Transformation of Sugar Maple Bark through Catalytic Organosolv Pulping
Abstract
:1. Introduction
2. Results and Discussion
2.1. Bark Extractives from Ethanol–Water Pretreatment
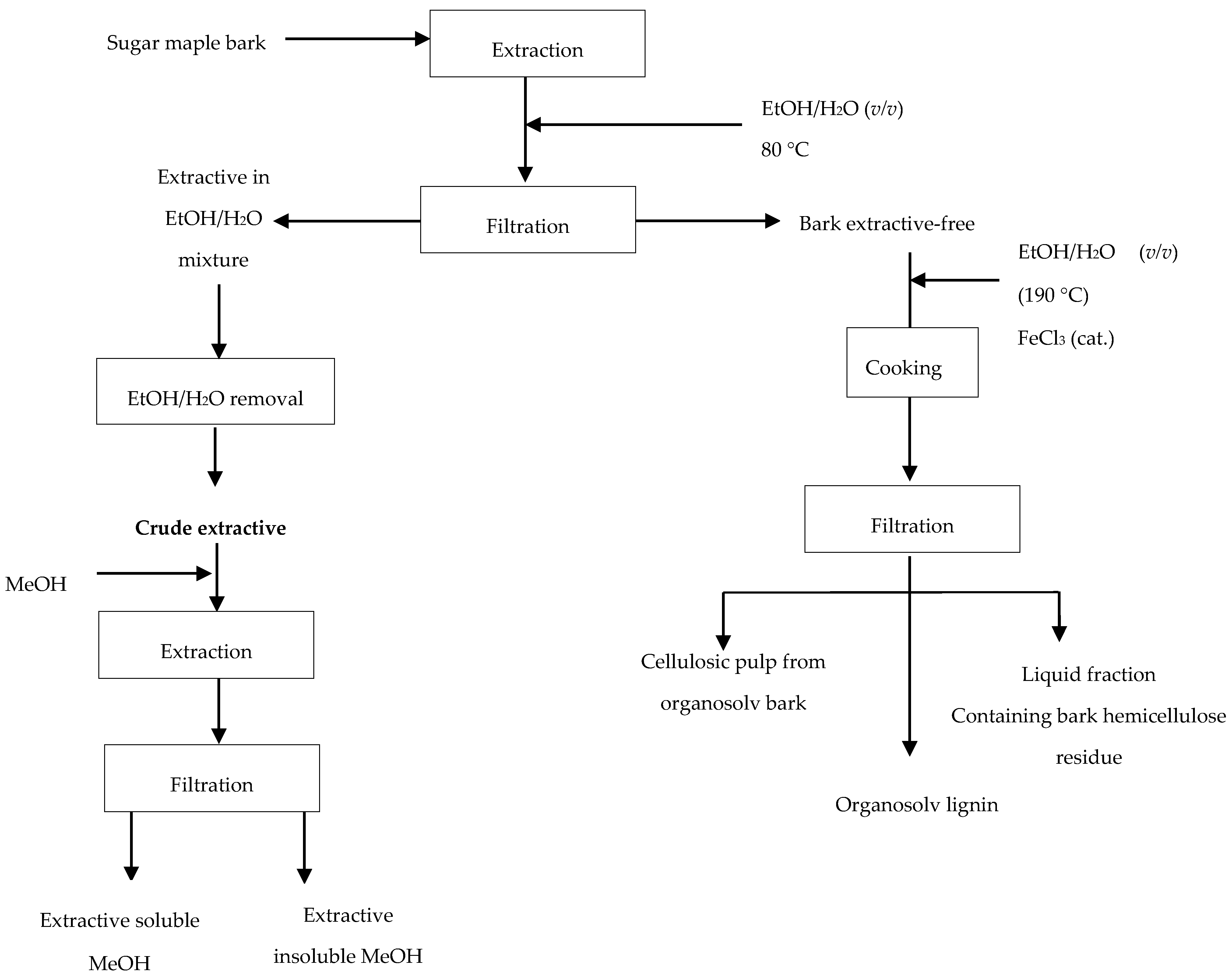
2.2. Organosolv Fractionation
2.2.1. Organosolv Lignin
2.2.2. Cellulosic Pulp from Sugar Maple Bark
2.2.3. Water Soluble Fraction of Residual Liquor
