3.1. Structural Properties
Table 2 gives the calculated equilibrium lattice constants and atomic Wyckoff positions of B19 TiAl in this work. From the selected-area electron diffraction patterns, Abe et al. [
2] measured the lattice parameters of B19 TiAl to be
a = 4.5 Å,
b = 2.8 Å, and
c = 4.9 Å. Ducher et al. [
5] estimated the lattice parameters of B19 TiAl to be
a = 4.65 Å,
b = 2.828 Å, and
c = 4.94 Å, in the unit cell of which Ti and Al atoms occupy the respective Wyckoff positions (1/4, 0, 5/6) and (1/4, 1/2, 1/3). Song et al. [
7] measured the lattice parameters of B19 phase to be
a = 4.64 Å,
b = 2.90 Å, and
c = 5.10 Å. From high-energy X-ray diffraction patterns, Schmoelzer et al. [
6] measured the lattice parameters of B19 phase to be
a = 4.65 Å,
b = 2.93 Å, and
c = 4.95 Å. These experimental data are also given in
Table 2 for comparison. It is found that the maximum deviations of the calculated equilibrium lattice constants
a,
b, and
c correspond to 2.94%, 2.29%, and 4.03% as compared with the respective experimental values. Furthermore, the calculated equilibrium atomic positions are in agreement with the respective experimental values [
5]. These indicate that our calculated results agree basically with the available experimental results. Moreover, the formation energy
Ef of B19 TiAl is defined as
Ef = [
E(TiAl) − 2
E(Ti) − 2
E(Al)]/4, where
E(TiAl) is the total energy of the whole unit cell, i.e., per 4 atoms, and
E(Ti) and
E(Al) correspond to the total energy per atom of Ti and Al elements in their standard states. The calculated formation energy is −0.416 eV/atom, indicating that B19 TiAl is energetically stable.
3.2. Elastic and Mechanical Properties
The relation between the variation in energy and the tensor parameter of orthorhombic B19 TiAl for each selected strain mode is fitted with a quadratic polynomial and a cubic polynomial, respectively. The second-order coefficient in the cubic polynomial fitting is found to be in agreement with that in the quadratic one because the small strains within ±0.015 may avoid the influence of higher-order terms on the computed coefficient. After numerically obtaining the second-order coefficients in the quadratic polynomials, the elastic constants
Cij can be determined using the equilibrium lattice parameters. The calculated results are listed in
Table 3. Obviously, the elastic constants obtained by using two ab initio techniques are in basic agreement with each other. The deviations are partially due to the calculation techniques and fitting errors. Unfortunately, there are no available experimental and theoretical values to check our calculated results. The obtained results in the present work could provide a useful reference for future studies. Moreover, an orthorhombic crystal will be mechanically unstable until its elastic constants satisfy the following necessary and sufficient conditions [
14]:
It is clear that the orthorhombic B19 TiAl is mechanically stable in terms of our calculated elastic constants.
From the single crystal elastic constants data, the polycrystalline bulk modulus
B and shear modulus
G can be usually calculated by using the Voigt approximation [
39] and the Reuss approximation [
40] methods. For an orthorhombic crystal, the Voigt bulk (
BV) and shear (
GV) moduli are given by
The Reuss bulk (
BR) and shear (
GR) moduli are given by
where the
Sij (=
Cij-1) are the elastic compliance constants. Hill [
41] proposed that the effective bulk (
B) and shear (
G) moduli should be the arithmetic averages of the Voigt and Reuss bulk (
BV,
BR) and shear (
GV,
GR) moduli in practice, namely
B = (
BV +
BR)/2 and
G = (
GV +
GR)/2. Further, the Young’s modulus
E and Poisson’s ratio
ν can be given by
Table 4 gives the calculated results of the polycrystalline bulk modulus
B, shear modulus
G, Young’s modulus
E, and Poisson ratio
ν of orthorhombic B19 TiAl. The
B measures the resistance of a material to a volume change. The
G measures the resistance of a material to a shape change. The
E is defined as the ratio of tensile stress to tensile strain and often used to provide a measure of the stiffness of the solid. The larger the value of
E, the stiffer the material. The calculated values of
B,
G, and
E of B19 TiAl from the EDM are lower 0.13%, 10.84%, and 9.01% lower than the corresponding theoretical values of γ-TiAl (113, 70.6, and 175 GPa) and 1.87%, 15.16%, and 12.98% lower than the corresponding theoretical values of α
2-Ti
3Al (115, 74.2, and 183 GPa) [
42]. These indicate that the plastic deformation of B19 TiAl is easier and its compliancy higher than γ-TiAl and α
2-Ti
3Al.
