Synthesis and Crystal Structures of New 5,5'-Azotetrazolates
Abstract
:1. Introduction
2. Results and Discussion
| Compound | 1 | 2 | 3 | 4 | 5 |
| CCDC no. | 846911 | 846912 | 846913 | 846914 | 846915 |
| Chemical formula | Ag2(NH3)2 (C2N10) | (C3H9S)2 (C2N10) | (C4H12P)2·(C2N10) | (C3H9OS)2·(C2N10) | (C5H14NO)2·(C2N10) |
| Mr | 413.89 | 318.43 | 346.31 | 350.42 | 372.43 |
| Crystal shape, color | plate, orange | prism, yellow | plate, orange | fragment, yellow | fragment, yellow |
| Crystal size/mm3 | 0.1 × 0.1 × 0.06 | 0.44 × 0.36 × 0.24 | 0.36 × 0.32 × 0.12 | 0.2 × 0.2 × 0.2 | 0.40 × 0.24 × 0.24 |
| Crystal system | monoclinic | triclinic | monoclinic | monoclinic | triclinic |
| Space group | C2/c | P 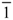 | P21/c | P21/n | P 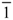 |
| a/Å | 18.0338(7) | 5.9032(7) | 5.9035(9) | 5.2452(2) | 5.4900(4) |
| b/Å | 3.601(2) | 7.4591(8) | 13.388(2) | 14.0290(4) | 8.4206(6) |
| c/Å | 14.906(3) | 9.2538(8) | 11.3173(19) | 10.7735(4) | 10.3003(6) |
| α/° | 90 | 113.598(9) | 90 | 90 | 78.564(5) |
| β/° | 91.94(1) | 98.370(8) | 93.941(17) | 102.036(3) | 85.796(6) |
| γ/° | 90 | 99.017(9) | 90 | 90 | 81.876(6) |
| V/Å3 | 967.4(6) | 358.88(7) | 892.4 (2) | 775.34 (5) | 461.53(6) |
| Z | 8 | 2 | 4 | 4 | 2 |
| Dx/g cm–3 | 2.84 | 1.47 | 1.29 | 1.50 | 1.34 |
| µ/mm–1 | 4.04 | 0.38 | 0.26 | 0.37 | 0.10 |
| F(000)/e | 784 | 168 | 368 | 368 | 200 |
| Diffractometer | Nonius KappaCCD | Gemini Ultra | Gemini-R Ultra | Gemini-R Ultra | Gemini-R Ultra |
| Data collection method | φand ω scans | ω scans | ω scans | Ω scans | ω scans |
| Temperature/K | 233 | 173 | 173 | 173 | 173 |
| θmax/° | 24 | 25.4 | 25 | 28 | 25 |
