Facile and Efficient Acetylation of Primary Alcohols and Phenols with Acetic Anhydride Catalyzed by Dried Sodium Bicarbonate
Abstract
:1. Introduction
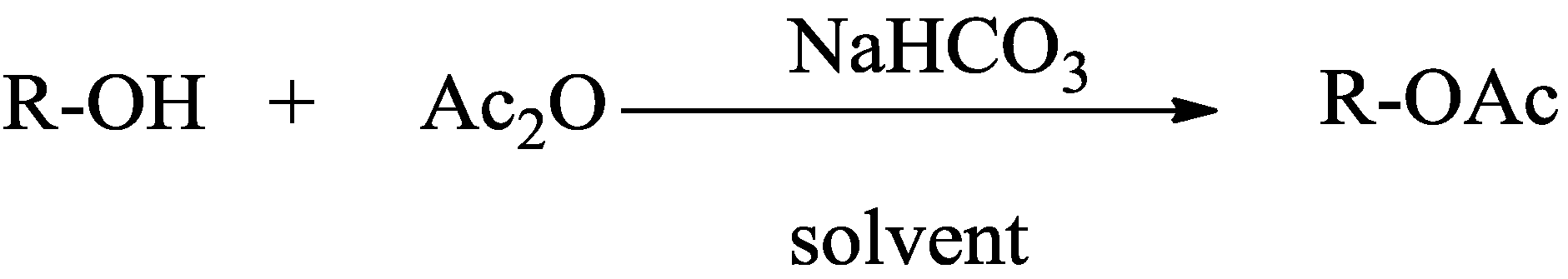
2. Results and Discussion
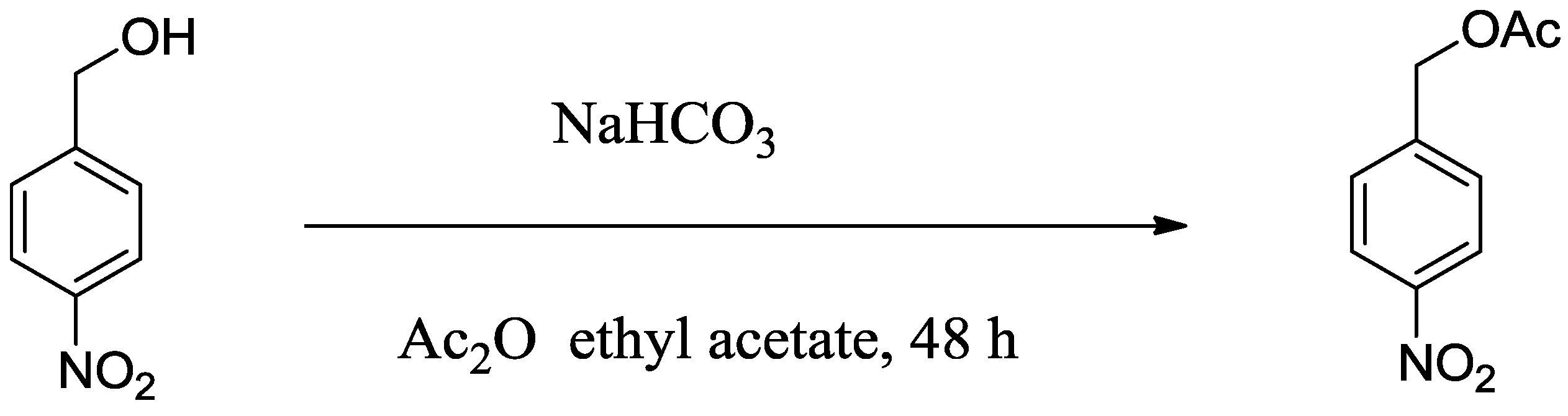
| Bicarbonate a | Yield (%) |
|---|---|
| None | 45 |
| NaHCO3 | 95 |
| KHCO3 | 80 |
| CsHCO3 | >99 |
| Li2CO3 | >99 |
| Na2CO3 | 90 |
| K2CO3 | 92 |
| Cs2CO3 | 85 |
| CaCO3 | 42 |
| CoCO3 | 45 |
| Solvent (6 mL) | Time (h) | Yield (%) |
|---|---|---|
| Ethyl acetate | 24 | 93 |
| THF a | 24 | 90 |
| Toluene | 24 | >99 |
| Diethyl ether | 48 | 75 |
| Anhydrous diethyl ether | 48 | 61 |
| Dichloromethane | 48 | 85 |
| Acetonitrile | 48 | 90 |
| Entry | Amount of water ( μL) | Yield (%) |
|---|---|---|
| 1 | No water added | 95 |
| 2 | 9 | 89 |
| 3 | 18 | 85 |
| 4 | 36 | 77 |
| 5 | 54 | 80 |
| Entry | Alcohol or phenol in 6 mL of solvent a | Product | Ref. h | Yield (%) |
|---|---|---|---|---|
| 1 |  |  | [39] | >99 |
| 2 |  |  | - | 90 b |
| 3 |  |  | [40] | 95 |
| 4 |  |  | 90 b,c | |
| 5 |  |  | [41] | >99 d |
| 6 |  |  | 95 b | |
| 7 |  |  | 88 b | |
| 8 |  |  | [40] | 92 |
| 9 |  |  | - | 87 b,c |
| 10 |  |  | [42] | 80 |
| 11 |  |  | [43] | >99 |
| 12 |  |  | [39,40] | 90 |
| 13 |  |  | [40,44] | >99 |
| 14 |  |  | [45] | 95 d |
| 15 |  |  | - | 93 b |
| 16 |  |  | - | 95 b |
| 17 |  |  | [46,47] | 90 e |
| 18 |  |  | [41,44] | >99 |
| 19 |  |  | [39,40] | 95 |
| 20 |  |  | - | 91 b |
| 21 |  |  | [48] | 83 f,g |
| 22 |  |  | [49] | 82 g |
| 23 |  |  | [39,44] | 92 f |
Mechanism
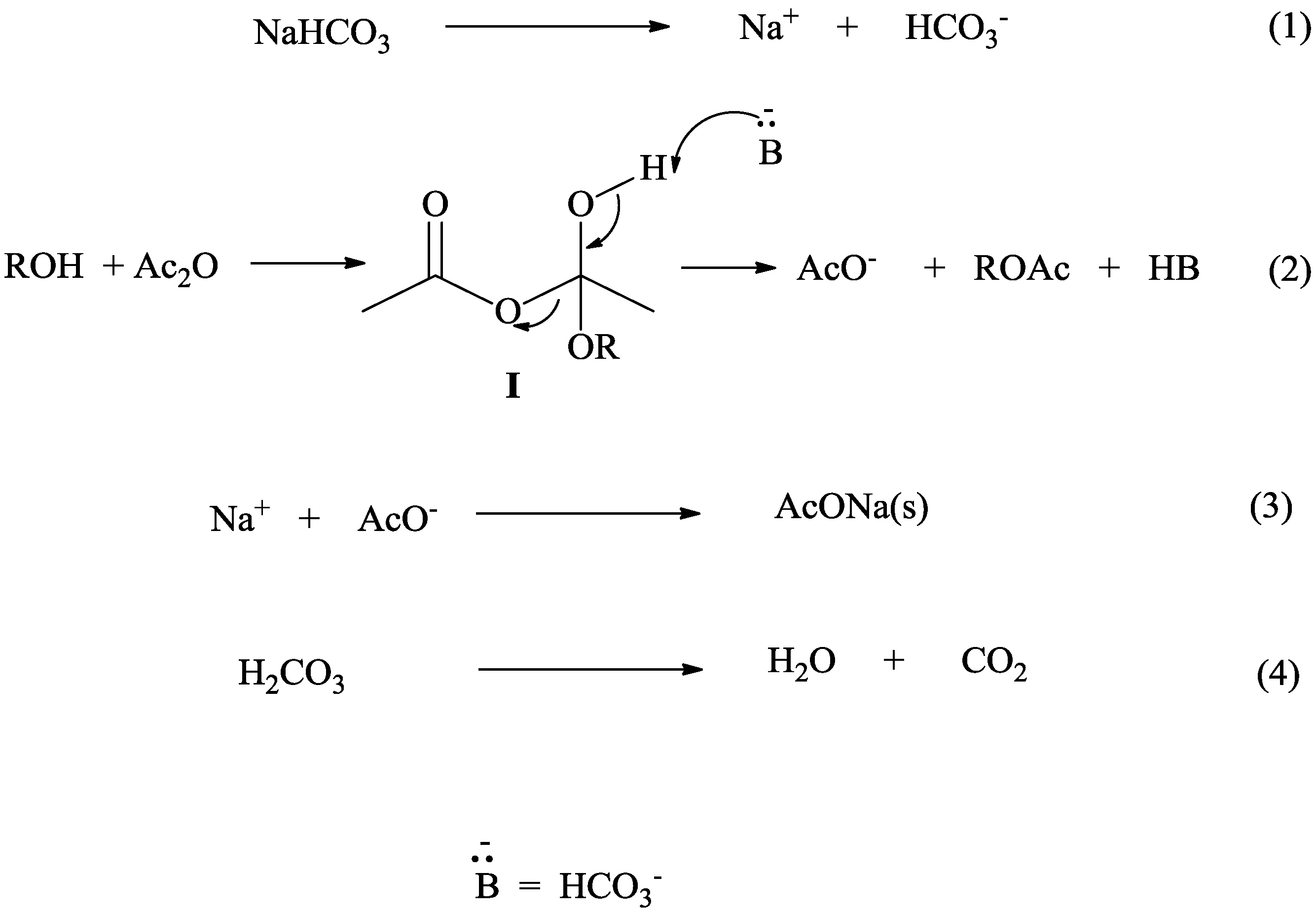
3. Experimental Section—General Procedure
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Greene, T.W.; Wuts, P.G.M. Protective Groups in Organic Synthesis, 4th ed.; Wiley Interscience: New York, NY, USA, 2007; pp. 223–238. [Google Scholar]
- Kocienski, P.J. Protecting Groups; Thieme: New York, NY, USA, 1994. [Google Scholar]
- Larock, R.C. Comprehensive Organic Transformations; VCH Publishers, Inc.: New York, NY, USA, 1989; p. 980. [Google Scholar]
