2.2. Single Crystal X-ray Structure Analysis
The low temperature determination of the crystal structures of
2–
5 was performed on an Oxford Xcalibur3 diffractometer with a Spellman generator (voltage 50 kV, current 40 mA) and a KappaCCD detector. The data collection and reduction was carried out using the C
RYSA
LISP
RO software [
7]. The structures were solved either with S
HELXS-97 [
8] or S
IR-92 [
9], refined with S
HELXL-97 [
10] and finally checked using the P
LATON [
11] software integrated in the W
INGX [
12] software suite. The non-hydrogen atoms were refined anisotropically and the hydrogen atoms were located and freely refined. The absorptions were corrected with a Scale3 Abspack multi-scan method [
13]. Selected data and parameters of the X-ray determinations are given in
Table 1. Crystallographic data for the structures have been deposited with the Cambridge Crystallographic Data Centre [
14].
Table 1.
X-ray data and parameters of 2–5.
Table 1.
X-ray data and parameters of 2–5.
| | 2 | 3 | 4 | 5 |
|---|
| Formula | CH6N5O2, H2O, Cl | CH6N5O2, H2O, Br | CH6N5O2, H2O, I | 2(CH6N5O2), SO4 |
| FW [g mol−1] | 173.56 | 218.03 | 265.02 | 336.29 |
| Crystal system | Monoclinic | Monoclinic | Triclinic | Orthorhombic |
| Space Group | P21/n | P21/c | P-1 | Fdd2 |
| Color/Habit | Colorless block | Colorless plate | Colorless block | Colorless block |
| Size [mm] | 0.37 × 0.44 × 0.49 | 0.04 × 0.21 × 0.50 | 0.38 × 0.42 × 0.50 | 0.14 × 0.22 × 0.26 |
| a [Å] | 7.6616(6) | 8.0261(5) | 6.6931(3) | 11.0638(8) |
| b [Å] | 12.7248(7) | 12.7412(7) | 7.0540(4) | 30.380(2) |
| c [Å] | 7.8116(8) | 7.9296(5) | 9.1359(5) | 6.7531(5) |
| α [°] | 90 | 90 | 97.754(5) | 90 |
| β [°] | 119.012(11) | 118.051(9) | 96.942(4) | 90 |
| γ [°] | 90 | 90 | 115.961(5) | 90 |
| V [Å3] | 666.01(12) | 715.64(7) | 376.35(4) | 2269.8(3) |
| Z | 4 | 4 | 2 | 8 |
| ρcalc. [g cm−3] | 1.731 | 2.024 | 2.339 | 1.968 |
| μ [mm−1] | 0.535 | 5.708 | 4.221 | 0.359 |
| F(000) | 360 | 432 | 252 | 1392 |
| λMoKα [Å] | 0.71073 | 0.71073 | 0.71073 | 0.71073 |
| T [K] | 173 | 173 | 173 | 173 |
| θ Min–Max [°] | 4.4, 28.6 | 4.3, 33.6 | 4.4, 32.4 | 4.6, 30.1 |
| Dataset | −10:7; −17:9; −8:9 | −9:9; −15:10; −9:9 | −9:9; −10:10; −13:13 | −15:15; −42:42; −9:9 |
| Reflections collected | 2525 | 5748 | 5175 | 10,853 |
| Independent refl. | 1437 | 2172 | 2457 | 1670 |
| Rint | 0.017 | 0.047 | 0.023 | 0.055 |
| Observed reflections | 1298 | 1395 | 2286 | 1392 |
| Parameters | 116 | 123 | 116 | 120 |
| R1 (obs) | 0.0292 | 0.0411 | 0.0220 | 0.0283 |
| wR2 (all data) | 0.0807 | 0.0697 | 0.0554 | 0.0607 |
| S(GooF) | 1.09 | 1.04 | 1.08 | 0.95 |
| Resd. Dens. [e Å−3] | −0.38, 0.43 | −0.77, 0.68 | −1.56, 0.76 | −0.35, 0.25 |
| Device type | Oxford Xcalibur3 | Oxford Xcalibur3 | Oxford Xcalibur3 | Oxford Xcalibur3 |
| CCD | CCD | CCD | CCD |
| Solution | SIR-92 | SHELXS-97 | SIR-92 | SHELXS-97 |
| Refinement | SHELXL-97 | SHELXL-97 | SHELXL-97 | SHELXL-97 |
| CCDC | 858928 | 858930 | 858931 | 858929 |
The structure of the 1-amino-3-nitroguanidinium cation is similar in all four structures investigated in this work. The cation is almost planar, controlled by the intramolecular hydrogen bond N5–H5···O1 (D···A: 2.589(2) Å–2.605(3) Å) [graph set [
15]:
![Crystals 02 00675 i001]()
(6)] but also by the strong angulated (~90°) hydrogen bond N4–H4
x···N1 (D···A: 2.547(5) Å–2.576(7) Å) [graph set:
![Crystals 02 00675 i001]()
(5)]. The C–N bond distances are between typical C–N single and C=N double bonds showing the delocalization of the positive charge. However, in all structures the bond length between C1 and N5 is observed to be the shortest one. Also the bonds N2–O1, N2–O2 and N1–N2 are significantly shorter than single bonds. These structural details are in agreement with our previously described investigations of this cation in the literature [
6]. Selected bond lengths and angles of the cation of all structurally investigated compounds
2–
5 are gathered in
Table 2. The halides, which were obtained crystalline under their monohydrated form, crystallize in common space groups (
2:
P2
1/
n,
3:
P2
1/
c,
4:
P-1) and follow the trend of rising densities (
2: 1.73 <
3: 2.02 <
4: 2.34 g cm
–3). All three structures are dominated by many hydrogen bonds [
16,
17,
18] involving the crystal water molecules.