| h, k, l range | –19 ≤ h ≤ 20 | –6 ≤ h ≤ 7 | –5 ≤ h ≤ 7 | –6 ≤ h ≤ 6 | –6 ≤ h ≤ 5 |
| –4 ≤ k ≤ 3 | –8 ≤ k ≤ 5 | –14 ≤ k ≤ 16 | –18 ≤ k ≤ 17 | –10 ≤ k ≤ 10 | |
| –16 ≤ l ≤ 17 | –10 ≤ l ≤ 11 | –15 ≤ l ≤ 13 | –13 ≤ l ≤ 13 | –12 ≤ l ≤ 10 | |
| Absorption correction | none | multi-scan | multi-scan | multi-scan | none |
| Measured reflections | 1927 | 2168 | 3626 | 6332 | 3070 |
| Independent reflections (Rint) | 739 (0.028) | 1288 (0.024) | 1760 (0.021) | 1723 (0.021) | 1686 (0.030) |
| Observed reflections [I≥ 2σ(I)] | 620 | 1162 | 1461 | 1573 | 1449 |
| Restraints / parameters | 0/75 | 0/94 | 0/104 | 0/103 | 0/123 |
| R1/wR2[I ≥ 2σ(I)] | 0.037/0.097 | 0.030/0.070 | 0.030/0.079 | 0.028/0.070 | 0.035/0.095 |
| R1/wR2 (all data) | 0.046/0.104 | 0.035/ 0.073 | 0.039/0.082 | 0.031/0.072 | 0.042/0.098 |
| Goodness of fit | 1.09 | 1.06 | 1.08 | 1.05 | 1.03 |
| Δρmax/min/e Å–3 | 1.38/–0.81 | 0.24/–0.25 | 0.27/–0.24 | 0.34/0.31 | 0.19/–0.17 |
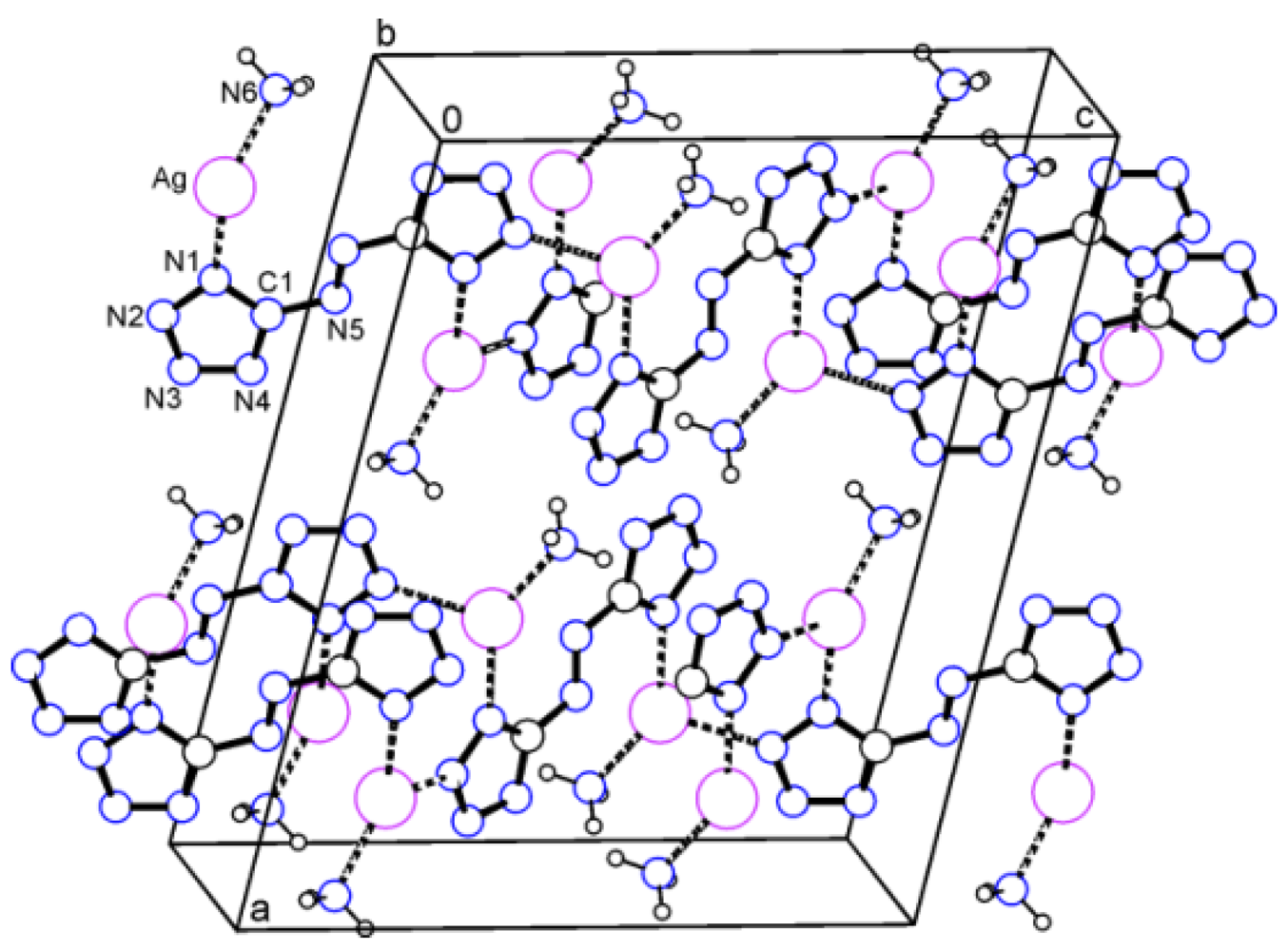
2.1. Bis(amminsilver(I)) 5,5'-Azotetrazolate (1)
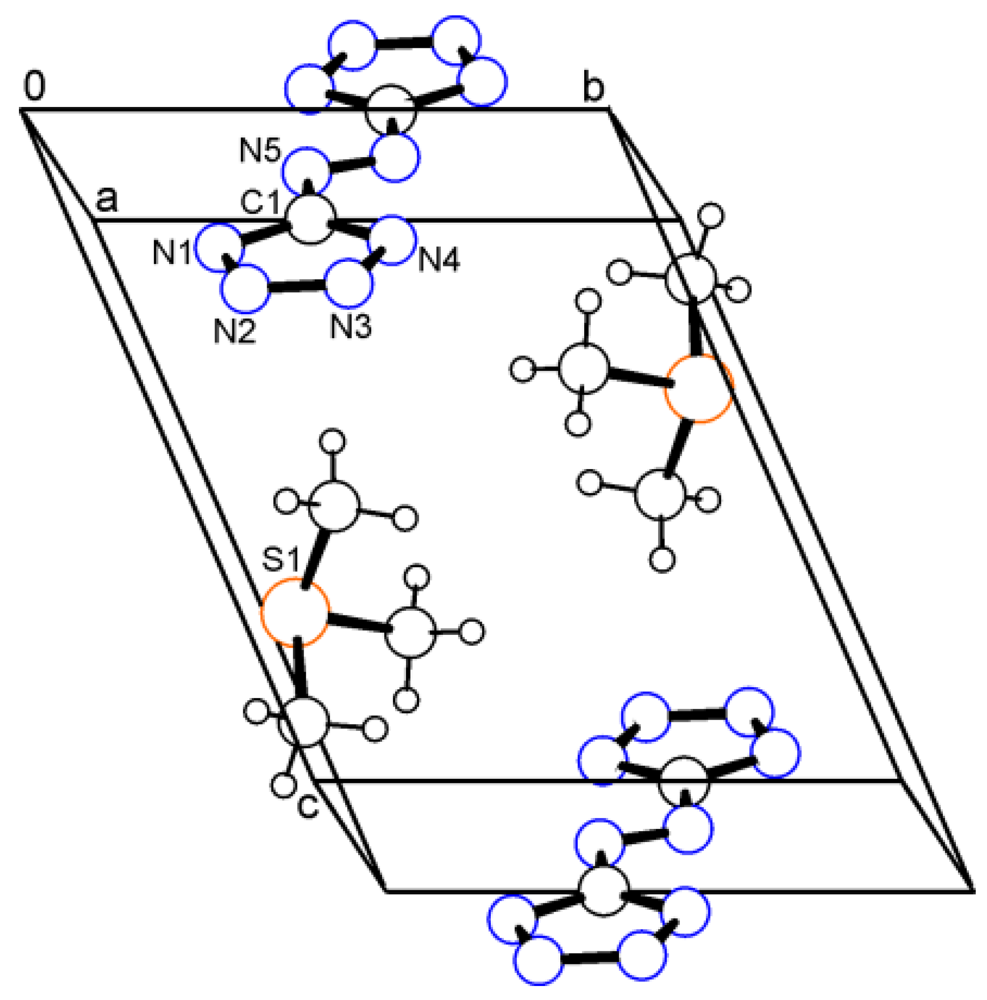
2.2. Bis(trimethylsulfonium) 5,5'-Azotetrazolate (2)
2.3. Bis(tetramethylphosphonium) 5,5'-Azotetrazolate (3)
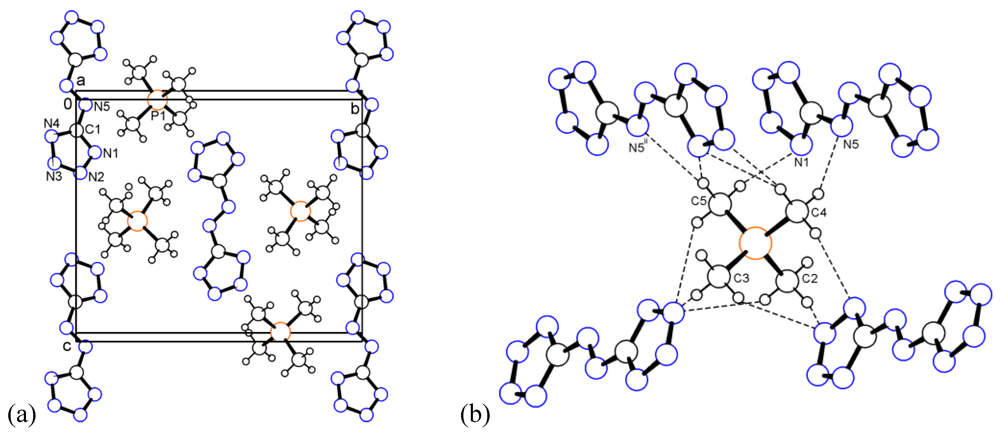
2.4. Bis(trimethylsulfoxonium) 5,5'-Azotetrazolate (4)
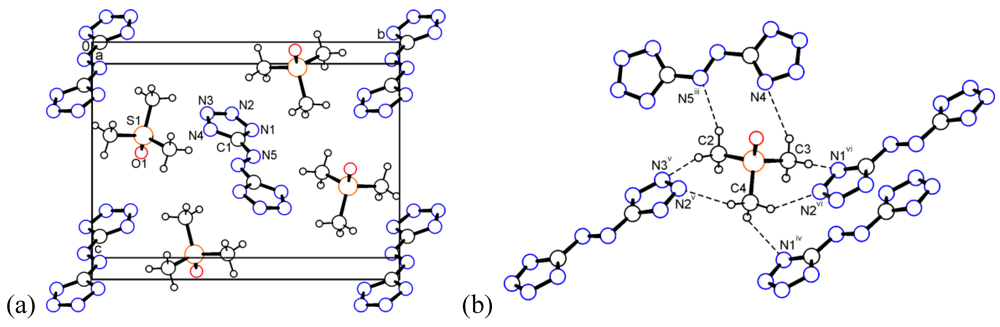
2.5. Bis(2-(hydroxyethyl)trimethylammonium) 5,5'-azotetrazolate (5)
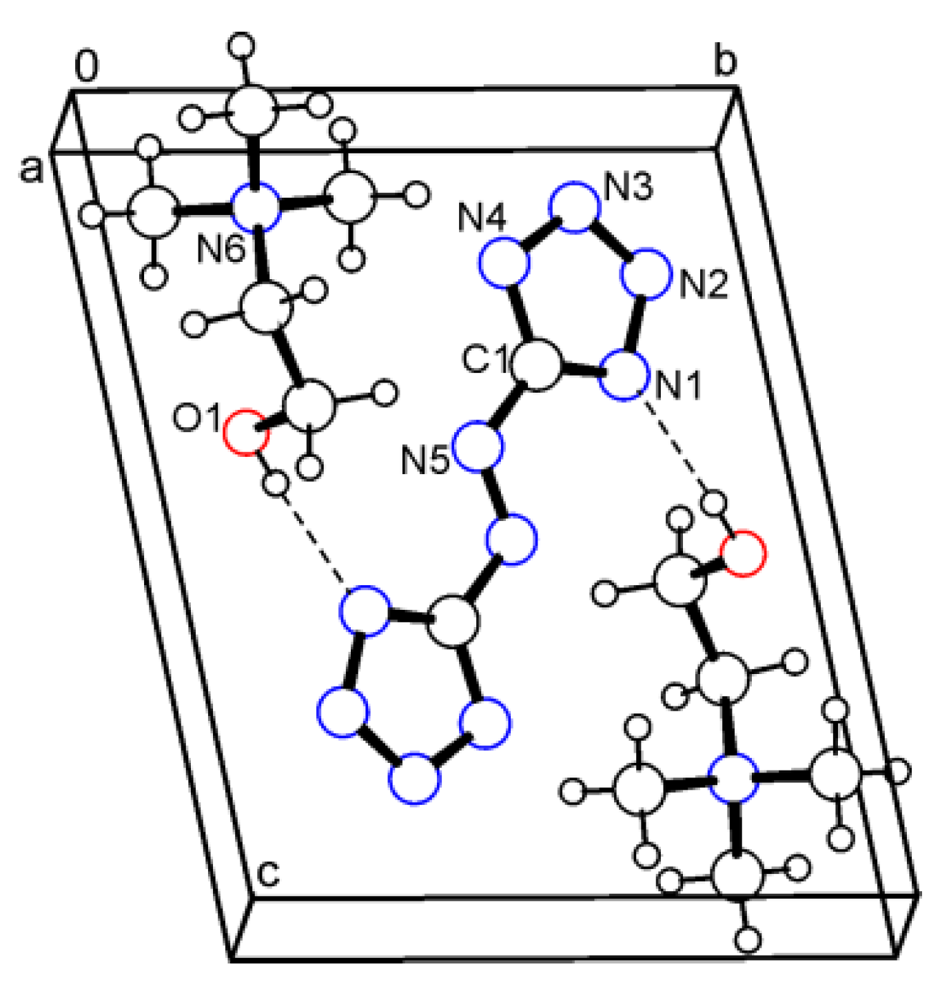
3. Experimental Section
3.1. Bis(amminsilver(I)) 5,5'-Azotetrazolate (1)
3.2. Preparation of 5,5'-Azotetrazolates (2–5) (General Procedure)
3.3. Bis(trimethylsulfonium) 5,5'-Azotetrazolate (2)
3.4. Bis(tetramethylphosphonium) 5,5'-Azotetrazolate (3)
3.5. Bis(trimethylsulfoxonium) 5,5'-Azotetrazolate (4)
