2.2. Kinetic Model
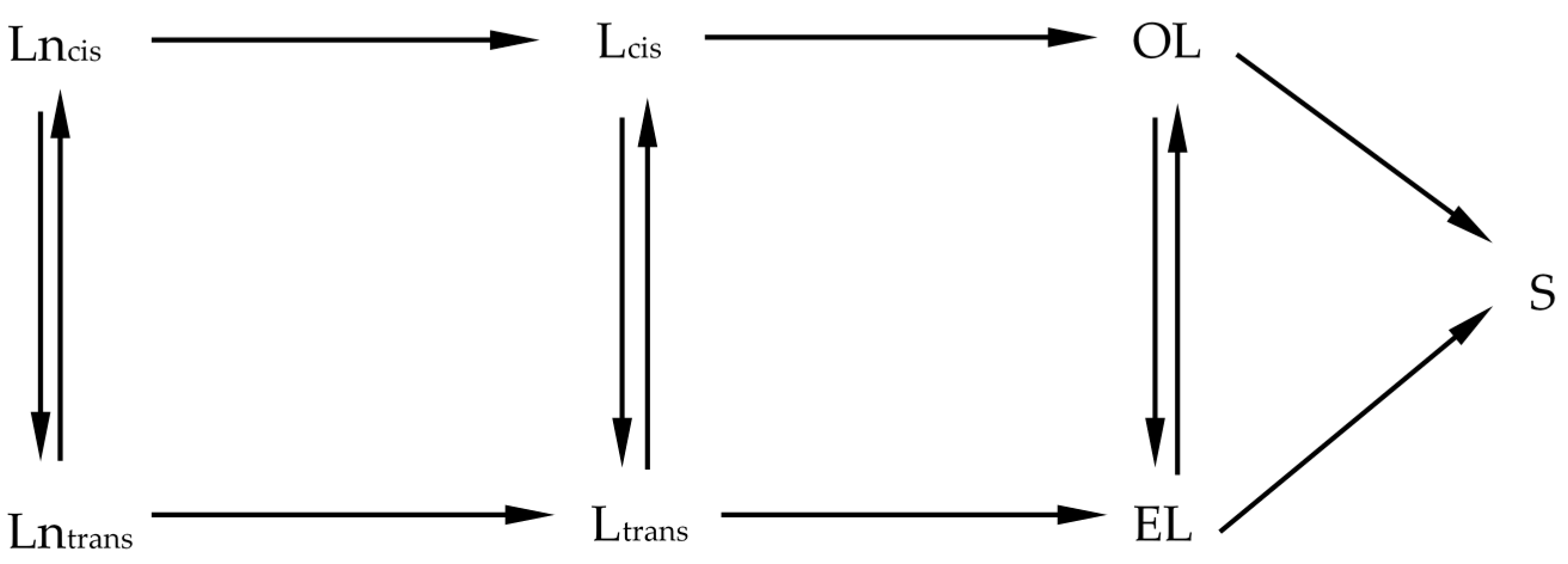
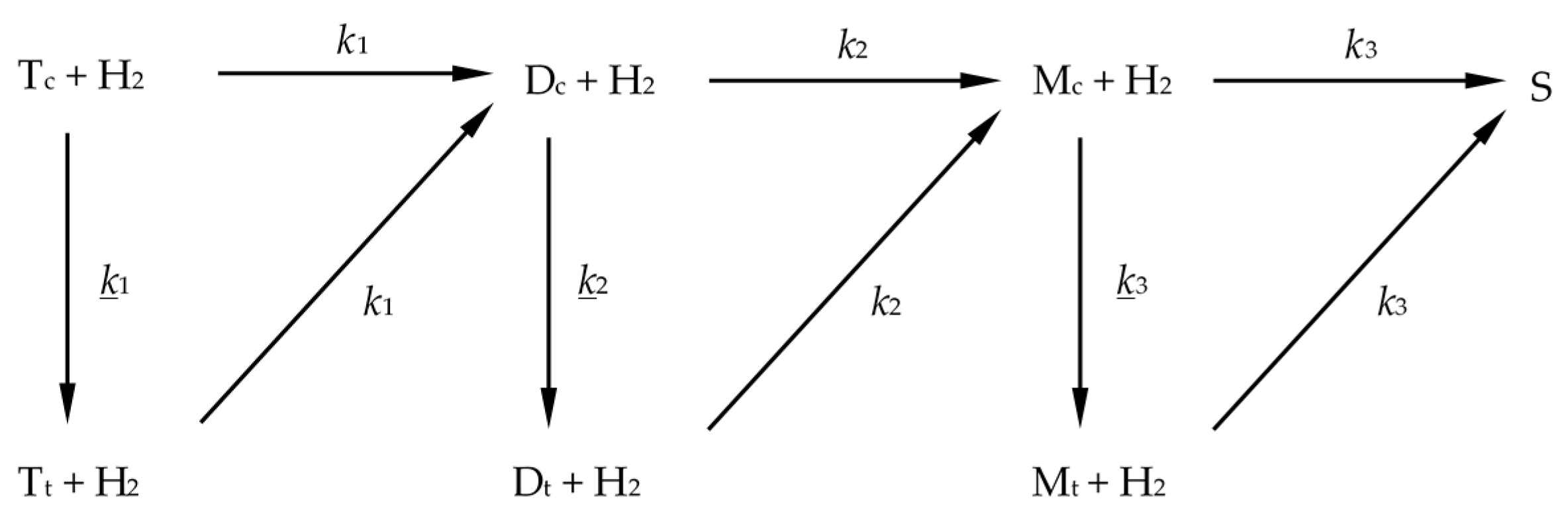
Based on the literature reports, the reaction network shown in
Scheme 5 was proposed.
The overall hydrogenation pathway involves the consecutive saturation of
cis C18:3 to
cis C18:2,
cis C18:2 to
cis C18:1, and
cis C18:1 to C18:0, as well as the parallel irreversible isomerization of
cis forms to
trans ones. The reaction pathway may also involve the partial saturation of
trans C18:3 to
cis C18:2,
trans C18:2 to
cis C18:1, and hydrogenation of
trans C18:1 to C18:0. The
trans C18:3, C18:2, and C18:1 notations include all of the positional and geometrical isomers of linolenic, linoleic, and oleic acids, respectively. Such an approach to simplify the complex reaction network has also been used by other researchers [
10,
23]. Moreover, several other assumptions were made: (a) at each hydrogenation stage a
cis form may isomerize to a
trans form in an irreversible manner; (b) at each step both forms of a fatty acid react with hydrogen with the same reaction rate constant leading to a
cis unsaturated fatty acid or saturated stearic acid in the last step; and (c) all reactions were considered to follow pseudo first-order kinetics with respect to fatty acids. Furthermore, under experimental conditions used in this work, hydrogen remained in a large excess so that its concentration was constant during the hydrogenation process and this concentration is embedded in the rate constant
k.
The assumption that all reactions are the first order in fatty acids provides a possibility to express the reaction rate
r for all reactions considered with an equation
r =
k ×
c, where
c is a molar concentration of the corresponding fatty acid. Since fatty acids are liquids and hydrogen is supplied as gas, it can be assumed that the volume of the liquid phase remains constant through the hydrogenation process. The number of moles of fatty acids also remains constant according to the
Scheme 5. Therefore, since the measurements of the particular fatty acids are performed in a known reaction volume and in the presence of a known amount of a catalyst , the molar concentration
c can reasonably be replaced by the number of moles of fatty acids
n in the reaction system.
Taking into account the abovementioned assumptions, the rates of reactions shown in
Scheme 5 can be expressed by means of a set of differential Equations (1a–f), where
m is a Ni concentration in the oil:
In the case of studies of the hydrogenation process yielding the fatty acids profiles after various time intervals, the formulas being a solution of this set of differential equations can be used to determine the rate constants for a particular reaction step.