- Saravanan, P.; Singh, V.K. An efficient method for acylation reactions. Tetrahedron Lett. 1999, 40, 2611–2614. [Google Scholar] [CrossRef]
- Chandrasekhar, S.; Ramachander, T.; Takhi, M. Acylation of Alcohols with Acetic Anhydride Catalyzed by TaCl5: Some Implications in Kinetic Resolution. Tetrahedron Lett. 1998, 39, 3263–3266. [Google Scholar] [CrossRef]
- Damen, E.W.P.; Braamer, L.; Scheeren, H.W. Lanthanide Trifluoromethanesulfonate Catalysed Selective Acylation of 10-Deacetylbaccatin III. Tetrahedron Lett. 1998, 39, 6081–6082. [Google Scholar] [CrossRef]
- Orita, A.; Mitsutome, A.; Otera, J. Distannoxane-Catalyzed Highly Selective Acylation of Alcohols. J. Org. Chem. 1998, 63, 2420–2421. [Google Scholar] [CrossRef]
- Brenton, G.W. Selective Monoacetylation of Unsymmetrical Diols using SiO2 Supported Sodium Hydrogen Sulfate. J. Org. Chem. 1997, 62, 8952–8954. [Google Scholar] [CrossRef]
- Orita, A.; Sakamoto, K.; Hamada, Y.; Mitsutome, A.; Otera, J. Mild and Practical Acylation of Alcohols with Esters or Acetic Anhydride Under Distannoxane Catalysis. Tetrahedron 1999, 55, 2899–2910. [Google Scholar] [CrossRef]
- Karimi, B.; Seradj, H. N-bromosuccinimide (NBS), A Novel and Highly Effective Catalyst for Acetylation of Alcohols Under Mild Reaction Conditions. Synlett 2001, 519–520. [Google Scholar]
- Iqbal, J.; Srivastava, R.R. Cobalt (II) Chloride Catalyzed Acylation of Alcohols with Acetic Anhydride: Scope and Mechanism. J. Org. Chem. 1992, 57, 2001–2007. [Google Scholar] [CrossRef]
- Li, T.S.; Li, A.X. Montmorillonite clay catalysis. Part 10. K-10 and KSF-Catalysed Acylation of Alcohols, Phenols, Thiols and Amines: Scope and Limitation. J. Chem. Soc. Perkin Trans. 1998, 1, 1913–1917. [Google Scholar]
- Ballini, R.; Bosica, G.; Carloni, L.; Maggi, R.; Sartori, G. Zeolite HSZ-360 as a New Reusable Catalyst for the Direct Acetylation of Alcohols and Phenols Under Solventless Conditions. Tetrahedron Lett. 1998, 39, 6049–6052. [Google Scholar] [CrossRef]
- Bhaskar, P.M.; Loganathan, D. Per-O-acetylation of Sugars Catalysed by Montmorillonite K-10. Tetrahedron Lett. 1998, 39, 2215–2218. [Google Scholar]
