Electroless Nickel-Based Catalyst for Diffusion Limited Hydrogen Generation through Hydrolysis of Borohydride
Abstract
:1. Introduction
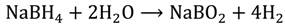
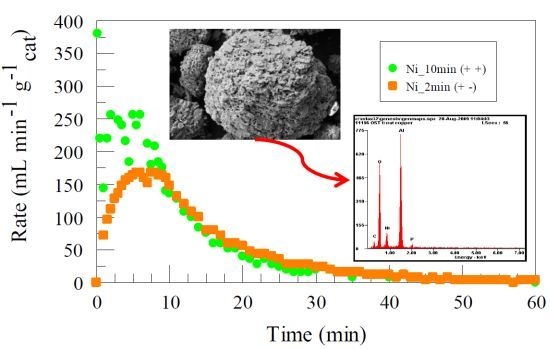
2. Results and Discussion
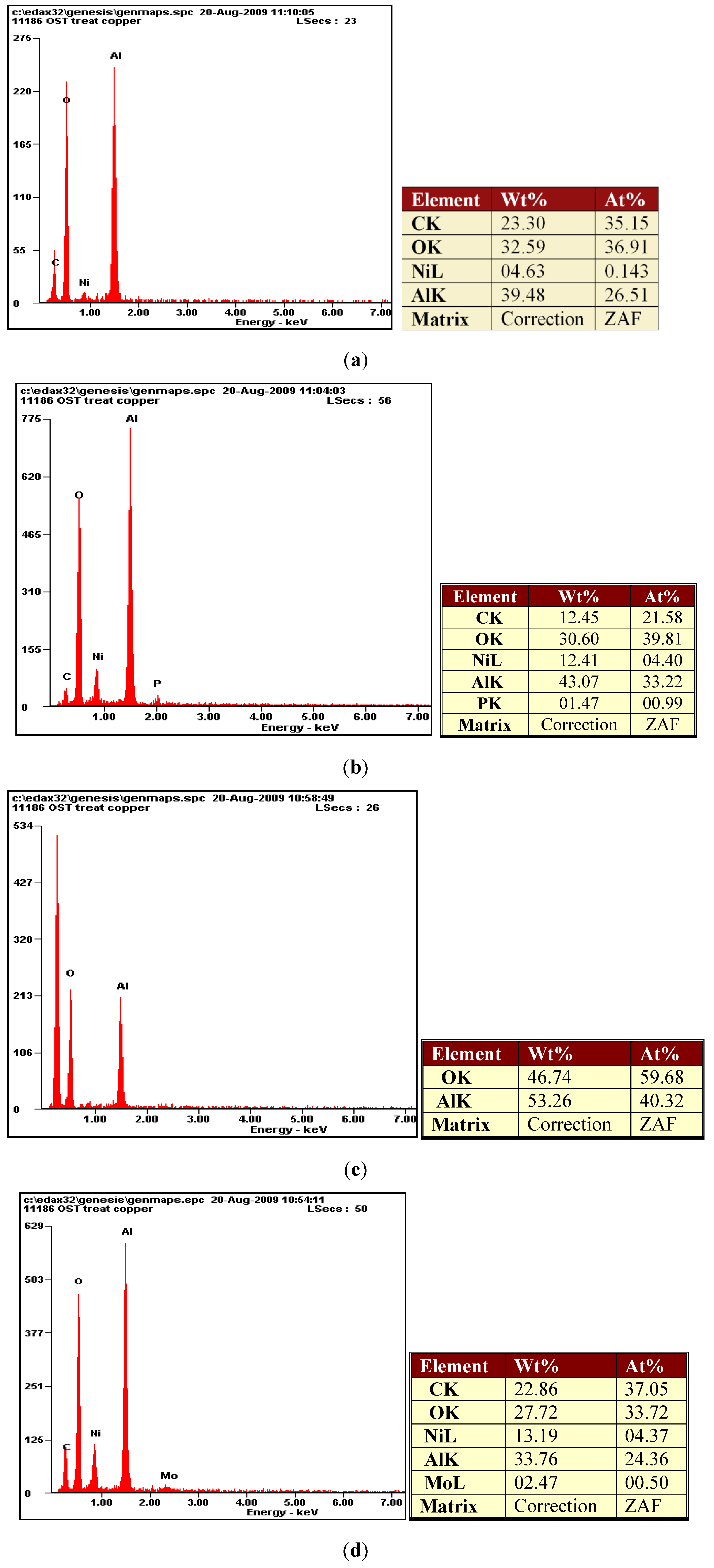
2.1. Surface Characterization and Composition

2.2. Design of Experiment
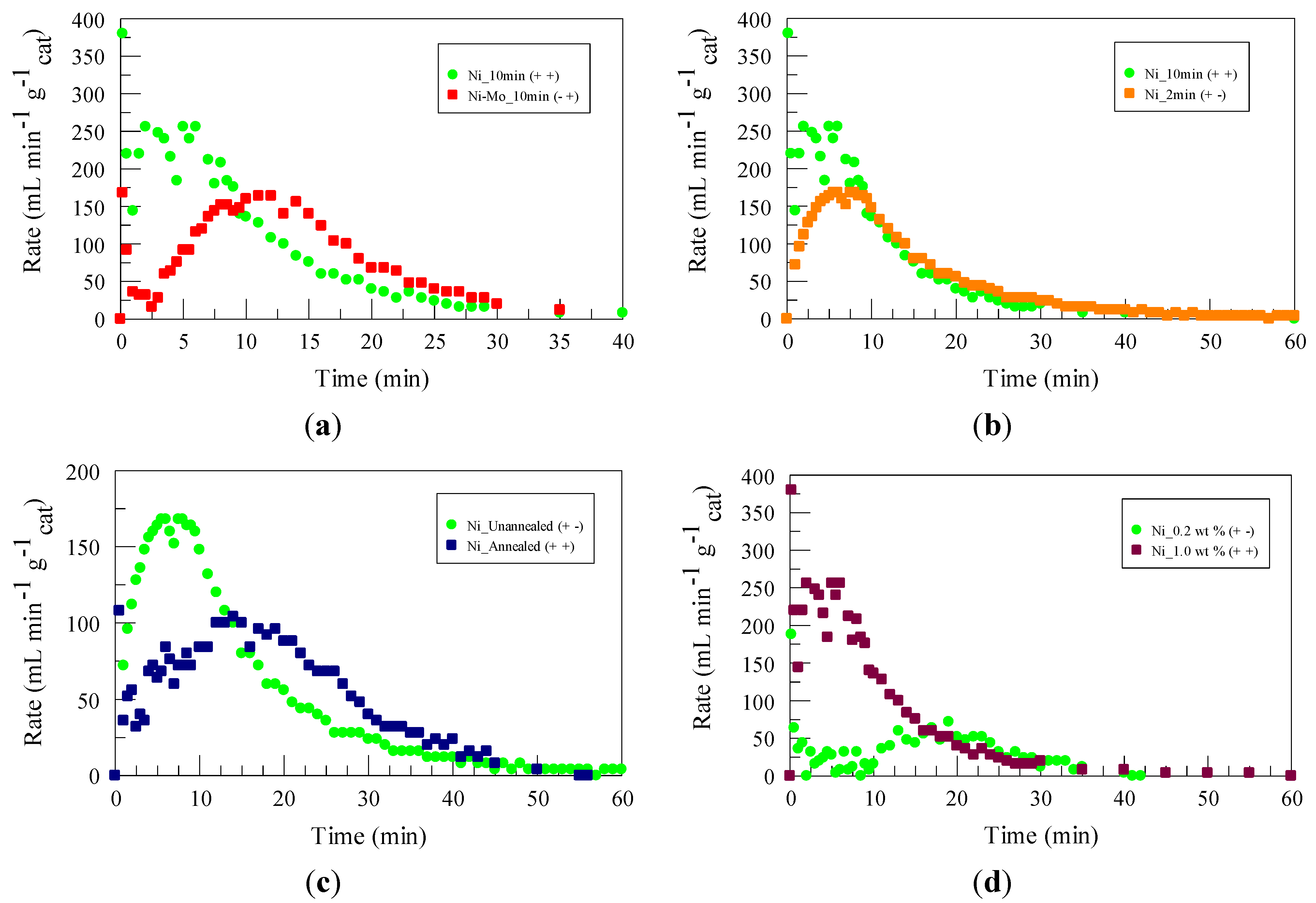
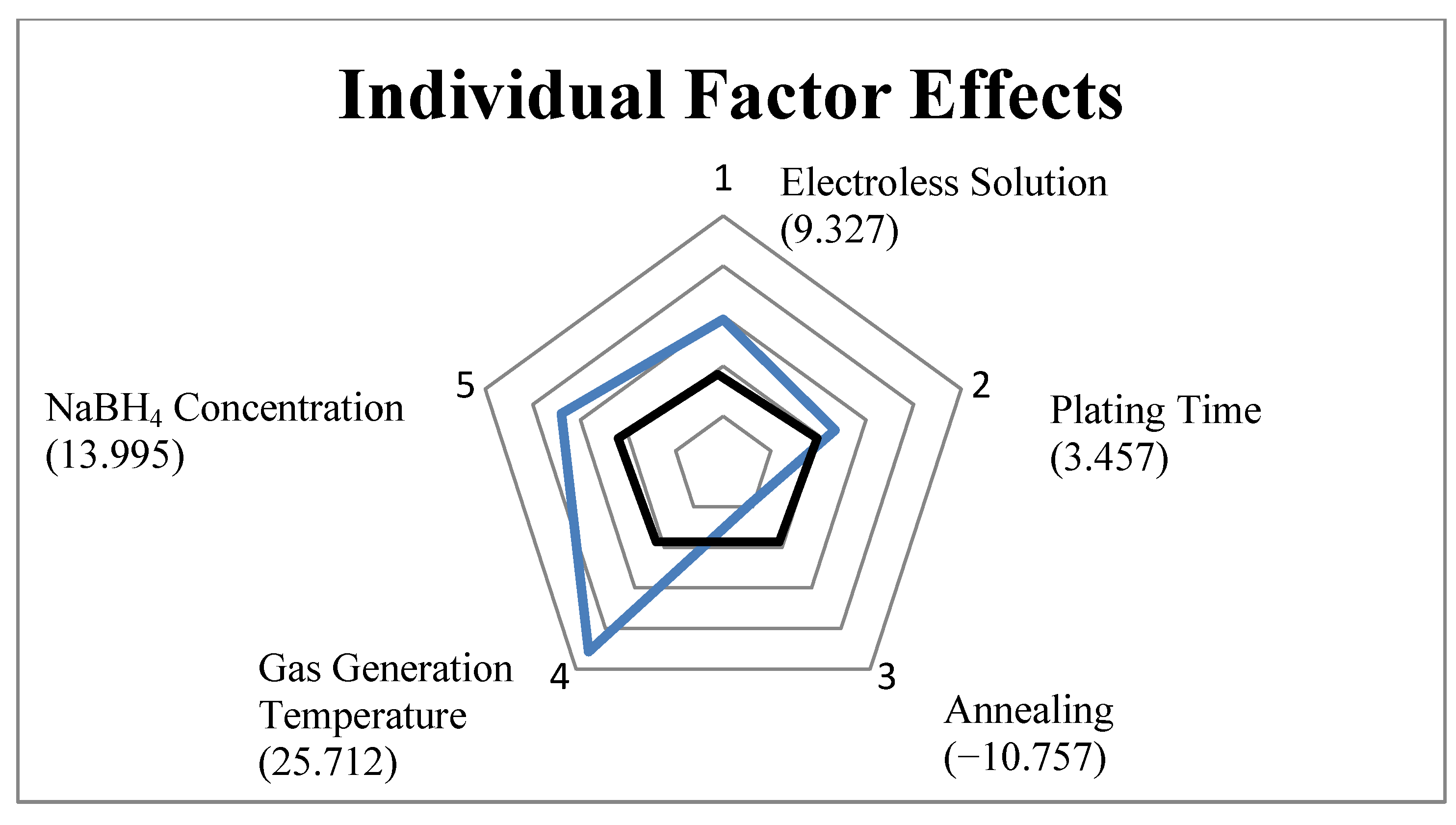
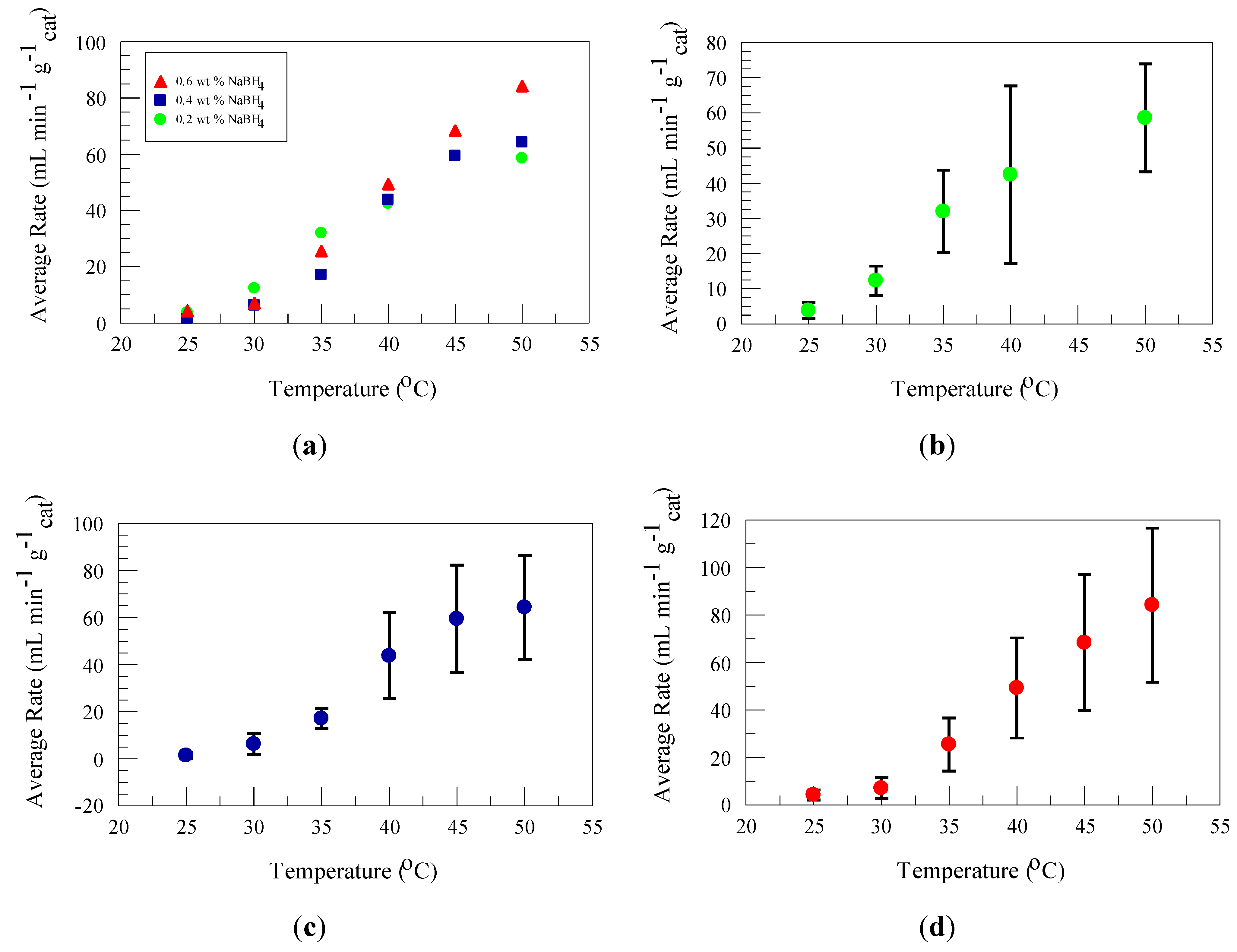
2.3. Temperature and Concentration Effects
2.4. Kinetic Parameters