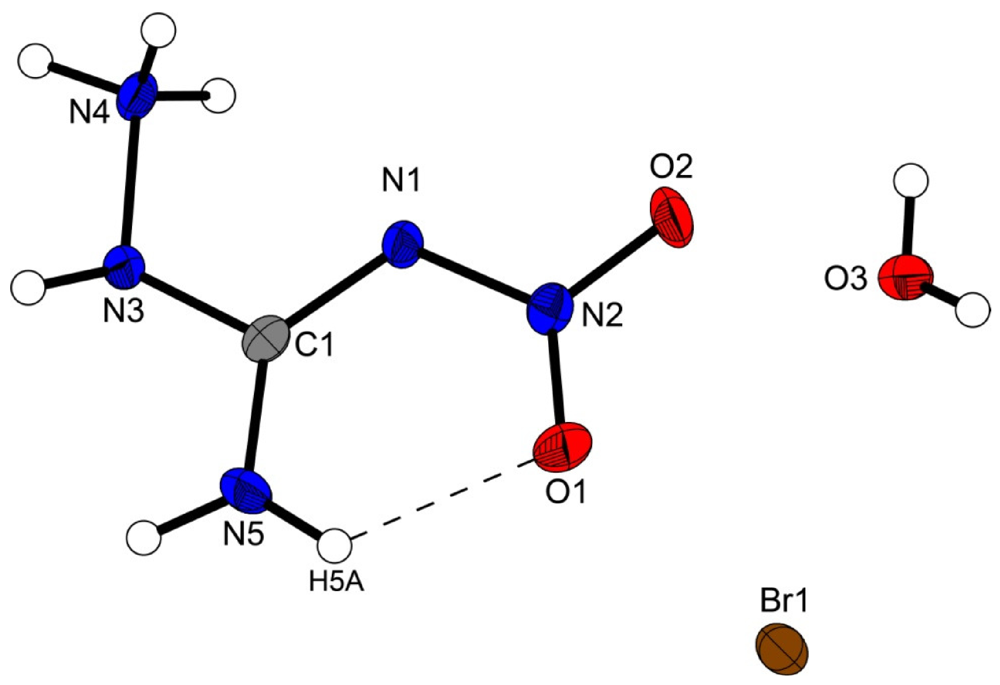
Figure 2 shows the molecular structure of 2.
Table 2.
Selected bond lengths [Å] of compounds 2–5.
Table 2.
Selected bond lengths [Å] of compounds 2–5.
| bond lengths [Å] | 2 | 3 | 4 | 5 |
|---|
| O1–N2 | 1.235(2) | 1.241(4) | 1.236(3) | 1.239(2) |
| O2–N2 | 1.234(2) | 1.229(4) | 1.242(3) | 1.255(2) |
| N1–N2 | 1.361(2) | 1.366(4) | 1.345(3) | 1.330(2) |
| N3–N4 | 1.423(2) | 1.433(4) | 1.419(3) | 1.417(2) |
| N1–C1 | 1.339(2) | 1.337(4) | 1.348(3) | 1.356(3) |
| N3–C1 | 1.368(2) | 1.374(4) | 1.355(4) | 1.349(3) |
| N5–C1 | 1.317(2) | 1.318(4) | 1.315(3) | 1.311(2) |
| bond angles [°] | 2 | 3 | 4 | 5 |
| C1–N1–N2 | 117.7(1) | 117.5(3) | 118.0(2) | 117.5(1) |
| C1–N3–N4 | 117.8(1) | 116.6(3) | 118.1(2) | 119.4(2) |
| O2–N2–O1 | 121.8(1) | 121.8(3) | 121.2(2) | 120.7(1) |
| O2–N2–N1 | 114.2(1) | 114.3(3) | 114.4(2) | 115.7(1) |
| O1–N2–N1 | 124.0(2) | 123.8(3) | 124.5(2) | 123.6(1) |
| N5–C1–N1 | 130.5(2) | 130.9(3) | 130.6(2) | 130.0(2) |
| N5–C1–N3 | 116.0(1) | 115.1(3) | 116.7(2) | 118.0(2) |
| N1–C1–N3 | 113.5(1) | 114.0(3) | 112.7(2) | 111.8(2) |
| Torsion angles [°] | | 2 | 4 | 5 |
| N2–N1–C1–N5 | 0.5(2) | −0.8(5) | 4.6(3) | 10.9(3) |
| N4–N3–C1–N5 | 171.1(1) | 174.0(3) | −177.1(2) | −174.0(2) |
Figure 2.
Molecular structure of amino-nitro-guanidinium chloride monohydrate (2). Ellipsoids are drawn at the 50% probability level. Symmetry code: (i) 0.5 + x, 0.5 − y, 0.5 + z.
Figure 2.
Molecular structure of amino-nitro-guanidinium chloride monohydrate (2). Ellipsoids are drawn at the 50% probability level. Symmetry code: (i) 0.5 + x, 0.5 − y, 0.5 + z.
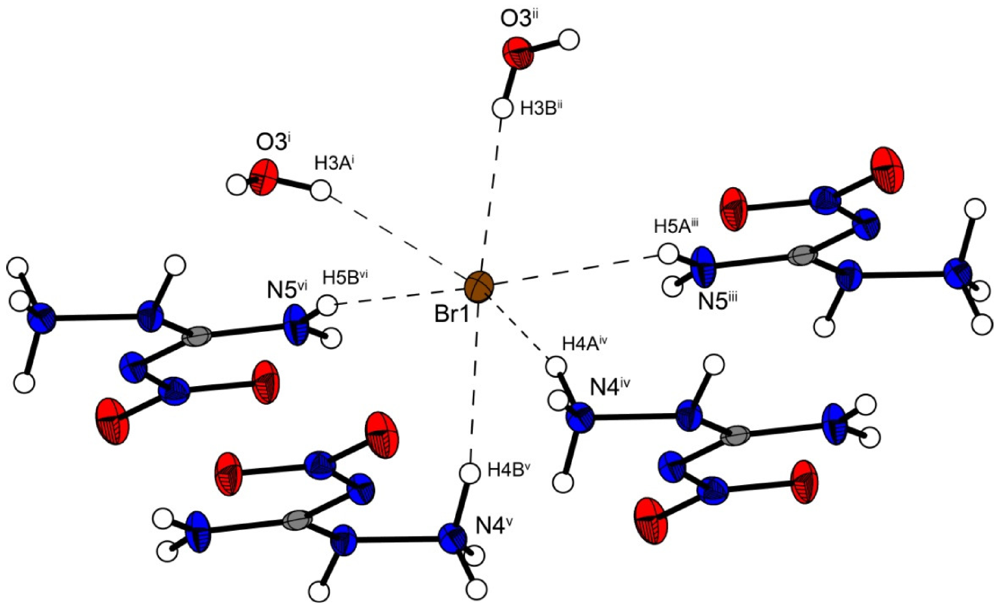
In the crystal structure the ions are held together by a three dimensional network of moderate N–H···O (d(N–O) ~2.83–3.04 Å), N–H···Cl (d(N–Cl) ~3.24–3.30 Å) and O–H···Cl (d(O–Cl ~3.08–3.14 Å) hydrogen bonds. The distorted pseudo-octahedral coordination sphere of the chloride anions is shown in
Figure 3. The chloride anion is coordinated by two water molecules and four cations (two times by the amine group and two times by the ammonium group). The bond lengths and angles of selected hydrogen bonds are listed in
Table 3 and
Table 4. The water molecules bridge between the chloride anions and 1-amino-3-nitroguanidinium cation, and act both as acceptor and donor. The cations build a chain via the hydrogen bridge N3
vi–H3
vi···O2 along the b-axis [graph set: C(6)]. Two cations build dimers via the hydrogen bridge N4
ii–H4c
ii···O2 parallel to the c-axis [graph set:
![Crystals 02 00675 i002]()
(14)]. The oxygen atom (O2) of the nitro group acts as bifurcated acceptor.
