Ru-(Mn-M)OX Solid Base Catalysts for the Upgrading of Xylitol to Glycols in Water
Abstract
:1. Introduction
2. Results and Discussion
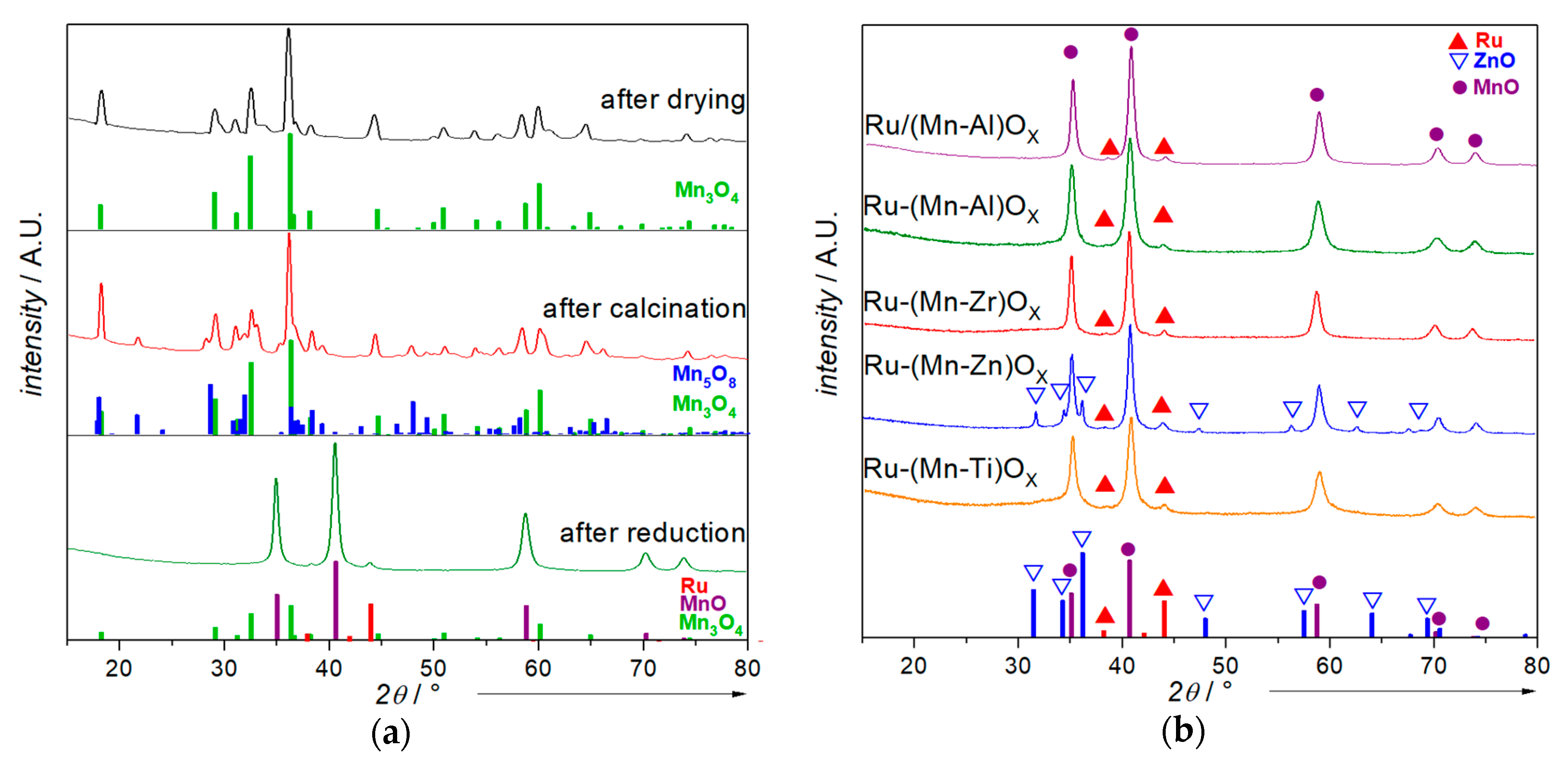
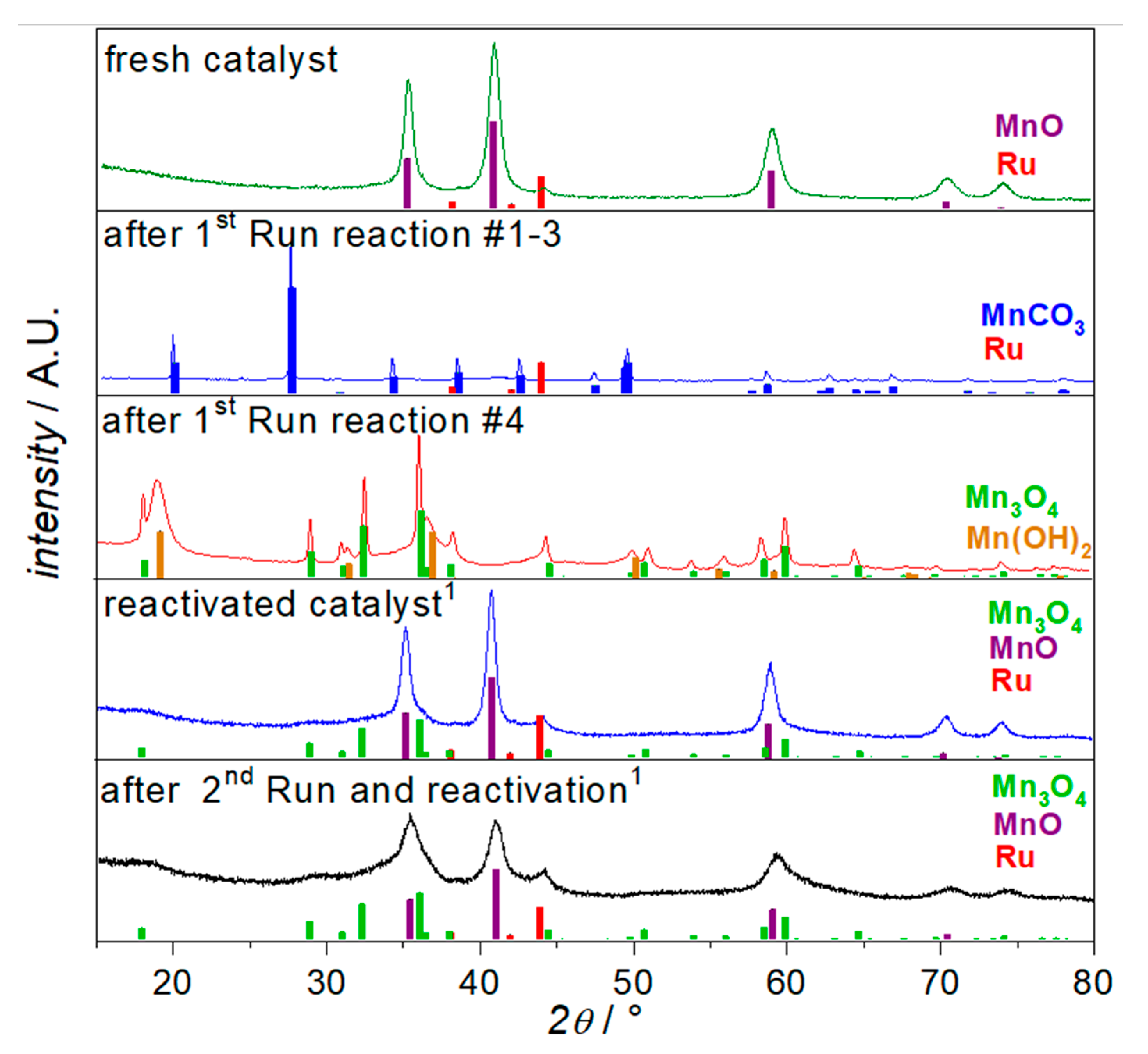
2.1. Catalyst Characterization
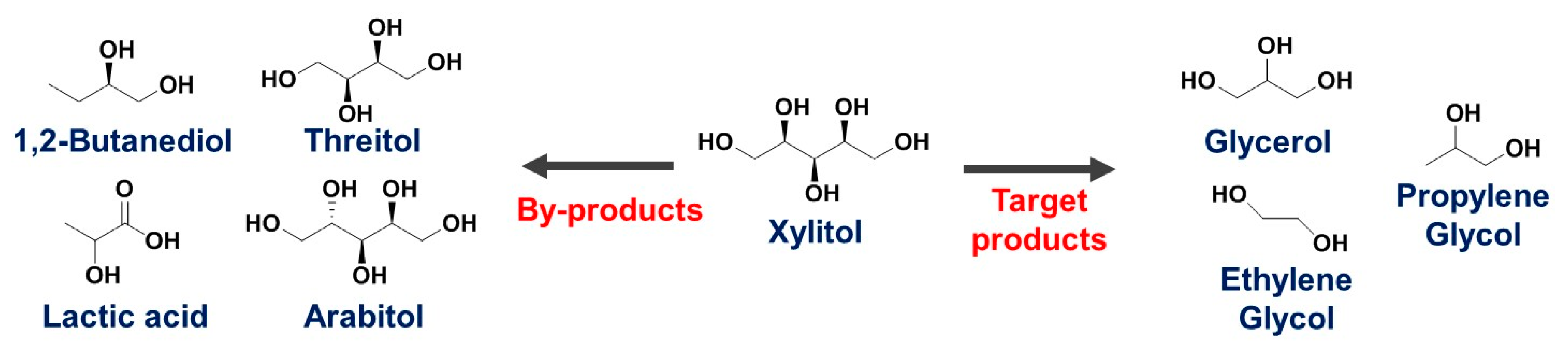
2.2. Hydrogenolysis of Xylitol
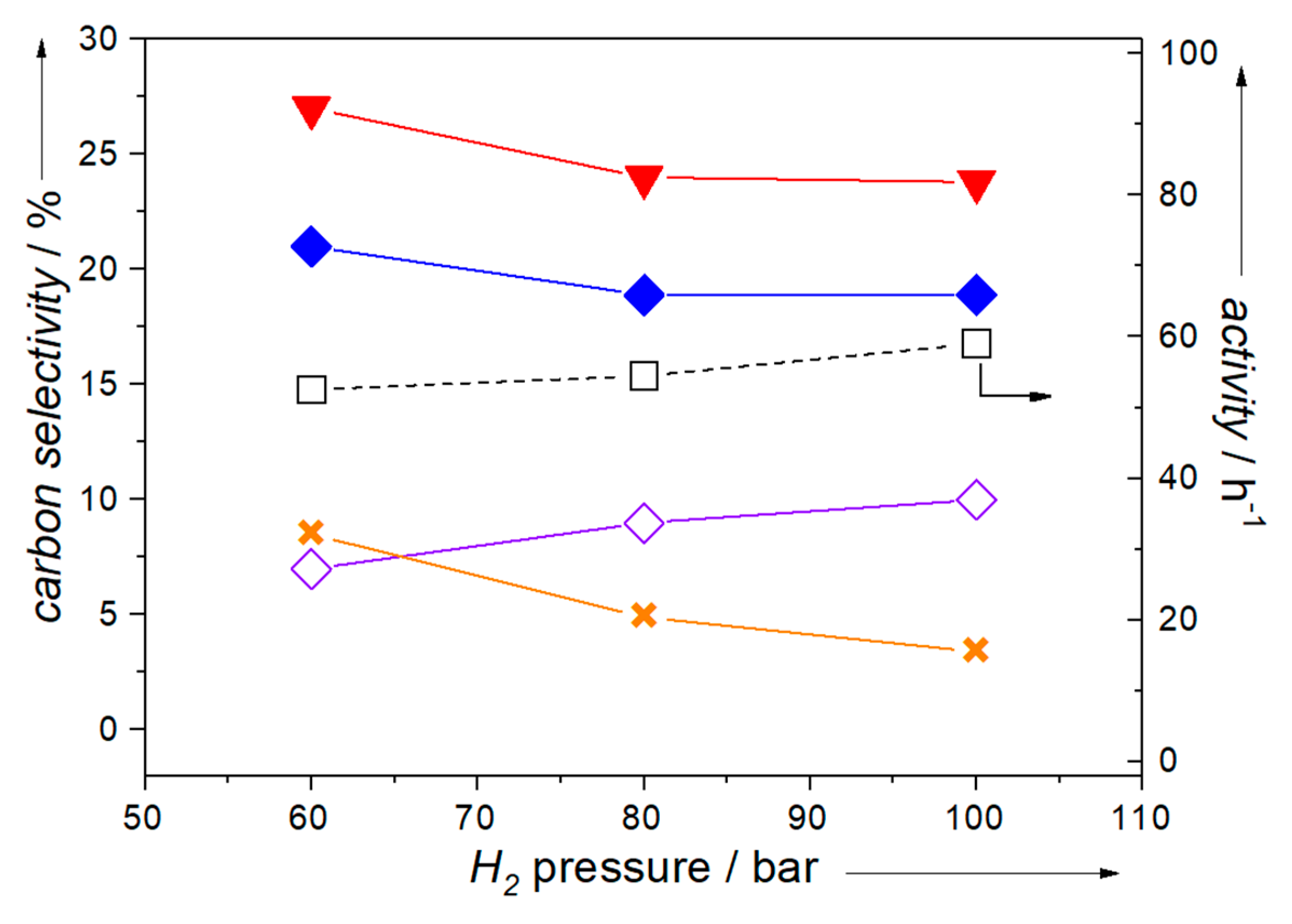
2.3. Influence of Operating Conditions
3. Experimental Section
3.1. Materials
3.2. Catalysts Preparation
3.3. Catalysts Characterization
3.4. Catalytic Testing
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A

References
- Yue, H.; Zhao, Y.; Ma, X.; Gong, J. Ethylene glycol: Properties, synthesis, and applications. Chem. Soc. Rev. 2012, 41, 4218–4244. [Google Scholar] [CrossRef] [PubMed]
- Dasari, M.A.; Kiatsimkul, P.-P.; Sutterlin, W.R.; Suppes, G.J. Low-pressure hydrogenolysis of glycerol to propylene glycol. Appl. Catal. A Gen. 2005, 281, 225–231. [Google Scholar] [CrossRef]
- Zheng, M.; Pang, J.; Sun, R.; Wang, A.; Zhang, T. Selectivity control for cellulose to diols: Dancing on the eggs. ACS Catal. 2017, 7, 1939–1954. [Google Scholar] [CrossRef]
- Climent, M.J.; Corma, A.; Iborra, S. Converting carbohydrates to bulk chemicals and fine chemicals over heterogeneous catalysts. Green Chem. 2011, 13, 520–540. [Google Scholar] [CrossRef]
- Besson, M.; Gallezot, P.; Pinel, C. Conversion of biomass into chemicals over metal catalysts. Chem. Rev. 2014, 114, 1827–1870. [Google Scholar] [CrossRef] [PubMed]
- Ruppert, A.M.; Weinberg, K.; Palkovits, R. Hydrogenolysis goes bio: From carbohydrates and sugar alcohols to platform chemicals. Angew. Chem. Int. Ed. Engl. 2012, 51, 2564–2601. [Google Scholar] [CrossRef] [PubMed]