2.3. Effect of Hydrogen Pressure on Kinetics of the Canola Oil Hydrogenation
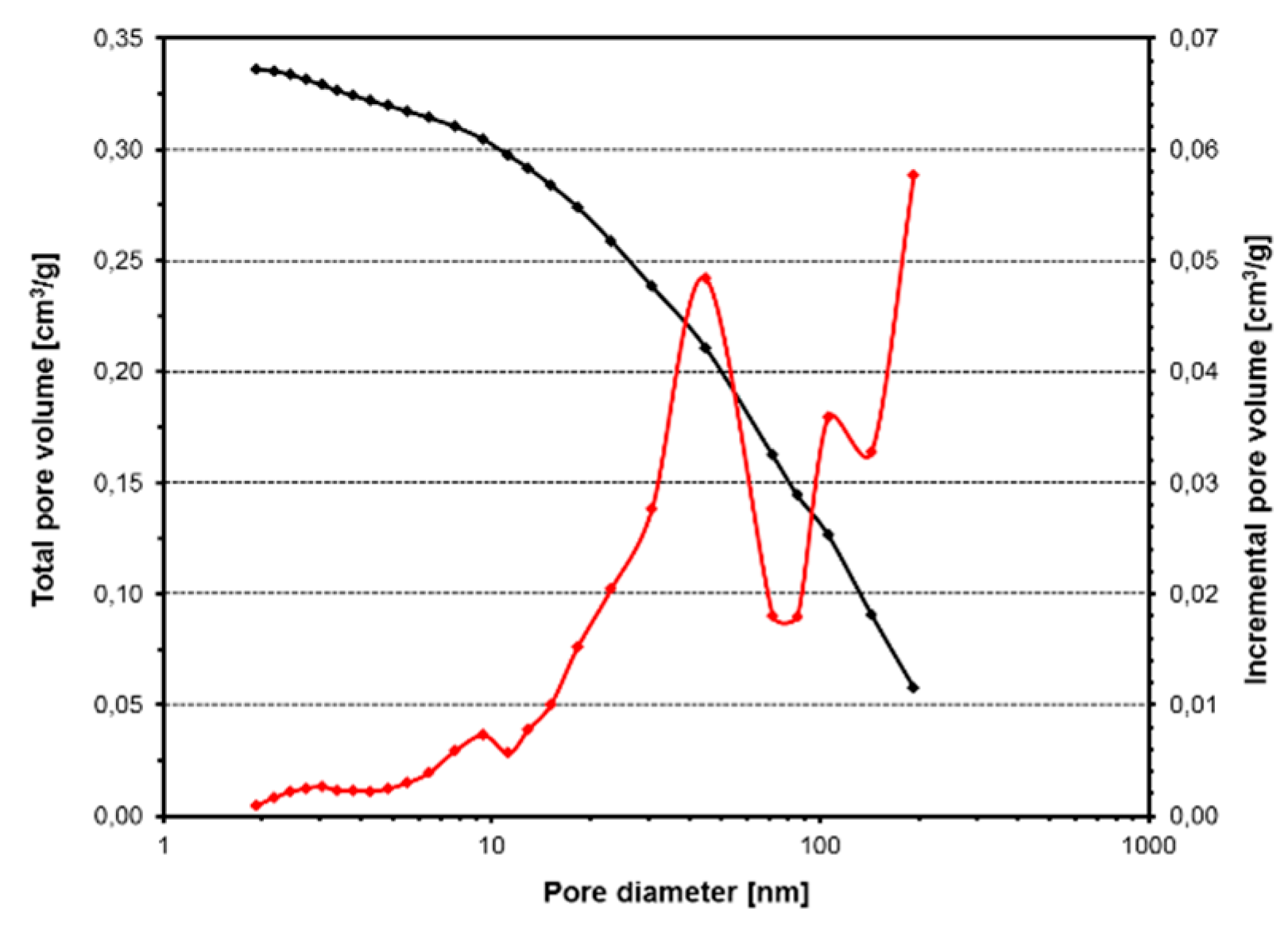
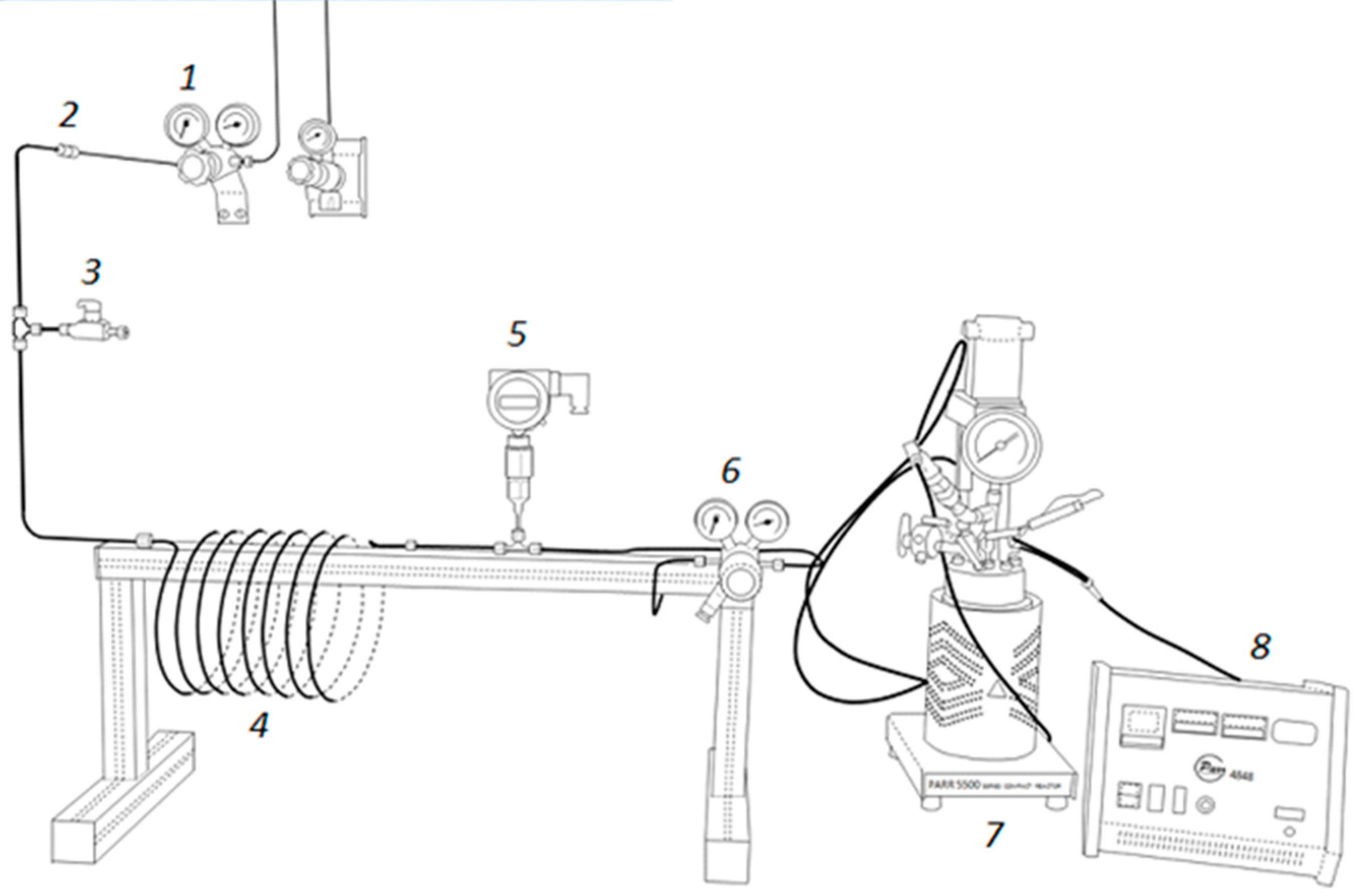
Canola oil was hydrogenated in a batch reactor using an industrial nickel catalyst denoted as INS Ni_C, containing 5.64 wt% Ni. The reactions were performed at 180 °C in a wide range of hydrogen pressures of 1.5 to 21 bar(a). This range corresponds to pressures used in the oil industry for hydrogenation of triglycerides and free fatty acids. It was verified that the agitation rate above 950 rpm had no effect on the hydrogenation rate. This indicates that above the agitation rate of 950 rpm there are no diffusional resistances for a transfer of dissolved hydrogen to the bulk liquid phase and then to the catalyst surface. Moreover, it is known that only nickel located in pores having a diameter of at least 2 nm participates in the hydrogenation reaction [
7]. The pore distribution analysis of the INS Ni precursor revealed pores with a diameter of 10–100 nm and the average pore diameter was
ca. 20 nm. According to the literature data [
23,
28], hampering of the hydrogenation reaction by the diffusion of reactants into the catalyst pores is low for medium-pore and wide-pore catalysts, especially with a diameter of over 4 nm (approx. twice the dimension of triglycerides).