As mentioned in
Section 1, Pugh’s ratio (
G/
B), Cauchy pressure, and Poisson’s ratio can allow us to know the ductile/brittle nature of a material. According to the Pugh’s ratio [
11], a material behaves in a ductile manner if
G/
B < 0.5; otherwise, it should be brittle. The
G/
B ratios of 0.558 and 0.574 can be obtained from two ab initio techniques for B19 TiAl. It is clear that both
G/
B ratios are larger than 0.5. The calculated
G/
B ratios of B19 TiAl from the EDM is 10.03% and 14.18% lower than the theoretical data of γ-TiAl (0.62) and α
2-Ti
3Al (0.65) [
42], respectively, indicating that B19 TiAl is more ductile than γ-TiAl and α
2-Ti
3Al. Experimentally, Appel et al. [
3,
8,
9,
10] have measured the mechanical properties of γ-TiAl-based intermetallics with the microstructure containing B19 phase and found that the existence of B19 structure can give the intermetallics relatively high tensile ductility. According to Pettifor’s rule [
12], a material has more metallic (angular) bonds and thus is more ductile (brittle) if it has a larger positive (negative) Cauchy pressure. For the orthorhombic system, the Cauchy pressures can be defined as
C23−
C44 for the (100) plane,
C13−
C55 for the (010) plane, and
C12−
C66 for the (001) plane. Based on the single crystal elastic constants obtained from the two techniques, the calculated values are 28.91 GPa and 23.50 GPa for the pressure (
C23−
C44), −7.22 GPa and −6.77 GPa for the pressure (
C13−
C55), and −2.34 GPa and −6.63 GPa for the pressure (
C12−
C66) for B19 TiAl. The results indicate that the bonding in the (100) plane have a strong metallic character, whereas those in the (010) and (010) planes have an angular character. According to Frantsevich’s rule [
13], a material is brittle if its Poisson’s ratio is less than 1/3, otherwise the material is ductile. The Poisson’s ratios of 0.265 and 0.259 can be obtained from the two techniques for B19 TiAl. It is clear that both of the Poisson’s ratios are also less than 1/3. From the above, it can be clearly shown that B19 TiAl is brittle in nature.
The elastic constants
C11,
C22, and
C33 and
C44,
C55,
C66,
C12,
C13, and
C23 are closely related to the elasticity of length and shape, respectively. As shown in
Table 2, the elastic constant
C11 is larger than
C22 and smaller than
C33 for B19 TiAl, implying the anisotropy of its elasticity. The elastic anisotropy can be described by the bulk modulus (
Ba,
Bb, and
Bc) along the axes
a,
b, and
c [
34]. The axial bulk modulus are given by
With respect to the
b axis, the anisotropies of the bulk modulus along the axes
a and
c are defined as
The calculated values of
Ba,
Bb,
Bc, and
Aa,
Ac are listed in
Table 5. It is clear that the value of
Ba is significantly smaller than those of
Bb and
Bc for two different ab initio techniques, whereas in both cases the value of
Bb is very close to that of
Bc. The results indicate that the compressibility along the
a axis is the largest whereas those along the axes
b and
c are very close to each other. Additionally, it is found that the value of
Aa is significantly smaller than one, whereas that of
Ac is very close to one. For an isotropic crystal the values of
Aa and
Ac must be one, while any departures from one measure the anisotropic degree of the crystal. Hence, B19 TiAl is an elastic anisotropic crystal.
In addition to the axial bulk modulus, the shear anisotropy factors (
A{100},
A{010}, and
A{001}) in the atomic bonding for {100}, {010}, and {001} planes can be calculated in terms of the elastic constants [
34], which are given by
For an isotropic crystal the values of
A{100},
A{010}, and
A{001} must be one, while any departures from one are a measure of the anisotropic degree of the crystal. Chung and Buessem [
43] have introduced the percent anisotropy factors of bulk modulus (
AB) and shear modulus (
AG) to measure the crystal anisotropy, which are defined as
Ranganathan and Ostoja-Starzewski [
44] have introduced a universal anisotropy factor to measure the crystal anisotropy, which is defined as
For an isotropic crystal the values of
AB,
AG, and
AU must be zero, while any departures from zero are a measure of the anisotropic degree of the crystal. The calculated values of various anisotropy factors are listed in
Table 6. It is clear that the values of
A{100},
A{010}, and
A{001} are all departed from one and the
A{100} has the largest absolute deviation from one, which indicates that the shear anisotropy for the {100} plane is the strongest. Additionally, it is observed that all of
AB,
AG, and
AU have nonzero positive values, and the value of
AG is significantly larger than that of
AB. These show that B19 TiAl is elastic anisotropic and exhibits a small anisotropy of bulk modulus but a high anisotropic degree of shear modulus.