- Jin, X.; Thapa, P.S.; Subramaniam, B.; Chaudhari, R.V. Kinetic Modeling of Sorbitol Hydrogenolysis over Bimetallic RuRe/C Catalyst. ACS Sustain. Chem. Eng. 2016, 4, 6037–6047. [Google Scholar] [CrossRef]
- Hausoul, P.J.C.; Beine, A.K.; Neghadar, L.; Palkovits, R. Kinetics study of the Ru/C-catalysed hydrogenolysis of polyols—Insight into the interactions with the metal surface. Catal. Sci. Technol. 2017, 7, 56–63. [Google Scholar] [CrossRef]
- Sun, J.; Liu, H. Selective hydrogenolysis of biomass-derived xylitol to ethylene glycol and propylene glycol on supported Ru catalysts. Green Chem. 2011, 13, 135–142. [Google Scholar] [CrossRef]
- Rivière, M.; Perret, N.; Cabiac, A.; Delcroix, D.; Pinel, C.; Besson, M. Xylitol Hydrogenolysis over Ruthenium-Based Catalysts: Effect of Alkaline Promoters and Basic Oxide-Modified Catalysts. ChemCatChem 2017, 9, 2145–2159. [Google Scholar] [CrossRef] [Green Version]
- Deutsch, K.L.; Lahr, D.G.; Shanks, B.H. Probing the ruthenium-catalyzed higher polyol hydrogenolysis reaction through the use of stereoisomers. Green Chem. 2012, 14, 1635–1642. [Google Scholar] [CrossRef]
- Hausoul, P.J.C.; Negahdar, L.; Schute, K.; Palkovits, R. Unravelling the Ru-Catalyzed Hydrogenolysis of Biomass-Based Polyols under Neutral and Acidic Conditions. ChemSusChem 2015, 8, 3323–3330. [Google Scholar] [CrossRef] [PubMed]
- Huang, Z.; Chen, J.; Jia, Y.; Liu, H.; Xia, C.; Liu, H. Selective hydrogenolysis of xylitol to ethylene glycol and propylene glycol over copper catalysts. Appl. Catal. B Environ. 2014, 147, 377–386. [Google Scholar] [CrossRef]