The nickel concentration for each catalytic run was equal to 0.03 wt%. The depth of hydrogenation was controlled by measurement of the iodine value. The iodine value (IV) of an oil or a fatty acid is the mass of iodine in grams that is consumed by 100 grams of the oil/fatty acid. For all tests the hydrogenation depth of canola oil was comparable, as indicated by similar changes of iodine value (monitored by changes of refractive index) and the final iodine value of the hydrogenated canola oil of
ca. 69–71 (determined by Wijs method). Such depth of hydrogenation corresponds to that in the production of fatty acids components for margarine and shortenings on an industrial scale. The results of the hydrogenation tests are compiled in
Table 2. Two approaches for the estimation of the hydrogenation activity were used. In the first one, the activity is expressed by the hydrogen consumption per 1 g of Ni per min. The second approach is based on the overall reaction constant
kr, according to [
23]. It is well-known that the saturation of double bonds follows first-order kinetics with respect to the iodine value drop as shown in Equation (2):
The integration of Equation (2) using the initial conditions that (IV)
t = (IV)
0 at
t = 0 gives:
It is known that the increase of pressure causes a linear growth of hydrogen solubility in accordance with Henry’s Law and, thus, it causes the increase of hydrogen concentration on the catalyst surface. This results in higher hydrogenation rates and lower amounts of
trans isomers and other positional isomers. Wisniak and Albright measured the solubility of hydrogen in cottonseed oil at pressures of 6.8–100 atm and temperatures in the range 50–140 °C and reported that the solubility increased with increased temperature and that Henry’s Law was satisfied up to
ca. 35 atm [
12]. Moreover, Andersson
et al. reported that the solubility of hydrogen in cottonseed oil was nearly independent of the iodine value of the oil, that means independent of a hydrogenation degree [
29]. The effect of pressure on the solubility of hydrogen in canola oil was calculated according to [
29] and is presented in
Figure 2.
In line with expectations, the increase of the hydrogenation activity of the INS Ni_C catalyst was observed with increasing hydrogen pressure. The reaction time required for a decrease of iodine value by
ca. 46–47 units (this corresponds to the hydrogen consumption of
ca. 1000 cm
3) decreased from 39 min for 2.5 bar(a) to 7 min for 21 bar(a). A 5.5-fold increase of the activity was found for two analogous hydrogenation reactions carried out under 2.5 and 21 bar(a). The hydrogenated canola oil samples with iodine value of
ca. 70 were analyzed by means of chromatography for the profile of fatty acids. The contents of monoenes, dienes, trienes, and saturated fatty acids are presented in
Table 3.
