Structures and Magnetic Properties of Iron(III) Complexes with Long Alkyl Chains
Abstract
:1. Introduction
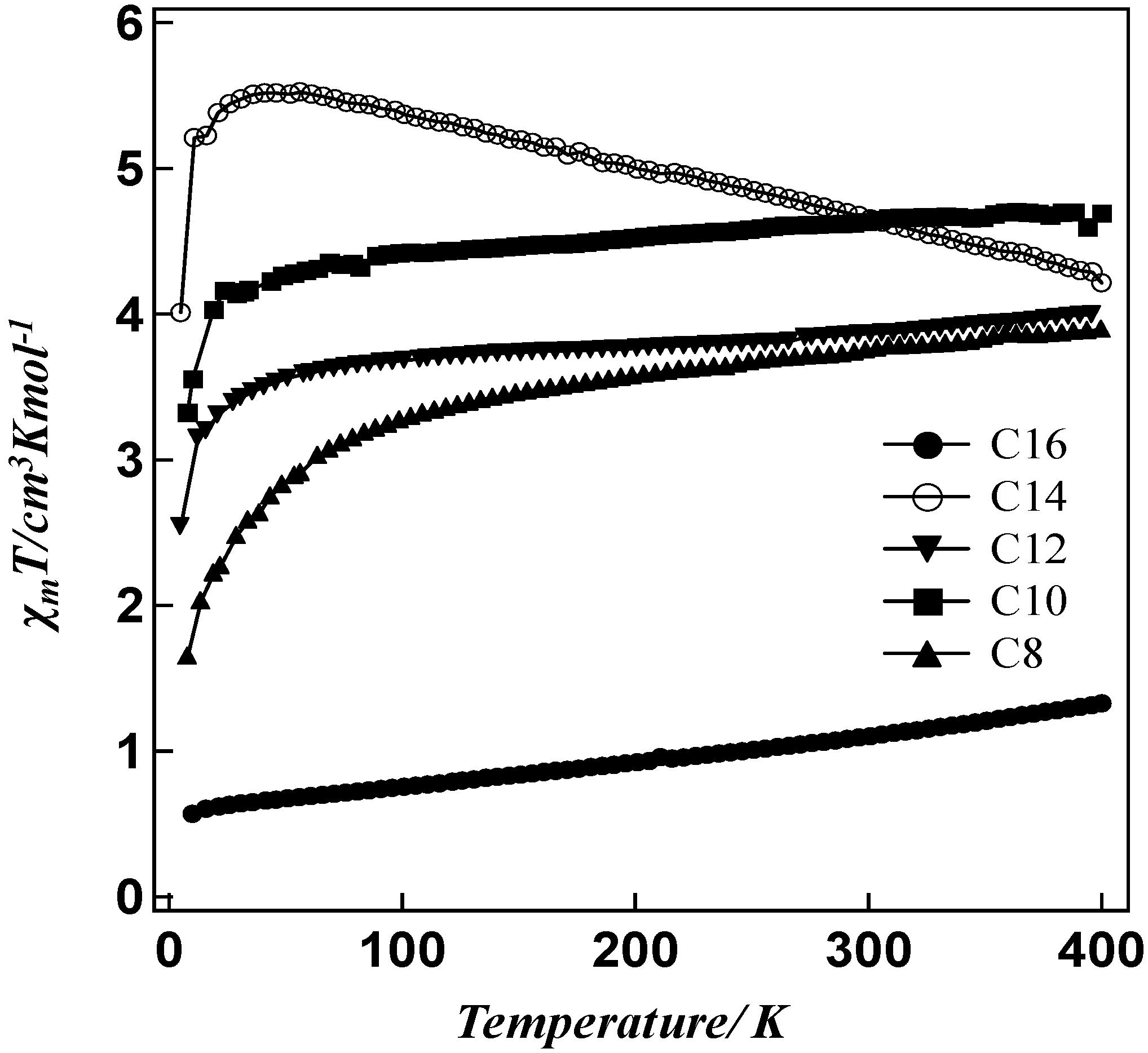
2. Results and Discussion
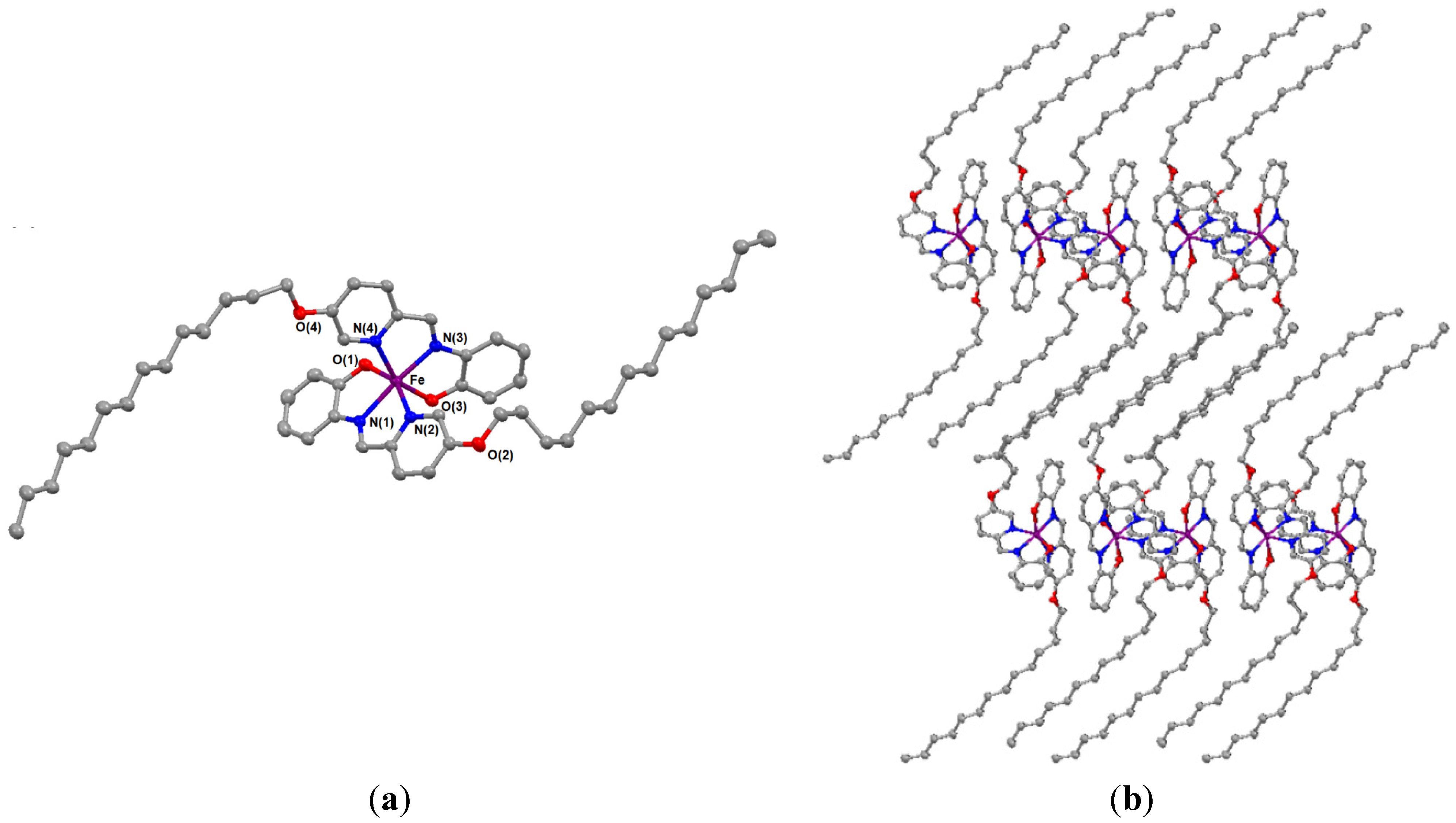
| Compound | 4 | Metal-ligand Ligand-metal-ligand | Bond lengths (Å) and angles (°) |
|---|---|---|---|
| CCDC Number [17] | 988486 | Fe–N(1) | 2.109 |
| Chemical form | C52H74ClFeN4O8 | Fe–N(2) | 2.207 |
| Mr | 974.48 | Fe–N(3) | 2.112 |
| Crystal size/mm3 | 0.200 × 0.180 × 0.020 | Fe–N(4) | 2.221 |
| Crystal system | triclinic | Fe–O(1) | 1.935 |
| Space group | P-1 (#2) | Fe–O(3) | 1.936 |
| a/Å | 10.6395(10) | – | – |
| b/Å | 12.2937(10) | O(1)–Fe–N(1) | 78.75(8) |
| c/Å | 22.129(3) | O(1)–Fe–N(2) | 152.76(8) |
| α/°C | 74.026(9) | O(1)–Fe–N(3) | 103.78(9) |
| β/°C | 84.735(11) | O(1)–Fe–N(4) | 90.98(7) |
| γ/°C | 65.255(9) | O(1)–Fe–O(3) | 99.79(7) |
| V/Å3 | 2526.2(5) | O(3)–Fe–N(1) | 113.24(7) |
| Z | 2 | O(3)–Fe–N(2) | 88.55(8) |
| Dχ/g·cm−3 | 1.281 | O(3)–Fe–N(3) | 78.87(7) |
| μ/mm−1 | 0.41 | O(3)–Fe–N(4) | 152.42(7) |
| F(000)/e | 1042 | N(1)–Fe–N(2) | 74.15(8) |
| T/K | 143 | N(1)–Fe–N(3) | 167.26(6) |
| θmax/° | 27.4 | N(1)–Fe–N(4) | 93.67(7) |
| h, k, l range | −13 ≤ h ≤ 13 | N(2)–Fe–N(3) | 103.26(8) |
| −15 ≤ k ≤ 15 | N(2)–Fe–N(4) | 93.37(8) | |
| −28 ≤ l ≤ 28 | N(3)–Fe–N(4) | 73.90(7) | |
| Measured reflections | 28551 | – | – |
| Independent reflections (Rint) | 11,396 (0.0499) | – | – |