- Banu, M.; Sivasanker, S.; Sankaranarayanan, T.M.; Venuvanalingam, P. Hydrogenolysis of sorbitol over Ni and Pt loaded on NaY. Catal. Commun. 2011, 12, 673–677. [Google Scholar] [CrossRef]
- Liu, H.H.; Huang, Z.; Kang, H.; Li, X.; Xia, C.; Chen, J.; Liu, H.H. Efficient bimetallic NiCu-SiO2 catalysts for selective hydrogenolysis of xylitol to ethylene glycol and propylene glycol. Appl. Catal. B Environ. 2018, 220, 251–263. [Google Scholar] [CrossRef]
- Zhao, L.; Zhou, J.H.; Sui, Z.J.; Zhou, X.G. Hydrogenolysis of sorbitol to glycols over carbon nanofiber supported ruthenium catalyst. Chem. Eng. Sci. 2010, 65, 30–35. [Google Scholar] [CrossRef]
- Guo, X.; Dong, H.; Li, B.; Dong, L.; Mu, X.; Chen, X. Influence of the functional groups of multiwalled carbon nanotubes on performance of Ru catalysts in sorbitol hydrogenolysis to glycols. J. Mol. Catal. A Chem. 2016. [Google Scholar] [CrossRef]
- Beine, A.K.; Krüger, A.J.D.; Artz, J.; Weidenthaler, C.; Glotzbach, C.; Hausoul, P.J.C.; Palkovits, R. Selective production of glycols from xylitol over Ru on covalent triazine frameworks—Suppressing decarbonylation reactions. Green Chem. 2018, 20, 1316–1322. [Google Scholar] [CrossRef]
- Chen, X.; Wang, X.; Yao, S.; Mu, X. Hydrogenolysis of biomass-derived sorbitol to glycols and glycerol over Ni-MgO catalysts. Catal. Commun. 2013, 39, 86–89. [Google Scholar] [CrossRef] [Green Version]
