Effects of Small-Scale Gold Mining Tailings on the Underwater Light Field in the Tapajós River Basin, Brazilian Amazon
Abstract
:1. Introduction
2. Material and Methods
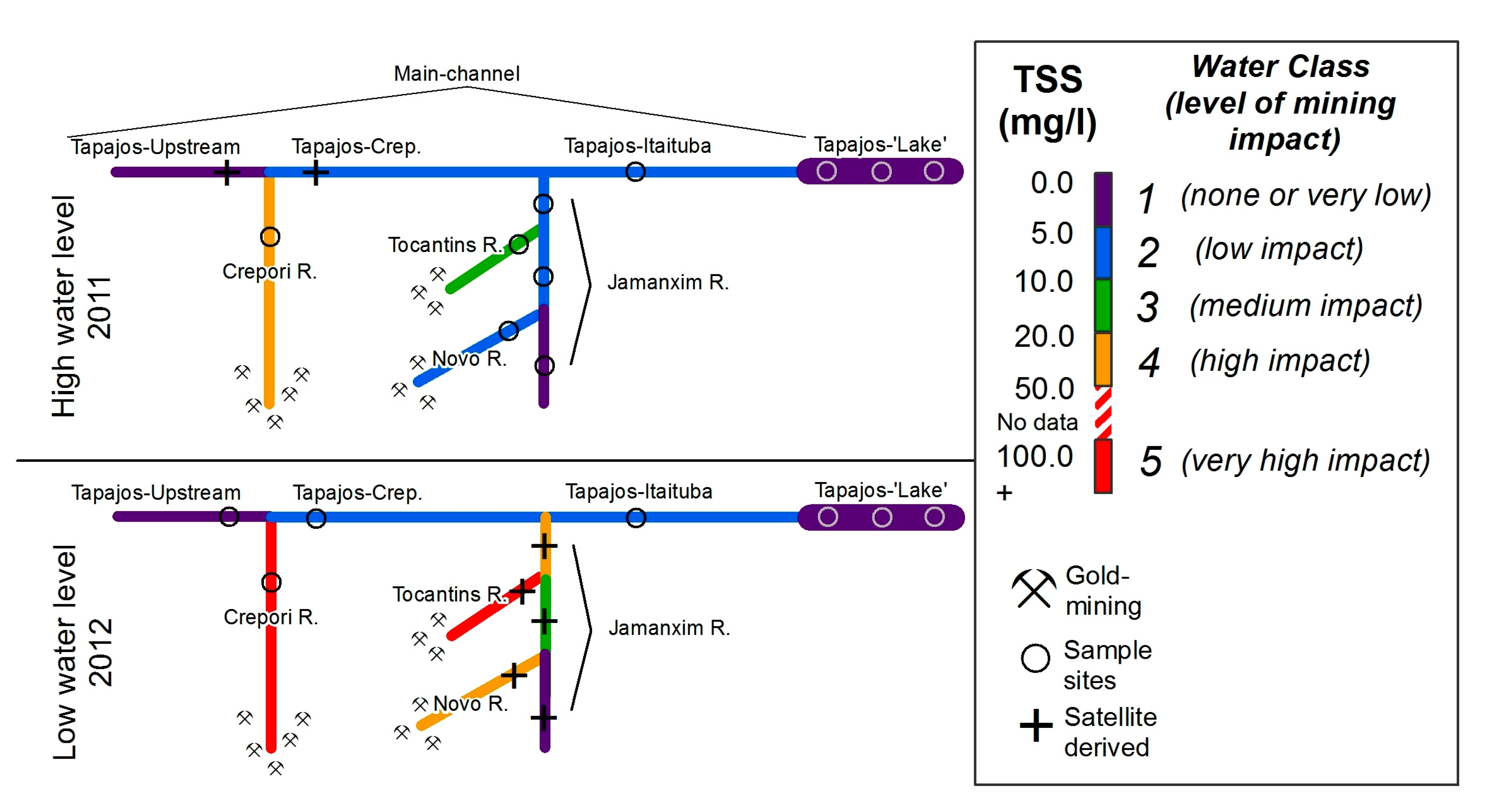
2.1. Study Area
2.2. Methods
2.2.1. Sampling
2.2.2. Biogeochemical Data
2.2.3. Optical Data
2.2.4. Critical Depth for Photosynthesis
2.2.5. Data Analysis
3. Results
3.1. Biogeochemical Data
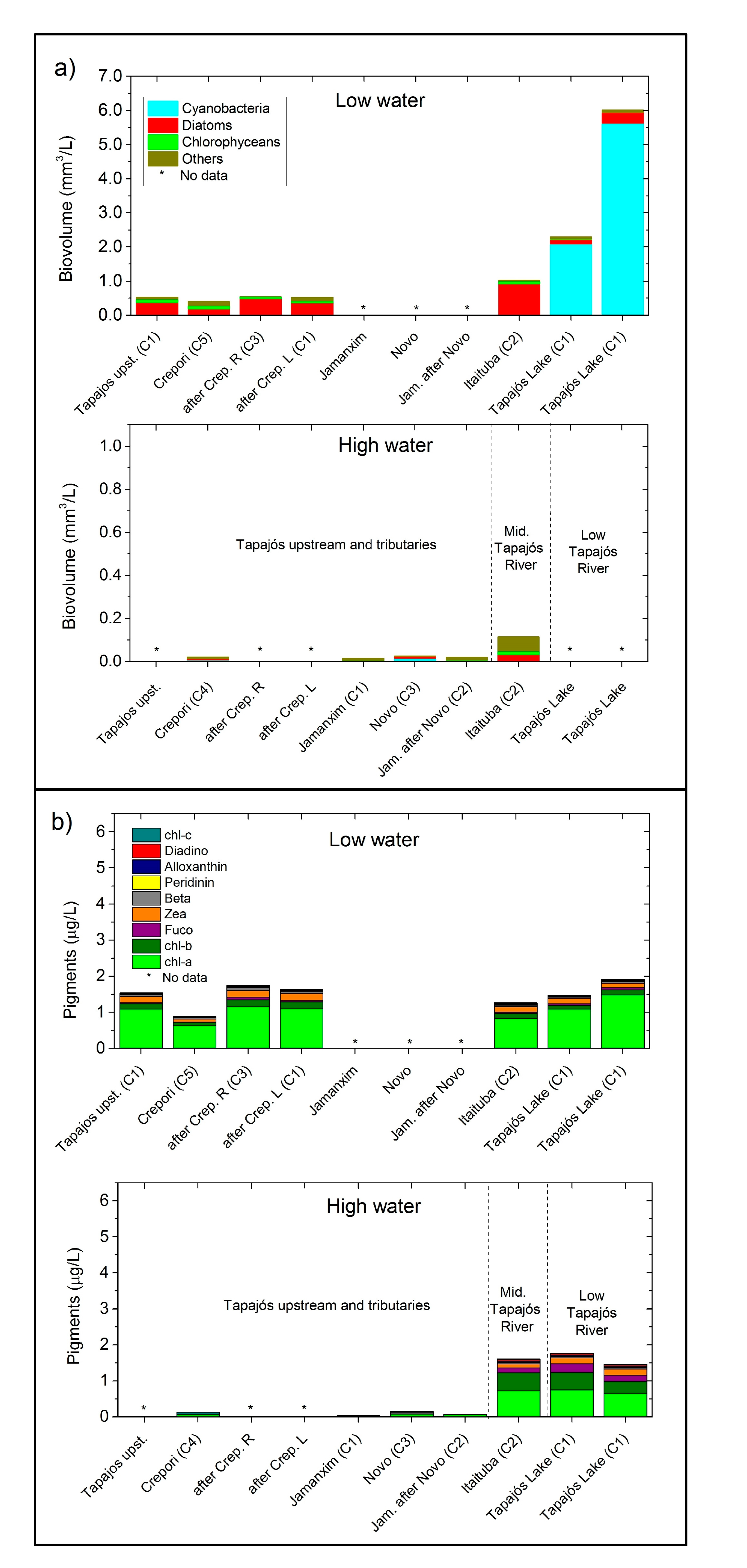
3.2. Phytoplankton and Pigments
3.3. Bio-Optical Data
3.3.1. Inherent Optical Properties (IOPs)
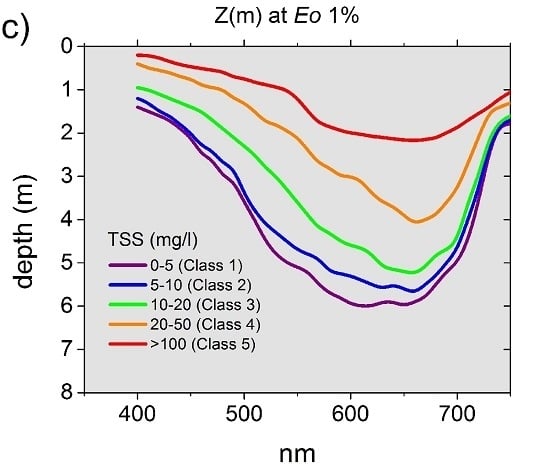
3.3.2. Apparent Optical Properties (AOPs) and Underwater Light Field
4. Discussion
4.1. Mining-Derived TSS as the Main Factor Changing the Water Optical Properties and Light Field
4.2. Underwater Light Field and Phytoplankton
5. Conclusions
Supplementary Materials
Supplementary File 1Acknowledgments
Author Contributions
Conflicts of Interests
Appendix A
| Valid N | Valid N | Rank Sum | Rank Sum | U | Z | p-Value | |
|---|---|---|---|---|---|---|---|
| Class 1 vs. Class 2 | class 1 | class 2 | class 1 | class 2 | |||
| 16 | 17 | 136.00 | 425.00 | 0.00 | −4.881 | 0.000 | |
| Class 2 vs. Class 3 | class 2 | class 3 | class 2 | class 3 | |||
| 17 | 2 | 153.00 | 37.00 | 0.00 | −2.192 | 0.028 | |
| Class 3 vs. Class 4 | class 3 | class 4 | class 3 | class 4 | |||
| 2 | 3 | 3.00 | 12.00 | 0.00 | −1.443 | 0.096 | |
| Class 4 vs. Class 5 | class 4 | class 5 | class 4 | class 5 | |||
| 3 | 3 | 6.00 | 15.00 | 0.00 | −1.746 | 0.081 |
References
- Seccatore, J. An estimation of the artisanal small-scale production of gold in the world. Sci. Total Environ. 2014, 496, 662–667. [Google Scholar] [CrossRef] [PubMed]
- Telmer, K.; Veiga, M. World emissions of mercury from artisanal and small scale gold mining. In Mercury Fate and Transport in the Global Atmosphere: Emissions, Measurements and Models; Springer: Boston, MA, USA, 2009; Chapter 6; pp. 131–172. ISBN 978-0-387-93958-2. [Google Scholar]
- Grätz, T. Moralities, risk and rules in West African artisanal gold mining communities: A case study of northern Benin. Resour. Policy 2009, 34, 12–17. [Google Scholar] [CrossRef]