3.6. Bis(2-(hydroxyethyl)trimethylammonium) 5,5'-Azotetrazolate (5)
4. Conclusions
References and Notes
- Singh, R.P.; Verma, R.D.; Meshri, D.T.; Shreeve, J.M. Energetic Nitrogen-Rich Salts and Ionic Liquids. Angew. Chem. Int. Ed. 2006, 45, 3584–3601. [Google Scholar]
- Steinhauser, G.; Klapötke, T.M. “Green” Pyrotechnics: A Chemists’ Challenge. Angew. Chem. Int. Ed. 2008, 47, 3330–3347. [Google Scholar] [CrossRef]
- Hammerl, A.; Holl, G.; Kaiser, M.; Klapötke, T.M.; Mayer, P.; Piotrowski, H.; Vogt, M. Methylated Ammonium and Hydrazinium Salts of 5,5'-Azotetrazolate. Z. Naturforsch. 2001, 56b, 847–856. [Google Scholar]
- Hammerl, A.; Holl, G.; Kaiser, M.; Klapötke, T.M.; Mayer, P.; Nöth, H.; Piotrowski, H.; Suter, M. New Hydrazinium Salts of 5,5'-Azotetrazolate. Z. Naturforsch. 2001, 56b, 857–870. [Google Scholar]
- Hammerl, A.; Klapötke, T.M.; Nöth, H.; Warchhold, M.; Holl, G.; Kaiser, M.; Ticmanis, U. [N2H5]+2[N4C-N=N-CN4]2–: A New High-Nitrogen High-Energetic Material. Inorg. Chem. 2001, 40, 3570–3575. [Google Scholar]
- Hammerl, A.; Hiskey, M.A.; Holl, G.; Klapötke, T.M.; Polborn, K.; Stierstorfer, J.; Weigand, J.J. Azidoformamidinium and Guanidinium 5,5'-Azotetrazolate Salts. Chem. Mater. 2005, 17, 3784–3793. [Google Scholar]
- Darwich, C.; Klapötke, T.M.; Sabaté, C.M. 1,2,4-Triazolium-Cation-Based Energetic Salts. Chem. Eur. J. 2008, 14, 5756–5771. [Google Scholar]
- Klapötke, T.M.; Sabaté, C.M. Bistetrazoles: Nitrogen-Rich, High-Performing, Insensitive Energetic Compounds. Chem. Mater. 2008, 20, 3629–3637. [Google Scholar]
- Xue, H.; Arritt, S.W.; Twamley, B.; Shreeve, J.M. Energetic Salts from N-Aminoazoles. Inorg. Chem. 2004, 43, 7972–7977. [Google Scholar]