- Vijaya Shanthi, R.; Sankaranarayanan, T.M.; Mahalakshmy, R.; Sivasanker, S. Fly ash based Ni catalyst for conversion of sorbitol into glycols. J. Environ. Chem. Eng. 2015, 3, 1752–1757. [Google Scholar] [CrossRef]
- Jin, X.; Shen, J.; Yan, W.; Zhao, M.; Thapa, P.S.; Subramaniam, B.; Chaudhari, R.V. Sorbitol Hydrogenolysis over Hybrid Cu/CaO-Al2O3 Catalysts: Tunable Activity and Selectivity with Solid Base Incorporation. ACS Catal. 2015, 5, 6545–6558. [Google Scholar] [CrossRef]
- Murillo Leo, I.; López Granados, M.; Fierro, J.L.G.; Mariscal, R. Selective conversion of sorbitol to glycols and stability of nickel–ruthenium supported on calcium hydroxide catalysts. Appl. Catal. B Environ. 2016, 185, 141–149. [Google Scholar] [CrossRef]
- Vijaya Shanthi, R.; Mahalakshmy, R.; Thirunavukkarasu, K.; Sivasanker, S. Hydrogenolysis of sorbitol over Ni supported on Ca- and Ca(Sr)-hydroxyapatites. Mol. Catal. 2018, 451, 170–177. [Google Scholar] [CrossRef]
- Du, W.C.; Zheng, L.P.; Shi, J.J.; Xia, S.X.; Hou, Z.Y. Production of C2 and C3 polyols from D-sorbitol over hydrotalcite-like compounds mediated bi-functional Ni-Mg-Al-Ox catalysts. Fuel Process. Technol. 2015, 139, 86–90. [Google Scholar] [CrossRef]
- Rivière, M.; Perret, N.; Delcroix, D.; Cabiac, A.; Pinel, C.; Besson, M. Solvent Effect in Hydrogenolysis of Xylitol over Bifunctional Ru/MnO/C Catalysts under Alkaline-Free Conditions. ACS Sustain. Chem. Eng. 2018, 6, 4076–4085. [Google Scholar] [CrossRef]
- Neaţu, F.; Petrea, N.; Petre, R.; Somoghi, V.; Florea, M.; Parvulescu, V.I. Oxidation of 5-hydroxymethyl furfural to 2,5-diformylfuran in aqueous media over heterogeneous manganese based catalysts. Catal. Today 2016, 278, 66–73. [Google Scholar] [CrossRef]
