Simple and Efficient One-Pot Synthesis, Spectroscopic Characterization and Crystal Structure of Methyl 5-(4-Chlorobenzoyloxy)-1-phenyl-1H-pyrazole-3-carboxylate
Abstract
:1. Introduction
2. Results and Discussion
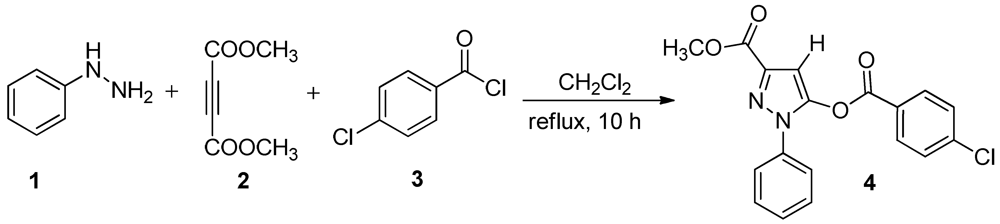
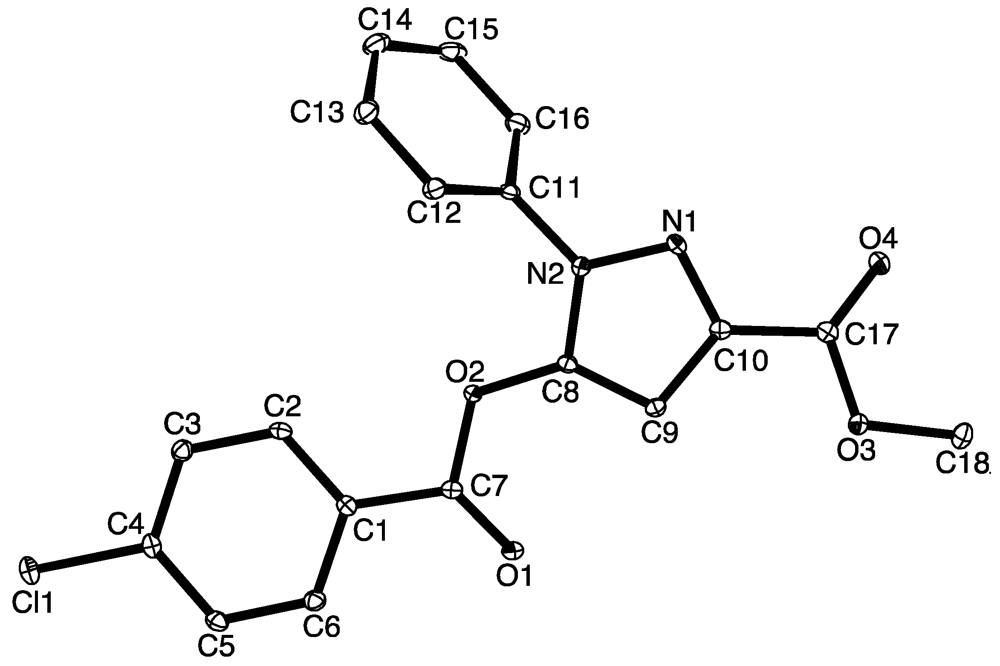
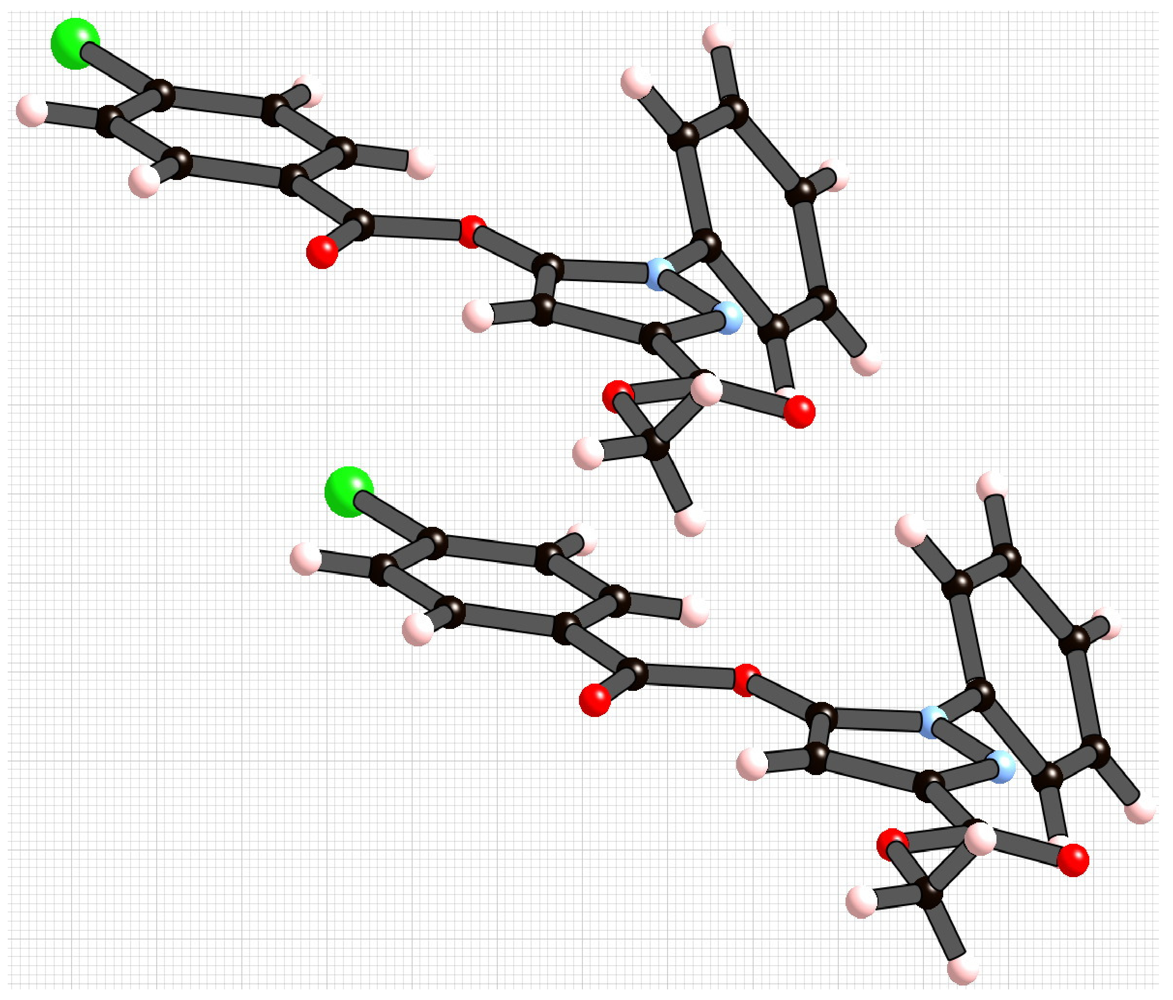
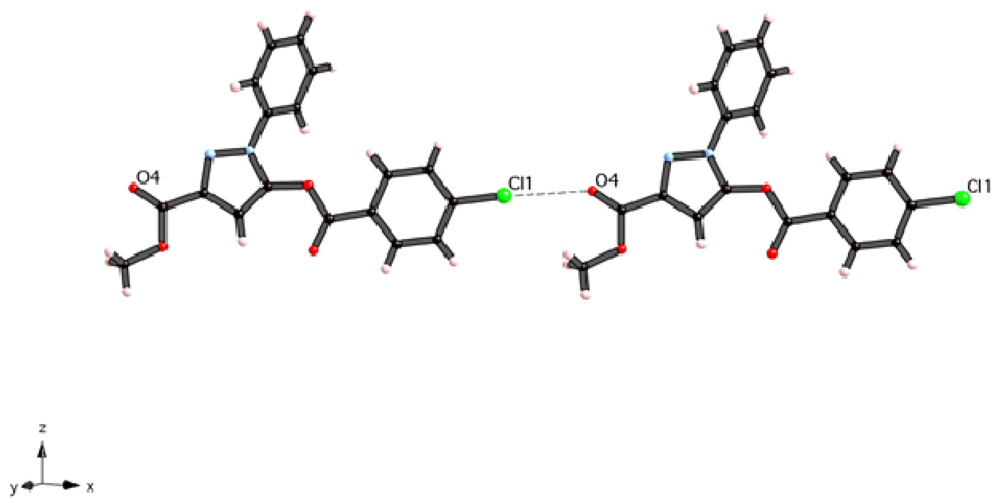
3. Experimental Section
3.1. General
3.2. Synthesis of Methyl 5-(4-Chlorobenzoyloxy)-1-phenyl-1H-pyrazole-3-carboxylate (4)
3.3. Data Collection and Structure Solution
4. Conclusions
Acknowledgements
Conflict of Interest
References
- Noga, E.J.; Barthalmus, G.T.; Mitchell, M.K. Cyclic amines are selective cytotoxic agents for pigmented cells. Cell. Biol. Int. Rep. 1986, 10, 239–247. [Google Scholar] [CrossRef]
- Van Herk, T.; Brussee, J.; Van den Nieuwendijk, A.M.; Van der Klein, P.A.; Ijzerman, A.P.; Stannek, C.; Burmeister, A.; Lorenzen, A. Pyrazole derivatives as partial agonists for the nicotinic acid receptor. J. Med. Chem. 2003, 46, 3945–3951. [Google Scholar]
- Dvorak, C.A.; Rudolph, D.A.; Ma, S.; Carruthers, N.I. Palladium-Catalyzed coupling of pyrazole triflates with arylboronic acids. J. Org. Chem. 2005, 70, 4188–4190. [Google Scholar] [CrossRef]
- Elguero, J. Comprehensive Heterocyclic Chemistry; Katritzky, A.R., Rees, C.W., Scriven, E.F.V., Eds.; Pergamon Press: Oxford, UK, 1996; pp. 1–75. [Google Scholar]
- Sugiura, S.; Ohno, S.; Ohtani, O.; Izumi, K.; Kitamikado, T.; Asai, H.; Kato, K. Syntheses and anti-inflammatory and hypnotic activity of 5-alkoxy-3-(N-substituted carbamoyl)-1-phenylpyrazoles. J. Med. Chem. 1997, 20, 80–85. [Google Scholar]
- Behr, L.C.; Fusco, R.; Jarboe, C.H. The Chemistry of Heterocyclic Compounds, Pyrazoles, Pyrazolines, Pyrazolidines, Indazoles and Condensed Rings; Interscience: New York, NY, USA, 1967; p. 1. [Google Scholar]