Figure 3.
Coordination sphere of one chloride anion in 2. Symmetry codes: (i) 0.5 + x, 0.5 − y, 0.5 + z; (ii) 1.5 − x, 0.5 + y, 1.5 − z; (iii) −0.5 + x, 0.5 − y, 0.5 + z; (iv) 0.5 − x, 0.5 + y, 0.5 − z; (v) −0.5 + x, 0.5 − y, −0.5 + z.
Figure 3.
Coordination sphere of one chloride anion in 2. Symmetry codes: (i) 0.5 + x, 0.5 − y, 0.5 + z; (ii) 1.5 − x, 0.5 + y, 1.5 − z; (iii) −0.5 + x, 0.5 − y, 0.5 + z; (iv) 0.5 − x, 0.5 + y, 0.5 − z; (v) −0.5 + x, 0.5 − y, −0.5 + z.
Table 3.
Selected hydrogen bonds in the structure of 2. Symmetry codes: (vi) 1.5 − x, −0.5 + y, 0.5 − z; (vii) 1 − x, −y, −z.
Table 3.
Selected hydrogen bonds in the structure of 2. Symmetry codes: (vi) 1.5 − x, −0.5 + y, 0.5 − z; (vii) 1 − x, −y, −z.
| D–H···A | d(D–H) | d(H···A) | d(D···A) | <(D–H···A) | <(H···A–X) |
|---|
| N3vi–H3vi···O2 | 0.83(2) | 2.57(2) | 3.043(2) | 117.5(15) | 150.7(5) |
| N4vii–H4cvii···O2 | 0.89(2) | 2.417(18) | 2.881(2) | 112.6(14) | 126.9(5) |
Table 4.
Selected hydrogen bonds in the structure of 2. Symmetry codes: (i) 0.5 + x, 0.5 − y, 0.5 + z; (ii) 1.5 − x, 0.5 + y, 1.5 − z; (iii) −0.5 + x, 0.5 − y, 0.5 + z; (iv) 0.5 − x, 0.5 + y, 0.5 − z; (v) −0.5 + x, 0.5 − y, −0.5 + z.
Table 4.
Selected hydrogen bonds in the structure of 2. Symmetry codes: (i) 0.5 + x, 0.5 − y, 0.5 + z; (ii) 1.5 − x, 0.5 + y, 1.5 − z; (iii) −0.5 + x, 0.5 − y, 0.5 + z; (iv) 0.5 − x, 0.5 + y, 0.5 − z; (v) −0.5 + x, 0.5 − y, −0.5 + z.
| D–H···A | d(D–H) | d(H···A) | d(D···A) | <(D–H···A) |
|---|
| N4iii–H4aiii···Cl1 | 0.88(2) | 2.42(2) | 3.279(1) | 168.8(2) |
| N4iv–H4biv···Cl1 | 0.88(2) | 2.37(2) | 3.242(1) | 159.1(2) |
| N5ii–H5aii···Cl1 | 0.85(2) | 2.59(2) | 3.291(1) | 141.2(2) |
| N5v–H5bv···Cl1 | 0.82(2) | 2.54(2) | 3.299(1) | 155.5(2) |
| O3–H3a···Cl1 | 0.78(2) | 2.34(2) | 3.084(1) | 160(2) |
| O3i–H3bi···Cl1 | 0.80(2) | 2.33(2) | 3.135(1) | 175(2) |
Figure 4 shows the molecular moiety of
3 while
Figure 5 shows the same pseudo-octahedral coordination sphere of the bromide anion as we could observe in the chloride structure. The bond lengths and angles of selected hydrogen bonds are listed in
Table 5,
Table 6. Also in this structure the ions are held together by a three dimensional network of moderate N–H···O (d(N–O) ~2.82–3.08 Å), N–H···Cl (d(N–Br) ~3.36–3.49 Å) and O–···Cl (d(O–Cl ~3.20–3.28 Å) hydrogen bonds. The crystal packing is identical to the chloride structure
2. The N–Br and O–Br hydrogen bonds are approximately 0.15 Å longer than those in the chloride structure
2.
Figure 4.
Molecular structure of amino-nitro-guanidinium bromide monohydrate (3). Ellipsoids are drawn at the 50% probability level.
Figure 4.
Molecular structure of amino-nitro-guanidinium bromide monohydrate (3). Ellipsoids are drawn at the 50% probability level.
Figure 5.
Coordination sphere of one bromide anion in 3. Symmetry codes: (i) −1 + x, 0.5 − y, −0.5 + z; (ii) −1 + x, y, z; (iii) x, 0.5 − y, 0.5 + z; (iv) 1 − x, 0.5 + y, 1.5 − z; (v) x, 0.5 − y, −0.5 + z, (vi) −x, 0.5 + y, 0.5 − z.
Figure 5.
Coordination sphere of one bromide anion in 3. Symmetry codes: (i) −1 + x, 0.5 − y, −0.5 + z; (ii) −1 + x, y, z; (iii) x, 0.5 − y, 0.5 + z; (iv) 1 − x, 0.5 + y, 1.5 − z; (v) x, 0.5 − y, −0.5 + z, (vi) −x, 0.5 + y, 0.5 − z.
Table 5.
Selected hydrogen bonds in the structure of 3. Symmetry codes: (vi) 1 − x, 0.5 + y, 1.5 − z; (vii) 1 − x, −y, 2 − z.
Table 5.
Selected hydrogen bonds in the structure of 3. Symmetry codes: (vi) 1 − x, 0.5 + y, 1.5 − z; (vii) 1 − x, −y, 2 − z.
| D–H···A | d(D–H) | d(H···A) | d(D···A) | <(D–H···A) | <(H···A–X) |
|---|
| N3vi–H3vi···O2 | 0.89(1) | 2.52(4) | 3.075(4) | 121(3) | 151.1(7) |
| N4vii–H4cvii···O2 | 0.90(4) | 2.40(4) | 2.871(4) | 113(3) | 128.0(10) |
Table 6.