- Jeong, D.; Jin, K.; Jerng, S.E.; Seo, H.; Kim, D.; Nahm, S.H.; Kim, S.H.; Nam, K.T. Mn5O8 Nanoparticles as Efficient Water Oxidation Catalysts at Neutral pH. ACS Catal. 2015, 5, 4624–4628. [Google Scholar] [CrossRef]
- Hem, J. Chemical Equilibria and Rates of Manganese Oxidation; Water-Supply Paper 1667-A; United States Government Publishing Office: Washington, DC, USA, 1963. [CrossRef]
- Robinson, D.M.; Go, Y.B.; Mui, M.; Gardner, G.; Zhang, Z.; Mastrogiovanni, D.; Garfunkel, E.; Li, J.; Greenblatt, M.; Dismukes, G.C. Photochemical Water Oxidation by Crystalline Polymorphs of Manganese Oxides: Structural Requirements for Catalysis. J. Am. Chem. Soc. 2013, 135, 3494–3501. [Google Scholar] [CrossRef] [PubMed]
- Gao, T.; Norby, P.; Krumeich, F.; Okamoto, H.; Nesper, R.; Fjellvåg, H. Synthesis and Properties of Layered-Structured Mn5O8 Nanorods. J. Phys. Chem. C 2010, 114, 922–928. [Google Scholar] [CrossRef]
- Rekha, V.; Raju, N.; Sumana, C.; Paul Douglas, S.; Lingaiah, N. Selective Hydrogenolysis of Glycerol Over Cu-ZrO2-MgO Catalysts. Catal. Lett. 2016, 146, 1487–1496. [Google Scholar] [CrossRef]
- Mazzieri, V.; Coloma-Pascual, F.; Arcoya, A.; L’Argentière, P.C.; Figoli, N.S. XPS, FTIR and TPR characterization of Ru/Al2O3 catalysts. Appl. Surf. Sci. 2003, 210, 222–230. [Google Scholar] [CrossRef]
- Ishikawa, M.; Tamura, M.; Nakagawa, Y.; Tomishige, K. Demethoxylation of guaiacol and methoxybenzenes over carbon-supported Ru–Mn catalyst. Appl. Catal. B Environ. 2016, 182, 193–203. [Google Scholar] [CrossRef]
