Abstract
The kinetics of hydrogen absorption/desorption in magnesium can be improved without any catalysis assistance and MgO was found to be more effective than the best catalyst reported so far, i.e., Nb2O5. Herein, a quantitative analysis of the hydrogen kinetics in magnesium modified with MgO was performed in order to identify possible rate controlling mechanisms. While hydrogen absorption was found to be diffusion controlled as commonly reported, hydrogen desorption evolved from nucleation and growth to an interface controlled process depending on the desorption temperature. Comparison with the effect of Nb2O5 indicates that similar rate limiting steps occur regardless of the oxide additive. These findings are reconciled by considering the tribological effect of solid oxide additives, as a correlation between oxides electronegativity and improvement in hydrogen kinetics was found. Such a correlation clearly highlights the mechanical effect of solid oxides in facilitating the grinding and stabilisation of small magnesium particles for efficient and fast hydrogen kinetics.
Keywords:
magnesium; hydrogen storage; metal oxides; lubricious oxides; electronegativity; tribology 1. Introduction
Magnesium has been the centre of extensive investigations for its potential use as a medium for storing hydrogen on board vehicles because it is an abundant and low cost metal that can store up to 7.6 mass % of hydrogen [1,2]. However, the practical use of magnesium as a hydrogen store has always been restrained by the high stability of its hydride (ΔH300 °Cdes = −74 kJ.mol−1 H2) and poor kinetics when it comes to release or absorb hydrogen [3,4].
Over the last decades, multiple attempts have aimed at solving these problems through alloying methods targeting the thermodynamic issues, i.e., reduce hydride stability, and/or catalyst addition addressing the kinetic problems [2,5]. When considering hydrogen kinetics in magnesium, two main hypotheses are generally accepted with respect to the mechanisms limiting hydrogen absorption/desorption rates. During absorption the reaction rate is controlled by the diffusion of hydrogen through the hydride phase (β-MgH2), whereas during desorption the reaction is controlled by the nucleation and growth of the magnesium phase [5]. To some extent, the use of mechanical milling has alleviated a number of the problems related to kinetics, and allowed fast hydrogen absorption/desorption kinetics, e.g., full hydrogen desorption in 30 min instead of hours at a temperature of 300 °C [4,6]. The reduction of particle size achieved upon mechanical milling (inducing shorter hydrogen diffusion paths) as well as the formation of fresh surfaces free from an oxide layer may explain such an improvement. Several groups have also advanced the formation of a nanostructure upon mechanical milling as a possible ground for the kinetics improvements [7,8,9]; however some investigations have shown that the nanostructure and associated strain and grain boundary defects may not play a crucial role in the overall kinetic enhancement observed [10,11,12]. The high temperature of 300 °C required for hydrogen absorption/desorption leads to strain relaxation and defects annihilation.
Further improvement in kinetics have also been obtained upon the addition of transition metals such as Cr, V, Nb and their respective oxides during the milling process, with now hydrogen desorption occurring in a few minutes at temperatures above 250 °C [13,14,15,16,17]. Analysis of the kinetics obtained revealed a potential shift of the step limiting the desorption from the nucleation and growth of the magnesium phase to an interface controlled process, whereby the hydrogen desorption is controlled by the diffusion of hydrogen through the magnesium phase [18,19]. Transition metals would reduce the energy barrier for the desorption of hydrogen by acting as gateways facilitating hydrogen adsorption/desorption at the surface of magnesium and/or enhancing the hydrogen dissociation/recombination processes at the magnesium surface [4,19,20].
If in the case of transition metals, the formation of an intermediate hydride phase could explain the enhancement in kinetics observed, the role of transition metal oxides is however still controversial. Although these transition metal oxides would be partially reduced by magnesium upon heating and lead to oxygen defective surface sites supposedly acting as active sites for hydrogen dissociation/recombination [10,21]; the activity of transition metal oxides as hydrogenation catalysts has not been reported in the literature. For example, Nb2O5 is known as an acid catalyst and a promoter reinforcing the catalytic performances of metals such as molybdenum used for the dehydrogenation of alkanes [22,23]. However, the ability of niobium oxides in splitting/recombining hydrogen molecules at rates sufficiently high to speed up hydrogen kinetics in magnesium as previously claimed is unknown [24,25]. The possible role of Nb2O5 acting as a pathway for hydrogen diffusion within the magnesium matrix is similarly controversial [26,27,28]. Some metal oxides such as WO3, MoO3, V2O5, capable of forming a so-called hydrogen bronze phase have been shown to be good proton conductors [21,29,30,31], however a noble catalyst is needed at the surface of the oxide to split hydrogen [29,30].
Recently, we have shown that in fact there is no need for transition metals and catalysis to effectively enhance the kinetics of hydrogen absorption/desorption in magnesium. By simply adding a diamond powder or MgO during mechanical milling to MgH2, it was possible to achieve hydrogen kinetics similar to those observed upon transition metal additions [32,33]. In this case, the improvement of hydrogen kinetics cannot be explained by the formation of new active phases, catalytic or chemical effects. Diamond is totally inert toward hydrogen and MgO is usually avoided as it inhibits hydrogen absorption/desorption [34]. These results opened new pathways and models toward the use of non-transition metals as effective promoters of hydrogen kinetics in magnesium. We proposed a new hypothesis whereby MgO would act as a dispersive agent during mechanical milling leading to an efficient particle size reduction and stabilisation and therefore enhanced hydrogen kinetics due to small diffusion paths [32]. However, similarities of the hydrogen kinetic curves obtained with transition metal oxides and MgO point toward more correlation between the effects of these oxides at the macroscopic level. Herein, we present a more detailed analysis of the hydrogen kinetics of MgH2 modified with MgO and propose a novel description rationalising the effects observed with all metal oxides.
2. Material and Methods
MgH2 powder (95%, 20–60 µm) was purchased from Goldschmidt AG. MgO powder (99.99%, 0.5–30 µm) was purchased from Sigma–Aldrich. MgH2 was milled in a ceramic vial to reduce the problems associated with the contamination from the milling tools. Lower contamination occurs in ceramic vials compared to stainless steel vials [35]. MgH2 was milled with 10 wt% of MgO on a Frischt P5 planetary mill hosted in a glove box under Ar atmosphere. The milling was carried out for 100 h with a 300 mL ceramic vial containing 10 g of MgH2 and 100 g of zirconium oxide balls (Ø 10 mm, 3 g). To clarify the effect of oxidation on the milling process and hydrogen desorption, 10 g of MgH2 powder was pre-oxidised in air for 1 h before milling in order to build-up the oxide layer at the surface of MgH2 particles [27,36]. Pre-oxidised MgH2 was then milled under the conditions described above.
