity Magnetic property | Oxidative stress Toxicity Hydrophobicity Additional synthesis step |
| Methods | Advantages | Disadvantages |
|---|---|---|
| Blending synthesis | Easy and simple process No additional techniques High reproducibility High stability of conductivity | Low conductivity of hydrogel Weaken hydrogel mechanical strength Difficulty of gelation Heterogeneous conductivity |
| In situ synthesis | Homogeneous conductivity in hydrogel Enhance hydrogel strength Uniform processability High conductivity of hydrogel High stability of conductivity | Additional techniques are needed Additional step can be needed Low reproducibility |
| Coating process | Simple process Giving conductivity easily in various shapes of hydrogel | Potential for coating damage Low stability of conductivity Heterogeneous conductivity |
| Application Category | Synthesizing Method | Conductive Materials | References |
|---|---|---|---|
| Cardiac Tissue Engineering | In situ Process | poly (thiophene-3-acetic acid), TiO2NPs, CNTs | [155,162,165,167] |
| Blending Process | AuNPs, rGO-PAAm, GO, CNTs, GNR | [153,157,159,160,161,163,164] | |
| Nerve Tissue Engineering | In situ Process | PPy, rGO-CNTs | [168,170,175,176] |
| Coating Process | PPy | [169] | |
| Blending Process | CNTs, GO, FeO2, PSS-PEDOT, GO | [171,172,173,174,177] | |
| Bone Tissue Engineering | In situ Process | AuNPs, AgNPs, PANi | [184,187] |
| Coating Process | PPy | [185] | |
| Blending Process | GNPs, Graphene, PANi, PPy, Graphene | [181,182,183,186] | |
| Skin Tissue Engineering | Blending Process | PPy, PANi | [191,193] |
| In situ Process | PANi, PPy | [192] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Min, J.H.; Patel, M.; Koh, W.-G. Incorporation of Conductive Materials into Hydrogels for Tissue Engineering Applications. Polymers 2018, 10, 1078. https://doi.org/10.3390/polym10101078
Min JH, Patel M, Koh W-G. Incorporation of Conductive Materials into Hydrogels for Tissue Engineering Applications. Polymers. 2018; 10(10):1078. https://doi.org/10.3390/polym10101078
Chicago/Turabian StyleMin, Ji Hong, Madhumita Patel, and Won-Gun Koh. 2018. "Incorporation of Conductive Materials into Hydrogels for Tissue Engineering Applications" Polymers 10, no. 10: 1078. https://doi.org/10.3390/polym10101078