| Observed reflections [I ≥ 2σ(I)] | 9,058 | – | – |
| Restraints/parameters | 597 | – | – |
| R1/wR2 [I ≥ 2σ(I)] | 0.0685/0.1811 | – | – |
| R1/wR2 (all data) | 0.0847/0.1811 | – | – |
| Δρmax/min/e Å−3 | 0.74/−0.108 | – | – |
3. Experimental Section
3.1. Synthesis of 5-Hydroxy-2-Methylpyridine N-Oxide
3.2. Synthesis of 2-Pyridylmethanol Acetate
3.3. Synthesis of 5-Hydroxy-2-Hydroxymethylpyridine
3.4. Synthesis of 5-Hydroxypyridine 2-Carbaldehyde
3.5. Synthesis of 5-(n-alkoxy)Pyridine 2-Carbaldehyde (n = 8, 10, 12, 14, 16)
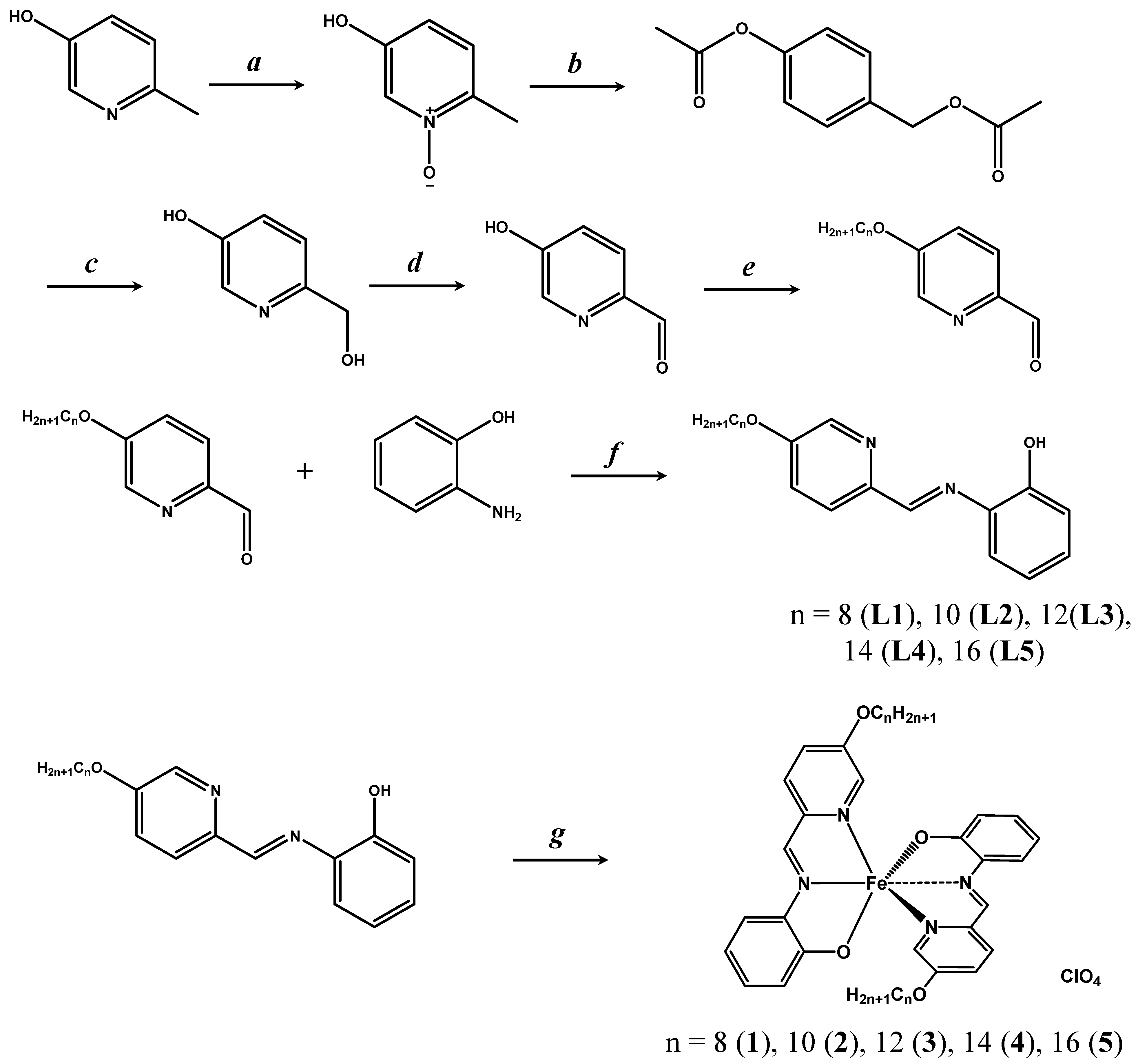
3.6. Synthesis of Cn-pap (n = 8, 10, 12, 14, 16) (L1)–(L5)
3.7. Synthesis of [Fe(Cn-pap)2]ClO4 (n = 8, 10, 12, 14, 16) (1)–(5)
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References and Notes
- Mizutani, Y.; Kitagawa, T. Ultrafast structural relaxation of myoglobin following photodissociation of carbon monoxide probed by time-resolved resonance Raman spectroscopy. J. Phys. Chem. B. 2001, 105, 10992–10999. [Google Scholar] [CrossRef]
- Galyametdinov, Y.; Ksenofontov, V.; Prosvirin, A.; Ovchinnikov, I.; Ivanova, G.; Gütlich, P.; Haase, W. First example of coexistence of thermal spin transition and liquid-crystal properties. Angew. Chem. Int. Ed. 2001, 40, 4269–4271. [Google Scholar] [CrossRef]
- Hayami, S.; Danjobara, K.; Inoue, K.; Ogawa, Y.; Matsumoto, N.; Maeda, Y. A photoinduced spin transition iron(II) complex with liquid-crystal properties. Adv. Mater. 2004, 16, 869–872. [Google Scholar] [CrossRef]