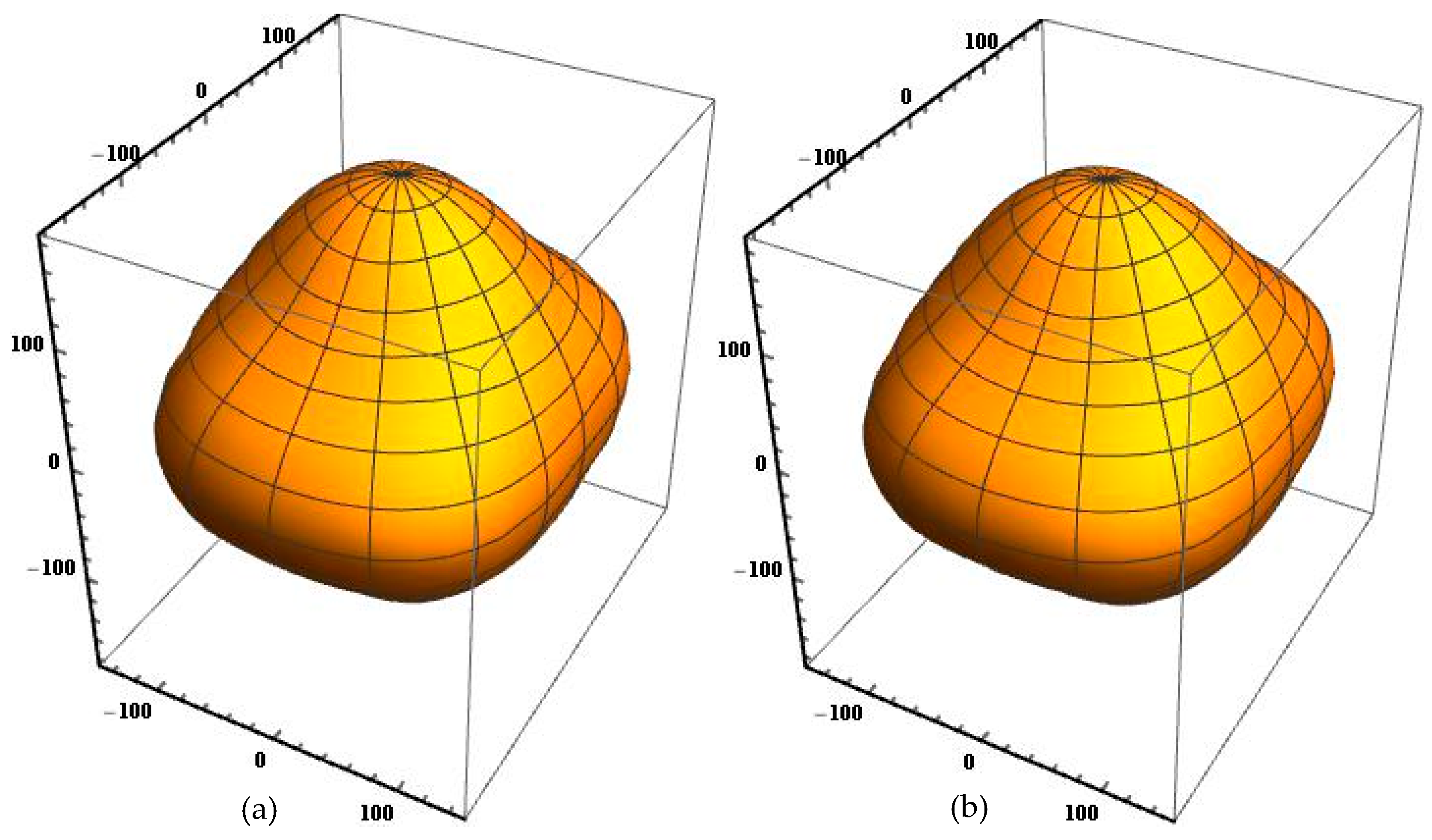
Nevertheless, these factors are not enough for the complete description of the elastic anisotropic behavior of the crystals. As a matter of fact, the orientation dependence of Young’s modulus
E is usually employed to analyze the elastic anisotropy of the crystals. For an orthorhombic crystal, the Young’s modulus in any orientation is given by [
45]
where
l1,
l2, and
l3 are direction cosines.