- Coelho, M.C.N.; Wanderley, L.J.; Costa, R.C. Small scale gold mining in the XXI century. Examples in the south-west Brazilian Amazon. Anu. Inst. Geociênc. UFRJ 2016, 39, 10. [Google Scholar]
- Nevado, J.J.B.; Martin-Doimeadios, R.C.R.; Bernardo, F.J.G.; Moreno, M.J.; Herculano, A.M.; do Nascimento, J.L.M.; Crespo-Lopez, M.E. Mercury in the Tapajos River Basin, Brazilian Amazon: A review. Environ. Int. 2010, 36, 593–608. [Google Scholar] [CrossRef] [PubMed]
- Dorea, J.G.; Barbosa, A.C. Anthropogenic impact of mercury accumulation in fish from the Rio Madeira and Rio Negro rivers (Amazonia). Biol. Trace Elem. Res. 2007, 115, 243–254. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, R.M.; Mascarenhas, A.F.S.; Ichihara, A.H.; Souza, T.M.C. Estudo dos Impactos Ambientais Decorrentes do Extrativismo Mineral e Poluição Mercurial no Tapajós—Pré-Diagnóstico; CETEM/CNPq: Rio de Janeiro, Brazil, 1994; p. 220. [Google Scholar]
- Dambacher, J.M.; Brewer, D.T.; Dennis, D.M.; Macintyre, M.; Foale, S. Qualitative modelling of gold mine impacts on Lihir island’s socioeconomic system and reef-edge fish community. Environ. Sci. Technol. 2007, 41, 555–562. [Google Scholar] [CrossRef] [PubMed]
- Schueler, V.; Kuemmerle, T.; Schroeder, H. Impacts of surface gold mining on land use systems in western Ghana. Ambio 2011, 40, 528–539. [Google Scholar] [CrossRef] [PubMed]
- Tudesque, L.; Grenouillet, G.; Gevrey, M.; Khazraie, K.; Brosse, S. Influence of small-scale gold mining on French Guiana streams: Are diatom assemblages valid disturbance sensors? Ecol. Indic. 2012, 14, 100–106. [Google Scholar] [CrossRef]
- Finer, M.; Novoa, S. Gold Mining Deforestation Continues to Expand in la Pampa (Madre De Dios, Peru). Available online: http://maaproject.org/2015/03/gold-mining-deforestation-expand-peruvian-amazon/ (accessed on 26 January 2016).
- Mol, J.H.; Ouboter, P.E. Downstream effects of erosion from small-scale gold mining on the instream habitat and fish community of a small neotropical rainforest stream. Conserv. Biol. 2004, 18, 201–214. [Google Scholar] [CrossRef]