- Xue, H.; Shreeve, J.M. Energetic Ionic Liquids from Azido Derivatives of 1,2,4-Triazole. Adv. Mater. 2005, 17, 2142–2146. [Google Scholar]
- Klapötke, T.M.; Sabaté, C.M. New energetic compounds based on the nitrogen-rich 5,5’-azotetrazolate anion ([C2N10]2-). New J. Chem. 2009, 33, 1605–1617. [Google Scholar] [CrossRef]
- Klapötke, T.M.; Sabaté, C.M. Nitrogen-Rich TetrazoliumAzotetrazolate Salts: A New Family of Insensitive Energetic Materials. Chem. Mater. 2008, 20, 1750–1763. [Google Scholar]
- Gao, H.; Huang, Y.; Twamley, B.; Ye, C.; Shreeve, J.M. Energetic N,N,N',N'-Tetraaminopiperazinium Salts. ChemSusChem 2008, 2, 222–227. [Google Scholar]
- Katritzky, A.R.; Singh, S.; Kirichenko, K.; Holbrey, J.D.; Smiglak, M.; Reichert, W.M.; Rogers, R.D. 1-Butyl-3-methylimidazolium 3,5-dinitro-1,2,4-triazolate: a novel ionic liquid containing a rigid, planar energetic anion. Chem. Commun. 2005, 868–870. [Google Scholar]
- Gao, H.; Ye, C.; Gupta, O.D.; Xiao, J.C.; Hiskey, M.A.; Twamley, B.; Shreeve, J.M. 2,4,5-Trinitroimidazole-Based Energetic Salts. Chem. Eur. J. 2007, 13, 3853–3860. [Google Scholar]
- Ye, C.; Xiao, J.-C.; Twamley, B.; Shreeve, J.M. Energetic salts of azotetrazolate, iminobis(5-tetrazolate) and 5,5'-bis(tetrazolate). Chem. Commun. 2005, 2750–2752. [Google Scholar]
- Thiele, J. Ueber Azo- und Hydrazoverbindungen des Tetrazols. Ann. Chem. 1898, 303, 57–75. [Google Scholar] [CrossRef]
- Weibel, N.; Grunder, S.; Mayor, M. Functional molecules in electronic circuits. Org. Biomol. Chem. 2007, 5, 2343–2353. [Google Scholar]
- Tappan, B.C.; Huynh, M.H.; Hiskey, M.A.; Chavez, D.E.; Luther, E.P.; Mang, J.T.; Son, S.F. Ultralow-Density Nanostructured Metal Foams: Combustion Synthesis, Morphology, and Composition. J. Am. Chem. Soc. 2006, 128, 6589–6594. [Google Scholar]