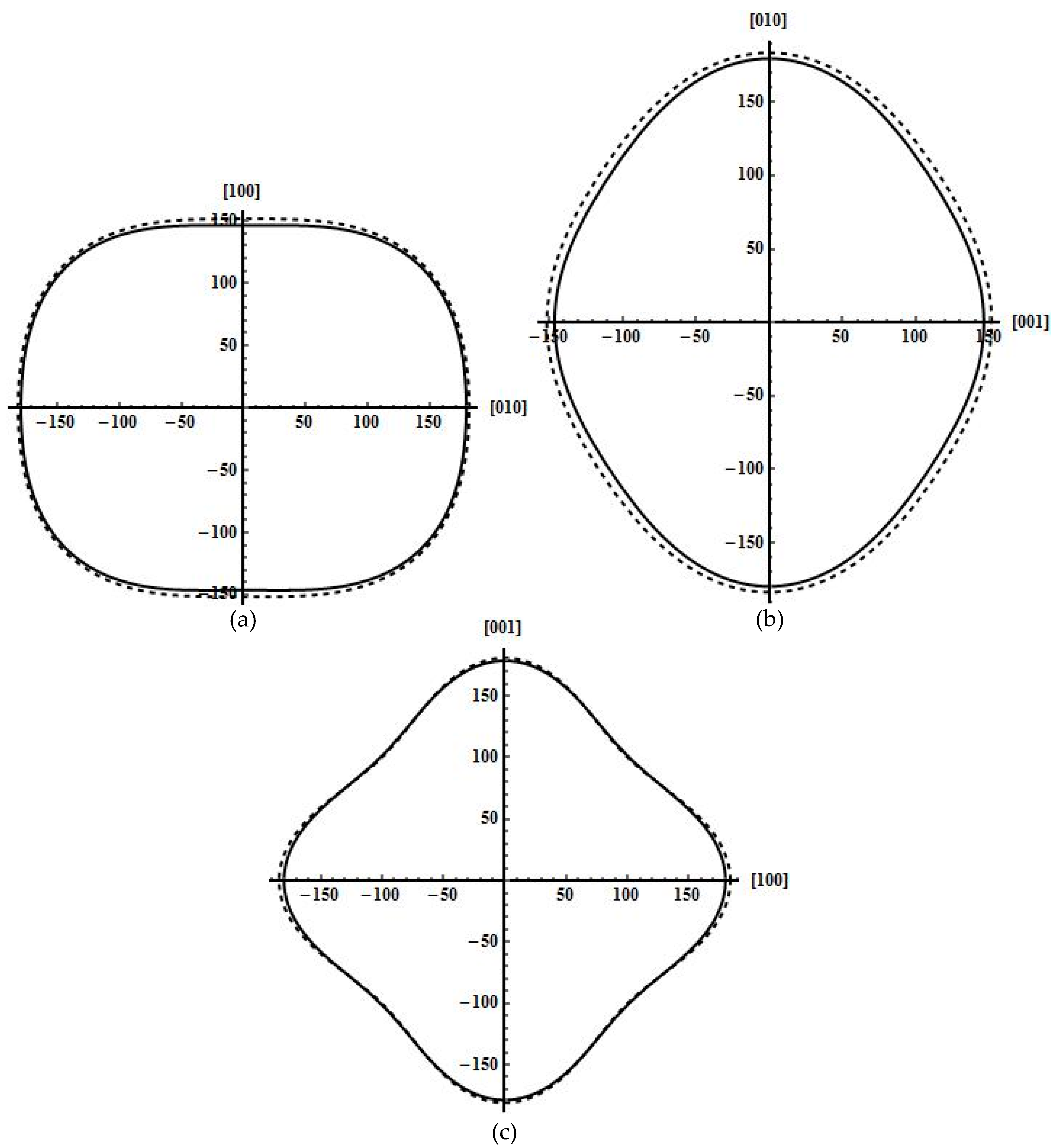
Figure 1 shows the orientation dependent Young’s modulus of B19 TiAl calculated by using the elastic compliance constants obtained from two different ab initio techniques. It is clear that the Young’s modulus surface of the energy density method is overall similar in contour to that of the strain–stress method. The Young’s modulus surface is a perfect spherical surface for an isotropic crystal, but are both not the case for B19 TiAl. Furthermore, the orientation dependence of Young’s modulus in the
ab,
bc, and
ca planes are shown in
Figure 2a–c, respectively. The orbit of Young’s modulus in any plane should be a perfect circle for an isotropic crystal, but are also not the case for B19 TiAl. These clearly show that B19 TiAl exhibit elastic anisotropy.