- Sousa, R.N.; Veiga, M.M. Using performance indicators to evaluate an environmental education program in artisanal gold mining communities in the Brazilian Amazon. Ambio 2009, 38, 40–46. [Google Scholar] [CrossRef] [PubMed]
- Couceiro, S.R.M.; Hamada, N.; Forsberg, B.R.; Padovesi-Fonseca, C. Trophic structure of macroinvertebrates in amazonian streams impacted by anthropogenic siltation. Austral Ecol. 2011, 36, 628–637. [Google Scholar] [CrossRef]
- Boudou, A.; Maury-Brachet, R.; Coquery, M.; Durrieu, G.; Cossa, D. Synergic effect of gold mining and damming on mercury contamination in fish. Environ. Sci. Technol. 2005, 39, 2448–2454. [Google Scholar] [CrossRef] [PubMed]
- Roland, F.; Esteves, F.D. Effects of bauxite tailing on par attenuation in an Amazonian crystalline water lake. Hydrobiologia 1998, 377, 1–7. [Google Scholar] [CrossRef]
- Guenther, M.; Bozelli, R. Effects of inorganic turbidity on the phytoplankton of an Amazonian lake impacted by bauxite tailings. Hydrobiologia 2004, 511, 151–159. [Google Scholar] [CrossRef]
- Kirk, J.T.O. Light and Photosynthesis in Aquatic Ecosystems, 3rd ed.; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2011; ISBN 9780521151757. [Google Scholar]
- Markager, S.; Vincent, W.F. Light absorption by phytoplankton: Development of a matching parameter for algal photosynthesis under different spectral regimes. J. Plankton Res. 2001, 23, 1373–1384. [Google Scholar] [CrossRef]
- Araújo Neto, H. Perfil do Ouro; Ministério de Minas e Energia: Brasília, Brazil, 2009; p. 50.
- Telmer, K.; Costa, M.; Simões Angélica, R.; Araujo, E.S.; Maurice, Y. The source and fate of sediment and mercury in the Tapajós River, Pará, Brazilian Amazon: Ground- and space-based evidence. J. Environ. Manag. 2006, 81, 101–113. [Google Scholar] [CrossRef] [PubMed]
- INPE. Prodes Project. Available online: www.obt.inpe.br/prodes (accessed on 1 June 2011).
- CPRM. Província Mineral do Tapajós: Geologia, Metalogenia e Mapa Previsional Para Ouro em SIG. 2009. Available online: http://www.cprm.gov.br (accessed on 1 April 2013).