Selected hydrogen bonds in the structure of 3.
Table 6.
Selected hydrogen bonds in the structure of 3.
| D–H···A | d(D–H) | d(H···A) | d(D···A) | <(D–H···A) |
|---|
| N4iv–H4aiv···Br1 | 0.91(4) | 2.57(4) | 3.427(3) | 156(3) |
| N4v–H4bv··· Br1 | 0.92(4) | 2.47(4) | 3.360(3) | 163(3) |
| N5iii–H5aiii··· Br1 | 0.90(1) | 2.71(3) | 3.428(3) | 138(3) |
| N5vi–H5bvi··· Br1 | 0.90(1) | 2.65(2) | 3.493(3) | 156(3) |
| O3i–H3ai··· Br1 | 0.78(4) | 2.48(5) | 3.203(3) | 156(4) |
| O3ii–H3bii··· Br1 | 0.78(4) | 2.50(4) | 3.275(3) | 172(4) |
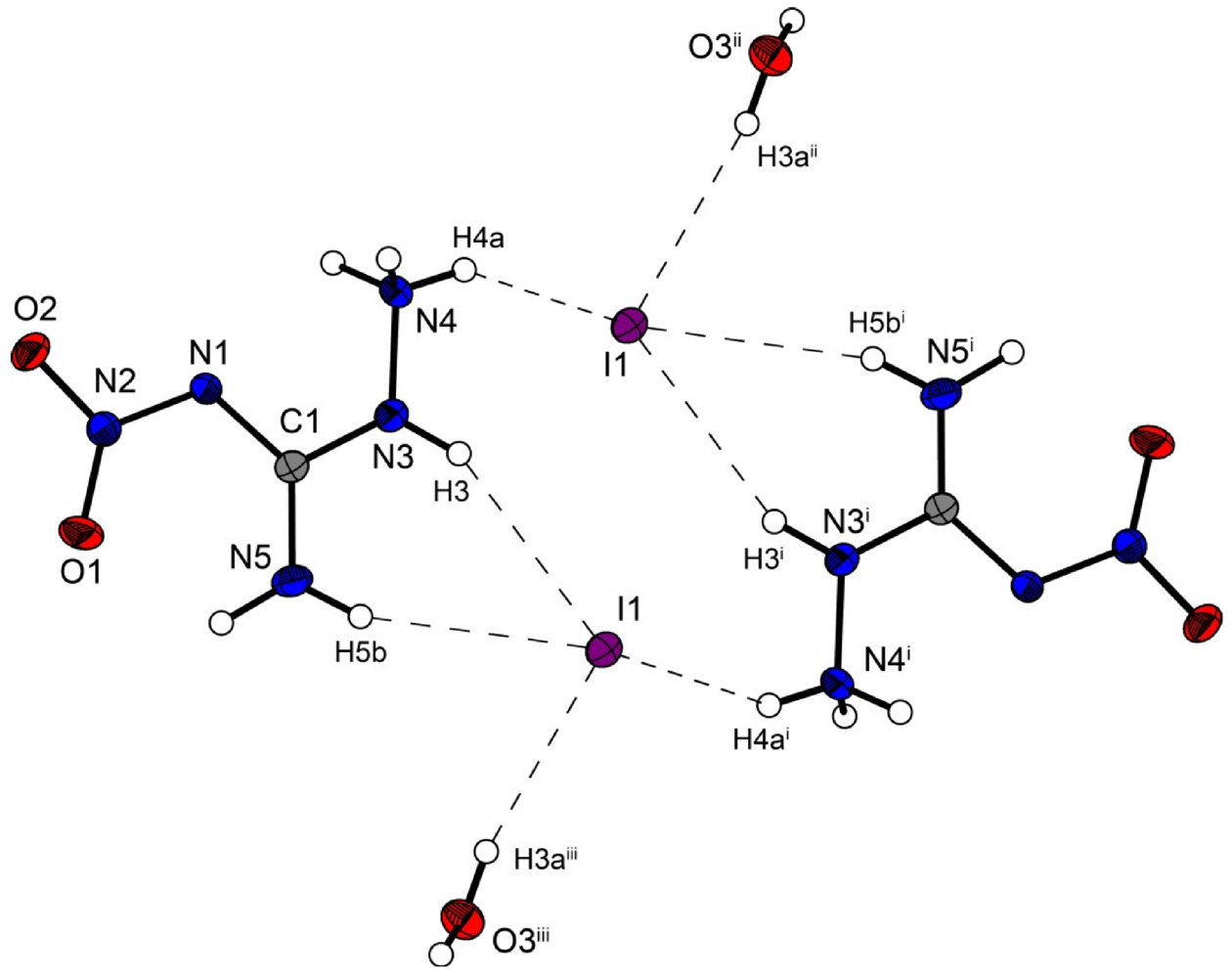
However the iodine structure
4 crystallizes in the triclinic space group
P–1 with two formula units in the unit cell. The crystal packing is different from the described chloride (
2) and bromide (
3) structure. Two molecular moieties of
4 connected to dimers are depicted in
Figure 6. These dimers are packed in layers which are linked by the water molecules.
The iodide anions participate in four hydrogen bonds listed in
Table 7. The N···I and O···I distances observed reveal ordinary values comparable to those found in 3-Cyano-anilinium iodide monohydrate [
19].
Figure 6.
Molecular structure of amino-nitro-guanidinium iodide monohydrate (4). Ellipsoids are drawn at the 50% probability level. Symmetry codes: (i) 2 − x, 1 − y, 1 − z; (ii) 1 + x, y, z; (iii) 1 − x, 1 − y, 1 − z.
Figure 6.
Molecular structure of amino-nitro-guanidinium iodide monohydrate (4). Ellipsoids are drawn at the 50% probability level. Symmetry codes: (i) 2 − x, 1 − y, 1 − z; (ii) 1 + x, y, z; (iii) 1 − x, 1 − y, 1 − z.
Table 7.
Selected hydrogen bonds in the structure of 4.
Table 7.