- Watanabe, M.; Aizawa, Y.; Iida, T.; Nishimura, R.; Inomata, H. Catalytic glucose and fructose conversions with TiO2 and ZrO2 in water at 473K: Relationship between reactivity and acid–base property determined by TPD measurement. Appl. Catal. A Gen. 2005, 295, 150–156. [Google Scholar] [CrossRef]
- Bancquart, S.; Vanhove, C.; Pouilloux, Y.; Barrault, J. Glycerol transesterification with methyl stearate over solid basic catalysts: I. Relationship between activity and basicity. Appl. Catal. A Gen. 2001, 218, 1–11. [Google Scholar] [CrossRef]
- Tajvidi, K.; Hausoul, P.J.C.; Palkovits, R. Hydrogenolysis of cellulose over Cu-based catalysts-analysis of the reaction network. ChemSusChem 2014, 7, 1311–1317. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.; Li, X.; Zeng, T.; Hong, W.; Cheng, Z.; Yuan, W. Kinetics of Hydrogenolysis of Glycerol to Propylene Glycol over Cu-ZnO-Al2O3 Catalysts. Chin. J. Chem. Eng. 2010, 18, 384–390. [Google Scholar] [CrossRef]
- Hirano, Y.; Sagata, K.; Kita, Y. Selective transformation of glucose into propylene glycol on Ru/C catalysts combined with ZnO under low hydrogen pressures. Appl. Catal. A Gen. 2015, 502, 1–7. [Google Scholar] [CrossRef]
- Sun, J.; Liu, H. Selective hydrogenolysis of biomass-derived xylitol to ethylene glycol and propylene glycol on Ni/C and basic oxide-promoted Ni/C catalysts. Catal. Today 2014, 234, 75–82. [Google Scholar] [CrossRef]
- Ketchie, W.C.; Maris, E.P.; Davis, R.J. In-situ X-ray Absorption Spectroscopy of Supported Ru Catalysts in the Aqueous Phase. Chem. Mater. 2007, 19, 3406–3411. [Google Scholar] [CrossRef]