- Fujigaya, T.; Jiang, D.L.; Aida, T. Spin-crossover dendrimers: Generation number-dependent cooperativity for thermal spin transition. J. Am. Chem. Soc. 2005, 127, 5484–5489. [Google Scholar] [CrossRef]
- Kuroiwa, K.; Shibata, T.; Takada, A.; Nemoto, N.; Kimizuka, N. Heat-set gel-like networks of lipophilic Co(II) triazole complexes in organic media and their thermochromic structural transitions. J. Am. Chem. Soc. 2004, 126, 2016–2021. [Google Scholar] [CrossRef]
- Bodenthin, Y.; Pietsch, U.; Möhwald, H.; Kurth, D.G. Inducing spin crossover in metallo-supramolecular polyelectrolytes through an amphiphilic phase transition. J. Am. Chem. Soc. 2005, 127, 3110–3114. [Google Scholar] [CrossRef]
- Armand, F.; Badoux, C.; Bonville, P.; Ruaudel-Teixier, A.; Kahn, O. Blodgett films of spin transition iron(II) metalloorganic polymers. 1. Iron (II) complexes of octadecyl-1,2,4-triazole. Langmuir 1995, 11, 3467–3472. [Google Scholar] [CrossRef]
- Létard, J.F.; Nguyen, O.; Soyer, H.; Mingotaud, C.; Delhaès, P.; Kahn, O. First evidence of the LIESST effect in a Langmuir-Blodgett film. Inorg. Chem. 1999, 38, 3020–3021. [Google Scholar] [CrossRef]
- Jiang, D.L.; Aida, T. Photoisomerization in dendrimers by harvesting of low-energy photons. Nature 1997, 388, 454–456. [Google Scholar] [CrossRef]
