The Synthesis and Molecular Structure of 2-(4-Methoxybenzyl)-4-nitro-2H-indazole
Abstract
:1. Introduction
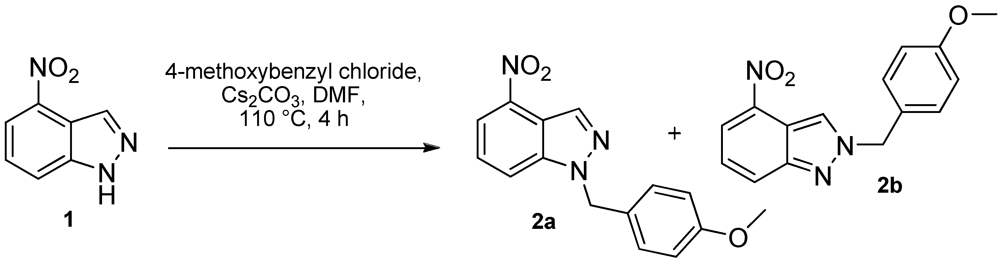
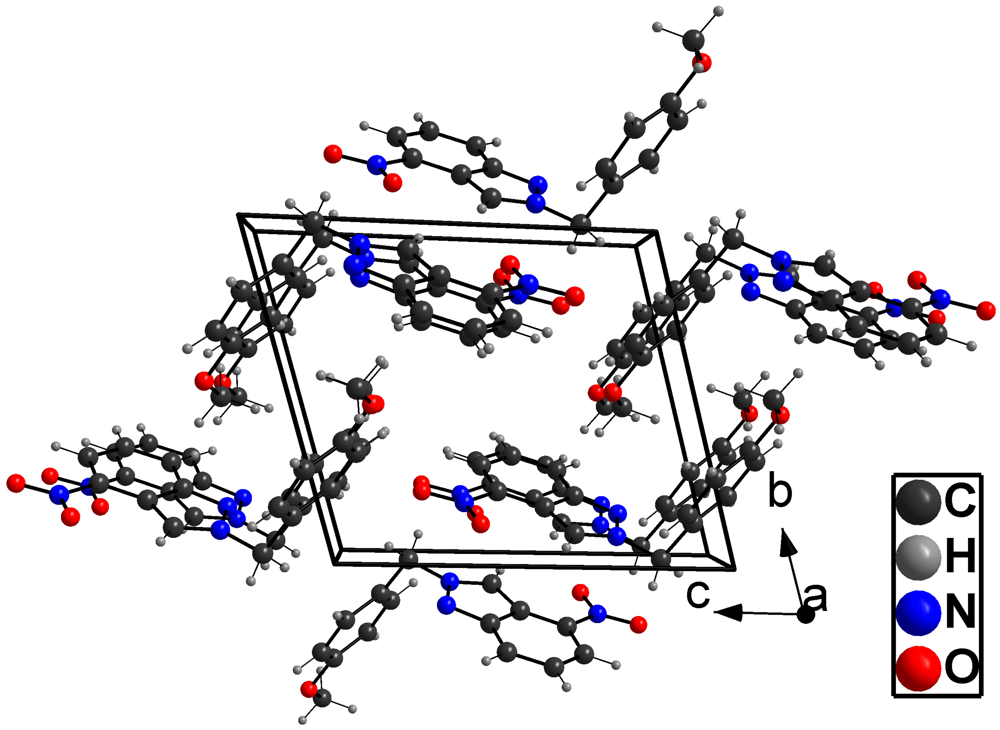
2. Results and Discussion
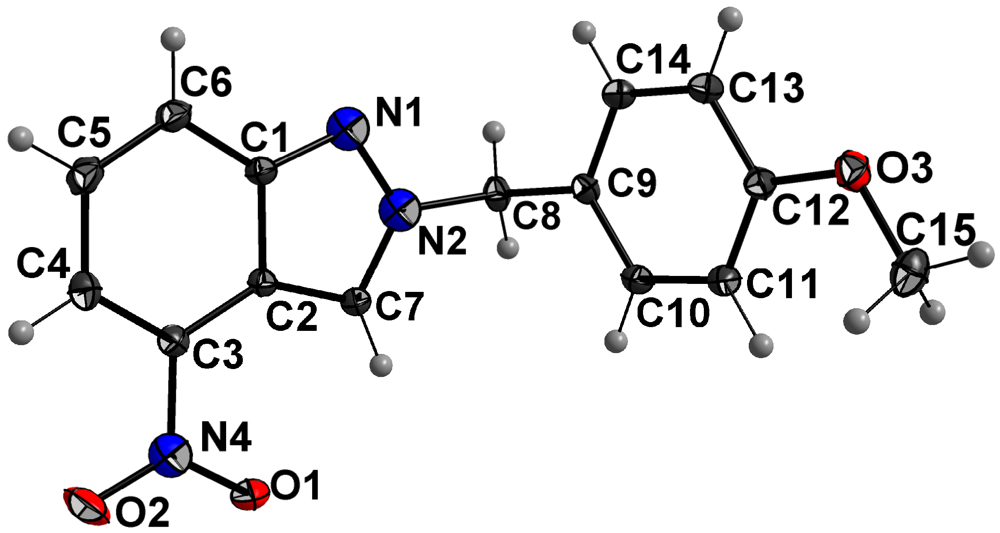
| Crystal data | Refinement | ||
|---|---|---|---|
| Formula | C15H13N3O3 | Refinement method | Full-matrix least- |
| Formula weight | 283.28 g·mol−1 | squares on F2 | |
| Temperature | 296 K | Data/restraints/parameters | 10467/0/190 |
| Wavelength | 0.71073 Å | Measured reflections | 30930 |
| Crystal system | triclinic | Goodness-of-fit on F2 | 1.02 |
| Space group | P ī | Final R indices | R1 = 0.0428 |
| Unit cell dimensions | a = 6.8994(1) Å | [I > 2σ(I)] | wR2 = 0.1238 |
| b = 9.8052(2) Å | R indices (all data) | R1 = 0.0580 | |
| c = 11.1525(2) Å | wR2 = 0.1352 | ||
| α = 71.729(1)° | |||
| β = 79.436(1)° | |||
| γ = 74.349(1)° | |||
| Volume | 685.83(2) Å3 | Largest diff. peak/hole | 0.66/-0.51 e Å-3 |
| Z | 2 | ||
| Density (calcd.) | 1.372 g·cm−3 | ||
| Absorption coefficient | 0.10 mm−1 | ||
| F(000) | 296 | ||
| Crystal size | 0.36 × 0.34 × 0.28 mm | ||
| atoms | distance | atoms | distance |
|---|---|---|---|
| N1−N2 | 1.3447(6) | N2−C7 | 1.3499(6) |
| C1−N1 | 1.3536(7) | N2−C8 | 1.4716(6) |
| C1−C2 | 1.4303(6) | C8−C9 | 1.5083(7) |
| C2−C3 | 1.4137(6) | C9−C10 | 1.3925(7) |
| C3−C4 | 1.3768(7) | C9−C14 | 1.3976(7) |
| C3−N4 | 1.4541(7) | C10−C11 | 1.3949(7) |
| N4−O2 | 1.2291(7) | C11−C12 | 1.3950(7) |
| N4−O1 | 1.2321(7) | C12−C13 | 1.3995(7) |
| C1−C6 | 1.4132(7) | C13−C14 | 1.3884(8) |
| C2−C7 | 1.3973(7) | C12−O3 | 1.3664(6) |
| C4−C5 | 1.4178(8) | O3−C15 | 1.4290(8) |
| C5−C6 | 1.3760(8) |
3. Experimental Section
3.1. General
3.2. Synthesis of Compounds 2a and 2b
3.3. Data Collection and Refinement
4. Conclusions
Acknowledgments
References
- Li, X.; Chu, S.; Feher, V.A.; Khalili, M.; Nie, Z.; Margosiak, S.; Nikulin, V.; Levin, J.; Sprankle, K.G.; Tedder, M.; Almassy, R.; Appelt, K.; Yager, K.M. Structure-Based Design, Synthesis, and Antimicrobial Activity of Indazole-Derived SAH/MTA Nucleosidase Inhibitors. J. Med. Chem. 2003, 46, 5663–5673. [Google Scholar]