- Rosiere, C.E.; Grossman, M.I. An analog of histamine that stimulates gastric acid secretion without other actions of histamine. Science 1951, 113, 651. [Google Scholar]
- Bailey, D.M.; Hansen, P.E.; Hlavac, A.G.; Baizman, E.R.; Pearl, J.; Defelice, A.F.; Feigenson, M.E. 3,4-Diphenyl-1H-pyrazole-1-propanamine antidepressants. J. Med. Chem. 1985, 28, 256–260. [Google Scholar] [CrossRef]
- Mahajan, R.N.; Havaldar, F.H.; Fernandes, P.S. Syntheses and biological activity of heterocycles derived from 3-methoxy-1-phenyl-1H-pyrazole-5-carboxylate. J. Ind. Chem. Soc. 1991, 68, 245–249. [Google Scholar]
- Chauhan, P.M.S.; Singh, S.; Chatterjee, R.K. Antifilarial profile of substituted pyrazoles: A new class of antifilarial agents. Ind. J. Chem. Sect. B 1993, 32, 858–861. [Google Scholar]
- Srivastava, B.K.; Joharapurkar, A.; Raval, S.; Patel, J.Z.; Soni, R.; Raval, P.; Gite, A.; Goswami, A.; Sadhwani, N.; Gandhi, N.; et al. Diaryl dihydropyrazole-3-carboxamides with significant in vivo antiobesity activity related to CB1 receptor antagonism: Synthesis, biological evaluation, and molecular modeling in the homology model. J. Med. Chem. 2007, 50, 5951–5966. [Google Scholar] [CrossRef]
- Hashimoto, H.; Imamura, K.; Haruta, J.I.; Wakitani, K. 4-(4-Cycloalkyl/aryl-oxazol-5-yl)benzenesulfonamides as selective cyclooxygenase-2 inhibitors: Enhancement of the selectivity by introduction of a fluorine atom and identification of a potent, highly selective, and orally active COX-2 inhibitor JTE-522. J. Med. Chem. 2002, 45, 1511–1517. [Google Scholar] [CrossRef]