Once milled for 100 h, 100 mg of powder was loaded in a hermetically sealed sample holder and hydrogen kinetic measurements were carried-out with a Sievert apparatus designed by HERA Hydrogen Storage Systems. The measurements were performed at 300 °C and under 1 MPa of hydrogen for absorption and 0.1 kPa for desorption. High purity hydrogen was used (99.999%). DSC measurements were performed with a DSC 404 C Pegasus from Netzsch. The measurements were carried out under a flow of high purity Ar (50 mL min−1) with a heating rate of 5 °C min−1 from 25 °C to 500 °C.
3. Results and Discussions
3.1. Oxidation Effect on Hydrogen Desorption
To outline the effect of mechanical milling and the additional effects of MgO, the materials were characterised by DSC measurements. As shown Figure 1, as-received MgH2 released hydrogen in a single endothermic peak at 414 °C. Upon milling MgH2, the hydrogen desorption temperature was reduced to 336 °C. However, a significant decrease of the hydrogen desorption temperature was obtained when MgH2 was milled with MgO as the desorption peak shifted from 336 °C to 262 °C. In our previous report, such a drastic improvement of hydrogen desorption was related to an important reduction in the size of MgH2 particles as MgO would act as a process control agent facilitating the grinding of MgH2 particles [32]. Further investigations on the potential effect of MgO have raised the possibility of a defective oxide layer at the surface of MgH2 particles catalysing the splitting/recombination of hydrogen [37]. However, such a hypothesis challenges common assumptions as an oxide layer would seriously impede hydrogen absorption/desorption. In fact, pre-oxidising MgH2 before milling in an attempt to enhance the number of potential oxygen vacancies resulted in poor hydrogen desorption characteristics as demonstrated by the DSC measurement carried out after milling (Figure 1). In this case, it is most likely that the oxide layer generated during the pre-oxidation of MgH2 still inhibits hydrogen desorption despites the high energy involved in the milling process and the possibility of forming fresh surfaces and defective surface sites facilitating the desorption of hydrogen. Other investigations have also found similar results with hydrogen kinetics deteriorating as an oxide layer is allowed to grow at the surface of MgH2 particles [27,34]. Although hydrogen could strongly absorb at defective oxide sites and dissociate, such a heterolytic dissociation would most likely result in the formation of surface hydroxyls that would be further reduced upon heating [38,39,40]. Accordingly, a catalytic activity of MgO should not be expected.
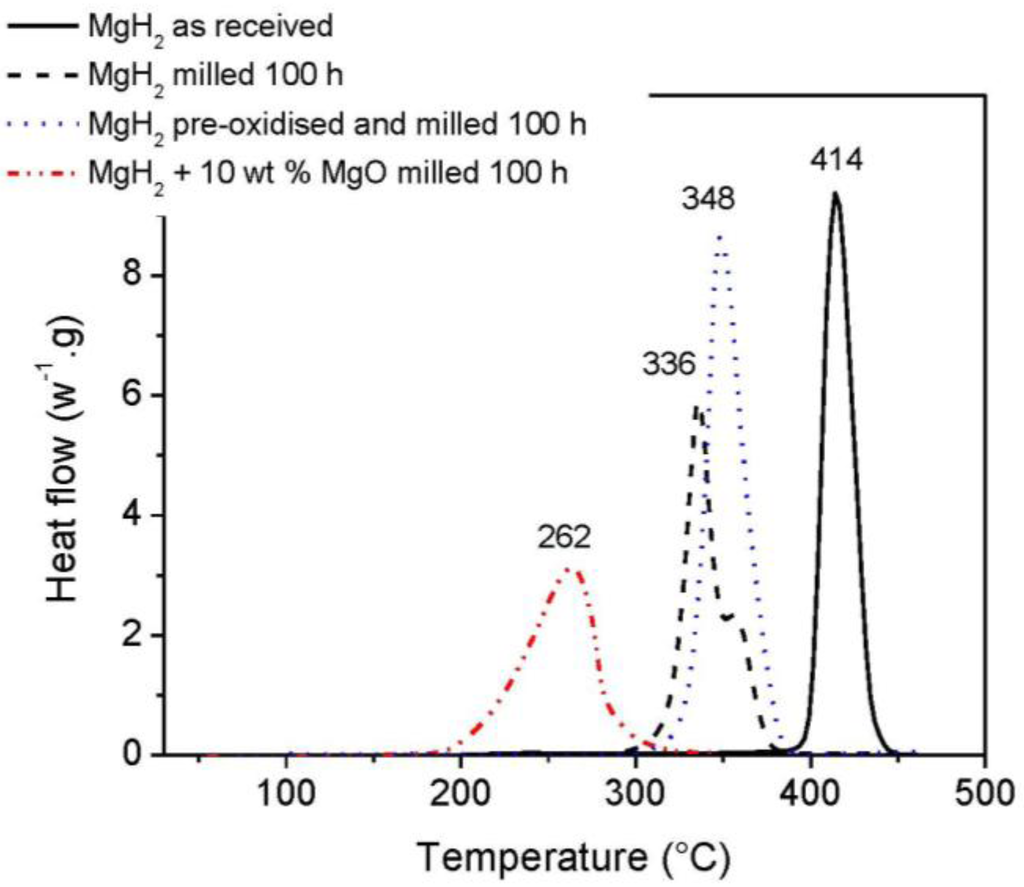
Figure 1.
DSC curves of MgH2 as-received, MgH2 and MgH2 pre-oxidised in air after milling and MgH2 after milling with MgO. The lower intensity of the peak for MgH2 milled with 10 wt % MgO as compared to pure MgH2 milled is due to the lower amount of the sample loaded in the instrument.
3.2. Kinetics and Rate Limiting Steps
Figure 2 shows the hydrogen kinetics of MgH2 milled for 100 h at 300 °C. As previously reported, hydrogen absorption is faster than desorption with ~30 min necessary to desorb MgH2 whereas only ~8 min are required to complete the absorption process up to 90% of the total capacity. Hydrogen kinetics were drastically improved when MgH2 was milled with MgO (Figure 3). Hydrogen absorption and desorption could be achieved in less than 100 s at 300 °C. At a lower temperature of 250 °C, full absorption was also completed in less than 200 s, and a fast desorption rate with full hydrogen release occurring in less than 800 s was still achievable. However, below 250 °C the desorption rate of hydrogen drastically decreased and no significant release of hydrogen could be observed in agreement with the DSC measurements. It is noteworthy that the shape of hydrogen absorption curves was not influenced by the temperature or the addition of MgO. Conversely, the hydrogen desorption curves evolved from a sigmoidal shape for milled MgH2 to a non-sigmoidal curve when MgO was used as a milling additive.