- Trinh, T.-K.-H.; de Hemptinne, J.-C.; Lugo, R.; Ferrando, N.; Passarello, J.-P. Hydrogen Solubility in Hydrocarbon and Oxygenated Organic Compounds. J. Chem. Eng. Data 2016, 61, 19–34. [Google Scholar] [CrossRef]
- Hu, C.-C.; Wu, Y.-T.; Chang, K.-H. Low-Temperature Hydrothermal Synthesis of Mn3O4 and MnOOH Single Crystals: Determinant Influence of Oxidants. Chem. Mater. 2008, 20, 2890–2894. [Google Scholar] [CrossRef]
- Menezes, P.W.; Indra, A.; Littlewood, P.; Schwarze, M.; Göbel, C.; Schomäcker, R.; Driess, M. Nanostructured manganese oxides as highly active water oxidation catalysts: A boost from manganese precursor chemistry. ChemSusChem 2014, 7, 2202–2211. [Google Scholar] [CrossRef] [PubMed]



| Entry | Catalysts | Nominal Loading [wt %] | Experimental Loading 1 [wt %] | Δ 2 [%] | SBET 3 [m2 g−1] | Crystallite Size of Ru 4 [nm] | Total Number of Sites [µmol g−1] | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Ru | Mn | M | Ru | Mn | M | Basic 5 | Acid 6 | |||||
| 1 | Ru/(Mn-Al)OX | 3 | 56 | 13 | n.m. | 66.5 | 0.5 | 97 | 13 | 6 | 28 (2.2) | 18 (1.3) |
| 2 | Ru-(Mn-Al)OX | 3 | 70 | 4 | n.m. | 68.4 | 1.4 | 60 | 26 | 29 | 44 (1.7) | 49 (1.8) |
| 3 | Ru-(Mn-Ti)OX | 3 | 64 | 11 | n.m. | 55.9 | 7.6 | 32 | 47 | 29 | 127 (2.7) | 66 (1.4) |
| 4 | Ru-(Mn-Zr)OX | 3 | 60 | 15 | n.m. | 56.7 | 10.0 | 29 | 61 | 27 | 123 (2.0) | 81 (1.3) |
| 5 | Ru-(Mn-Zn)OX | 3 | 60 | 15 | n.m. | 54.7 | 13.4 | 10 | 37 | 13 | 123 (3.3) | 83 (2.2) |
| Catalyst | Activity [h−1] | Carbon Selectivity 2 [%] | CBT 4 [%] | ||||||
|---|---|---|---|---|---|---|---|---|---|
| EG | PG | GLY | LA | C4 Products 3 | C5 Alditols | Gas Phase Products | |||