- Hofle, G.; Stelich, V.; Vobruggen, H. 4-Dialkylaminopyridines as Highly Active Acylation Catalysts. Angew Chem. Int. Ed. Engl. 1987, 17, 569–583. [Google Scholar]
- Scriven, E.F.V. 4-Dialkylaminopyridines: Super Acylation and Alkylation Catalysts. Chem. Soc. Rev. 1983, 12, 129–161. [Google Scholar] [CrossRef]
- Vedejs, E.; Diver, S.T. Tributylphosphine: A remarkable Acylation Catalyst. J. Am. Chem. Soc. 1993, 115, 3358–3359. [Google Scholar] [CrossRef]
- Vedejs, E.; Bennett, N.S.; Conn, L.M.; Diver, S.T.; Gingras, M.; Lin, S.; Oliver, P.A.; Peterson, M.J. Tributylphosphine-catalyzed Acylations of Alcohols: Scope and Related Reactions. J. Org. Chem. 1993, 58, 7286–7288. [Google Scholar] [CrossRef]
- Chauhan, K.K.; Frost, C.G.; Love, I.; Waite, D. Indium triflate: An Efficient Catalyst for the Friedel-Crafts Acylation of Aromatics. Synlett 1999, 1743–1744. [Google Scholar] [CrossRef]
- Orita, A.; Tanahashi, C.; Kakuda, A.; Otera, J. Highly Efficient and Versatile Acylation of Alcohols with Bi(OTf)3 as Catalyst. Angrew. Chem. Int. Ed. 2000, 39, 2877–2879. [Google Scholar]
- Moghadam, M.; Tangestaninejad, S.; Mirkhani, V.; Mohammadpoor-Baltork, I.; Babghanbari, M.; Zarea, L. Zirconyl triflate: A New, Highly Efficient and Reusable Catalyst for Acetylation and Benzoylation of Alcohols, Phenols, Amines and Thiols with Acetic and Benzoic Anhydrides. J. Iran. Chem. Soc. 2009, 6, 523–532. [Google Scholar] [CrossRef]
- Iranpoor, N.; Shekarrize, M. Catalytic Esterification of Alcohols, Carboxylic Acids and Transesterification Reactions with Cerium(IV) Triflate. Bull. Chem. Soc. Jpn. 1999, 72, 455–458. [Google Scholar] [CrossRef]
- Alleti, R.; Perambuduru, M.; Samantha, S.; Prakash, V.; Reddy, V.P. Gadolinium Triflate: an Efficient and Convenient Catalyst for Acetylation of Alcohols and Amines. J. Mol. Catal. A Chem. 2005, 226, 57–59. [Google Scholar] [CrossRef]
- Ishihara, K.; Kubota, M.; Kurihara, H.; Yamamoto, H. Scandium Trifluoromethanesulfonate as an Extremely Active Acylation Catalyst. J. Am. Chem. Soc. 1995, 117, 6639–6639. [Google Scholar]