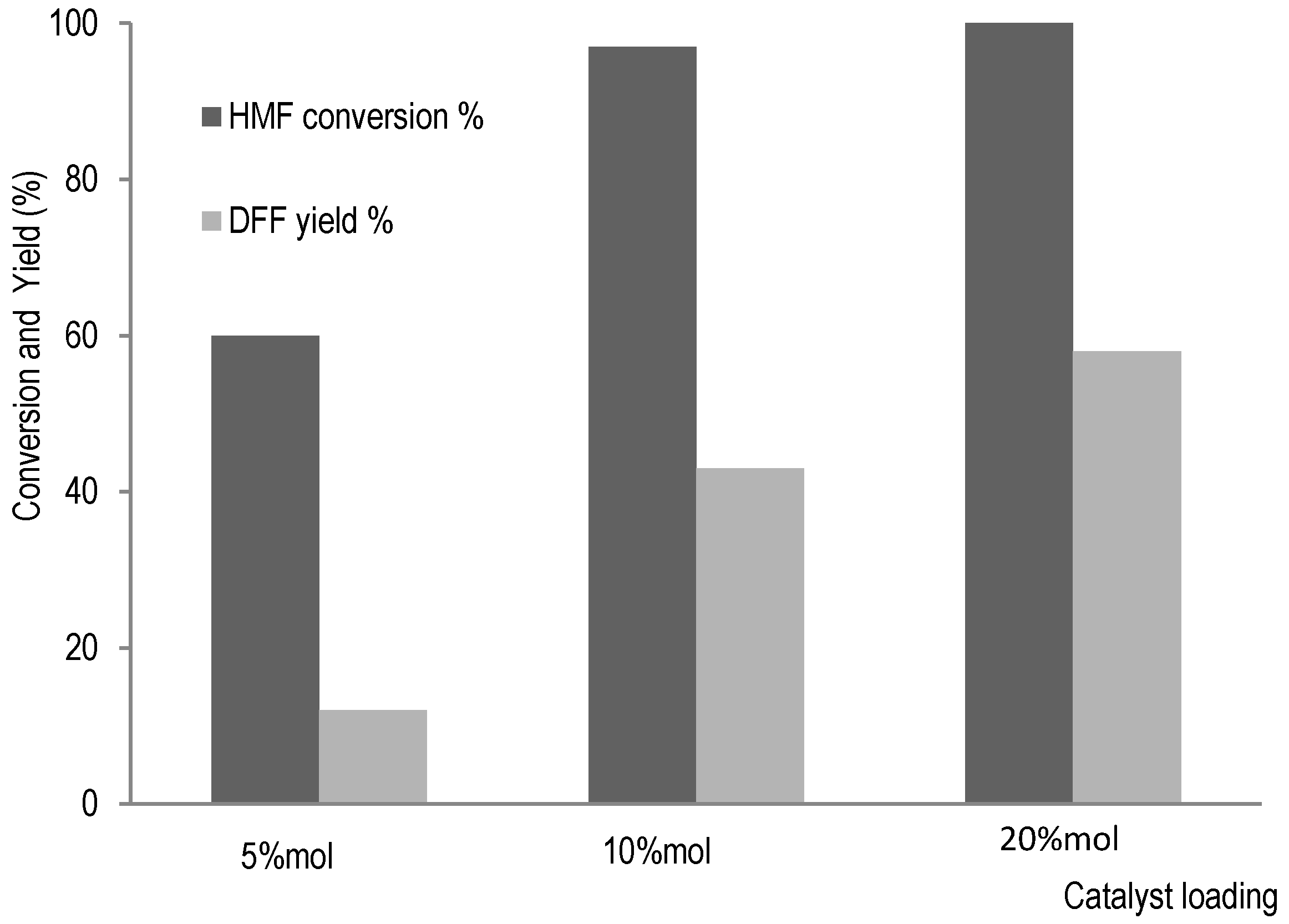
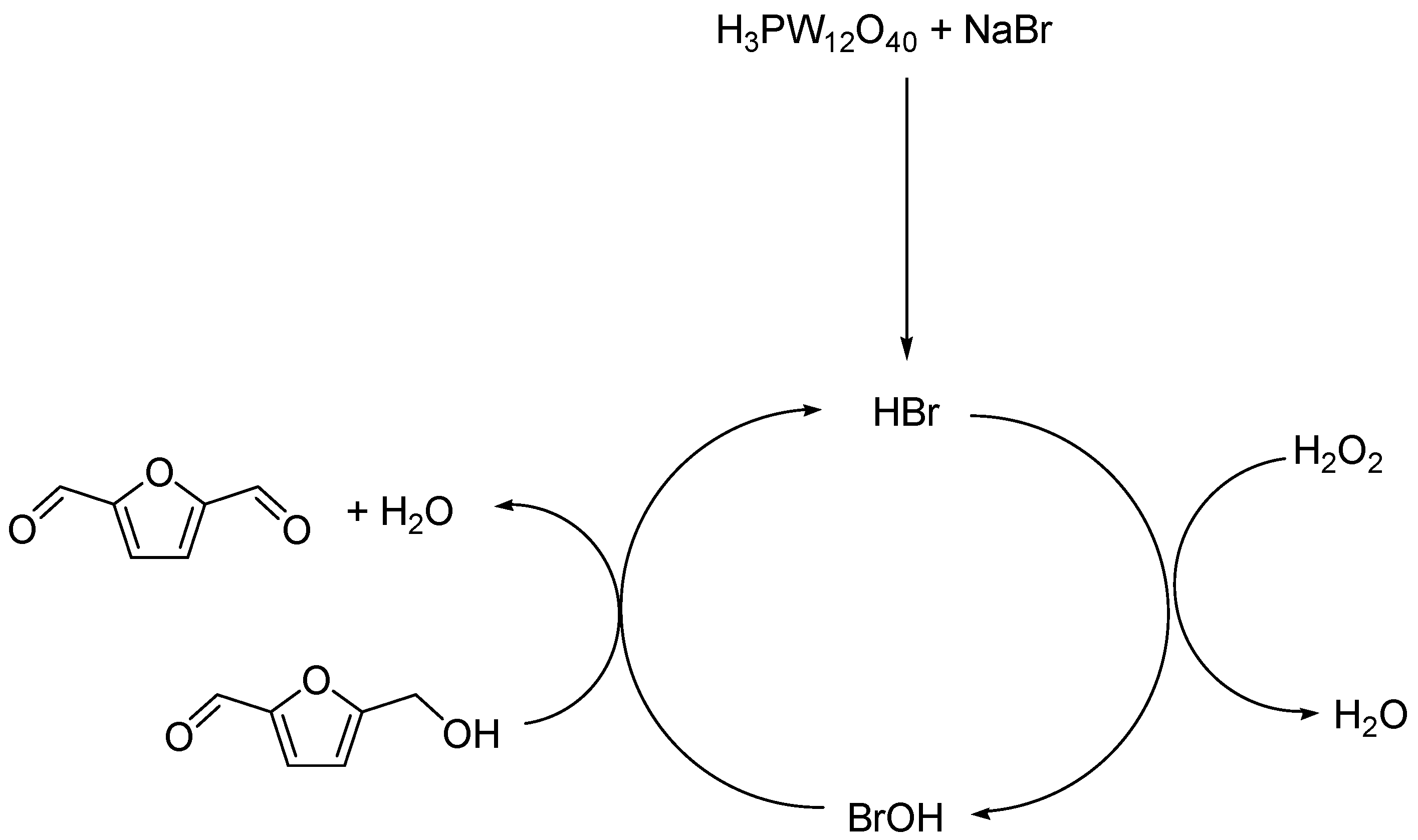
2.2.4. Hemicellulose Valorisation Pathway from HMF Transformation
3. Materials and Methods
3.1. Material
Chemical Composition of Sugar Maple Wood and Bark
3.2. Methods
3.2.1. Pre-Extraction Procedure
3.2.2. Determination of Total Phenol Content of the Bark Extracts
3.2.3. Determination of Proanthocyanidin Content of the Bark Extracts
3.2.4. GC-MS Analyses
3.2.5. XPS Analyses
3.2.6. Nitrogen Analysis
3.2.7. DFF Synthesis from HMF
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- State of Canada’s Forests: Annual Report for 2016; Natural Resources Canada: Library and Archives Canada Cataloguing in Publication. Cat. No. Fo1-6E-PDF (Online); Library and Archives Canada: Gatineau, QC, Canada, 2016.