 , versus the natural log of the NaBH4 concentration obtained at each temperature was slightly greater than zero, but considering the errors inherent in measuring system, we assumed that the kinetics of the hydrolysis is a zero order reaction, in agreement with published literature work [25,26]. Thus, the reaction rate equation, with a modified rate constant, k′, and a modified Arrhenius exponential factor, ko′, is:
, versus the natural log of the NaBH4 concentration obtained at each temperature was slightly greater than zero, but considering the errors inherent in measuring system, we assumed that the kinetics of the hydrolysis is a zero order reaction, in agreement with published literature work [25,26]. Thus, the reaction rate equation, with a modified rate constant, k′, and a modified Arrhenius exponential factor, ko′, is:
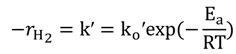
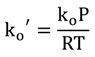

3. Experimental Section
3.1. Preparing Ni-P/Pd-Al2O3 and Ni-Mo-P/Pd-Al2O3
| Material | Quantity (g/L) |
|---|---|
| Sodium Hypophosphite | 10 |
| Gluconic Acid | 10 |
| NaK Tartrate | 10 |
| Nickel Sulfate | 7 |
| Boric Acid | 10 |
| EDTA | 5 |
| Glycine | 10 |
| Material | Quantity (g/L) |
|---|---|
| Sodium Hypophosphite | 10 |
| Gluconic Acid | 15 |
| NaK Tartrate | 10 |
| Nickel Sulfate | 7 |
| Sodium Molybdenum Oxide | 3.94 |
3.2. Testing Catalytic Activity
| Factor | Electroless Solution | Plating Time (min) | Annealing (under N2) | Gas Generation Temperature (°C) | NaBH4 Concentration |
|---|---|---|---|---|---|
| Factor number | 1 | 2 | 3 | 4 | 5 |
| + | Ni | 10 | Yes | 50 | 1.0 wt % |
| − | Ni-Mo | 2 | No | 25 | 0.2 wt % |
4. Conclusions
Acknowledgments
Conflict of Interest
References and Notes
- Chamoun, R.; Demirci, U.B.; Zaatar, Y.; Khoury, A.; Miele, P. Co-αAl2O3-Cu as shaped catalyst in NaBH4 hydrolysis. Int. J. Hydrog. Energy 2010, 35, 6583–6591. [Google Scholar] [CrossRef]
- In accordance with the USDOE definitions, the term “gravimetric hydrogen storage capacity” (GHSC) is used for storage system taken as a whole (i.e., including all components such as the hydride, tank, valves and so on). Hence, we use the term “hydrogen density” (HD) as the theoretical storage capacity of the hydride (wt %).