- Junk, W. General aspects of floodplain ecology with special reference to Amazonian floodplains. In The Central Amazon Floodplain; Junk, W., Ed.; Springer: Berlin, Germany, 1997; Volume 126, pp. 3–20. [Google Scholar]
- Sioli, H. The amazon and its main affluents: Hydrography, morphology of the river courses, and river types. In The Amazon; Sioli, H., Ed.; Springer: Dordrecht, The Netherlands, 1984; Volume 56, pp. 127–165. [Google Scholar]
- Rudorff, C.M.; Galvao, L.S.; Novo, E.M.L.M. Reflectance of floodplain waterbodies using EO-1 Hyperion data from high and receding flood periods of the Amazon River. Int. J. Remote Sens. 2009, 30, 2713–2720. [Google Scholar] [CrossRef]
- Novo, E.; Barbosa, C.C.D.; de Freitas, R.M.; Shimabukuro, Y.E.; Melack, J.M.; Pereira, W. Seasonal changes in chlorophyll distributions in amazon floodplain lakes derived from MODIS images. Limnology 2006, 7, 153–161. [Google Scholar] [CrossRef]
- Sá, L.L.C.D.; Vieira, J.M.D.S.; Mendes, R.D.A.; Pinheiro, S.C.C.; Vale, E.R.; Alves, F.A.D.S.; Jesus, I.M.D.; Santos, E.C.D.O.; Costa, V.B.D. Ocorrência de uma floração de cianobactérias tóxicas na margem direita do rio Tapajós, no município de Santarém (Pará, Brasil). Rev. Pan-Amazôn. Saúde 2010, 1, 159–166. [Google Scholar] [CrossRef]
- Roulet, M.; Lucotte, M.; Canuel, R.; Farella, N.; Goch, Y.G.D.; Peleja, J.R.P.; Guimaraes, J.R.D.; Mergler, D.; Amorim, M. Spatio-temporal geochemistry of mercury in waters of the Tapajos and Amazon rivers, Brazil. Limnol. Oceanogr. 2001, 46, 1141–1157. [Google Scholar] [CrossRef]
- Farella, N.; Lucotte, M.; Louchouarn, P.; Roulet, M. Deforestation modifying terrestrial organic transport in the rio Tapajós, Brazilian Amazon. Organ. Geochem. 2001, 32, 1443–1458. [Google Scholar] [CrossRef]
- Gibbs, R.J. Amazon river: Environmental factors that control its dissolved and suspended load. Science 1967, 156, 1734–1737. [Google Scholar] [CrossRef] [PubMed]
- Costa, M.P.F.; Novo, E.M.L.M.; Telmer, K.H. Spatial and temporal variability of light attenuation in large rivers of the Amazon. Hydrobiologia 2013, 702, 171–190. [Google Scholar] [CrossRef]
- Roulet, M.; Lucotte, M.; Canuel, R.; Rheault, I.; Tran, S.; Gog, Y.G.D.; Farella, N.; do Vale, R.S.; Passos, C.J.S.; da Silva, E.D.; et al. Distribution and partition of total mercury in waters of the Tapajos River Basin, Brazilian Amazon. Sci. Total Environ. 1998, 213, 203–211. [Google Scholar] [CrossRef]
- Reynolds, C.S. The Ecology of Phytoplankton; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2006; ISBN 9780521844130. [Google Scholar]
- Bernardes, M.C.; Martinelli, L.A.; Krusche, A.V.; Gudeman, J.; Moreira, M.; Victoria, R.L.; Ometto, J.; Ballester, M.V.R.; Aufdenkampe, A.K.; Richey, J.E.; et al. Riverine organic matter composition as a function of land use changes, southwest amazon. Ecol. Appl. 2004, 14, S263–S279. [Google Scholar] [CrossRef]
- APHA. Standard Methods for the Examination of Water and Wastewater; APHA-AWWA-WEF: Washington, DC, USA, 2005; ISBN 0875530478 9780875530475. [Google Scholar]
- Hansell, D.A. Biogeochemistry of total organic carbon and nitrogen in the Sargasso Sea: Control by convective overturn. Deep-Sea Res. Part II Top. Stud. Oceanogr. 2001, 48, 1649–1667. [Google Scholar] [CrossRef]
- Buchaca, T. A comparison of HPLC pigment analyses and biovolume estimates of phytoplankton groups in an oligotrophic lake. J. Plankton Res. 2004, 27, 91–101. [Google Scholar] [CrossRef]
- Utermöhl, H. Zur vervollkommung der quantitative phytoplankton—Methodik. Mitt. Int. Ver. Theor. Angew. Limnol. 1958, 9, 1–38. [Google Scholar]
- Lund, J.; Kipling, C.; LeCren, E. The inverted microscope method of estimating algal number and the statistical basis of estimation by count. Hydrobiologia 1958, 11, 143–170. [Google Scholar] [CrossRef]
- Hillebrand, H.; Dürselen, C.; Kirschtel, D.; Pollingher, U.; Zohary, T. Biovolume calculation for pelagic and benthic algae. J. Phycol. 1999, 35, 403–424. [Google Scholar] [CrossRef]
- Mobley, C.D. Light and Water: Radiative Transfer in Natural Waters; Academic Press: Cambridge, MA, USA, 1994; ISBN 0125027508. [Google Scholar]
- Wet Labs, I. Spectral Absorption and Attenuation Sensor (ac-s) User’s Guide; WET Labs, Inc.: Philomath, OR, USA, 2013; p. 33. [Google Scholar]
- Tilstone, G.H.; Peters, S.W.M.; van der Woerd, H.J.; Eleveld, M.A.; Ruddick, K.; Schönfeld, W.; Krasemann, H.; Martinez-Vicente, V.; Blondeau-Patissier, D.; Röttgers, R.; et al. Variability in specific-absorption properties and their use in a semi-analytical ocean colour algorithm for meris in North sea and western English channel coastal waters. Remote Sens. Environ. 2012, 118, 320–338. [Google Scholar] [CrossRef]
- Boss, E.; Pegau, W.S. Relationship of light scattering at an angle in the backward direction to the backscattering coefficient. Appl. Opt. 2001, 40, 5503–5507. [Google Scholar] [CrossRef] [PubMed]
- Satlantic. Prosoft 7.7 User Manual; Satlantic Inc.: Halifax, NS, Canada, 2011. [Google Scholar]
- Deblois, C.P.; Marchand, A.; Juneau, P. Comparison of photoacclimation in twelve freshwater photoautotrophs (chlorophyte, bacillaryophyte, cryptophyte and cyanophyte) isolated from a natural community. PLoS ONE 2013, 8, e57139. [Google Scholar] [CrossRef] [PubMed]