- Palazzo, G.; Corsi, G.; Baiocchi, L.; Silvestrini, B. Synthesis and Pharmacological Properties of 1-Substituted 3-Dimethylaminoalkoxy-1H-indazoles. J. Med. Chem. 1966, 9, 38–41. [Google Scholar] [CrossRef]
- Bardelle, C.; Barlaam, B.; Brooks, N.; Coleman, T.; Cross, D.; Ducray, R.; Green, I.; Lambert-van der Brempt, C.; Olivier, A.; Read, J. Inhibitors of the tyrosine kinase EphB4. Part 3: Identification of non-benzodioxole-based kinase inhibitors. Bioorg. Med. Chem. Lett. 2010, 20, 6242–6245. [Google Scholar]
- Slade, D.J.; Pelz, N.F.; Bodnar, W.; Lampe, J.W.; Watson, P.S. Indazoles: Regioselective Protection and Subsequent Amine Coupling Reactions. J. Org. Chem. 2009, 74, 6331–6334. [Google Scholar]
- Kocienski, P.J. Protecting Groups; Georg Thieme Verlag: Stuttgart, Germany, 1994; p. 52. [Google Scholar]
- Greene, T.W.; Wuts, P.G.M. Protective Groups in Organic Synthesis, 2nd ed; John Wiley & Sons: New York, NY, USA, 1991; p. 390. [Google Scholar]
- Miloudi, A.; Abed, D.E.; Boyer, G.; Galy, J.-P. Reduction of Nitroindazoles: Preparation of New Amino and Chloroamino Derivatives. Heterocycles 2006, 68, 2595–2605. [Google Scholar] [CrossRef]
- Sheldrick, G.M. SHELXS/L-97, Programs for the Solution and Refinement of Crystal Structures; University of Göttingen: Göttingen, Germany, 1997. [Google Scholar]
- Sheldrick, G.M. A short history of SHELX. Acta Cryst. 2008, A64, 112–122. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ebert, K.; Köckerling, M.; Mamat, C. The Synthesis and Molecular Structure of 2-(4-Methoxybenzyl)-4-nitro-2H-indazole. Crystals 2012, 2, 176-181. https://doi.org/10.3390/cryst2020176
Ebert K, Köckerling M, Mamat C. The Synthesis and Molecular Structure of 2-(4-Methoxybenzyl)-4-nitro-2H-indazole. Crystals. 2012; 2(2):176-181. https://doi.org/10.3390/cryst2020176
Chicago/Turabian StyleEbert, Kristin, Martin Köckerling, and Constantin Mamat. 2012. "The Synthesis and Molecular Structure of 2-(4-Methoxybenzyl)-4-nitro-2H-indazole" Crystals 2, no. 2: 176-181. https://doi.org/10.3390/cryst2020176