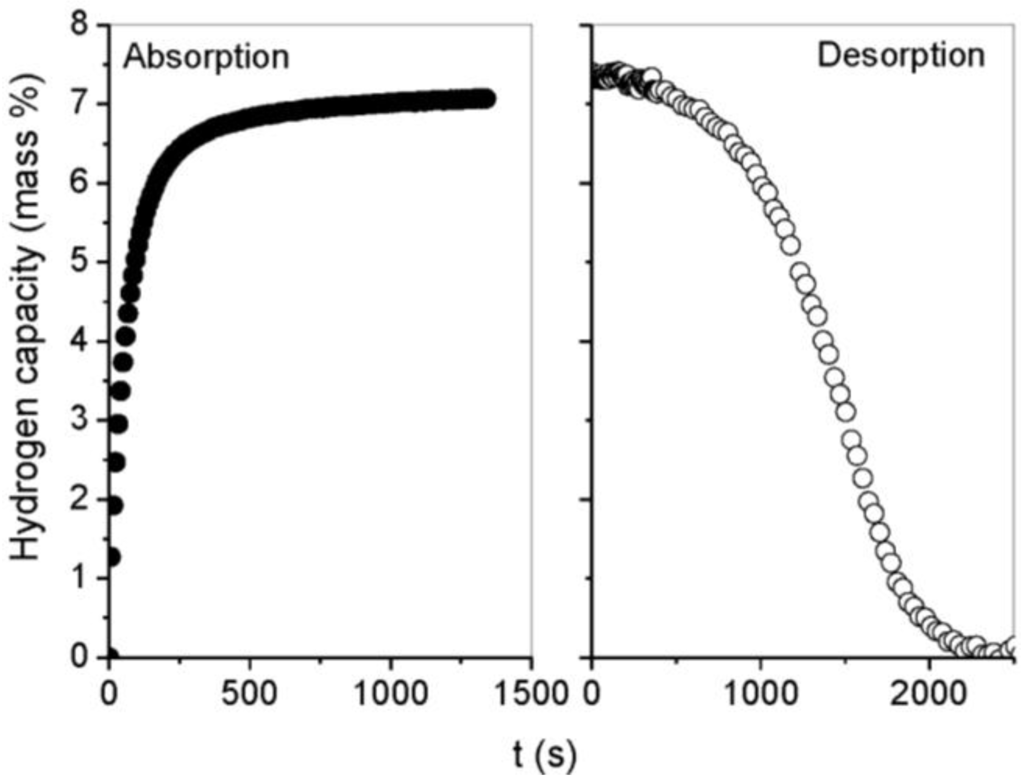
Figure 2.
Hydrogen absorption/desorption curves at 300 °C for MgH2 milled for 100 h.
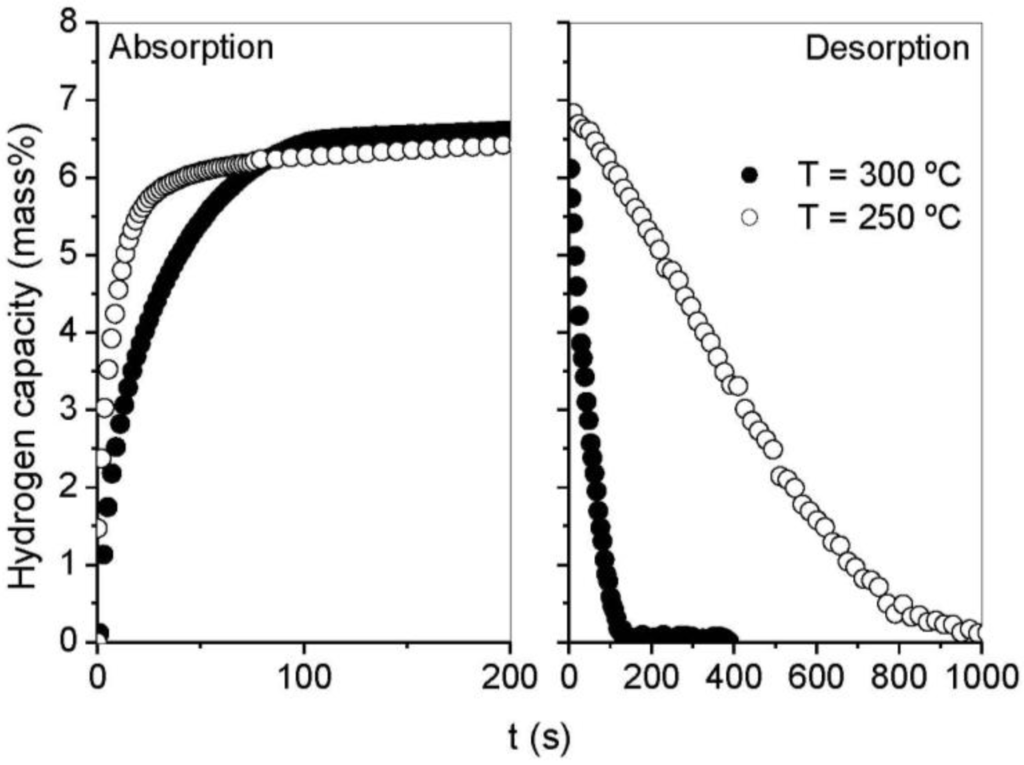
Figure 3.
Hydrogen absorption/desorption curves at 300 °C and 250 °C for MgH2 milled with MgO.
In order to identify possible mechanisms governing the hydrogen kinetics of MgH2 modified with MgO and better understand the improvements observed, the kinetic curves obtained under isothermal conditions were fitted using the general equation for solid-state reactions [41]:
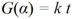
where α is the fraction reacted in time t, k = k(T,P,r) is the reaction rate and G(α) is a functiondepending on the mechanism controlling the reaction. The main theoretical functions G(α) are summarized in Table 1 and grouped according to four major reaction mechanisms governed by: (i) the surface; (ii) the nucleation and growth of the new phase; (iii) the reaction at the new phase/shrinking core interface with the interface reaction proceeding at constant velocity; and (iv) the reaction at the new phase/shrinking core interface with the interface reaction controlled by diffusion. In order to facilitate the fitting of the hydrogen curves measured and ease the distinction of reaction mechanisms, the method of Hancok and Sharp was used [42]. Since nucleation and growth processes in condensed systems follow the almost universal Equation (2), the method consists of plotting ln(−ln(1 − α)) vs. ln(t).
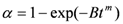
where B is a constant that depends on the nucleation frequency and linear rate of the grain growth, and m is a constant that varies according to the geometry of the system.
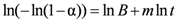
upon plotting Equation (3), the determination of m allows the identification of the reaction mechanism by comparison with tabulated theoretical values (Table 1).

Table 1.