- Sverdrup, H. On conditions for the vernal blooming of phytoplankton. J. Conseil 1953, 18, 287–295. [Google Scholar] [CrossRef]
- Siegel, D.A.; Doney, S.C.; Yoder, J.A. The North Atlantic spring phytoplankton bloom and Sverdrup’s critical depth hypothesis. Science 2002, 296, 730–733. [Google Scholar] [CrossRef] [PubMed]
- Dustan, P. Terrestrial limitation of Amazon River productivity: Why the Amazon river is not green. Evol. Ecol. Res. 2009, 11, 421. [Google Scholar]
- Lobo, F.L.; Costa, M.P.F.; Novo, E.M. Time-series analysis of Landsat-MSS/TM/OLI images over Amazonian waters impacted by gold mining activities. Remote Sens. Environ. 2015, 157, 170–184. [Google Scholar] [CrossRef]
- Junk, W.J.; Piedade, M.T.F.; Schöngart, J.; Cohn-Haft, M.; Adeney, J.M.; Wittmann, F. A classification of major naturally-occurring Amazonian lowland wetlands. Wetlands 2011, 31, 623–640. [Google Scholar] [CrossRef]
- Mackey, M.D.; Mackey, D.J.; Higgins, H.W.; Wright, S.W. Chemtax—A program for estimating class abundances from chemical markers: Application to HPLC measurements of phytoplankton. Mar. Ecol. Prog. Ser. 1996, 144, 265–283. [Google Scholar] [CrossRef]
- Kuhl, M. Light penetration and light-intensity in sandy marine-sediments measured with irradiance and scalar irradiance fiberoptic microprobes rid a-1977–2009. Mar. Ecol. Prog. Ser. 1994, 105, 139. [Google Scholar] [CrossRef]
- Lobo, F.; Costa, M.; Novo, E.; Telmer, K. Distribution of artisanal and small-scale gold mining in the Tapajós River Basin (Brazilian Amazon) over the past 40 years and relationship with water siltation. Remote Sens. 2016, 8, 22. [Google Scholar] [CrossRef]
- Melack, J.; Forsberg, B. Biogeochemistry of Amazon floodplain lakes and associated wetlands. In The Biogeochemistry of the Amazon Basin and Its Role in a Changing World; Oxford University Press: Oxford, UK, 2001; pp. 235–276. [Google Scholar]
- Bezerra, O.; Veríssimo, A.; Uhl, C. Impactos da Garimpagem de Ouro na Amazônia Oriental; Amazon: Belém, Brazil, 1998; p. 25. [Google Scholar]
- Abe, C.; Novo, E.; Lobo, F.; Costa, M.P.F.; Dibike, Y. Impact of land use change on the hydrology and sediment load in an Amazonian Basin using SWAT. In Water Resources Management (In Review); Springer: Berlin, Germany, 2013. [Google Scholar]
- Veiga, M.M. Mercury in Artisanal Gold Mining in Latin America: Facts, Fantasies, and Solutions. In Proceedings of the UNIDO—Expert Group Meeting—Introducing New Technologies for Abatement of Global Mercury Pollution Deriving from Artisanal Gold Mining, Vienna, Auatria, 1–3 July 1997. [Google Scholar]
- Bowers, D.G.; Binding, C.E. The optical properties of mineral suspended particles: A review and synthesis. Estuar. Coast. Shelf Sci. 2006, 67, 219–230. [Google Scholar] [CrossRef]
- Binding, C.E.; Bowers, D.G.; Mitchelson-Jacob, E.G. Estimating suspended sediment concentrations from ocean colour measurements in moderately turbid waters; the impact of variable particle scattering properties. Remote Sens. Environ. 2005, 94, 373–383. [Google Scholar] [CrossRef]
- Slade, W.H.; Boss, E. Spectral attenuation and backscattering as indicators of average particle size. Appl. Opt. 2015, 54, 7264–7277. [Google Scholar] [CrossRef] [PubMed]
- Sun, D.; Li, Y.; Wang, Q.; Gao, J.; Lv, H.; Le, C.; Huang, C. Light scattering properties and their relation to the biogeochemical composition of turbid productive waters: A case study of Lake Taihu. Appl. Opt. 2009, 48, 1979–1989. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, L.A.S.D.; Barbosa, C.C.F.; Novo, E.M.L.D.M.; Rudorff, C.D.M. Implications of scatter corrections for absorption measurements on optical closure of Amazon floodplain lakes using the spectral absorption and attenuation meter (ac-S-Wetlabs). Remote Sens. Environ. 2015, 157, 123–137. [Google Scholar] [CrossRef]
- Stramski, D.; Boss, E.; Bogucki, D.; Voss, K.J. The role of seawater constituents in light backscattering in the ocean. Prog. Oceanogr. 2004, 61, 27–56. [Google Scholar] [CrossRef]
- Bergmann, T. Impacts of a recurrent resuspension event and variable phytoplankton community composition on remote sensing reflectance. J. Geophys. Res. 2004, 109. [Google Scholar] [CrossRef]
- Doxaran, D.; Ehn, J.; Bélanger, S.; Matsuoka, A.; Hooker, S.; Babin, M. Optical characterisation of suspended particles in the Mackenzie River plume (Canadian Arctic ocean) and implications for ocean colour remote sensing. Biogeosciences 2012, 9, 3213–3229. [Google Scholar] [CrossRef]
- Lorthiois, T.; Doxaran, D.; Chami, M. Daily and seasonal dynamics of suspended particles in the Rhône River plume based on remote sensing and field optical measurements. Geo-Mar. Lett. 2012, 32, 89–101. [Google Scholar] [CrossRef]
- Davies-Colley, R.; Hickey, C.; Quinn, J.; Ryan, P. Effects of clay discharges on streams. Hydrobiologia 1992, 248, 215–234. [Google Scholar] [CrossRef]
- Robert, A. River Processes: An Introduction to Fluvial Dynamics; Routledge: Abingdon, UK, 2014; ISBN 1444118927. [Google Scholar]
- Okogwu, O.I.; Ugwumba, A.O. Seasonal dynamics of phytoplankton in two tropical rivers of varying size and human impact in southeast Nigeria. Rev. Biol. Trop. 2013, 61, 1827–1840. [Google Scholar] [PubMed]
- ANA. Hidroweb: Sistemas de Informações Hidrológicas. Available online: http://hidroweb.ana.gov.br/HidroWeb (accessed on 1 May 2013).