- Bentivoglio, G.; Laus, G.; Kahlenberg, V.; Nauer, G.; Schottenberger, H. Crystal structure of bis(hydroxylammonium) 5,5'-azotetrazolate dihydrate, (NH3OH)2(C2N10)·2H2O. Z. Kristallogr. NCS 2008, 223, 425–426. [Google Scholar]
- Pan, W.-L.; Chena, X.-Y.; Hu, C.-W. Bis(amantadinium) 5,5'-diazenediylditetrazolate dihydrate. Acta Crystallogr. 2007, E63, o1606–o1608. [Google Scholar]
- Hammerl, A.; Holl, G.; Klapötke, T.M.; Mayer, P.; Nöth, H.; Piotrowski, H.; Warchhold, M. Salts of 5,5'-Azotetrazolate. Eur. J. Inorg. Chem. 2002, 834–845. [Google Scholar]
- Meng, Y. Poly[l-(5,5'-diazenediylditetrazolido)dicaesium]. Acta Crystallogr. 2011, E67, m453. [Google Scholar]
- Steinhauser, G.; Giester, G.; Leopold, N.; Wagner, C.; Villa, M. Nitrogen-Rich Compounds of the Lanthanoids: The 5,5'-Azobis[1H-tetrazol-1-ides] of the Light Rare Earths (Ce, Pr, Nd, Sm, Eu, Gd). Helv. Chim. Acta 2009, 92, 2038–2051. [Google Scholar] [CrossRef]
- Steinhauser, G.; Giester, G.; Wagner, C.; Leopold, N.; Sterba, J.H.; Lendl, B.; Bichler, M. Nitrogen-Rich Compounds of the Lanthanoids: The 5,5'-Azobis[1H-tetrazol-1-ides] of some Yttric Earths (Tb, Dy, Ho, Er, Tm, Yb, and Lu). Helv. Chim. Acta 2009, 92, 1371–1384. [Google Scholar] [CrossRef]
- Steinhauser, G.; Giester, G.; Leopold, N.; Wagner, C.; Villa, M.; Musilek, A. Nitrogen-Rich Compounds of the Lanthanoids: Highlights and Summary. Helv. Chim. Acta 2010, 93, 183–202. [Google Scholar] [CrossRef]
- Pierce-Butler, M.A. Structure of Bis[hydroxolead(II)] 5,5'-Azotetrazolediide. Acta Crystallogr. 1982, B38, 2681–2683. [Google Scholar]