- Baptista, I.; Miranda, I.; Quilhó, T.; Gominho, J.; Pereira, H. Characterisation and fractioning of Tectona grandis bark in view of its valorisation as a biorefinery raw-material. Ind. Crop. Prod. 2013, 50, 166–175. [Google Scholar] [CrossRef]
- Ruiz-Aquinoa, F.; González-Pena, M.M.; Valdez-Hernández, J.I.; Revilla, U.S.; Romero-Manzanares, A. Chemical characterization and fuel properties of wood and bark of two oaks from Oaxaca, Mexico. Ind. Crop. Prod. 2015, 65, 90–95. [Google Scholar] [CrossRef]
- Diouf, P.N.; Stevanovic, T.; Cloutier, A. Antioxidant properties and polyphenol contents of trembling aspen bark extracts. Wood Sci. Technol. 2009, 43, 457–470. [Google Scholar] [CrossRef]
- Royer, M.; Diouf, P.N.; Stevanovic, T. Polyphenol contents and radical scavenging capacities of red maple (Acer rubrum L.) extracts. Food Chem. Toxicol. 2011, 49, 2180–2188. [Google Scholar] [CrossRef] [PubMed]
- St-Pierre, F.; Achim, A.; Stevanovic, T. Composition of ethanolic extracts of wood and bark from Acer saccharum and Betula alleghaniensis trees of different vigor classes. Ind. Crop. Prod. 2013, 41, 179–187. [Google Scholar] [CrossRef]
- Zeng, X.-L.; Fu, G.M.; Tian, K.; Sun, J.X.; Xiong, H.B.; Huang, X.Z.; Jiang, Z.Y. Acutissimanide, a new lignan with antioxidant activity isolated from the bark of Quercus acutissima Carruth. Nat. Prod. Res. 2014, 28, 1364–1370. [Google Scholar] [CrossRef] [PubMed]
- Miranda, I.; Gominho, J.; Mirra, I.; Pereira, H. Chemical characterization of barks from Picea abies and Pinus sylvestris after fractioning into different particle sizes. Ind. Crop. Prod. 2012, 36, 395–400. [Google Scholar] [CrossRef]
- Kofujita, H.; Ettyu, K.; Ota, M. Characterization of the major components in bark from five Japanese tree species for chemical utilization. Wood Sci. Technol. 1999, 33, 223–228. [Google Scholar] [CrossRef]
- Costa, E.C.A.; Pinto, P.C.R.; Rodrigues, A.E. Evaluation of chemical processing impact on E. globulus wood lignin and comparison with bark lignin. Ind. Crop. Prod. 2014, 61, 479–491. [Google Scholar] [CrossRef]
- Churms, S.C.; Stephen, A.M. Chromatographic separation and examination of carbohydrate and phenolic components of the non-tannin fraction of black wattle (Acacia mearnsii) bark extract. J. Chromatogr. 1991, 550, 519–537. [Google Scholar] [CrossRef]
- Nair, S.S.; Yan, N. Bark derived submicron-sized and nano-sized cellulose fibers: Fromindustrial waste to high performance materials. Carbohyd. Polym. 2015, 134, 258–266. [Google Scholar] [CrossRef] [PubMed]