- Salmaso, N.; Zignin, A. At the extreme of physical gradients: Phytoplankton in highly flushed, large rivers. Hydrobiologia 2010, 639, 21–36. [Google Scholar] [CrossRef]
- Brando, P.M.; Goetz, S.J.; Baccini, A.; Nepstad, D.C.; Beck, P.S.A.; Christman, M.C.; DeFries, R.S. Seasonal and interannual variability of climate and vegetation indices across the Amazon. Proc. Natl. Acad. Sci. USA 2010, 107, 14685–14690. [Google Scholar] [CrossRef] [PubMed]
- Casali, S.; Calijuri, M.D.C.; Barbarisi, B.; Renó, V.F.; Affonso, A.G.; Barbosa, C.; Silva, T.S.F.; Novo, E.M.L.D.M. Impact of the 2009 extreme water level variation on phytoplankton community structure in lower Amazon floodplain lakes. Acta Limnol. Bras. 2011, 23, 260–270. [Google Scholar] [CrossRef]

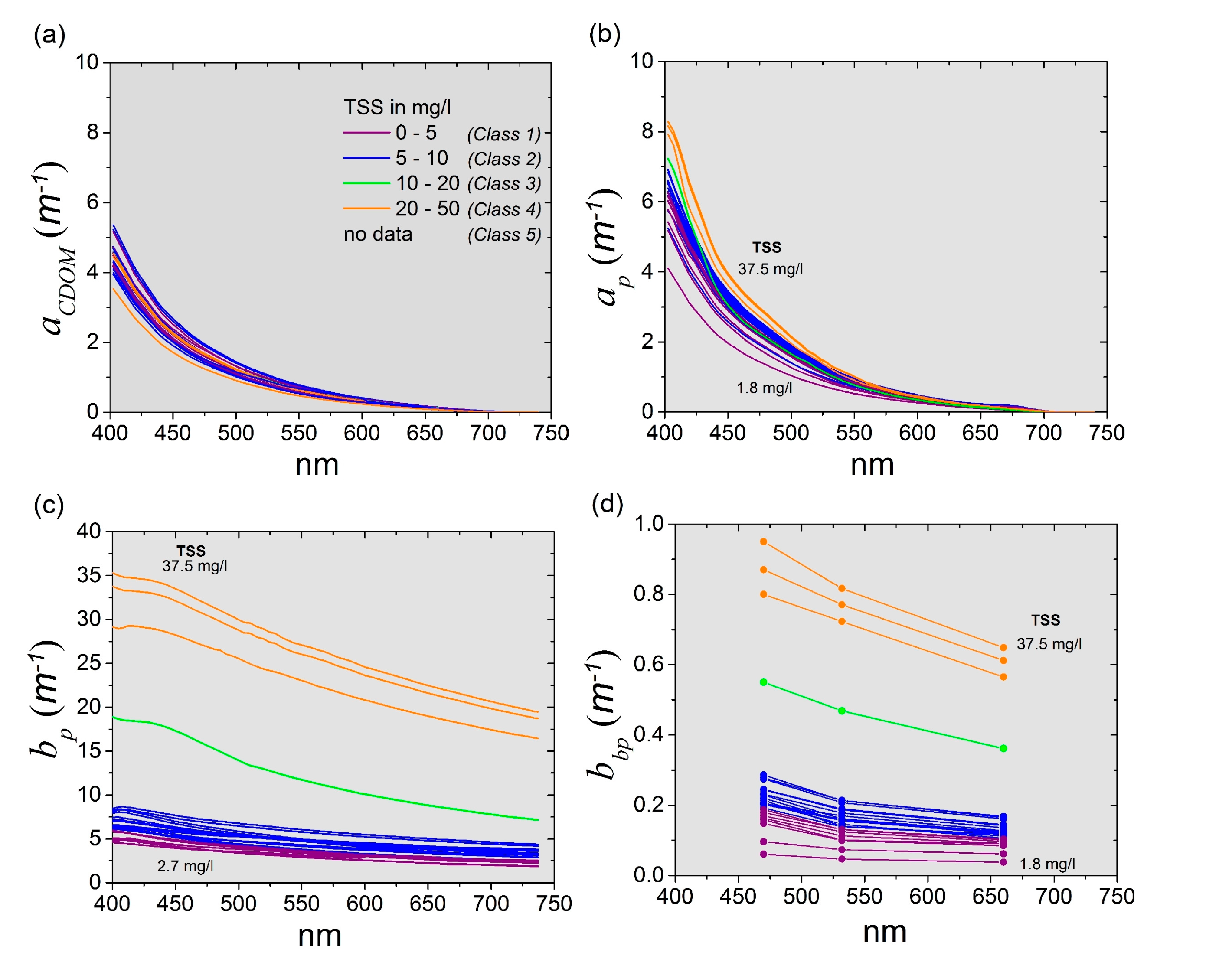

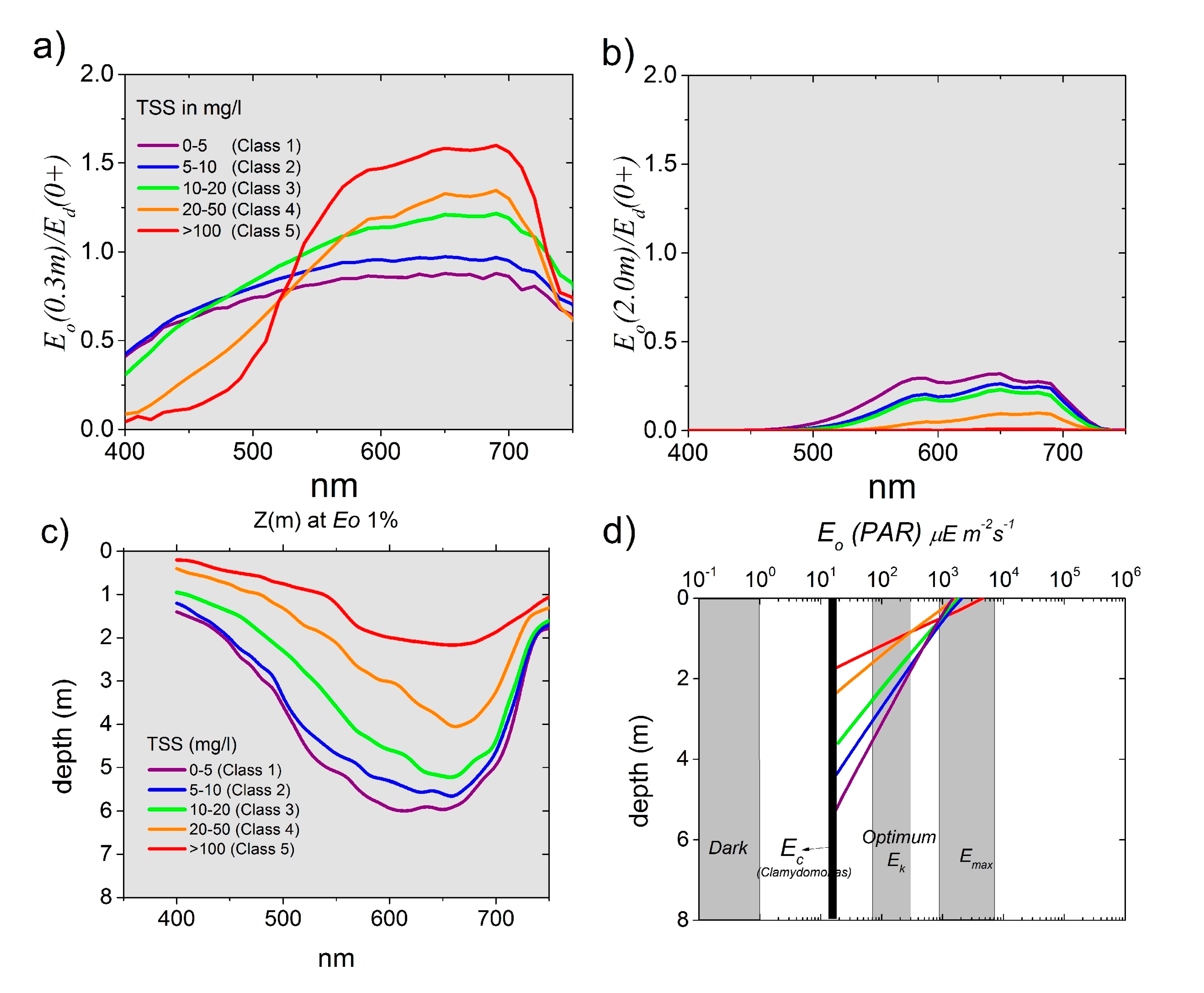
| Inherent Optical Property | Symbol | Unit | Formula |
|---|---|---|---|
| Attenuation coefficient | c(λ) | m−1 | Equation (1) |
| Absorption coefficient | a(λ) | m−1 | Equation (2) |
| Scattering coefficient | b(λ) | m−1 | Equation (3) |
| Backscattering coefficient | bb(λ) | m−1 | Equation (4) |
| Apparent Optical Property | |||
| Upwelling radiance | Lu | W·m−2·sr−1 | - |
| Downwelling irradiance | Ed | W·m−2 | - |
| Downwelling irradiance (above water) | Ed (0+) | W·m−2 | - |
| Upwelling irradiance | Eu | W·m−2 | Equation (5) |