- Ishihara, K.; Kubota, M.; Kurihara, H.; Yamamoto, H. Scandium Trifluoromethanesulfonate as an Extremely Active Lewis Acid Catalyst in Acylation of Alcohols with acid Anhydrides and Mixed Anhydrides. J. Org. Chem. 1996, 61, 4560–4567. [Google Scholar] [CrossRef]
- Ishihara, K.; Kubota, M.; Yamamoto, H. A New Scandium Complex as an Extremely Active Acylation Catalyst. Synlett 1996, 265–266. [Google Scholar] [CrossRef]
- Barrett, A.G.M.; Braddock, D.C. Scandium(III) or Lanthanide(III) Triflates as Recyclable Catalysts for the Direct Acetylation of Alcohols with Acetic Acid. Chem. Commun. 1997, 351–352. [Google Scholar] [CrossRef]
- Zhao, H.; Pendri, A.; Greenwald, R.B. General Procedure for Acylation of 3 Degrees Alcohols: Scandium Triflate/DMAP Reagent. J. Org. Chem. 1998, 63, 7559–7562. [Google Scholar] [CrossRef]
- Procopiou, P.A.; Baugh, S.P.D.; Flack, S.S.; Inglis, G.G.A. An Extremely Powerful Acylation Reaction of Alcohols with Acid Anhydrides Catalyzed by Trimethylsilyl Trifluoromethanesulfonate. J. Org. Chem. 1998, 63, 2342–2347. [Google Scholar] [CrossRef]
- Procopiou, P.A.; Baugh, S.P.D.; Flack, S.S.; Inglis, G.G.A. An Extremely Fast and Efficient Acylation Reaction of Alcohols with acid Anhydrides in the Presence of Trimethylsilyl Trifluoromethanesulfonate as Catalyst. J. Chem. Soc. Chem. Commun. 1996, 2625–2626. [Google Scholar]
- Miyashita, M.; Shiina, I.; Miyoshi, S.; Mukaiyama, T. A New and Efficient Esterification Reaction via Mixed Anhydrides by the Promotion of a Catalytic Amount of Lewis Acid. Bull. Chem. Soc. Jpn. 1993, 66, 1516–1527. [Google Scholar] [CrossRef]
- Nakae, Y.; Kusaki, I.; Sato, T. Lithium Perchlorate Catalyzed Acetylation of Alcohols Under Mild Reaction Conditions. Synlett 2001, 1584–1586. [Google Scholar] [CrossRef]
- Bartoli, G.; Bosco, M.; Dalpozzo, R.; Marcantoni, E.; Massaccesi, M.; Rinaldi, S.; Sambri, L. Mg(ClO4)(2) as a Powerful Catalyst for the Acylation of Alcohols Under Solvent-Free Conditions. Synlett 2003, 39–42. [Google Scholar]
- Phukan, P. Iodine as an Extremely Powerful Catalyst for the Acetylation of Alcohols Under Solvent-free conditions. Tetrahedron Lett. 2004, 45, 4785–4787. [Google Scholar] [CrossRef]