The chromatographic analysis results revealed that in the hydrogenated samples (iodine value ~70) the content of elaidic acid, 9t-C18:1, and other positional isomers decreased from 36.1 to 22.7 wt% with the increasing pressure from 2.5 to 21 bar(a). A concurrent increase of the stearic acid content from 13.1 to 22.1 wt% was also observed. Furthermore, some deterioration of the linolenic selectivity was observed at elevated hydrogen pressures as evidenced by an increase of the
cis C18:3 content from 0 to 0.5 wt%. Similar observations were reported by List
et al. for the supported nickel catalyst in the soybean oil hydrogenation at various pressures [
27].
The experimental data of the FA contents in hydrogenated samples of canola oil were used to determine the apparent rate constants
kx of the particular hydrogenation steps depending on the hydrogen pressure. The results are summarized in
Table 4. Since no
trans isomers of linolenic acid in hydrogenated samples were found by means of the chromatographic analysis, the
k1 constant for isomerization of
cis C18:3 to
trans C18:3 could be neglected as equal to 0.
It is known that the rates at which various chains of fatty acids are hydrogenated depend on the number of double bonds. The situation is additionally complicated by the fact that only some double bonds may undergo hydrogenation or isomerization and at different rates depending on the position in the carbon chain. The presented results provide a quantitative measure of the influence of hydrogen pressure on hydrogenation rates of particular reaction steps. The results reveal that, under the pressure of 1.5 bar(a), trienes are hydrogenated more than 2.5 times faster than dienes, and over 65 times faster than monoenes to stearic acid (
k1~2.5×
k2~65×
k3). The increase of values of all apparent rate constants was observed with the increased pressure, with the largest increase being observed for the
k3 constant of hydrogenation of monoenes to stearic acid (about 20 times). As a consequence, under 21 bar(a) the following relations of rate constants were obtained:
k1~1.5×
k2~9×
k3, showing a distinct increase of the monoenes hydrogenation rate. Furthermore, in case of dienes, the
cis to
trans isomerisation rate increased ca. 2.5 times with the increased pressure. However, this increase of the
cis/trans isomerization rate of dienes was lower than the corresponding increase of the dienes hydrogenation rate (1.5 bar(a),
k2/
k2 = 1.6; 21 bar(a),
k2/
k2 = 3.2). This means that higher hydrogen pressure facilitates hydrogenation to a larger extent than
cis/trans isomerization. Similarly, the
cis/trans monoenes isomerization rate was also found to decrease with the increased pressure, the
k3/
k3 ratio changed from 6.1 to 1.6 under 21 bar(a). This observation can be rationalized on the basis of the Horiuti-Polanyi mechanism [
30]. According to this mechanism, the hydrogenation proceeds in two stages through a partially-hydrogenated transition state, in which only one hydrogen atom is bound. Thus, a high concentration of hydrogen on nickel surface favors hydrogenation of the partially-hydrogenated transition state at the expense of isomerization and decreases the contact time of the adsorbed molecule on the catalyst surface. The addition of the second hydrogen atom (saturation) becomes more likely than cleavage of the already bound hydrogen atom that may lead to positional or geometric isomers.