- Vispute, T.P.; Huber, G.W. Production of hydrogen, alkanes and polyols by aqueous phase processing of wood-derived pyrolysis oils. Green Chem. 2009, 11, 1433–1445. [Google Scholar] [CrossRef]
- Lima, M.A.; Lavorente, G.B.; da Silva, H.K.P.; Bragatto, J.; Rezende, C.A.; Bernardinelli, O.D.; de Azevedo, E.R.; Gomez, L.D.; McQueen-Mason, S.J.; Labate, C.A.; et al. Effects of pretreatment on morphology, chemical composition and enzymatic digestibility of eucalyptus bark: A potentially valuable source of fermentable sugars for biofuel production—Part 1. Biotechnol. Biofuels 2013, 6, 75. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vicente, A.I.; Coelho, J.A.S.; Simeonov, S.P.; Lazarova, H.I.; Popova, M.D.; Afonso, C.A.M. Oxidation of 5-Chloromethylfurfural (CMF) to 2,5-Diformylfuran (DFF). Molecules 2017, 22, 329. [Google Scholar] [CrossRef] [PubMed]
- Karnaouri, A.; Rova, U.; Christakopoulos, P. Effect of Different Pretreatment Methods on Birch Outer Bark: New Biorefinery Routes. Molecules 2016, 21, 427. [Google Scholar] [CrossRef] [PubMed]
- Koumba-Yoya, G.; Stevanovic, T. New biorefinery strategy for high purity lignin production. Chem. Select 2016, 1, 6562–6570. [Google Scholar] [CrossRef]
- Galkin, M.V.; Samec, J.S.M. Lignin Valorization through Catalytic Lignocellulose Fractionation: A Fundamental Platform for the Future Biorefinery. ChemSusChem 2016, 9, 1544–1558. [Google Scholar] [CrossRef] [PubMed]
- Stevanovic, T.; Koumba-Yoya, G. Organosolv Process for the Extraction of Highly Pure Lignin and Products Comprising the Same. WO 2016197233 A1, 15 December 2016. [Google Scholar]
- Koumba-Yoya, G.; Stevanovic, T. Study of Organosolv Lignins as Adhesives in Wood Panel Production. Polymers 2017, 9, 46. [Google Scholar] [CrossRef]
- Kirk, T.K.; Farrell, R.L. Enzymatic Combustion: The Microbial-Degradation of Lignin. Annu. Rev. Microbiol. 1987, 41, 465–505. [Google Scholar] [CrossRef] [PubMed]
- Dashtban, M.; Schraft, H.; Syed, T.A.; Qin, W. Fungal biodegradation and enzymatic modification of lignin. Int. J. Biochem. Mol. Biol. 2010, 1, 36–50. [Google Scholar] [PubMed]
- Jayasekara, T.K.; Stevenson, P.C.; Belmain, S.R.; Farman, D.I.; Hall, D.R. Identification of methyl salicylate as the principal volatile component in the methanol extract of root bark of Securidaca longepedunculata Fers. J. Mass. Spectrom. 2002, 37, 577–580. [Google Scholar] [CrossRef] [PubMed]