- Nishiguchi, T.; Taya, H. Facile Acylation of Alcohols Using Esters and Silica Gel-Supported Metallic Sulphates and Hydrogen Sulphates. J. Chem Soc. Perkin Trans. 1990, 1, 172–173. [Google Scholar] [CrossRef]
- Ambika; Singh, P.P.; Chauhan, S.M.S. Chemoselective Esterification of Phenolic Acids in the Presence of Sodium Bicarbonate in Ionic Liquids. Synth. Commun. 2008, 38, 928–936. [Google Scholar] [CrossRef]
- Mallesha, N.; Rao, S.P.; Suhas, R.; Gowda, D.C. A Green Method for Selective Acetylation of Primary Alcohols Using Ethyl acetate and Solid Potassium Carbonate. J. Chem Res. 2011, 35, 536–539. [Google Scholar] [CrossRef]
- Yan, X.; Xueqiang, Z. Significant Heterogeneous Carbonate Salt Catalyzed Acetylation of Alcohols via a Transesterification Process with Carbonate Salt-Activated Alcohol 1H NMR Evidence. Chin. J. Chem. 2011, 29, 1143–1148. [Google Scholar] [CrossRef]
- Zarei, A.; Hajipour, A.R.; Khazdooz, L. P2O5/Al2O3 As an Efficient Heterogeneous Catalyst for the Acetylation of Alcohols, Phenols, Thiols, and Amines Under Solvent-Free Conditions. Synth. Commun. 2011, 41, 1772–1785. [Google Scholar] [CrossRef]
- Das, R.; Chakraborty, D. Silver Triflate catalyzed Acetylation of Alcohols, Thiols, and Amines. Synthesis 2011, 10, 1621–1625. [Google Scholar] [CrossRef]
- Xiao, Z.-P.; Fang, R.-Q.; Lei, S.; Ding, H.; Xu, C.; Zhu, H.-L. Synthesis, Crystal Structure, and Growth Inhibiton of Human Heptoma Cell (HepG2) of Polyphenolic Compound Based on Gallates. Can. J. Chem. 2007, 85, 951–957. [Google Scholar] [CrossRef]
- Carrasco, H.A.; Espinoza, L.C.; Cardile, V.; Gallardo, C.; Cardona, W.; Lombardo, L.; Catalán, K.M.; Cuellar, M.F.; Russo, A. Eugenol and its synthetic analogues inhibit cell growth of human cancer cells (Part I). J. Braz. Chem. Soc. 2008, 19, 543–548. [Google Scholar] [CrossRef]
- Kozlov, N.G.; Gusak, K.N.; Tereshko, A.B.; Dikusar, E.A. Vanillin esters of aliphatic acids in the synthesis of 4,7-phenanthroline derivatives. Russ. J. Organ. Chem. 2004, 40, 705–710. [Google Scholar] [CrossRef]