Selected hydrogen bonds in the structure of 4.
| D–H···A | d(D–H) | d(H···A) | d(D···A) | <(D–H···A) |
|---|
| N3i–H3i ···I1 | 0.90(3) | 2.80(3) | 3.635(2) | 156(3) |
| N4–H4a···I1 | 0.87(3) | 2.78(3) | 3.501(2) | 141(3) |
| N5i–H5bi ···I1 | 0.88(3) | 2.83(3) | 3.669(2) | 160(3) |
| O3ii–H3aii ···I1 | 0.84(4) | 2.71(4) | 3.540(2) | 169(3) |
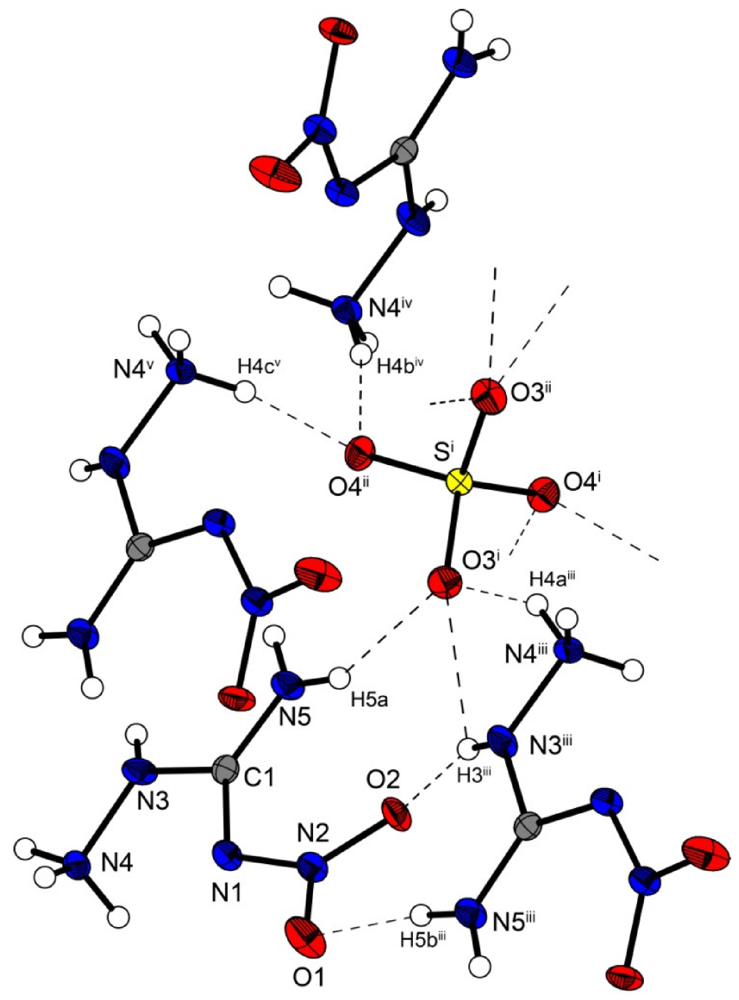
Bis(amino-nitroguanidinium) sulfate crystallizes in the non-centrosymmetric orthorhombic space group
Fdd2 with eight formula units in the unit cell. The density of 1.97 g cm
−3 is slightly lower than that of the bromide structure
3 but higher than that of
2. The molecular structure of
5 is depicted in
Figure 7. The sulfur atoms lie on the special position ½,0,
z. The S–O bonds are uniform with a length of 1.48 Å, which is in agreement to many sulfate structures in the literature, e.g., that of potassium sulfate [
20].
Figure 7.
Molecular structure of bis(amino-nitro-guanidinium) sulfate (5). Ellipsoids are drawn at the 50% probability level. Symmetry codes: (i) 0.75 − x, 0.25 + y, 0.75 + z; (ii) −0.25 + x, 0.25 − y, 0.75 + z; (iii) 0.25 + x, 0.25 − y, −0.75 + z; (iv) 0.25 − x, 0.25 + y, 0.25 + z; (v) −0.25 + x, 0.25 − y, −0.25 + z.
Figure 7.
Molecular structure of bis(amino-nitro-guanidinium) sulfate (5). Ellipsoids are drawn at the 50% probability level. Symmetry codes: (i) 0.75 − x, 0.25 + y, 0.75 + z; (ii) −0.25 + x, 0.25 − y, 0.75 + z; (iii) 0.25 + x, 0.25 − y, −0.75 + z; (iv) 0.25 − x, 0.25 + y, 0.25 + z; (v) −0.25 + x, 0.25 − y, −0.25 + z.
In the crystal structure of 5, the amino-nitro-guanidinium cations and the sulfate anion are held together by a three dimensional network of moderate N–H···O (d(N–O) ~2.60–3.08 Å) hydrogen bonds. Selected bond lengths and angles of the hydrogen bonds are listed in
Table 8 In the crystal structure there are two layers of cations which twisted around ~60° against each other. The layers are connected via hydrogen bonds with the sulfate anions. The oxygen atoms of the sulfate anions act as bifurcated acceptors. The oxygen atom O4 bridges two cations via the hydrogen bonds N4
iv–H4b
iv ····O4
ii and N4
v–H4c
v ····O4
ii, which form zig-zag chains along the c-axis [graph set:
![Crystals 02 00675 i003]()
(4)]. Also the oxygen atom O3 bridges two cations via the hydrogen bonds N5–H5a····O3
i, N4
iii–H4a
iii····O3
i and N3
iii–H3
iii····O3
i. This hydrogen bonding pattern forms a “chain of rings” along the ab-diagonal [graph set:
![Crystals 02 00675 i004]()
(7)
![Crystals 02 00675 i005]()
(5)]]. However this chain is also held together by the hydrogen bonds N3
iii–H3
iii····O2 and N5
iii–H5b
iii····O1 of the nitrate group [graph set:
![Crystals 02 00675 i006]()
(6)
![Crystals 02 00675 i007]()
(8)]]. In the crystal structure one observes a short O···O contact (O1···O4: 2.733(2) Å), which is generated by the existing hydrogen bonds. Such a short O···O contact has also been observed in the literature [
21,
22].
Table 8.
Selected hydrogen bonds in the structure of 5.
Table 8.
