Photorefractive Properties of Molybdenum and Hafnium Co-Doped LiNbO3 Crystals
Abstract
:1. Introduction
2. Materials and Methods
3. Results
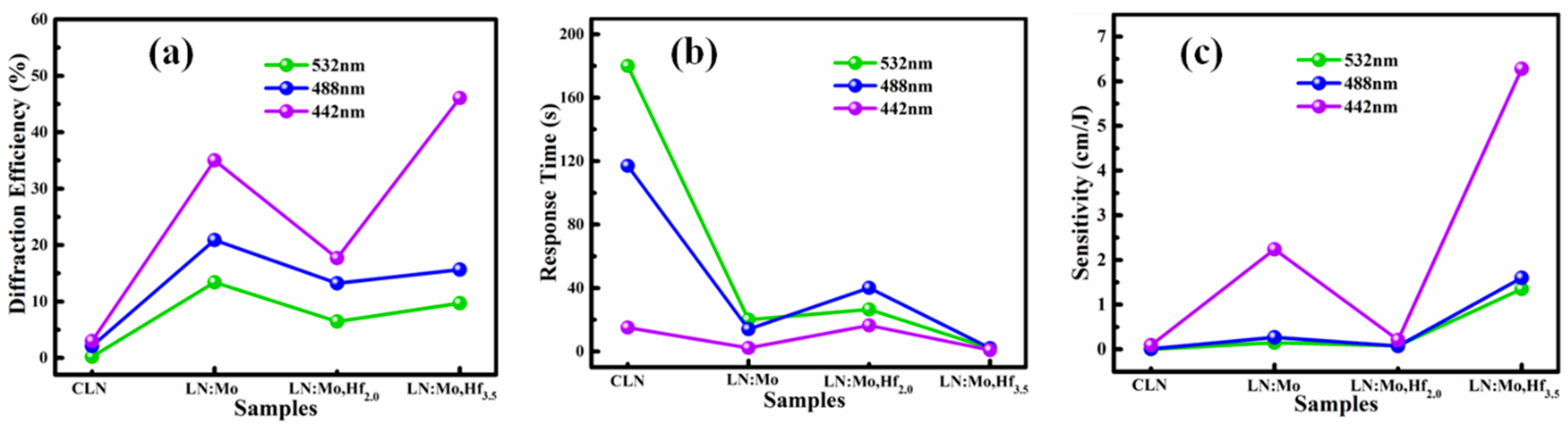
3.1. Two-Wave Coupling Measurements
3.2. Optical Damage Resistance
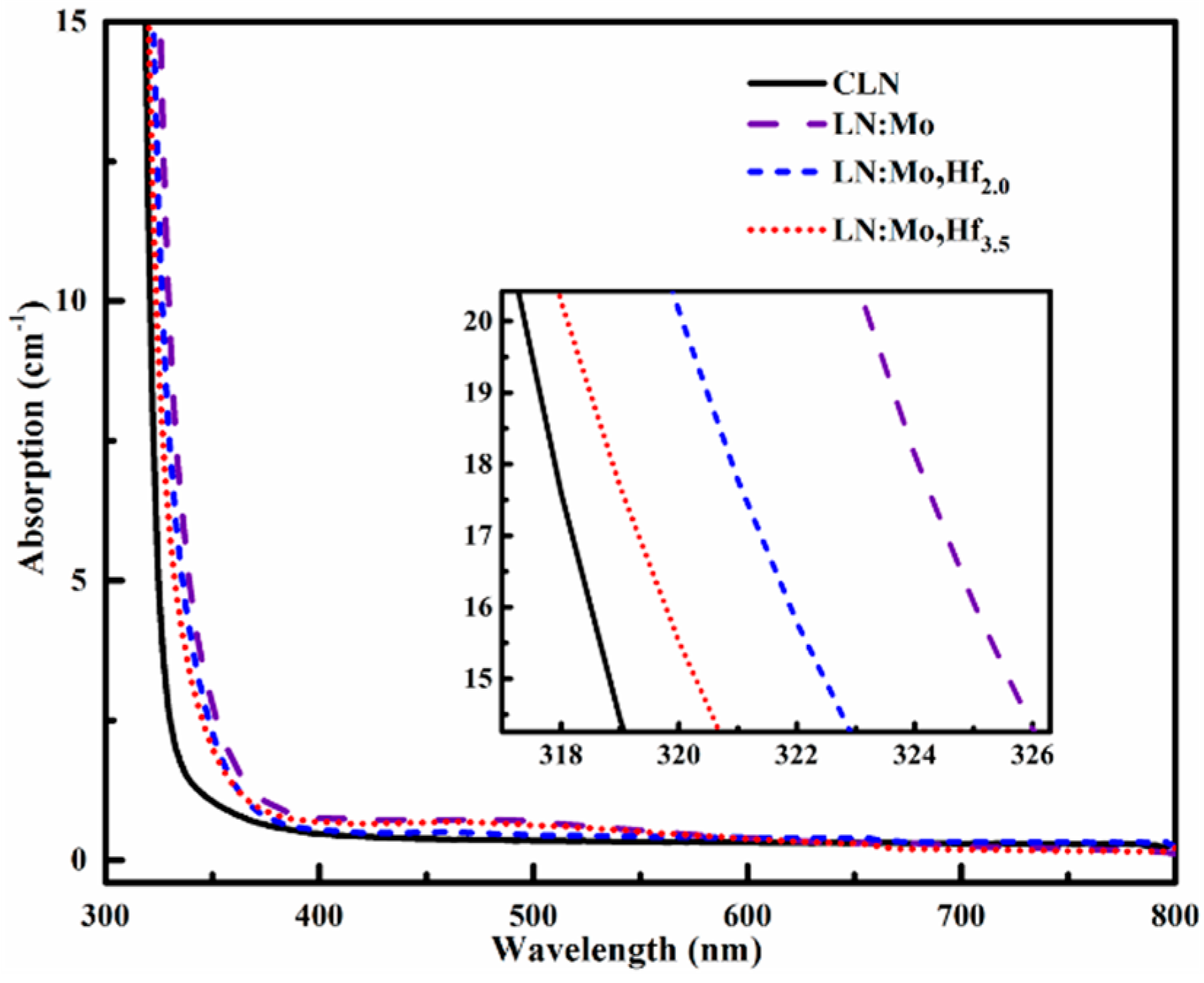
3.3. UV-Visible Absorption Spectra
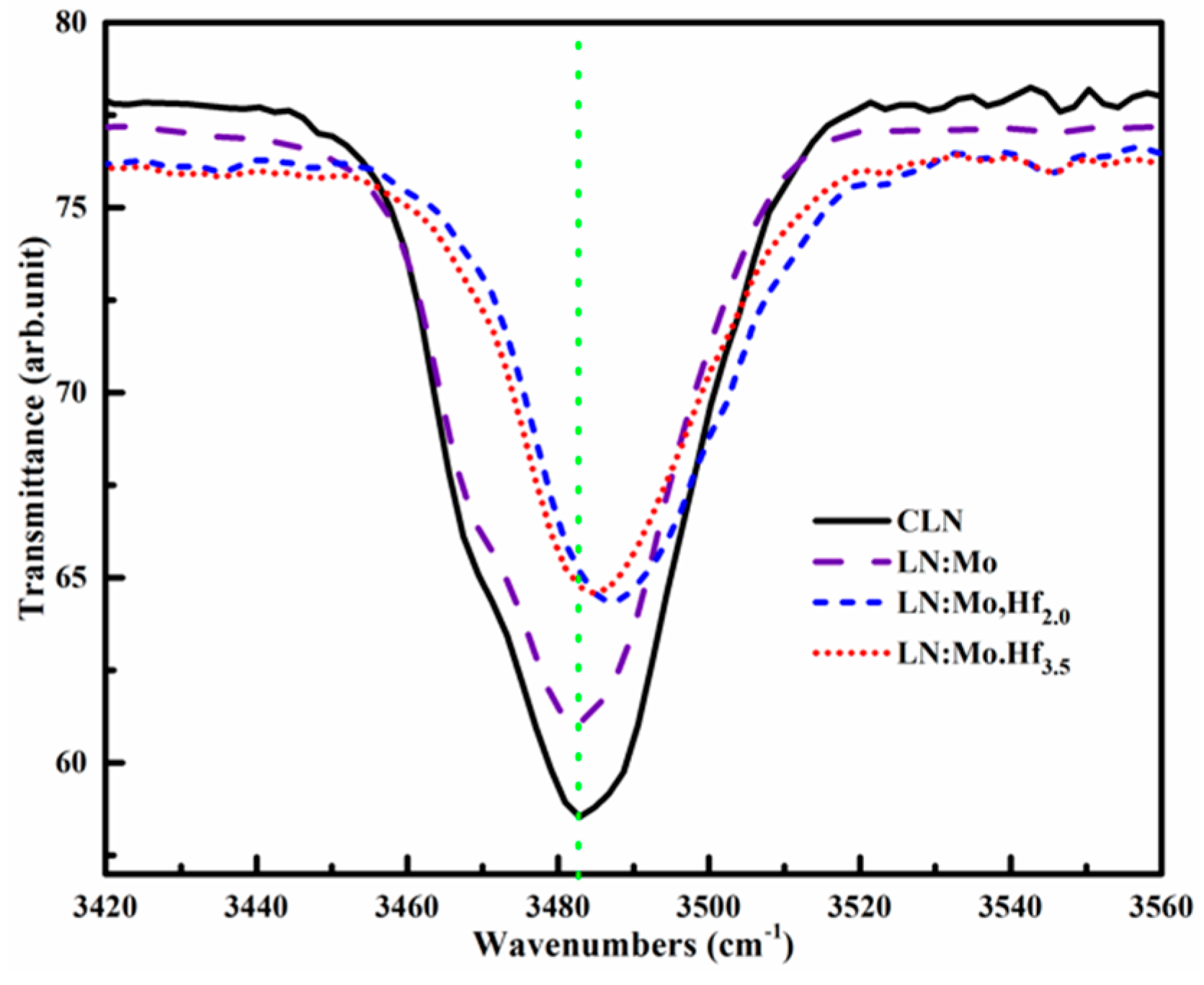
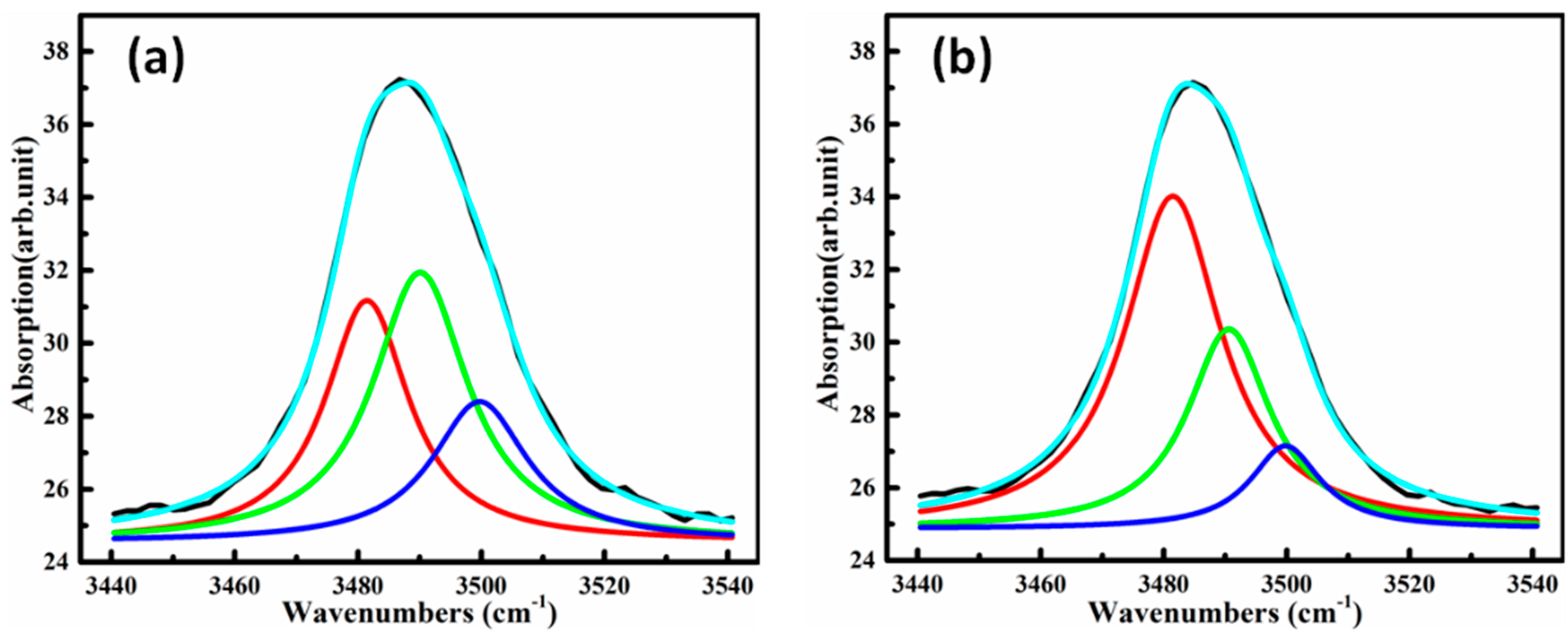
3.4. OH− Absorption Spectra
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Wang, C.; Zhang, M.; Stern, B.; Lipson, M.; Lončar, M. Nanophotonic lithium niobate electro-optic modulators. Opt. Express 2018, 26, 1547–1555. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dhar, L.; Curtis, K.; Fäcke, T. Holographic data storage: coming of age. Nat. Photonics 2008, 2, 403–405. [Google Scholar] [CrossRef]
- Wang, Y.; Zhou, S.X.; He, D.H.; Hu, Y.; Chen, H.X.; Liang, W.G.; Yu, J.H.; Guan, H.Y.; Luo, Y.H.; Zhang, J.; et al. Electro-optic beam deflection based on a lithium niobate waveguide with microstructured serrated electrodes. Opt. Lett. 2016, 41, 4739–4742. [Google Scholar] [CrossRef] [PubMed]
- Bazzan, M.; Sada, C. Optical waveguides in lithium niobate: Recent developments and applications. Appl. Phys. Rev. 2015, 2, 040603. [Google Scholar] [CrossRef]
- Jiang, H.W.; Luo, R.; Liang, H.X.; Chen, X.F.; Chen, Y.P.; Lin, Q. Fast response of photorefraction in lithium niobate microresonators. Opt. Lett. 2017, 42, 3267–3270. [Google Scholar] [CrossRef] [PubMed]