| Scalar irradiance | Eo | W·m−2 | Equation (7) |
| Downwelling irradiance attenuation coefficient | Kd | m−1 | Equation (6) |
| Normalized scalar irradiance | - | Equation (8) | |
| Critical depth | m | Equation (9) |
| Parameter (nm) | Unit | Class 1 | Class 2 | Class 3 | Class 4 | Class 5 | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Min | Max | Average (n = 17) | SD | Min | Max | Average (n = 15) | SD | Min | Max | Average (n = 2) | Min | Max | Average (n = 3) | SD | Min | Max | Average (n = 3) | SD | |||
| Particulate Matter | TSS | mg·L−1 | 1.8 | 4.2 | 3.3 | 0.7 | 5.6 | 6.8 | 6.3 | 0.5 | 13.4 | 14.7 | 14.0 | 33.4 | 35.8 | 35.7 | 2.3 | 111.1 | 113.6 | 111.3 | 2.1 |
| Org. Matter | % of TSS | 24.0 | 58.0 | 35.0 | 11.0 | 16.0 | 28.0 | 24.0 | 4.0 | 11.0 | 16.0 | 13.0 | 12.0 | 14.0 | 12.0 | 1.0 | 2.0 | 2.0 | 2.0 | 1.0 | |
| POC | % of TSS | 10.0 | 28.0 | 15.0 | 6.0 | 7.0 | 10.0 | 9.0 | 1.0 | 4.0 | 5.0 | 4.0 | 3.0 | 3.0 | 3.0 | - | 2.0 | 2.0 | 2.0 | - | |
| chl-a | µg·L−1 | 0.0 | 1.2 | 0.7 | 0.5 | 0.1 | 1.1 | 0.5 | 0.4 | 1.1 | 0.0 | 0.6 | 0.1 | 0.1 | 0.1 | 0.0 | 0.6 | 0.7 | 0.7 | 0.1 | |
| Measured IOPs * | aCDOM (440) | m−1 | 2.5 | 3.2 | 2.9 | 0.3 | 2.9 | 3.5 | 3.2 | 0.2 | 3.0 | 3.1 | 3.1 | 2.0 | 2.1 | 2.1 | 0.1 | 2.4 | 2.8 | 2.5 | 0.3 |
| ap (440) | m−1 | 0.4 | 1.8 | 1.5 | 0.4 | 1.7 | 2.1 | 1.8 | 0.2 | 2.0 | 2.0 | 2.0 | 2.6 | 3.1 | 2.9 | 0.3 | 6.1 | 6.2 | 6.1 | 0.1 | |