Selected hydrogen bonds in the structure of 5.
| D–H···A | d(D–H) | d(H···A) | d(D···A) | <(D–H···A) |
|---|
| N5–H5a····O3i | 0.89(3) | 2.17(2) | 2.979(2) | 152(2) |
| N4iii–H4aiii····O3i | 0.88(2) | 1.95(2) | 2.731(2) | 149(2) |
| N3iii–H3iii····O3i | 0.74(2) | 2.57(2) | 2.964(2) | 115(2) |
| N3iii–H3iii····O2 | 0.74(2) | 2.15(2) | 2.869(2) | 163(2) |
| N5iii–H5biii····O1 | 0.83(2) | 2.09(2) | 2.916(2) | 173(2) |
| N4iv–H4biv ····O4ii | 0.87(3) | 2.23(3) | 3.081(2) | 167(2) |
| N4v–H4cv ····O4ii | 0.89(2) | 1.81(2) | 2.688(2) | 168(2) |
 (6)] but also by the strong angulated (~90°) hydrogen bond N4–H4x···N1 (D···A: 2.547(5) Å–2.576(7) Å) [graph set:
(6)] but also by the strong angulated (~90°) hydrogen bond N4–H4x···N1 (D···A: 2.547(5) Å–2.576(7) Å) [graph set:  (5)]. The C–N bond distances are between typical C–N single and C=N double bonds showing the delocalization of the positive charge. However, in all structures the bond length between C1 and N5 is observed to be the shortest one. Also the bonds N2–O1, N2–O2 and N1–N2 are significantly shorter than single bonds. These structural details are in agreement with our previously described investigations of this cation in the literature [6]. Selected bond lengths and angles of the cation of all structurally investigated compounds 2–5 are gathered in Table 2. The halides, which were obtained crystalline under their monohydrated form, crystallize in common space groups (2: P21/n, 3: P21/c, 4: P-1) and follow the trend of rising densities (2: 1.73 < 3: 2.02 < 4: 2.34 g cm–3). All three structures are dominated by many hydrogen bonds [16,17,18] involving the crystal water molecules. Figure 2 shows the molecular structure of 2.
(5)]. The C–N bond distances are between typical C–N single and C=N double bonds showing the delocalization of the positive charge. However, in all structures the bond length between C1 and N5 is observed to be the shortest one. Also the bonds N2–O1, N2–O2 and N1–N2 are significantly shorter than single bonds. These structural details are in agreement with our previously described investigations of this cation in the literature [6]. Selected bond lengths and angles of the cation of all structurally investigated compounds 2–5 are gathered in Table 2. The halides, which were obtained crystalline under their monohydrated form, crystallize in common space groups (2: P21/n, 3: P21/c, 4: P-1) and follow the trend of rising densities (2: 1.73 < 3: 2.02 < 4: 2.34 g cm–3). All three structures are dominated by many hydrogen bonds [16,17,18] involving the crystal water molecules. Figure 2 shows the molecular structure of 2. 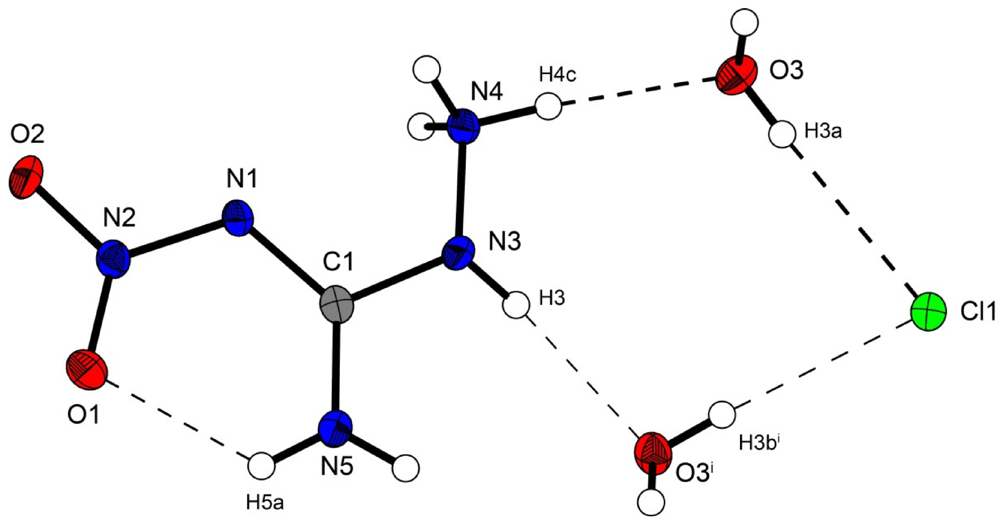
 (14)]. The oxygen atom (O2) of the nitro group acts as bifurcated acceptor.
(14)]. The oxygen atom (O2) of the nitro group acts as bifurcated acceptor. 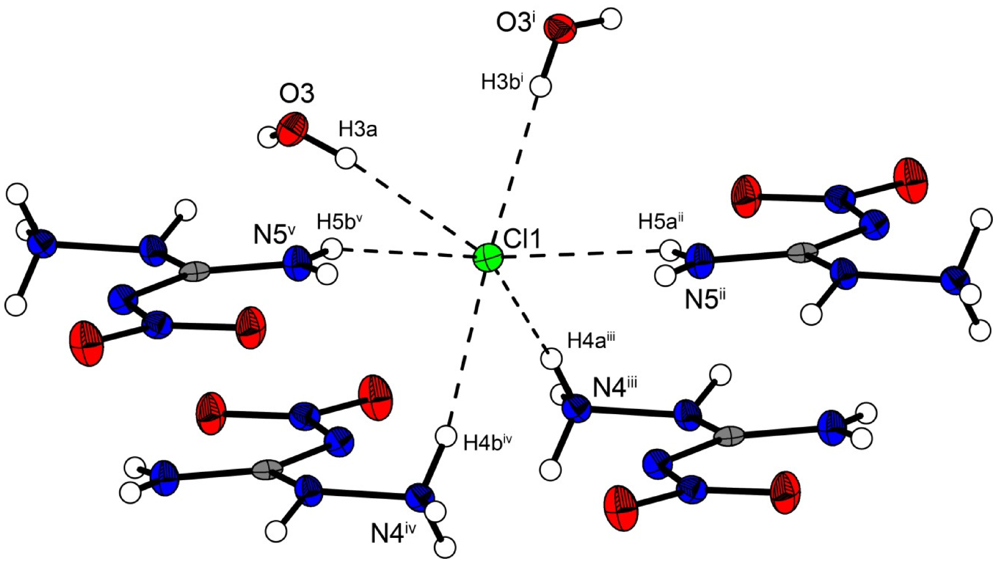
 (4)]. Also the oxygen atom O3 bridges two cations via the hydrogen bonds N5–H5a····O3i, N4iii–H4aiii····O3i and N3iii–H3iii····O3i. This hydrogen bonding pattern forms a “chain of rings” along the ab-diagonal [graph set:
(4)]. Also the oxygen atom O3 bridges two cations via the hydrogen bonds N5–H5a····O3i, N4iii–H4aiii····O3i and N3iii–H3iii····O3i. This hydrogen bonding pattern forms a “chain of rings” along the ab-diagonal [graph set:  (7)
(7)  (5)]]. However this chain is also held together by the hydrogen bonds N3iii–H3iii····O2 and N5iii–H5biii····O1 of the nitrate group [graph set:
(5)]]. However this chain is also held together by the hydrogen bonds N3iii–H3iii····O2 and N5iii–H5biii····O1 of the nitrate group [graph set:  (6)
(6)  (8)]]. In the crystal structure one observes a short O···O contact (O1···O4: 2.733(2) Å), which is generated by the existing hydrogen bonds. Such a short O···O contact has also been observed in the literature [21,22].