Summary of main model functions describing solid-gas kinetics.
| Mechanism | Functional time dependence G(α) | m |
|---|---|---|
| Surface control | ||
| S1 | α | 1.24 |
| Random nucleation and growth (Avrami equations) | ||
| A1 | −ln(1 − α)1/4 | 4.00 |
| A2 | −ln(1 − α)1/3 | 3.00 |
| A3 | −ln(1 − α)2/5 | 2.50 |
| A4 | −ln(1 − α)1/2 | 2.00 |
| A5 | −ln(1 − α)2/3 | 1.50 |
| Shrinking Core with constant velocity: controlled by interface reaction | ||
| IP2–contracting surface | 1 − (1 − α)1/2 | 1.11 |
| IP3–contracting volume | 1 − (1 − α)1/3 | 1.07 |
| Shrinking Core with decelerating velocity: controlled by diffusion | ||
| D1-1-D diffusion | α2 | 0.62 |
| D2-2-D diffusion | (1 − α)ln(1 − α) + α | 0.57 |
| D3-Jander, 3-D diffusion | (1 −(1 − α)1/3)2 | 0.54 |
Figure 4 shows the fits for the hydrogen absorption and desorption kinetic curves obtained for Mg and Mg milled with MgO. In both cases, a slope close to 0.62 for the absorption kinetic indicates that the absorption process was controlled by the diffusion of hydrogen into MgH2 in a one dimensional process. Despite the low diffusion coefficient of hydrogen in the MgH2 phase, such behaviour is possibly due to the high temperatures and pressures used to perform the absorptions. However, upon hydrogen desorption different slopes of 1.04 and 2.86 were obtained for MgH2 and MgH2 milled with MgO, respectively. This would indicate a shift from a mechanism where hydrogen desorption is controlled by the nucleation and growth of the Mg phase to a desorption process controlled by the Mg/MgH2 interface as the MgH2 core is shrinking. It is widely accepted that the hydrogen desorption of milled MgH2 follows a nucleation and growth process [5,6]. Accordingly, the results obtained for milled MgH2 are in agreement with the general trends observed.
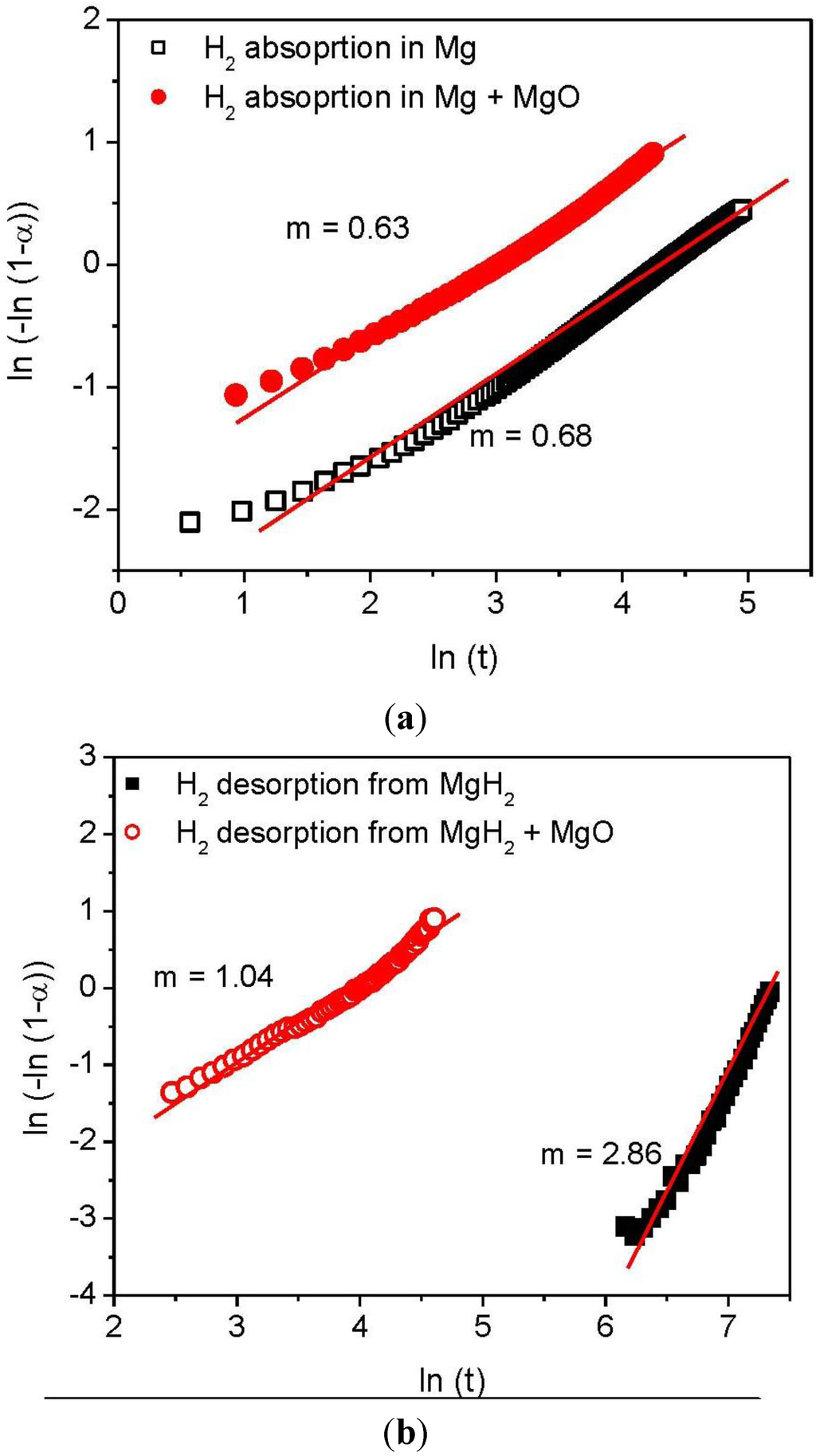
Figure 4.
Plot of ln(ln(1 − α)) vs. ln(t) for the hydrogen absorption/desorption curves obtained at 300 °C for MgH2 milled with and without MgO.
As the temperature for hydrogen absorption/desorption in MgH2 milled with MgO is decreased to 250 °C, the hydrogen absorption kinetic was found to be once again controlled by the diffusion of hydrogen, however hydrogen desorption shifted back to a reaction mechanism controlled by a nucleation and growth process (Figure 5). At such a low temperature, the energy provided may not be sufficient to overcome the activation energy required for the nucleation of the magnesium phase. Therefore, the mechanism would be controlled again by nucleation and growth instead of interface control. Liang et al. reported similar findings with hydrogen desorption from MgH2 modified with 5 at% of V shifting from a nucleation and growth process to a interface process for temperatures above 250 °C [19]. In this case, the shift observed was explained by the catalytic activity of V taking place above 250 °C by reducing the barrier for nucleation and thus making hydrogen desorption occurring at low driving forces. However, with MgH2 modified with MgO, it is unlikely that MgO would catalyse the recombination of hydrogen and thus the nucleation of a magnesium phase as previously discussed. The shift in controlling mechanisms observed would therefore correspond to the energy required to effectively recombine hydrogen at the surface of milled MgH2. After milling, magnesium showed smaller particle sizes and increased specific surface area resulting in a higher density of nucleation sites. As a significant reduction of activation energy for hydrogen desorption has been observed for decreasing particles sizes [43], the smaller particles generated upon milling MgH2 with MgO would result in faster hydrogen kinetics and depending on the driving forces different reaction mechanisms.