- Zhang, W.; Zhao, F.; Liu, T.; Yuan, M.; Wang, Z.-M.; Gao, S. Spin crossover in a series of iron(II) complexes of 2-(2-alkyl-2H-tetrazol-5-yl)-1,10-phenanthroline: Effect of alkyl side chain, solvent, and anion. Inorg. Chem. 2007, 46, 2541–2555. [Google Scholar] [CrossRef]
- Hayami, S.; Gu, Z.-Z.; Shiro, M.; Einaga, Y.; Fujishima, A.; Sato, O. First observation of light-induced excited spin state trapping for an iron(III) complex. J. Am. Chem. Soc. 2000, 122, 7126–7127. [Google Scholar] [CrossRef]
- Hayami, S.; Gu, Z.-Z.; Yoshiki, H.; Fujishima, A.; Sato, O. Iron(III) spin-crossover compounds with a wide apparent thermal hysteresis around room temperature. J. Am. Chem. Soc. 2001, 123, 11644–11650. [Google Scholar] [CrossRef]
- Juhasz, G.; Hayami, S.; Sato, O.; Maeda, Y. Photo-induced spin transition for iron(III) compounds with π–π interactions. Chem. Phys. Lett. 2002, 364, 164–170. [Google Scholar] [CrossRef]
- Hayami, S.; Hiki, K.; Kawahara, T.; Maeda, Y.; Urakami, D.; Inoue, K.; Ohama, M.; Kawata, S.; Sato, O. Photo-induced spin transition of iron(III) compounds with π–π intermolecular interactions. Chem. Eur. J. 2009, 15, 3497–3508. [Google Scholar] [CrossRef]
- Harding, D.J.; Phonsri, W.; Harding, P.; Gass, I.A.; Murray, K.S.; Moubaraki, B.; Cashion, J.D.; Liu, L.; Telfer, S.G. Abrupt spin crossover in an iron(III) quinolylsalicylaldimine complex: Structural insights and solvent effects. Chem. Commun. 2013; 49, 6340–6342. [Google Scholar]