(8)]]. In the crystal structure one observes a short O···O contact (O1···O4: 2.733(2) Å), which is generated by the existing hydrogen bonds. Such a short O···O contact has also been observed in the literature [21,22].  = 3549 (s), 3414 (vs), 3236 (s), 2964 (m), 2719 (m), 2675 (m), 2109 (w), 1636 (s), 1618 (s), 1561 (m), 1488 (m), 1384 (m), 1278 (s), 1223 (m), 1195 (m), 908 (w), 783 (w), 621 (w), 483 (w); Raman (1064 nm, 200 mW, 25 °C, cm−1):
= 3549 (s), 3414 (vs), 3236 (s), 2964 (m), 2719 (m), 2675 (m), 2109 (w), 1636 (s), 1618 (s), 1561 (m), 1488 (m), 1384 (m), 1278 (s), 1223 (m), 1195 (m), 908 (w), 783 (w), 621 (w), 483 (w); Raman (1064 nm, 200 mW, 25 °C, cm−1):  = 3335 (14), 3250 (27), 3159 (6), 2965 (16), 1625 (7), 1584 (6), 1565 (18), 1551 (5), 1478 (15), 1402 (10), 1258 (98), 1194 (16), 992 (12), 913 (26), 800 (32), 616 (30), 432 (7), 345 (15); 1H NMR (DMSO-d6, 25 °C, ppm) δ: 8.44 (s, 1H, NH), 6.24 (s, 5H, –NH3+, –NH2); 13C NMR (DMSO-d6, 25 °C, ppm) δ: 159.5 (C(NNO2)(N2H4+)(NH2)); m/z (FAB+): 120.1 [CH6N5O2+]; m/z (FAB−): 35.0 [Cl−]; EA (CH5N5O2·H2O·HCl, 173.56) calc.: C 6.92, H 4.65, N 40.35%; found: C 6.82, H 4.51, N 39.82%; BAM drophammer: 40 J; friction tester: 288 N; ESD: 0.15 J (at grain size 100–500 µm).
= 3335 (14), 3250 (27), 3159 (6), 2965 (16), 1625 (7), 1584 (6), 1565 (18), 1551 (5), 1478 (15), 1402 (10), 1258 (98), 1194 (16), 992 (12), 913 (26), 800 (32), 616 (30), 432 (7), 345 (15); 1H NMR (DMSO-d6, 25 °C, ppm) δ: 8.44 (s, 1H, NH), 6.24 (s, 5H, –NH3+, –NH2); 13C NMR (DMSO-d6, 25 °C, ppm) δ: 159.5 (C(NNO2)(N2H4+)(NH2)); m/z (FAB+): 120.1 [CH6N5O2+]; m/z (FAB−): 35.0 [Cl−]; EA (CH5N5O2·H2O·HCl, 173.56) calc.: C 6.92, H 4.65, N 40.35%; found: C 6.82, H 4.51, N 39.82%; BAM drophammer: 40 J; friction tester: 288 N; ESD: 0.15 J (at grain size 100–500 µm). = 3379 (vs), 2996 (s), 2681 (m), 2375 (w), 2086 (w), 1633 (s), 1586 (s), 1557 (m), 1538 (m), 1490 (m), 1475 (m), 1399 (m), 1383 (m), 1323 (m), 1279 (s), 1223 (s), 1190 (m), 1155 (m), 1121 (m), 1096 (m), 1037 (w), 908 (w), 783 (w), 662 (w), 581 (w), 532 (w), 473 (w); Raman (1064 nm, 300 mW, 25 °C, cm−1):
= 3379 (vs), 2996 (s), 2681 (m), 2375 (w), 2086 (w), 1633 (s), 1586 (s), 1557 (m), 1538 (m), 1490 (m), 1475 (m), 1399 (m), 1383 (m), 1323 (m), 1279 (s), 1223 (s), 1190 (m), 1155 (m), 1121 (m), 1096 (m), 1037 (w), 908 (w), 783 (w), 662 (w), 581 (w), 532 (w), 473 (w); Raman (1064 nm, 300 mW, 25 °C, cm−1):  = 3318 (1), 3254 (8), 2974 (11), 1609 (8), 1580 (3), 1567 (2), 1556 (15), 1533 (2), 1485 (10), 1406 (7), 1255 (100), 1186 (13), 1155 (4), 1094 (5), 994 (15), 913 (24), 800 (45), 720 (2), 614 (37), 587 (2), 519 (2), 467 (1), 431 (8); 1H NMR (DMSO-d6, 25 °C, ppm) δ: 9.64 (s, 1H, -NHH), 9.17 (s, 1H, -NHH), 6.31 (s, 4H, NHNH3+); 13C NMR (DMSO-d6, 25 °C, ppm) δ: 160.8 (C(NNO2)(N2H3)(NH2)), 161.0 (C(NNO2)(N2H4+)(NH2)); m/z (FAB+): 120.1 [CH6N5O2+]; m/z (FAB−): 78.9 [79Br−], 80.9 [81Br−]; EA (CH5N5O2·H2O·HBr, 218.01) calc.: C 5.51, H 3.70, N 32.12%; found: C 5.43, H 3.57, N 31.76 %; BAM drophammer: 25 J; friction tester: 288 N; ESD: 0.20 J (at grain size 500–1000 µm).