- Chandra, M.; Xu, Q. A high-performance hydrogen generation system: Transition metal-catalyzed dissociation and hydrolysis of ammonia-borane. J. Power Sources 2006, 156, 190–194. [Google Scholar] [CrossRef]
- Jiang, H.L.; Xu, Q. Catalytic hydrolysis of ammonia borane for chemical hydrogen storage. Catal. Today 2011, 170, 56–63. [Google Scholar] [CrossRef]
- Yadav, M.; Xu, Q. Liquid-phase chemical hydrogen storage materials. Energy Environ. Sci. 2012, 5, 9698–9725. [Google Scholar] [CrossRef]
- Lu, Z.-H. Recent Progress in Boron- and Nitrogen-Based Chemical Hydrogen Storage. Funct. Mater. Lett. 2012, 5, 1230001:1–1230001:9. [Google Scholar]
- Kim, D.R.; Cho, K.W.; Choi, Y.I.; Park, C.J. Fabrication of porous Co-Ni-P catalysts by electrodeposition and their catalytic characteristics for the generation of hydrogen for an alkaline NaBH4 solution. Int. J. Hydrog. Energy 2009, 34, 2622–2623. [Google Scholar] [CrossRef]
- Eom, K.S.; Kwon, H.S. Effects of deposition time on the H2 generation kinetics of electroless-deposited cobalt-phosphorous catalyst from NaBH4 hydrolysis, and its cyclic durability. Int. J. Hydrog. Energy 2010, 35, 5220–5221. [Google Scholar] [CrossRef]
- Peng, B.; Chen, D. Ammonia borane as an efficient and lightweight hydrogen medium. Energy Environ. Sci. 2008, 1, 479–483. [Google Scholar]
- Umegaki, T.; Yan, J.M.; Zhang, X.B.; Shioyama, H.; Kuriyama, N.; Xu, Q. Boron- and nitrogen-based chemical hydrogen storage materials. Int. J. Hydrog. Energy 2008, 34, 2303–2311. [Google Scholar]
- Demirci, U.B.; Miele, P. Sodium borohydride versus ammonia borane, in hydrogen storage and direct fuel cell applications. Energy Environ. Sci. 2009, 2, 627–637. [Google Scholar] [CrossRef]
- Sahin, Ö.; Dolas, H.; Kaya, M.; Izgi, M.S.; Demir, H. Hydrogen production from sodium borohydride for fuel cells in presence of electrical field. Int. J. Energy Res. 2010, 34, 557–558. [Google Scholar]
- Liu, B.H.; Li, Z.P.; Morigasaki, N.; Suda, S. Kinetic characteristics of sodium borohydride formation when sodium meta-borate reacts with magnesium and hydrogen. Int. J. Hydrog. Energy 2008, 33, 1323–1328. [Google Scholar] [CrossRef]
- Kim, T. Hydrogen generation from sodium borohydride using microreactor for micro fuel cells. Int. J. Hydrog. Energy 2011, 36, 1404–1405. [Google Scholar] [CrossRef]
- Zhang, J.S.; Delgass, W.N.; Fisher, T.S.; Gore, J.P. Kinetics of Ru catalyzed sodium borohydride hydrolysis. J. Power Sources 2007, 164, 772–781. [Google Scholar] [CrossRef]
- Kojima, Y.; Suzuki, K.; Fukumoto, K.; Kawai, Y.; Kimbara, M.; Nakanishi, H. Development of 10 kW-scale hydrogen generator using chemical hydride. J. Power Sources 2004, 125, 22–26. [Google Scholar] [CrossRef]
- Patel, N.; Patton, B.; Zanchetta, C.; Fernandes, R.; Guella, G.; Kale, A.; Miotello, A. Pd-C powder and thin film catalysts for hydrogen production by hydrolysis of sodium borohydride. Int. J. Hydrog. Energy 2008, 33, 287–292. [Google Scholar] [CrossRef]
- Amendola, S.C.; Onnerud, P.; Kelly, M.T.; Petillo, P.J.; Sharp-Goldman, S.L.; Binder, M. An ultrasafe hydrogen generator: Aqueous, alkaline borohydride solutions and Ru catalyst. J. Power Sources 2000, 85, 186–189. [Google Scholar]