- Tu, D.; Xu, C.N.; Yoshida, A.; Fujihala, M.; Hirotsu, J.; Zheng, X.G. LiNbO3:Pr3+: A multipiezo material with simultaneous piezoelectricity and sensitive piezoluminescence. Adv. Mater. 2017, 29, 1606914. [Google Scholar] [CrossRef] [PubMed]
- Gopalan, K.K.; Janner, D.; Nanot, S.; Parret, R.; Lundeberg, M.B.; Koppens, F.H.L.; Pruneri, V. Mid-infrared pyroresistive graphene detector on LiNbO3. Adv. Opt. Mater. 2017, 5, 1600723. [Google Scholar] [CrossRef]
- Lengyel, K.; Péter, Á.; Kovács, L.; Corradi, G.; Pálfalvi, L.; Hebling, J.; Unferdorben, M.; Dravecz, G.; Hajdara, I.; Szaller, Z.; et al. Growth, defect structure, and THz application of stoichiometric lithium niobate. Appl. Phys. Rev. 2015, 2, 040601. [Google Scholar] [CrossRef] [Green Version]
- Hesselink, L.; Orlov, S.S.; Liu, A.; Akella, A.; Lande, D. Neurgaonkar, R.R. Photorefractive materials for nonvolatile volume holographic data storage. Science 1998, 282, 1089–1094. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, W.G.; Albrecht, M.; Wippermann, S.; Blankenburg, S.; Rauls, E.; Fuchs, F.; Rödl, C.; Furthmüller, J.; Hermann, A. LiNbO3 ground- and excited-state properties from first-principles calculations. Phys. Rev. B 2008, 77, 035106. [Google Scholar] [CrossRef]
- Bernert, C.; Hoppe, R.; Wittwer, F.; Woike, T.; Schroer, C.G. Ptychographic analysis of the photorefractive effect in LiNbO3:Fe. Opt. Express 2017, 25, 31640–31650. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.P.; Buse, K.; Psaltis, D. Photorefractive recording in LiNbO3:Mn. Opt. Lett. 2002, 27, 158–160. [Google Scholar] [CrossRef] [PubMed]