- Crystal data for 4 at 143 K: fw = 974.48; colorless prism, crystal dimensions of 0.200 mm × 0.180 mm × 0.020 mm, triclinic, space group P-1; a = 10.6395(10) Å, b = 12.2937(10) Å, c = 22.129(3) Å, α = 74.026(9)°, β = 84.735(11)°, γ = 65.255(9)°, V = 2526.2(5) Å3, Z = 2, Dcalc = 1.281 g·cm−3, R1 = 0.0685 for I > 2σ(I), R = 0.049 and Rw = 0.181 for all data with a linear absorption coefficient μ(Mo Kα) = 4.073 cm−1.
- CCDC 988486 contains the supplementary crystallographic data for this paper. These data can be obtained free of charge via http://www.ccdc.cam.ac.uk/conts/retrieving.html (or from the CCDC, 12 Union Road, Cambridge CB2 1EZ, UK; Fax: +44 1223 336033; E-mail: [email protected]).
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Nakaya, M.; Shimayama, K.; Takami, K.; Hirata, K.; Amolegbe, S.A.; Nakamura, M.; Lindoy, L.F.; Hayami, S. Structures and Magnetic Properties of Iron(III) Complexes with Long Alkyl Chains. Crystals 2014, 4, 104-112. https://doi.org/10.3390/cryst4020104
Nakaya M, Shimayama K, Takami K, Hirata K, Amolegbe SA, Nakamura M, Lindoy LF, Hayami S. Structures and Magnetic Properties of Iron(III) Complexes with Long Alkyl Chains. Crystals. 2014; 4(2):104-112. https://doi.org/10.3390/cryst4020104
Chicago/Turabian StyleNakaya, Manabu, Kodai Shimayama, Kohei Takami, Kazuya Hirata, Saliu Alao Amolegbe, Masaaki Nakamura, Leonald F. Lindoy, and Shinya Hayami. 2014. "Structures and Magnetic Properties of Iron(III) Complexes with Long Alkyl Chains" Crystals 4, no. 2: 104-112. https://doi.org/10.3390/cryst4020104