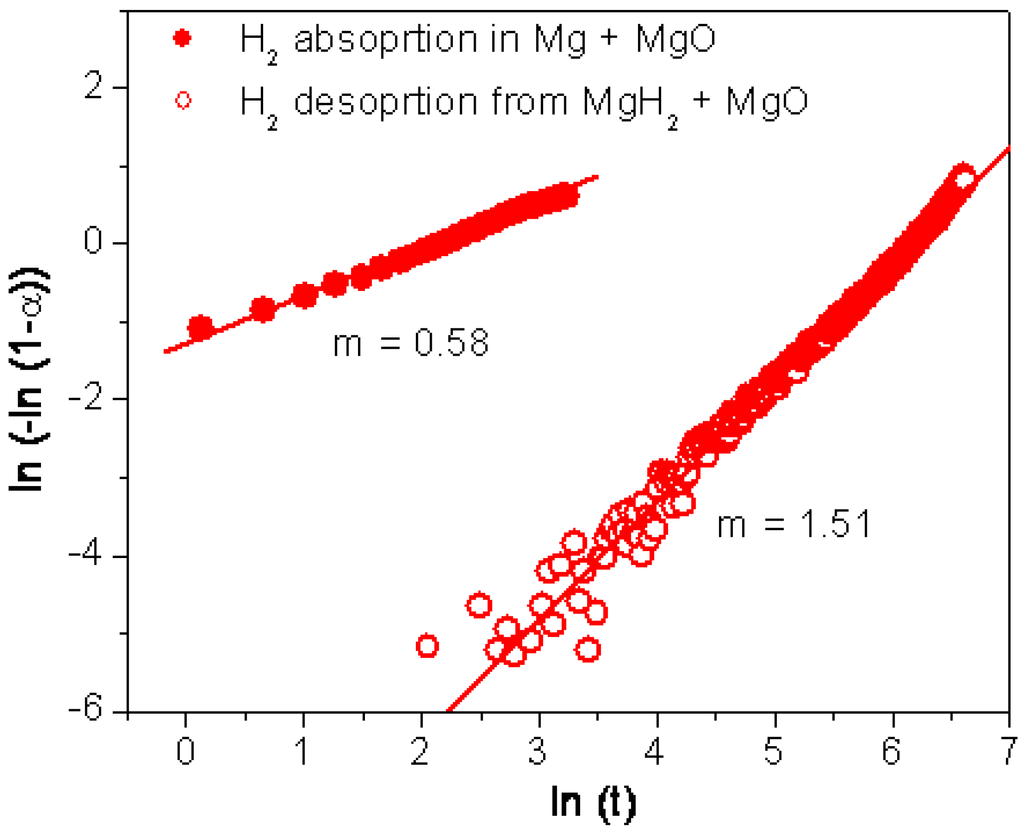
Figure 5.
Plot of ln(ln(1 − α)) vs. ln(t) for the hydrogen absorption/desorption curves obtained at 250 °C for MgH2 milled with MgO.
It is also striking that the rate limiting steps identified for MgH2 milled with MgO are similar to those observed with MgH2 modified with the best known catalyst, i.e., Nb2O5 [24]. At the macroscopic level, hydrogen desorption at 300 °C would proceed through an Mg/MgH2 interface-controlled growth process regardless the additive (Table 2). The hypothesis can be made that the use of MgO resulted in a better grinding process leading to smaller particle/crystal sizes and thus faster kinetics. However, comparison of the physical properties of the materials obtained in both cases does not revealed any significant differences in terms of particle and crystallite sizes (Table 2). Accordingly, such a finding contradicts current assumptions whereby Nb2O5 is considered to as a catalyst reducing the barriers for nucleation and thus facilitating hydrogen desorption at low driving forces [25,27]. The formation of mixed phases of MgxNbyO acting as pathways for hydrogen diffusion with the formation of metastable niobium hydride phases reducing the activation energy of the Mg/MgH2 transition is another possibility [27]. Alike Nb2O5, MgO would also be reduced by MgH2 upon heating to form new oxide phases. However MgO would not generate nucleation points of partial oxides that could induce the interface controlled mechanism observed with MgO and/or Nb2O5. It is noteworthy that the addition of Nb2O5 to MgH2 with fully optimised hydrogen kinetics does not lead to further kinetic improvements. Once fast hydrogen kinetics have been reached at 250 °C by effectively reducing particle size during the milling process, any catalyst addition could not further enhance the hydrogen kinetics [44]. Accordingly, if the modification of MgH2 with MgO or Nb2O5 has led to a final material with equivalent properties, a possible catalytic effect of Nb2O5 would be mirror in comparison to other potential effects such as particle size.

Table 2.
Rate limiting steps for hydrogen absorption/desorption in magnesium upon modification with MgO and Nb2O5.
| MgH2 [6,32,44] | MgH2 + MgO [32] | MgH2 + Nb2O5 [18,44] | ||
|---|---|---|---|---|
| Particle size (µm) | 1 ± 0.5 | 0.4 ± 0.3 | 0.5 ± 0.3 | |
| Surface area (m2·g−1) | 9.9 | - | 22.4 | |
| Crystallite size (nm) | 79 | 86 | 83 | |
| Absorption at 300 °C (250 °C) | ||||
| Rate constant (s−1) | 0.00068 | 0.0027 (0.0052) | - | |
| Rate limiting step | Diffusion | Diffusion (Diffusion) | Diffusion | |
| Desorption at 300 °C (250 °C) | ||||
| Rate constant (s−1) | 0.00065 | 0.0055 (0.0207) | - | |
| Rate limiting step | Nucleation and growth | Shrinking core controlled by interface(Nucleation and growth) | Shrinking core controlled by interface | |
3.3. Rationalisation of the Effect of Oxides
In the light of these findings, alternative explanations should be advanced to describe the role of the oxides used to improve the hydrogen kinetics of Mg/MgH2. In the initial model of Klassen et al., it is proposed that only transition metal oxides comprising defective oxide sites and high valance state metals capable of high electrons exchange rate during catalysis are suitable for improving Mg/MgH2 kinetics [15,25]. If such model can in part explain a potential contribution of transition metal oxides in improving the kinetics of the hydrogen absorption/desorption process, it fails to describe the positive effect of TiO2, MgO and other non-transition metals oxides.