- Vázquez, G.; Fontenla, E.; Santos, J.; Freire, M.S.; González-Álvarez, J.; Antorrena, G. Antioxidant activity and phenolic content of chestnut (Castanea sativa) shell and eucalyptus (Eucalyptus globulus) bark extracts. Ind. Crop. Prod. 2008, 28, 279–285. [Google Scholar] [CrossRef]
- Lee, D.G.; Park, Y.; Kim, M.R.; Jung, H.J.; Seu, Y.B.; Hahm, K.; Woo, E.R. Anti-fungal effects of phenolic amides isolated from the root bark of Lycium chinense. Biotechnol. Lett. 2004, 26, 1125–1130. [Google Scholar] [CrossRef] [PubMed]
- Pasztory, Z.; Mohacsine, I.R.; Gorbacheva, G.; Borcsok, Z. The utilization of tree bark. Bioresources 2016, 11, 7859–7888. [Google Scholar] [CrossRef]
- Plomion, C.; Leprovost, G.; Stokes, A. Wood Formation in Trees. Plant Physiol. 2001, 127, 1513–1523. [Google Scholar] [CrossRef] [PubMed]
- Rencoret, J.; Ralph, J.; Marques, G.; Gutiérrez, A.; Martínez, A.T.; Del Río, J.C. Structural Characterization of Lignin Isolated from Coconut (Cocos nucifera) Coir Fibers. J. Agric. Food Chem. 2013, 61, 2434–2445. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Silva, M.C.; Lopes, O.R.; Colodette, J.L.; Porto, A.O.; Rieumont, J.; Chaussy, D.; Belgacem, M.N.; Silva, G.G. Characterization of three non-product materials from a bleached eucalyptus kraft pulp mill, in view of valorising them as a source of cellulose fibres. Ind. Crop. Prod. 2008, 27, 288–295. [Google Scholar] [CrossRef]
- Higuchi, T. Biochemistry and Molecular Biology of Wood; Springer: Berlin/Heidelberg, Germany, 2012; p. 94. [Google Scholar]
- Hu, R.F.; Lin, L.; Liu, T.J.; Liu, S.J. Dilute sulfuric acid hydrolysis of sugar maple wood extract at atmospheric pressure. Bioresour. Technol. 2010, 101, 3586–3594. [Google Scholar] [CrossRef] [PubMed]
- Xie, R.; Tu, M.; Wu, Y.; Adhikari, S. Improvement in HPLC separation of acetic acid and levulinic acid in the profiling of biomass hydrolysate. Bioresour. Technol. 2011, 102, 4938–4942. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Qi, W.; Su, R.; He, Z. Selective synthesis of 2,5-diformylfuran and 2,5-Furandicarboxylic acid from 5-Hydroxymethylfurfural and fructose catalyzed by magnetically separable catalysts. Energy Fuels 2017, 31, 533–541. [Google Scholar] [CrossRef]
- Joshi, G.; Patil, R.D.; Adimurthy, S. Green bromine: In situ generated catalyst for the selective oxidation of alcohols using H2O2 as a benign oxidant. RSC Adv. 2012, 2, 2235–2239. [Google Scholar] [CrossRef]
- Girka, Q.; Estrine, B.; Hoffmann, N.; Le Bras, J.; Marinković, S.; Muzart, J. Simple efficient one-pot synthesis of 5-hydroxymethylfurfural and 2,5-diformylfuran from carbohydrates. React. Chem. Eng. 2016, 1, 176–182. [Google Scholar] [CrossRef]