- Bhandari, S.; Mahon, M.F.; Molloy, K.C.; Palmer, J.S.; Sayers, S.F. Thallium(I)- and organothallium(III)-substituted mono-, bis- and tris-tetrazoles: synthesis and supramolecular structures. J. Chem. Soc. Dalton Trans. 2000, 1053–1060. [Google Scholar]
- Jiao, B.; Chen, S.; Zhao, F.; Hu, R.; Gao, S. A new high-nitrogen compound [Mn(ATZ)(H2O)4]·2H2O: Synthesis and characterization. J. Hazard. Mater. 2007, 142, 550–554. [Google Scholar] [CrossRef]
- Jiao, B.; Yan, Z.; Fan, G.; Chen, S.; Gao, S. catena-Poly[[[tetraaquairon(II)]-µ-5,5'-diazenediyldi-tetrazolido] dihydrate]. Acta Crystallogr. 2010, E66, m1374. [Google Scholar]
- Tao, G.-H.; Twamley, B.; Shreeve, J.M. Energetic Nitrogen-Rich Cu(II) and Cd(II) 5,5'-Azobis(tetrazolate) Complexes. Inorg. Chem. 2009, 48, 9918–9923. [Google Scholar]
- Jannin, M.; Puget, R.; de Brauer, C.; Perret, R. Structures of Trimethylsulfonium Salts. I. Refinement of the Structure of the Iodide (CH3)3SI. Acta Cryst. 1991, C47, 982–984. [Google Scholar]
- Knop, O.; Linden, A.; Vincent, B.R.; Choi, S.C.; Cameron, T.S.; Boyd, R.J. The lone electron pair and crystal packing: observations on pyramidal YEL3 species, ab initio calculations, and the crystal structures of Me3SOI, Et3SI, (Me3S)2SnCl6, (Me3SO)2SnCl6, and (Et3S)2SnCl6. Can. J. Chem. 1989, 67, 1984–2008. [Google Scholar] [CrossRef]
- Jannin, M.; Puget, R.; de Brauer, C.; Perret, R. Structures of Trimethyloxosulfonium Salts. I. The Iodide and the Bromide. Acta Cryst. 1991, C47, 1687–1689. [Google Scholar]
- Available online: www.bam.de (accessed on 4 March 2012).
- Available online: www.reichel-partner.de (accessed on 4 March 2012).