- Ren, L.Y.; Liu, L.R.; Liu, D.A.; Zu, J.F.; Luan, Z. Optimal switching from recording to fixing for high diffraction from a LiNbO3:Ce:Cu photorefractive nonvolatile hologram. Opt. Lett. 2004, 29, 186–188. [Google Scholar] [CrossRef] [PubMed]
- Volk, T.R.; Rubinina, N.M.; Pryalkin, V.I.; Krasnikov, V.V.; Volkov, V.V. Optical and Nonlinear Optical Investigations in LiNbO3: Mg and LiNbO3:Zn. Frroelectrics 1990, 109, 345–350. [Google Scholar] [CrossRef]
- Fang, S.F.; Han, Z.X.; Qiao, Y.J.; Liu, Y.Y.; Jia, Q. Defect structure characteristics in near-stoichiometric In: LiNbO3 single crystals grown by K2O‑flux method. Cryst. Res. Technol. 2009, 44, 1211–1214. [Google Scholar] [CrossRef]
- Minzioni, P.; Cristiani, I.; Yu, J.; Parravicini, J.; Kokanyan, E.P.; Degiorgio, V. Linear and nonlinear optical properties of Hafnium-doped lithium-niobate crystals. Opt. Express 2007, 15, 14171–14176. [Google Scholar] [CrossRef] [PubMed]
- Tian, T.; Kong, Y.F.; Liu, S.G.; Li, W.; Wu, L.; Chen, S.L.; Xu, J.J. Photorefraction of molybdenum-doped lithium niobate crystals. Opt. Lett. 2012, 37, 2679–2681. [Google Scholar] [CrossRef] [PubMed]
- Shi, H.X.; Ren, C.Y. Investigation on photorefractive properties of Hf: Fe: LiNbO3 crystals. Opt. Int. J. Light Electron Opt. 2013, 124, 3170–3172. [Google Scholar] [CrossRef]