- Amendola, S.C.; Sharp-Goldman, S.L.; Janjua, M.S.; Spencer, N.C.; Kelly, M.T.; Petillo, P.J. Safe, portable, hydrogen gas generator using aqueous borohydride solution and Ru catalyst. Int. J. Hydrog. Energy 2000, 25, 969–975. [Google Scholar] [CrossRef]
- Andrieux, J.; Demirci, U.B.; Miele, P. Langmuir-Hinshelwood kinetic model to capture the cobalt nanoparticles-catalyzed hydrolysis of sodium borohydride over a wide temperature range. Catal. Today 2011, 170, 13–19. [Google Scholar] [CrossRef]
- Rakap, M.; Kalu, E.E.; Özkar, S. Hydrogen generation from the hydrolysis of ammonia borane using cobalt-nickel-phosphorous (Co-Ni-P) catalyst supported on Pd-activated TiO2 by electroless deposition. Int. J. Hydrog. Energy 2011, 36, 254–261. [Google Scholar] [CrossRef]
- Wu, C.; Bai, Y.; Liu, D.X.; Wu, F.; Pang, M.L.; Yi, B.L. Ni-Co-B catalyst-promoted hydrogen generation by hydrolysis NaBH4 solution for in situ hydrogen supply of portable fuel cells. Catal. Today 2011, 170, 33–39. [Google Scholar] [CrossRef]
- Oh, T.H.; Kwon, S. Effect of manufacturing conditions on properties of electroless deposited Co-P/Ni foam catalyst for hydrolysis of sodium borohydride solution. Int. J. Hydrog. Energy 2012, 37, 15925–15937. [Google Scholar] [CrossRef]
- Oh, T.H.; Kwon, S. Effect of bath composition on properties of electroless Co-P/Ni foam catalyst for hydrolysis of sodium borohydride solution. Int. J. Hydrog. Energy 2012, 37, 17027–17039. [Google Scholar] [CrossRef]
- Liu, C.H.; Chen, B.H.; Hsueh, C.L.; Ku, J.R.; Jeng, M.S.; Tsau, F. Hydrogen generation from hydrolysis of sodium borohydride using Ni-Ru nanocomposite as catalyst. Int. J. Hydrog. Energy 2009, 34, 2153–2163. [Google Scholar] [CrossRef]
- Ingersoll, J.C.; Mani, N.; Thenmozhiyal, J.C.; Muthaiah, A. Catalytic hydrolysis of sodium borohydride by a novel nickel-cobalt-boride catalyst. J. Power Sources 2007, 173, 450–457. [Google Scholar] [CrossRef]
- Rakap, M; Kalu, E.E.; Özkar, S. Cobalt-nickel-phosphorus supported on Pd-activated TiO2 (Co-Ni-P/Pd-TiO2) as cost-effective and reusable catalyst for hydrogen generation from hydrolysis of alkaline sodium borohydride solution. J. Alloys Compd. 2011, 509, 7016–7021. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Anderson, S.P.; Kalu, E.E. Electroless Nickel-Based Catalyst for Diffusion Limited Hydrogen Generation through Hydrolysis of Borohydride. Crystals 2013, 3, 405-418. https://doi.org/10.3390/cryst3030405
Anderson SP, Kalu EE. Electroless Nickel-Based Catalyst for Diffusion Limited Hydrogen Generation through Hydrolysis of Borohydride. Crystals. 2013; 3(3):405-418. https://doi.org/10.3390/cryst3030405
Chicago/Turabian StyleAnderson, Shannon P., and Egwu E. Kalu. 2013. "Electroless Nickel-Based Catalyst for Diffusion Limited Hydrogen Generation through Hydrolysis of Borohydride" Crystals 3, no. 3: 405-418. https://doi.org/10.3390/cryst3030405
APA StyleAnderson, S. P., & Kalu, E. E. (2013). Electroless Nickel-Based Catalyst for Diffusion Limited Hydrogen Generation through Hydrolysis of Borohydride. Crystals, 3(3), 405-418. https://doi.org/10.3390/cryst3030405