- Curini, M.; Epifano, F.; Marcotullio, M.C.; Rosati, O.; Rossi, M. Heterogeneous catalysis in acetylation of alcohols and phenols promoted by zirconium sulfophenyl phosphate. Synth. Commun. 2000, 30, 1319–1329. [Google Scholar] [CrossRef]
- Jayatilake, G.S.; Baker, B.J.; McClintock, J.B. Isolation and Identification of a Stilbene Derivative From The Antarctic Sponge Kirkpatrickia Variolosa. J. Nat. Prod. 1995, 58, 1958–1960. [Google Scholar] [CrossRef]
- Bodrikov, I.V.; Borisova, N.V.; Chiyanov, A.A.; Kurskii, Y.A.; Fukin, G.K. Vinylic substitution in the reaction of betulin diacetate with tert-butyl hypochlorite. Russ. J. Organ. Chem. 2013, 49, 78–82. [Google Scholar] [CrossRef]
- Deng, Y.; Snyder, J.K. Preparation of a 24-nor-1,4-dien-3-one triterpene derivative from betulin: A new route to 24-nortriterpene analogues. J. Org. Chem. 2002, 67, 2864–2873. [Google Scholar] [CrossRef]
- Gordon, A.J. Acetyl salicylamide O- to N-acetyl migration. Tetrahedron 1967, 23, 863–870. [Google Scholar] [CrossRef]
- Sasaki, T.; Yamakoshi, J.; Saito, M.; Kasai, K.; Matsudo, T.; Kikuchi, M.; Koga, T.; Mori, K. Synthesis of 4-hydroxy-3(2H)-furanone acyl derivatives and their anti-cataract effect on spontaneous cataract rats (ICR/f). Biosci. Biotechnol. Biochem. 1998, 62, 2145–2154. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Lugemwa, F.N.; Shaikh, K.; Hochstedt, E. Facile and Efficient Acetylation of Primary Alcohols and Phenols with Acetic Anhydride Catalyzed by Dried Sodium Bicarbonate. Catalysts 2013, 3, 954-965. https://doi.org/10.3390/catal3040954
Lugemwa FN, Shaikh K, Hochstedt E. Facile and Efficient Acetylation of Primary Alcohols and Phenols with Acetic Anhydride Catalyzed by Dried Sodium Bicarbonate. Catalysts. 2013; 3(4):954-965. https://doi.org/10.3390/catal3040954
Chicago/Turabian StyleLugemwa, Fulgentius Nelson, Koonj Shaikh, and Edwin Hochstedt. 2013. "Facile and Efficient Acetylation of Primary Alcohols and Phenols with Acetic Anhydride Catalyzed by Dried Sodium Bicarbonate" Catalysts 3, no. 4: 954-965. https://doi.org/10.3390/catal3040954