- Fujimura, R.; Shimura, T.; Kuroda, K. Two-color nonvolatile holographic recording and light-induced absorption in Ru and Fe codoped LiNbO crystals. Opt. Mater. 2009, 31, 1194–1199. [Google Scholar] [CrossRef]
- Fan, Y.X.; Xu, C.; Wang, Y.J.; Xia, S.X.; Guan, C.X.; Cao, L.C. Influence of ZnO codoping on growth and holographic properties of Ru/Fe double-doped LiNbO3 single crystals. J. Cryst. Growth 2011, 318, 657–660. [Google Scholar] [CrossRef]
- Tian, T.; Kong, Y.F.; Liu, S.G.; Li, W.; Chen, S.L.; Rupp, R.; Xu, J.J. Fast UV-Vis photorefractive response of Zr and Mg codoped LiNbO3:Mo. Opt. Express 2013, 21, 10460. [Google Scholar] [CrossRef] [PubMed]
- Danai, M.M. Czochralski method. In Dictionary of Gems and Gemology, 3rd ed.; Springer: Berlin/Heidelberg, Germany, 2009; p. 289. ISBN 978-3-540-72795-8. [Google Scholar]
- Yan, W.B.; Shi, L.H.; Chen, H.J.; Shen, X.N.; Kong, Y.F. Investigations of the OH− absorption bands in congruent and near-stoichiometric LiNbO3: Hf crystals. EPL 2010, 91, 36002. [Google Scholar] [CrossRef]
- Smith, R.G.; Fraser, D.B.; Denton, R.T.; Rich, T.C. Correlation of reduction in optically induced refractive-index inhomogeneity with OH content in LiTaO3 and LiNbO3. J. Appl. Phys. 1968, 39, 4600–4602. [Google Scholar] [CrossRef]
- Kong, Y.F.; Zhang, W.L.; Chen, X.J.; Xu, J.J.; Zhang, G.Y. OH− absorption spectra of pure lithium niobate crystals. J. Phys. Condens. Mater. 1999, 11, 2139. [Google Scholar] [CrossRef]