In our early investigations, we have reported that non transition metal oxides such as Al2O3 were also good at improving the hydrogen kinetics of Mg/MgH2 [45]. We also found that upon Nb2O5 addition to MgH2 during the milling process a significant improvement of the grinding process was achieved [44]. Taking into account the poor ability of metal oxides such as SiO2 to enhance the hydrogen kinetics of Mg [45], and considering the mechanical effect of metal oxides in facilitating the grinding and stabilisation of small MgH2 particles a correlation was found between the potency of a metal oxide in improving hydrogen kinetics in Mg and the nature of the metal-oxygen bond (Figure 6). Depending on the electronegativity of solid oxides envisaged lower hydrogen desorption temperatures were obtained for oxides of higher electronegativity such as MgO. Such a correlation is comprehensible when considering the processes occurring during high energy ball milling. As MgH2 particles are initially fractured, the generation of fresh surfaces would result in an excess of surface energy leading to particles agglomeration and cold welding upon balls impact. Hence depending on the experimental conditions, the milling process would become quickly inefficient in reducing particle sizes when the equilibrium between cold-welding and fracturing is established [32,44]. To delay this equilibrium and reach the smallest possible particle size, process control agents such as polar, non-polar organic solvents or polymers maintaining a low friction are usually employed to reduce agglomeration and cold-welding [46]. However, such additives are known to be relatively inefficient with MgH2 as they quickly degrade and lead to severe carbon contamination due to the high temperatures, i.e., >500 °C, locally generated during milling processes. At such high temperatures, oxides such as TiO2 and MgO are known to be effective lubricants providing low frictions coefficients to sliding tribological interfaces [47]. In a recently proposed model, Erdemir reported a correlation between friction coefficients and electronegativity [48]. The higher the electronegativity of a solid oxide is, the lower the friction coefficient would be. As metal oxides of higher electronegativity hold their bonding electrons more tightly and have a lower tendency to chemically bind with their neighbouring oxides, Erdemir found that higher electronegativity would lead to lower shearing at the interface of sliding oxide particles and thus lower friction coefficients. The favourable tribological properties of highly electronegative oxides would explain the correlation obtained with respect to the hydrogen kinetics of MgH2 milled with oxides.
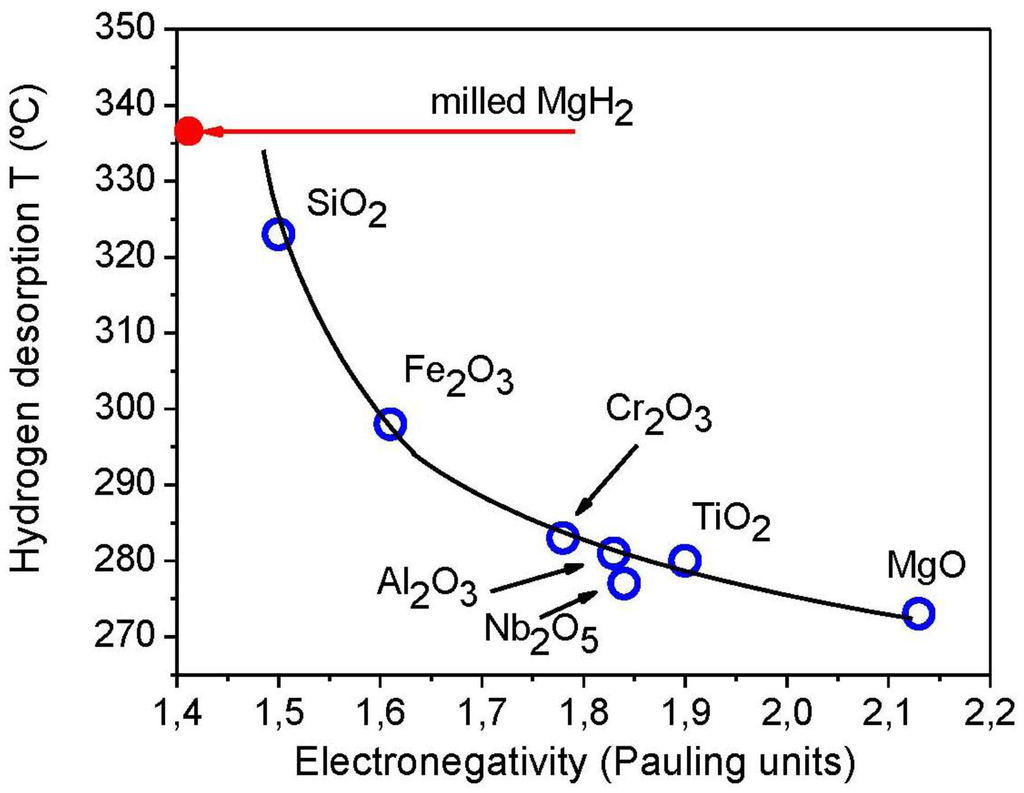
Figure 6.
Correlation between the desorption temperature of hydrogen from MgH2 achieved upon oxide addition during milling and the electronegativity of the oxide additives.
At high temperatures, as the oxide layer is depleted from the particle surface by wear, the surface of MgH2 particles would be oxidised to some extend at sliding interfaces. Accordingly, the mechanical process of MgH2 with solid oxides could involve the sliding of two oxidised surfaces: that of the partially oxidised MgH2 particles and the metal oxide additive. The formation of MgO observed during the milling process of MgH2 with Nb2O5, for example [26], would corroborate the existence of such an interface. As Nb2O5 is replaced by more electronegative oxides showing higher friction coefficients, efficient reduction of MgH2 particle size upon milling would thus become possible. Such a correlation clearly corroborates our observations as the use of MgO having the highest electronegativity of all metal oxides investigated so far led to the most advanced hydrogen kinetics in Mg. It also explains why a simple pre-oxidation of MgH2 prior to mechanical milling did not help in reducing particle sizes and improving hydrogen kinetics. Furthermore, the correlation found is a clear indicator of the mechanical effect of solid oxides in facilitating the grinding process of MgH2 and fast hydrogen kinetics through particle size reduction and stabilisation. Although further work is needed to establish the levels of “catalytic/mechanical” contribution of solid oxides to the hydrogen kinetics of magnesium, the correlation found evidently highlights the potential of particle size reduction as a mean for finally controlling the properties of the Mg/MgH2 system and we are currently exploring the potential of such a route [49].
4. Conclusions
Addition of MgO to MgH2 during the milling process led to a significant improvement of hydrogen kinetics with absorption/desorption rates as fast as those reported with the best catalyst found so far, i.e., Nb2O5. Quantitative analysis of the hydrogen kinetic curves obtained for MgH2 milled with MgO revealed that hydrogen absorption is diffusion controlled, whereas hydrogen desorption shifted from the typical nucleation and growth process characterising pristine MgH2 to an interface controlled process. Such modifications in desorption behaviour also observed upon Nb2O5 addition has previously been regarded as an indicator of the catalytic behaviour of Nb2O5. Although contradictory in first instance, since MgO is unable to catalyse the reaction of hydrogen with magnesium, these results can be reconciled by considering the tribological effects of solid oxides during the milling process of MgH2. Hence, a correlation between the electronegativity of metal oxides and achievable hydrogen desorption was found. The higher the oxide electronegativity, the lower the hydrogen desorption temperature. Such a correlation can be explained by the lower friction coefficient involved at the interface of solid oxides of high electronegativity reflecting effective gridding processes. Furthermore, the correlation found clearly points toward the mechanical effect of solid oxides in facilitating the grinding of MgH2 and the stabilisation of small particles sizes capable of fast hydrogen kinetics due to the shorter diffusion paths involved.