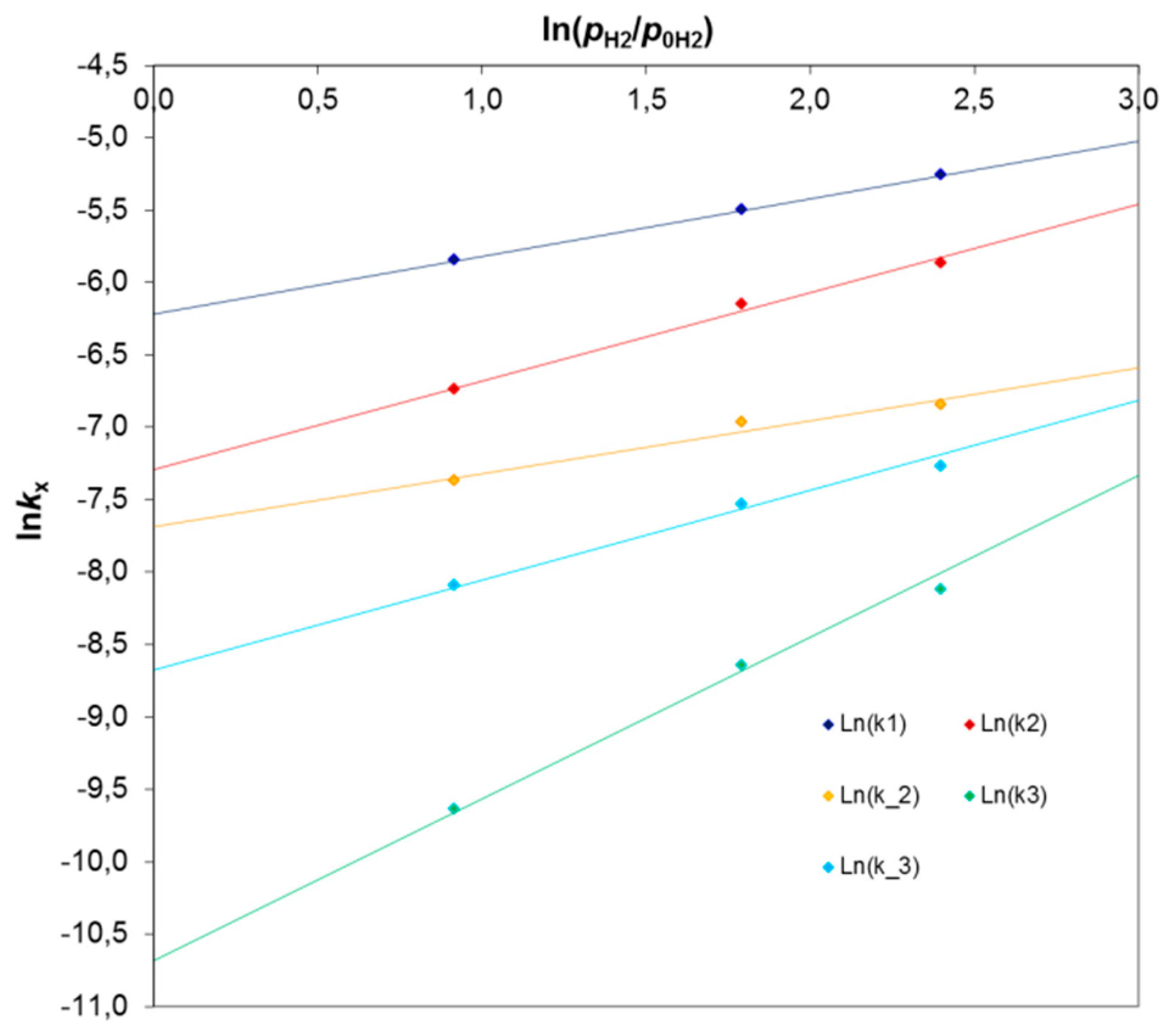
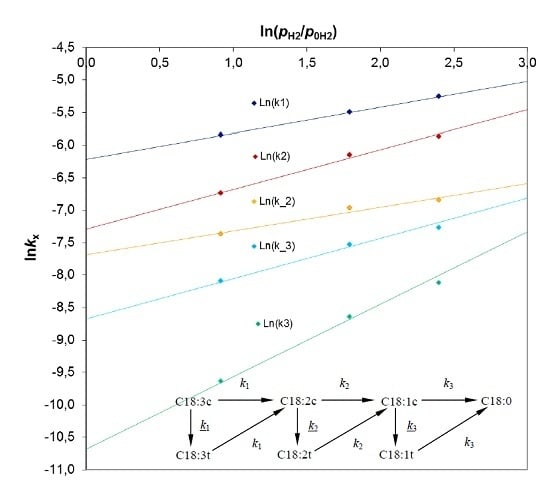
The rate constants at atmospheric pressure (
kx0) were determined from the following formula:
. The linear formula has a form:
where
. A dependence of ln
kx from
for the particular apparent rate constants is presented in
Figure 3. The slope of the line gives the
n exponent, whereas the intercept point provides ln(
kx0) values that are compiled in
Table 5.