- Li, S.Q.; Liu, S.G.; Kong, Y.F.; Deng, D.L.; Gao, G.Y.; Li, Y.B.; Gao, H.C.; Ling, Z.; Hang, Z.H.; Chen, S.L.; et al. The optical damage resistance and absorption spectra of LiNbO3:Hf crystals. J. Phys. Condens. Matter 2006, 18, 3527. [Google Scholar] [CrossRef]
- Kovács, L.; Szalay, V.; Capelletti, R. Stoichiometry dependence of the OH− absorption band in LiNbO3 crystals. Solid State Commun. 2015, 52, 1029–1031. [Google Scholar] [CrossRef]
- Kovács, L.; Szaller, Z.; Lengyel, K.; Corradi, G. Hydroxyl ions in stoichiometric LiNbO3, crystals doped with optical damage resistant ions. Opt. Mater. 2014, 37, 55–58. [Google Scholar] [CrossRef]

© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhu, L.; Zheng, D.; Saeed, S.; Wang, S.; Liu, H.; Kong, Y.; Liu, S.; Chen, S.; Zhang, L.; Xu, J. Photorefractive Properties of Molybdenum and Hafnium Co-Doped LiNbO3 Crystals. Crystals 2018, 8, 322. https://doi.org/10.3390/cryst8080322
Zhu L, Zheng D, Saeed S, Wang S, Liu H, Kong Y, Liu S, Chen S, Zhang L, Xu J. Photorefractive Properties of Molybdenum and Hafnium Co-Doped LiNbO3 Crystals. Crystals. 2018; 8(8):322. https://doi.org/10.3390/cryst8080322
Chicago/Turabian StyleZhu, Ling, Dahuai Zheng, Shahzad Saeed, Shuolin Wang, Hongde Liu, Yongfa Kong, Shiguo Liu, Shaolin Chen, Ling Zhang, and Jingjun Xu. 2018. "Photorefractive Properties of Molybdenum and Hafnium Co-Doped LiNbO3 Crystals" Crystals 8, no. 8: 322. https://doi.org/10.3390/cryst8080322