- Chatterjee, M.; Ishizaka, T.; Chatterjee, A.; Kawanami, H. Dehydrogenation of 5-hydroxymethylfurfural to diformylfuran in compressed carbon dioxide: An oxidant free approach. Green Chem. 2017, 19, 1315–1326. [Google Scholar] [CrossRef]
- Halliday, G.A.; Young, R.J., Jr.; Grushin, V.V. One-pot, two-step, practical catalytic synthesis of 2,5-diformylfuran from fructose. Org. Lett. 2003, 5, 2003–2005. [Google Scholar] [CrossRef] [PubMed]
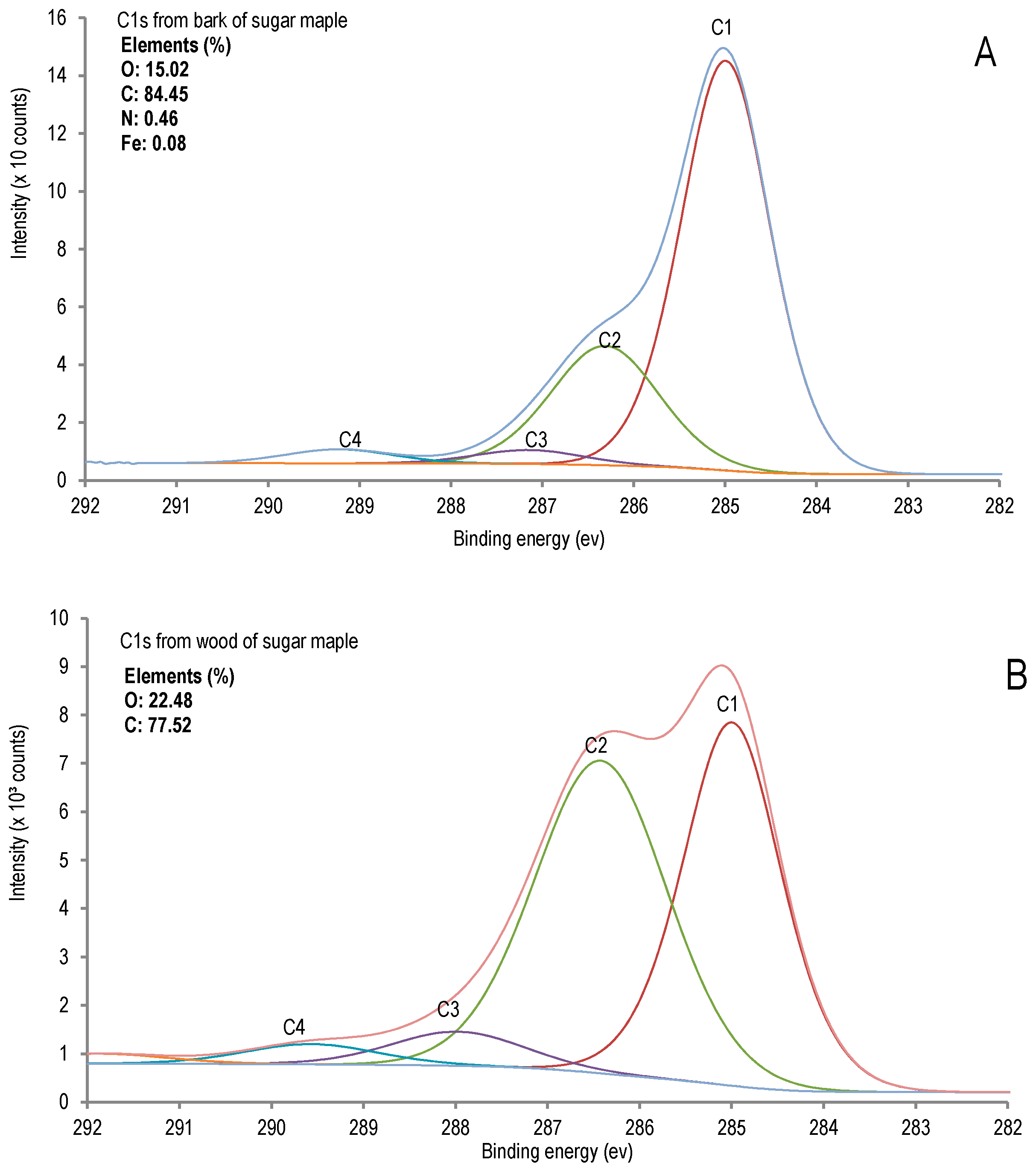


| Entry | Name | Bond Type | Position | Atomic % | |
|---|---|---|---|---|---|
| Wood | Bark | ||||
| 1 | C1 | C-C, C-H | 285 | 42.42 | 69.91 |
| 2 | C2 | C-O, C-O-C | 287 | 47.76 | 24.38 |
| 3 | C3 | O-C-O, C=O | 288 | 5.20 | 2.84 |
| 4 | C4 | O-C=O | 289 | 3.07 | 2.88 |
| Extracts | Yield (%) | Total Phenol Content (mg GAE/g) a | Proanthocyanidin Content (mg CChE/g) b |
|---|---|---|---|
| Total extractive from ethanol–water | 5.2 * | - | - |
| Halite (crystal fraction) | 9.1 ** | - | - |
| MeOH insoluble | 54.7 ** | 130.1 | - |
| MeOH soluble | 36.2 ** | 212.7 | 50.6 |
| Conditions | Lignin Yield * (wt % of O.D Bark) | KL ** (%) | ASL * (%) | Glucose * (%) | Xylose * (%) | Ash * (%) | Nitrogen Content % (Protein %) |
|---|---|---|---|---|---|---|---|
| EtOH/H2O (1:1; v/v); FeCl3 (cat.) | 24.1 | 92.6 ± 0.5 | 3.5 ± 0.1 | 1.5 ± 0.7 | N.D. | 0.08 ± 0.9 | 0.29 (1.78) |
| Conditions | Pulp Yield (wt % of O.D.) | KL * (%) | ASL ** (%) | Glucose ** (%) | Xylose ** (%) | Calcium Oxalate (%) | Nitrogen Content % (Protein %) | Ash (%) |
|---|---|---|---|---|---|---|---|---|
| EtOH/H2O (1:1; v/v): FeCl3 (cat.) | 42.3 | 27.5 ± 0.5 | 4.3 ± 0.1 | 43.2 ± 0.8 | N.D. | 10.3 ± 0.1 | 0.57 (3.5) | 9.2 ± 0.9 |
| Conditions | Water Soluble Fraction * (wt % Based on O.D Bark) | Glucose (%) | Xylose (%) | Furfural (%) | HMF (%) |
|---|---|---|---|---|---|
| EtOH/H2O (1:1; v/v); FeCl3 (cat.) | 23.2 | 0.91 ± 0.6 | 2.1 ± 0.3 | 0.35 ± 0.1 | 0.74 ± 0.2 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Koumba-Yoya, G.; Stevanovic, T. Transformation of Sugar Maple Bark through Catalytic Organosolv Pulping. Catalysts 2017, 7, 294. https://doi.org/10.3390/catal7100294
Koumba-Yoya G, Stevanovic T. Transformation of Sugar Maple Bark through Catalytic Organosolv Pulping. Catalysts. 2017; 7(10):294. https://doi.org/10.3390/catal7100294
Chicago/Turabian StyleKoumba-Yoya, Georges, and Tatjana Stevanovic. 2017. "Transformation of Sugar Maple Bark through Catalytic Organosolv Pulping" Catalysts 7, no. 10: 294. https://doi.org/10.3390/catal7100294