- Impact: insensitive > 40 J, less sensitive > 35 J, sensitive > 4 J, very sensitive < 3 J. Friction: insensitive > 360 N, less sensitive = 360 N, sensitive < 360 N and > 80 N, very sensitive < 80 N, extremely sensitive < 10 N. According to the UN Recommendations on the Transport of Dangerous Goods, (+) indicates not safe for transport.
- Available online: http://www.ozm.cz/en/sensitivity-tests/esd-2008a-small-scale-electrostatic-spark-sensitivity-test/ (accessed on 4 March 2012).
- Burla, M.C.; Carrozzini, B.; Cascarano, G.L.; Giacovazzo, C.; Polidori, G. More power for direct methods: SIR2002. Z. Kristallogr. 2002, 217, 629–635. [Google Scholar] [CrossRef]
- Sheldrick, G.M. A short history of SHELX. Acta Crystallogr. 2008, A64, 112–122. [Google Scholar]
- Gao, H.; Shreeve, J.M. Azole-Based Energetic Salts. Chem. Rev. 2011, 111, 7377–7436. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Laus, G.; Kahlenberg, V.; Wurst, K.; Schottenberger, H.; Fischer, N.; Stierstorfer, J.; Klapötke, T.M. Synthesis and Crystal Structures of New 5,5'-Azotetrazolates. Crystals 2012, 2, 127-136. https://doi.org/10.3390/cryst2010127
Laus G, Kahlenberg V, Wurst K, Schottenberger H, Fischer N, Stierstorfer J, Klapötke TM. Synthesis and Crystal Structures of New 5,5'-Azotetrazolates. Crystals. 2012; 2(1):127-136. https://doi.org/10.3390/cryst2010127
Chicago/Turabian StyleLaus, Gerhard, Volker Kahlenberg, Klaus Wurst, Herwig Schottenberger, Niko Fischer, Jörg Stierstorfer, and Thomas M. Klapötke. 2012. "Synthesis and Crystal Structures of New 5,5'-Azotetrazolates" Crystals 2, no. 1: 127-136. https://doi.org/10.3390/cryst2010127
APA StyleLaus, G., Kahlenberg, V., Wurst, K., Schottenberger, H., Fischer, N., Stierstorfer, J., & Klapötke, T. M. (2012). Synthesis and Crystal Structures of New 5,5'-Azotetrazolates. Crystals, 2(1), 127-136. https://doi.org/10.3390/cryst2010127