In all cases, very good values of R
2 coefficients in the range of 0.98–1.00 were obtained. The reaction orders (
n exponent) with respect to hydrogen were in the range of 0.35 to 1.1. These values are consistent with literature reports indicating that the rates of dienes to monoenes hydrogenation and the
cis/trans monoenes isomerization are half order with respect to the hydrogen concentration [
9], while the hydrogenation reaction of monoenes follows first-order kinetics [
9]. Other authors [
31] arrive at the first order in hydrogen for dienes and an order of 3/2 for monoenes when hydrogenating in the gas phase.
The lumped kinetic model was verified by comparsion of predicted fatty acid contents with the experimental data of FA profiles obtained for pressures 1.5 and 6 bar(a). The experimental points cover the iodine value range from 95 to 70, which is typically of interest during industrial hydrogenation of vegetable oils in order to obtain fatty acids components for the manufacture of margarine and shortenings. The results of predicted profiles of fatty acids (solid lines) along with those obtained experimentally are shown in
Figure 4 and
Figure 5. As can be seen, a reasonably good consistency was found between the experimental and predicted fatty acid contents from the proposed kinetic model. The observed deviations in the course of the hydrogenation reaction between the calculated model and measured experimental points for M-
cis (C18:1
cis) fraction result from measurement errors (deviations in FA analysis by means of GC-MS) and simplifications assumed during the process modelling. The first pair of rate constants
k1 and
k1 is a result of the analytical solution, whereas calculation of other pairs can be done only via numerical iterations. A character of the solved equations allows for determination of rate constants with an accuracy resulting only from constraints of floating-point calculations.
A very important catalyst feature in the hydrogenation reaction of vegetable oils is the catalyst selectivity. In hydrogenation studies there is a tendency to optimize catalyst properties and process parameters so that the most unsaturated fatty acids are hydrogenated to oleic acid with a minimal increase in the amount of stearic acid. According to the well-known and accepted method [
3,
28,
32] selectivity of the hydrogenation reaction of fats and vegetable oils can be determined from the ratio of the corresponding rate constants of consecutive reaction steps in
Scheme 5: linolenic selectivity S
12 =
k1/
k2 (preference for linolenic acid over linoleic acid hydrogenation) and linoleic selectivity S
23 =
k2/
k3 (preference for linoleic acid over oleic acid hydrogenation).
In the oil industry the linoleic selectivity S
23 is the most important parameter. Moreover, it is known that an increase of pressure results in a decrease of the linoleic selectivity.
Table 6 presents the quantitative data on the influence of the hydrogen pressure on various selectivities. Since both linolenic and linoleic selectivities do not take into account the
cis/trans isomerization, the specific isomerization index S
i = (
k2 +
k3)/
mkr as proposed by Plourde
et al. is also given [
23]. As can be seen, upon increasing the hydrogen pressure from 1.5 to 21 bar(a) the linolenic selectivity was found to decrease from 2.70 to 1.51, whereas the linoleic selectivity decreased four times from 24.17 to 6.05. These changes indicate suppression of the hydrogenation of linolenic and linoleic acids with the increased pressure. This observation is in agreement with the results of the analysis of the fatty acid profile revealing that with the increased pressure the C18:2
cis content was found to increase slightly from 0.6 wt% at 2.5 bar(a) to 3.3 wt% at 21 bar(a). Moreover, in line with the results and expectations the content of steric acid C18:0 increased considerably from 13.1 wt% at 2.5 bar(a) to 22.1 wt% at 21 bar(a). On the other hand, the increasing hydrogen pressure has a substantial effect on suppressing the
cis/trans isomerization. The corresponding S
i index, which is a ratio of the total isomerization activity to the saturation activity of the catalyst, was found to decrease from 14.72 at 2.5 bar(a) to 7.17 at 21 bar(a). It indicates that less
trans isomers are formed at higher pressures as is also reflected in the analysis of the fatty acid profile of the hydrogenated samples.