| Ru/(Mn-Al)OX | 111 | 12 | 17 | 8 | 4 | 11 | 6 | 32 | 90 |
| Ru-(Mn-Al)OX | 57 | 21 | 28 | 9 | 3 | 5 | 2 | 20 | 87 |
| Ru-(Mn-Zr)OX | 33 | 21 | 29 | 9 | 2 | 5 | 3 | 23 | 92 |
| Ru-(Mn-Ti)OX | 33 | 22 | 29 | 8 | 2 | 7 | 3 | 26 | 99 |
| Ru-(Mn-Zn)OX | 0 | - | - | - | - | - | - | - | - |
| Run | Activity [h−1] | Carbon Selectivity 2 [%] | CBT 4 [%] | ||||||
|---|---|---|---|---|---|---|---|---|---|
| EG | PG | GLY | LA | C4 Products 3 | C5 Alditols | Gas Phase Products | |||
| 1 | 26 | 20 | 25 | 12 | 1 | 8 | 3 | 10 | 85 |
| 2 | 17 | 16 | 24 | 10 | 1 | 12 | 5 | 15 | 83 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rivière, M.; Perret, N.; Delcroix, D.; Cabiac, A.; Pinel, C.; Besson, M. Ru-(Mn-M)OX Solid Base Catalysts for the Upgrading of Xylitol to Glycols in Water. Catalysts 2018, 8, 331. https://doi.org/10.3390/catal8080331
Rivière M, Perret N, Delcroix D, Cabiac A, Pinel C, Besson M. Ru-(Mn-M)OX Solid Base Catalysts for the Upgrading of Xylitol to Glycols in Water. Catalysts. 2018; 8(8):331. https://doi.org/10.3390/catal8080331
Chicago/Turabian StyleRivière, Maxime, Noémie Perret, Damien Delcroix, Amandine Cabiac, Catherine Pinel, and Michèle Besson. 2018. "Ru-(Mn-M)OX Solid Base Catalysts for the Upgrading of Xylitol to Glycols in Water" Catalysts 8, no. 8: 331. https://doi.org/10.3390/catal8080331
APA StyleRivière, M., Perret, N., Delcroix, D., Cabiac, A., Pinel, C., & Besson, M. (2018). Ru-(Mn-M)OX Solid Base Catalysts for the Upgrading of Xylitol to Glycols in Water. Catalysts, 8(8), 331. https://doi.org/10.3390/catal8080331