- Genin, M.J.; Biles, C.; Keiser, B.J.; Poppe, S.M.; Swaney, S.M.; Tarpley, W.G.; Yagi, Y.; Romero, D.L. Novel 1,5-diphenyl pyrazole non-nucleoside HIV-1. Reverse transcriptase inhibitors with enhanced activity versus the delaviridine-resistant P236L mutant: Lead identification and SAR of 3-and 4-substituted derivatives. J. Med. Chem. 2000, 43, 1034–1040. [Google Scholar]
- Kees, K.L.; Fitzgerald, J.J.; Steiner, K.E.; Mattes, J.F.; Mihan, B.; Tosi, T.; Mondoro, D.; McCaleb, M.L. Synthesis of pyrazolone derivatives. J. Med. Chem. 1996, 39, 3920–3928. [Google Scholar] [CrossRef]
- Ouyang, G.; Chen, Z.; Cai, X.J.; Song, B.A.; Bhadury, P.S.; Yang, S.; Jin, L.H.; Xue, W.; Hu, D.Y.; Zeng, S. Synthesis and antiviral activity of novel pyrazole derivatives containing oxime esters group. Bioorg. Med. Chem. 2008, 16, 9699–9707. [Google Scholar]
- Zohreh, N.; Alizadeh, A. Simple and efficient one-pot synthesis of N-phenyl-3,5-difunctionalized pyrazoles. Tetrahedron 2011, 67, 4595–4600. [Google Scholar] [CrossRef]
- Metrangolo, P.; Neukirch, H.; Pilati, T.; Resnati, G. Halogen bonding based recognition processes: A world parallel to hydrogen bonding. Acc. Chem. Res. 2005, 36, 386–395. [Google Scholar]
- Erdelyi, M. Halogen bonding in solution. Chem. Soc. Rev. 2012, 41, 3547–3557. [Google Scholar] [CrossRef]
- Murray, J.S.; Lane, P.; Clark, T.; Riley, K.E.; Politzer, P. Σ-holes, π-holes and electrostatically-driven interactions. J. Mol. Model. 2012, 18, 541–548. [Google Scholar] [CrossRef]
- Brycki, B.; Kowalczyk, I.; Zielinski, A.; Borowiak, T.; Wolska, I. Spectroscopic properties of N-n-hexyltetrachlorophthalimide and supramolecular interactions in its crystals. J. Mol. Struct. 2008, 874, 145–150. [Google Scholar] [CrossRef]
- Basavaiah, D.; Roy, S.; Das, U. Toward understanding the scope of Baylis-Hillman reaction: Synthesis of 3-(2-hydroxyphenyl)indolin-2-ones and polycyclic fused furans. Tetrahedron 2010, 66, 5612–5622. [Google Scholar] [CrossRef]