Acknowledgments
This research was under the Australian Research Council’s Discovery Projects funding scheme (project DP1095209) and supported by the Spanish Ministry of Science and Innovation, under the contract MICINN-MAT2011-22780 and Comunidad de Madrid under the contract CG10-UAM/ENE-5245.
References
- Niemann, M.U.; Srinivasan, S.S.; Phani, A.R.; Kumar, A.; Goswami, D.Y.; Stefanakos, E.K. Nanomaterials for hydrogen storage applications: A review. J. Nanomater. 2008, 2008, 950967:1–950967:9. [Google Scholar]
- Sakintuna, B.; Lamari-Darkrim, F.; Hirscher, M. Metal hydride materials for solid hydrogen storage: A review. Int. J. Hydr. Energy 2007, 32, 1121–1140. [Google Scholar] [CrossRef]
- Bogdanovic, B. Catalytic synthesis of organo-lithium and organomagnesium compounds and of lithium and magnesium hydrides—Applications in organic-synthesis and hydrogen storage. Angew. Chem. Int. Ed. 1985, 24, 262–273. [Google Scholar] [CrossRef]
- Huot, J.; Liang, G.; Schulz, R. Mechanically alloyed metal hydride systems. Appl. Phys. Mater. Sci. Proc. 2001, 72, 187–195. [Google Scholar] [CrossRef]
- Aguey-Zinsou, K.F.; Ares-Fernandez, J.R. Hydrogen in magnesium: New perspectives toward functional stores. Energy Environ. Sci. 2010, 3, 526–543. [Google Scholar] [CrossRef]
- Huot, J.; Liang, G.; Boily, S.; van Neste, A.; Schulz, R. Structural study and hydrogen sorption kinetics of ball-milled magnesium hydride. J. Alloy. Compd. 1999, 293, 495–500. [Google Scholar] [CrossRef]
- Schulz, R.; Huot, J.; Liang, G.; Boily, S.; Lalande, G.; Denis, M.C.; Dodelet, J.P. Recent developments in the applications of nanocrystalline materials to hydrogen technologies. Mater. Sci. Eng. 1999, 267, 240–245. [Google Scholar] [CrossRef]
- Klassen, T.; Bohn, R.; Fanta, G.; Oelerich, W.; Eigen, N.; Gartner, F.; Aust, E.; Bormann, R.; Kreye, H. Tailoring nanocrystalline materials towards potential applications. Z. Für Metall. 2003, 94, 610–614. [Google Scholar]
- Zaluska, A.; Zaluski, L.; Strom-Olsen, J.O. Nanocrystalline magnesium for hydrogen storage. J. Alloy. Compd. 1999, 288, 217–225. [Google Scholar] [CrossRef]
- Friedrichs, O.; Aguey-Zinsou, F.; Fernandez, J.R.A.; Sanchez-Lopez, J.C.; Justo, A.; Klassen, T.; Bormann, R.; Fernández, A. MgH2 with Nb2O5 as additive, for hydrogen storage: Chemical, structural and kinetic behavior with heating. Acta Mater. 2006, 54, 105–110. [Google Scholar]
- Schimmel, H.G.; Johnson, M.R.; Kearley, G.J.; Ramirez-Cuesta, A.J.; Huot, J.; Mulder, F.M. The vibrational spectrum of magnesium hydride from inelastic neutron scattering and density functional theory. Mater. Sci. Eng. 2004, 108, 38–41. [Google Scholar] [CrossRef]
- Schimmel, H.G.; Johnson, M.R.; Kearley, G.J.; Ramirez-Cuesta, A.J.; Huot, J.; Mulder, F.M. Structural information on ball milled magnesium hydride from vibrational spectroscopy and ab-initio calculations. J. Alloy. Compd. 2005, 393, 1–4. [Google Scholar] [CrossRef]
- Huot, J.; Pelletier, J.F.; Lurio, L.B.; Sutton, M.; Schulz, R. Investigation of dehydrogenation mechanism of MgH2-Nb nanocomposites. J. Alloy. Compd. 2003, 348, 319–324. [Google Scholar] [CrossRef]
- Schimmel, H.G.; Huot, J.; Chapon, L.C.; Tichelaar, F.D.; Mulder, F.M. Hydrogen cycling of niobium and vanadium catalyzed nanostructured magnesium. J. Am. Chem. Soc. 2005, 127, 14348–14354. [Google Scholar]
- Oelerich, W.; Klassen, T.; Bormann, R. Metal oxides as catalysts for improved hydrogen sorption in nanocrystalline Mg-based materials. J. Alloy. Compd. 2001, 315, 237–242. [Google Scholar] [CrossRef]
- Song, M.Y.; Bobet, J.L.; Darriet, B. Improvement in hydrogen sorption properties of Mg by reactive mechanical grinding with Cr2O3, Al2O3 and CeO2. J. Alloy. Compd. 2002, 340, 256–262. [Google Scholar] [CrossRef]
- Yan, Y.G.; Chen, Y.G.; Liang, H.; Wu, C.L.; Tao, M.D. Hydrogen storage properties of V30-Ti-Cr-Fe alloys. J. Alloy. Compd. 2007, 427, 110–114. [Google Scholar] [CrossRef]
- Barkhordarian, G.; Klassen, T.; Bormann, R. Kinetic investigation of the effect of milling time on the hydrogen sorption reaction of magnesium catalyzed with different Nb2O5 contents. J. Alloy. Compd. 2006, 407, 249–255. [Google Scholar] [CrossRef]
- Liang, G.; Huot, J.; Boily, S.; van Neste, A.; Schulz, R. Hydrogen storage properties of the mechanically milled MgH2-V nanocomposite. J. Alloy. Compd. 1999, 291, 295–299. [Google Scholar] [CrossRef]
- Bazzanella, N.; Checchetto, R.; Miotello, A. Catalytic effect on hydrogen desorption in Nb-doped microcrystalline MgH2. Appl. Phys. Lett. 2004, 85, 5212–5214. [Google Scholar]
- Dolci, F.; Di Chio, M.; Baricco, M.; Giamello, E. Niobium pentoxide as promoter in the mixed MgH2/Nb2O5 system for hydrogen storage: A multitechnique investigation of the H-2 uptake. J. Mater. Sci. 2007, 42, 7180–7185. [Google Scholar]
- Iizuka, T.; Ogasawara, K.; Tanabe, K. Acidic and catalytic properties of niobium pentaoxide. Bull. Chem. Soc. Jpn. 1983, 56, 2927–2931. [Google Scholar] [CrossRef]
- Ziolek, M.; Sobczak, I.; Lewandowska, A.; Nowak, I.; Decyk, P.; Renn, M.; Jankowska, B. Oxidative properties of niobium-containing mesoporous silica catalysts. Catal. Today 2001, 70, 169–181. [Google Scholar]