= 3318 (1), 3254 (8), 2974 (11), 1609 (8), 1580 (3), 1567 (2), 1556 (15), 1533 (2), 1485 (10), 1406 (7), 1255 (100), 1186 (13), 1155 (4), 1094 (5), 994 (15), 913 (24), 800 (45), 720 (2), 614 (37), 587 (2), 519 (2), 467 (1), 431 (8); 1H NMR (DMSO-d6, 25 °C, ppm) δ: 9.64 (s, 1H, -NHH), 9.17 (s, 1H, -NHH), 6.31 (s, 4H, NHNH3+); 13C NMR (DMSO-d6, 25 °C, ppm) δ: 160.8 (C(NNO2)(N2H3)(NH2)), 161.0 (C(NNO2)(N2H4+)(NH2)); m/z (FAB+): 120.1 [CH6N5O2+]; m/z (FAB−): 78.9 [79Br−], 80.9 [81Br−]; EA (CH5N5O2·H2O·HBr, 218.01) calc.: C 5.51, H 3.70, N 32.12%; found: C 5.43, H 3.57, N 31.76 %; BAM drophammer: 25 J; friction tester: 288 N; ESD: 0.20 J (at grain size 500–1000 µm). = 3384 (vs), 3278 (s), 3224 (s), 2927 (m), 2732 (m), 2371 (w), 1632 (vs), 1587 (s), 1475 (s), 1458 (m), 1369 (m), 1278 (vs), 1122 (m), 1099 (m), 1037 (w), 1000 (w), 913 (w), 835 (w), 783 (w), 701 (w), 618 (w), 581 (w); Raman (1064 nm, 300 mW, 25 °C, cm−1):
= 3384 (vs), 3278 (s), 3224 (s), 2927 (m), 2732 (m), 2371 (w), 1632 (vs), 1587 (s), 1475 (s), 1458 (m), 1369 (m), 1278 (vs), 1122 (m), 1099 (m), 1037 (w), 1000 (w), 913 (w), 835 (w), 783 (w), 701 (w), 618 (w), 581 (w); Raman (1064 nm, 300 mW, 25 °C, cm−1):  = 3393 (2), 3235 (7), 2978 (2), 1629 (7), 1604 (1), 1563 (44), 1531 (8), 1489 (19), 1456 (2), 1383 (24), 1264 (100), 1162 (21), 1110 (3), 999 (21), 921 (38), 797 (69), 699 (5), 620 (37), 571 (2), 433 (32); 1H NMR (DMSO-d6, 25 °C, ppm) δ: 8.30 (s, 3H, NH, NH2), 6.47 (s, 3H, -NH3+); 13C NMR (DMSO-d6, 25 °C, ppm) δ: 159.3 (C(NNO2)(N2H4+)(NH2)); m/z (FAB+): 120.1 [CH6N5O2+]; m/z (FAB−): 126.9 [I−]; EA (CH5N5O2·H2O·HI, 265.01) calc.: C 4.53, H 3.04, N 26.43 %; found: C 4.87, H 2.92, N 26.46%; BAM drophammer: 25 J; friction tester: 288 N; ESD: 0.20 J (at grain size 500–1000 µm).
= 3393 (2), 3235 (7), 2978 (2), 1629 (7), 1604 (1), 1563 (44), 1531 (8), 1489 (19), 1456 (2), 1383 (24), 1264 (100), 1162 (21), 1110 (3), 999 (21), 921 (38), 797 (69), 699 (5), 620 (37), 571 (2), 433 (32); 1H NMR (DMSO-d6, 25 °C, ppm) δ: 8.30 (s, 3H, NH, NH2), 6.47 (s, 3H, -NH3+); 13C NMR (DMSO-d6, 25 °C, ppm) δ: 159.3 (C(NNO2)(N2H4+)(NH2)); m/z (FAB+): 120.1 [CH6N5O2+]; m/z (FAB−): 126.9 [I−]; EA (CH5N5O2·H2O·HI, 265.01) calc.: C 4.53, H 3.04, N 26.43 %; found: C 4.87, H 2.92, N 26.46%; BAM drophammer: 25 J; friction tester: 288 N; ESD: 0.20 J (at grain size 500–1000 µm). = 3401 (s), 3307 (vs), 3153 (s), 2923 (m), 2647 (m), 2060 (w), 1684 (m), 1659 (m), 1639 (m), 1566 (m), 1485 (m), 1459 (m), 1384 (m), 1280 (s), 1256 (s), 1161 (m), 1124 (m), 1091 (m), 1065 (s), 1009 (m), 976 (m), 924 (w), 788 (w), 728 (w), 647 (w), 614 (m), 457 (w); Raman (1064 nm, 200 mW, 25 °C, cm−1):
= 3401 (s), 3307 (vs), 3153 (s), 2923 (m), 2647 (m), 2060 (w), 1684 (m), 1659 (m), 1639 (m), 1566 (m), 1485 (m), 1459 (m), 1384 (m), 1280 (s), 1256 (s), 1161 (m), 1124 (m), 1091 (m), 1065 (s), 1009 (m), 976 (m), 924 (w), 788 (w), 728 (w), 647 (w), 614 (m), 457 (w); Raman (1064 nm, 200 mW, 25 °C, cm−1):  = 3238 (4), 3165 (4), 1643 (10), 1580 (9), 1543 (8), 1485 (11), 1414 (5), 1280 (23), 1161 (19), 1131 (13), 1091 (7), 982 (100), 935 (35), 802 (31), 635 (28), 604 (10), 476 (11), 458 (15), 435 (12), 356 (30), 248 (14), 196 (30), 178 (29), 136 (44), 116 (43); 1H NMR (DMSO-d6, 25 °C, ppm) δ: 8.17 (s, NH), 7.64 (s, 5H, -NH3+, -NH2); 13C NMR (DMSO-d6, 25 °C, ppm) δ: 160.5 (C(NNO2)(N2H4+)(NH2)); m/z (FAB+): 120.1 [CH6N5O2+]; m/z (FAB–): 96.9 [HSO4–]; EA (C2H12N10O8S, 336.24) calc.: C 7.14, H 3.60, N 41.66%; found: C 7.47, H 3.44, N 41.35%; BAM drophammer: 6 J; friction tester: 120 N; ESD: 0.30 J (at grain size 500–1000 µm).
= 3238 (4), 3165 (4), 1643 (10), 1580 (9), 1543 (8), 1485 (11), 1414 (5), 1280 (23), 1161 (19), 1131 (13), 1091 (7), 982 (100), 935 (35), 802 (31), 635 (28), 604 (10), 476 (11), 458 (15), 435 (12), 356 (30), 248 (14), 196 (30), 178 (29), 136 (44), 116 (43); 1H NMR (DMSO-d6, 25 °C, ppm) δ: 8.17 (s, NH), 7.64 (s, 5H, -NH3+, -NH2); 13C NMR (DMSO-d6, 25 °C, ppm) δ: 160.5 (C(NNO2)(N2H4+)(NH2)); m/z (FAB+): 120.1 [CH6N5O2+]; m/z (FAB–): 96.9 [HSO4–]; EA (C2H12N10O8S, 336.24) calc.: C 7.14, H 3.60, N 41.66%; found: C 7.47, H 3.44, N 41.35%; BAM drophammer: 6 J; friction tester: 120 N; ESD: 0.30 J (at grain size 500–1000 µm).