- Lemouchi, C.; Simonov, S.; Zorina, L.; Gautier, C.; Hudhomme, P.; Batail, P. Amino acid derivatives of perylenediimide and their N–H…O peptide bond dipoles-templated solid state assembly into stacks. Org. Biomol. Chem. 2011, 9, 8096–8101. [Google Scholar]
- Zhang, Y.J.; Luo, C.T.; Wang, Y.G.; Wang, Z. 4,5,6,7-Tetrachloro-N-(2,3,4-trifluorophenyl)phthalimide. Acta Cryst. Sect. E 2011, 67, 1604. [Google Scholar]
- Metrangolo, P.; Meyer, F.; Pilati, T.; Resnati, G.; Terraneo, G. Halogen bonding in supramolecular chemistry. Angew. Chem. Int. Ed. 2008, 47, 6114–6127. [Google Scholar]
- Sheldrick, G.M. A short history of SHELX. Acta Cryst. Sect. A 2008, 64, 112–122. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Khan, I.; White, J.M. Simple and Efficient One-Pot Synthesis, Spectroscopic Characterization and Crystal Structure of Methyl 5-(4-Chlorobenzoyloxy)-1-phenyl-1H-pyrazole-3-carboxylate. Crystals 2012, 2, 967-973. https://doi.org/10.3390/cryst2030967
Khan I, White JM. Simple and Efficient One-Pot Synthesis, Spectroscopic Characterization and Crystal Structure of Methyl 5-(4-Chlorobenzoyloxy)-1-phenyl-1H-pyrazole-3-carboxylate. Crystals. 2012; 2(3):967-973. https://doi.org/10.3390/cryst2030967
Chicago/Turabian StyleKhan, Imtiaz, and Jonathan M. White. 2012. "Simple and Efficient One-Pot Synthesis, Spectroscopic Characterization and Crystal Structure of Methyl 5-(4-Chlorobenzoyloxy)-1-phenyl-1H-pyrazole-3-carboxylate" Crystals 2, no. 3: 967-973. https://doi.org/10.3390/cryst2030967
APA StyleKhan, I., & White, J. M. (2012). Simple and Efficient One-Pot Synthesis, Spectroscopic Characterization and Crystal Structure of Methyl 5-(4-Chlorobenzoyloxy)-1-phenyl-1H-pyrazole-3-carboxylate. Crystals, 2(3), 967-973. https://doi.org/10.3390/cryst2030967