- Barkhordarian, G.; Klassen, T.; Bormann, R. Effect of Nb2O5 content on hydrogen reaction kinetics of Mg. J. Alloy. Compd. 2004, 364, 242–246. [Google Scholar] [CrossRef]
- Barkhordarian, G.; Klassen, T.; Bormann, R. Catalytic mechanism of transition-metal compounds on Mg hydrogen sorption reaction. J. Phys. Chem. 2006, 110, 11020–11024. [Google Scholar]
- Friedrichs, O.; Martinez-Martinez, D.; Guilera, G.; Lopez, J.C.S.; Fernandez, A. In situ energy-dispersive XAS and XRD study of the superior hydrogen storage system MgH2/Nb2O5. J. Phys. Chem. 2007, 111, 10700–10706. [Google Scholar]
- Friedrichs, O.; Sanchez-Lopez, J.C.; Lopez-Cartes, C.; Klassen, T.; Bormann, R.; Fernandez, A. Nb2O5 “pathway effect” on hydrogen sorption in Mg. J. Phys. Chem. 2006, 110, 7845–7850. [Google Scholar]
- Aurora, A.; Mancini, M.R.; Gattia, D.M.; Montone, A.; Pilloni, L.; Todini, E.; Antisari, M.V. Microstructural and kinetic investigation of hydrogen sorption reaction of MgH2/Nb2O5 nanopowders. Mater. Manuf. Proc. 2009, 24, 1058–1063. [Google Scholar] [CrossRef]
- Noh, H.; Wang, D.; Luo, S.; Flanagan, T.B.; Balasubramaniam, R.; Sakamoto, Y. Hydrogen bronze formation within Pd/MoO3 composites. J. Phys. Chem. 2004, 108, 310–319. [Google Scholar]
- Sakaguchi, H.; Shirai, H.; Tanaka, H.; Adachi, G.Y. Hydrogen permeation characteristics for oxide/metal multilayered films. Chem. Mater. 1995, 7, 137–141. [Google Scholar] [CrossRef]
- Shirai, H.; Tanaka, H.; Sakaguchi, H.; Adachi, G. Hydrogen permeation characteristics for oxide/metal multilayered films. J. Phys. Chem. 1993, 97, 6007–6010. [Google Scholar]
- Aguey-Zinsou, K.F.; Fernandez, J.R.A.; Klassen, T.; Bormann, R. Using MgO to improve the (de)hydriding properties of magnesium. Mater. Res. Bull. 2006, 41, 1118–1126. [Google Scholar] [CrossRef]
- Aguey-Zinsou, K.F.; Ares, J.R.; Klassen, T.; Bormann, R. Production of Magnesium/Magnesium Alloy Powder Hydride Containing Hydrogen Storage Material for Power Supply of Technical Devices, Comprises Grinding the Magnesium Hydride Powder and Diamond Powder under Hydrogen Atmosphere; GKSS Forschungszentrum Geesthacht Gmbh: Geesthacht, Germany, 2006. [Google Scholar]
- Dehouche, Z.; Goyette, J.; Bose, T.K.; Huot, J.; Schulz, R. Sensitivity of nanocrystalline MgH2-V hydride composite to the carbon monoxide during a long-term cycling. Nano Lett. 2001, 1, 175–178. [Google Scholar] [CrossRef]
- Ares, J.R.; Aguey-Zinsou, K.F.; Klassen, T.; Bormann, R. Influence of impurities on the milling process of MgH2. J. Alloy. Compd. 2007, 434, 729–733. [Google Scholar] [CrossRef]
- Friedrichs, O.; Sanchez-Lopez, J.C.; Lopez-Cartes, C.; Dornheim, M.; Klassen, T.; Bormann, R.; Fernández, A. Chemical and microstructural study of the oxygen passivation behaviour of nanocrystalline Mg and MgH2. Appl. Surf. Sci. 2006, 252, 2334–2345. [Google Scholar]
- Borgschulte, A.; Bielmann, M.; Zuttel, A.; Barkhordarian, G.; Dornheim, M.; Bormann, R. Hydrogen dissociation on oxide covered MgH2 by catalytically active vacancies. Appl. Surf. Sci. 2008, 254, 2377–2384. [Google Scholar]
- Paganini, M.C.; Chiesa, M.; Giamello, E.; Coluccia, S.; Martra, G.; Murphy, D.M.; Pacchioni, G. Colour centres at the surface of alkali-earth oxides. A new hypothesis on the location of surface electron traps. Surf. Sci. 1999, 421, 246–262. [Google Scholar] [CrossRef]
- Sterrer, M.; Berger, T.; Diwald, O.; Knozinger, E.; Sushko, P.V.; Shluger, A.L. Chemistry at corners and edges: Generation and adsorption of H atoms on the surface of MgO nanocubes. J. Chem. Phys. 2005, 123, 064714:1–064714:7. [Google Scholar]
- Diwald, O.; Hofmann, P.; Knozinger, E. H-2 chemisorption and consecutive UV stimulated surface reactions on nanostructured MgO. Phys. Chem. Chem. Phys. 1999, 1, 713–721. [Google Scholar]
- Mintz, M.H.; Zeiri, Y. Hydriding kinetics of powders. J. Alloy. Compd. 1995, 216, 159–175. [Google Scholar] [CrossRef]
- Hancock, J.D.; Sharp, J.H. Method of comparing solid-state kinetic data and its application to decomposition of kaolinite, brucite, and BaCO3. J. Am. Ceram. Soc. 1972, 55, 74–77. [Google Scholar] [CrossRef]
- Li, W.Y.; Li, C.S.; Ma, H.; Chen, J. Magnesium nanowires: Enhanced kinetics for hydrogen absorption and desorption. J. Am. Chem. Soc. 2007, 129, 6710–6711. [Google Scholar] [CrossRef]
- Aguey-Zinsou, K.F.; Fernandez, J.R.A.; Klassen, T.; Bormann, R. Effect of Nb2O5 on MgH2 properties during mechanical milling. Int. J. Hydr. Energy 2007, 32, 2400–2407. [Google Scholar] [CrossRef]
- Aguey-Zinsou, K.F.; Nicolaisen, T.; Fernandez, J.R.A.; Klassen, T.; Bormann, R. Effect of nanosized oxides on MgH2 (de)hydriding kinetics. J. Alloy. Compd. 2007, 434, 738–742. [Google Scholar] [CrossRef]
- Suryanarayana, C. Mechanical alloying and milling. Prog. Mater. Sci. 2001, 46, 1–184. [Google Scholar] [CrossRef]
- Erdemir, A. A crystal-chemical approach to lubrication by solid oxides. Tribol. Lett. 2000, 8, 97–102. [Google Scholar] [CrossRef]
- Erdemir, A.; Li, S.H.; Jin, Y.S. Relation of certain quantum chemical parameters to lubrication behavior of solid oxides. Int. J. Mol. Sci. 2005, 6, 203–218. [Google Scholar] [CrossRef]
- Aguey-Zinsou, K.F.; Ares-Fernandez, J.R. Synthesis of colloidal magnesium: A near room temperature store for hydrogen. Chem. Mater. 2008, 20, 376–378. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).