| bp (660) | m−1 | 1.3 | 2.8 | 2.1 | 0.6 | 3.2 | 4.4 | 3.7 | 0.4 | 7.7 | 8.6 | 8.2 | 18.7 | 21.3 | 20.8 | 1.9 | 64.3 | 65.5 | 64.4 | 1.1 | |
| bbp (660) | m−1 | 0.1 | 0.1 | 0.1 | 0.0 | 0.1 | 0.2 | 0.1 | 0.0 | 0.3 | 0.3 | 0.3 | 0.6 | 0.6 | 0.6 | 0.0 | 1.9 | 2.0 | 1.9 | 0.0 | |
| Measured AOPs | Kd (−0) (440) | µE·−2·s−1 | 1.3 | 6.9 | 3.4 | 1.9 | 1.7 | 7.2 | 4.8 | 1.6 | 3.5 | 6.6 | 5.0 | 10.2 | 10.4 | 10.3 | 0.1 | 13.5 | 14.2 | 13.9 | 0.4 |
| Kd (−0) (560) | µE·−2·s−1 | 0.6 | 2.8 | 1.4 | 0.8 | 0.8 | 3.1 | 2.1 | 0.7 | 1.9 | 3.0 | 2.4 | 4.5 | 4.8 | 4.6 | 0.2 | 8.0 | 9.0 | 8.7 | 0.6 | |
| Kd (−0) (660) | µE·−2·s−1 | 0.7 | 1.9 | 1.2 | 0.4 | 0.9 | 2.2 | 1.6 | 0.4 | 1.7 | 1.9 | 1.8 | 3.0 | 3.4 | 3.2 | 0.2 | 6.0 | 6.3 | 6.2 | 0.2 | |
| Kd (−0, PAR) | m−1 | 1.0 | 2.8 | 2.0 | 0.9 | 1.1 | 3.7 | 2.7 | 1.1 | 3.2 | 3.8 | 3.5 | 6.0 | 6.2 | 6.0 | 0.2 | 9.0 | 9.8 | 9.6 | 0.5 | |
| Eo (0.3m)/Ed+(440) | µE·−2·s−1 | 1.6 | 2.3 | 1.9 | 0.3 | 1.5 | 2.2 | 1.8 | 0.3 | 0.9 | 1.7 | 1.3 | 0.6 | 0.8 | 0.7 | 0.1 | 0.4 | 0.5 | 0.4 | 0.3 | |
| Eo (0.3m)/Ed+(560) | µE·−2·s−1 | 4.8 | 5.4 | 5.1 | 0.3 | 4.4 | 5.2 | 4.7 | 0.4 | 3.3 | 3.9 | 3.6 | 1.9 | 2.3 | 2.1 | 0.2 | 1.0 | 1.4 | 1.2 | 0.2 | |
| Eo (0.3m)/Ed+(660) | µE·−2·s−1 | 5.1 | 6.6 | 5.9 | 0.4 | 5.5 | 6.3 | 5.7 | 0.4 | 5.0 | 5.5 | 5.3 | 3.9 | 4.3 | 4.1 | 0.2 | 2.0 | 2.4 | 2.2 | 0.2 | |
| Eo1% PAR | µE·−2·s−1 | 5.9 | 19.0 | 10.8 | 5.1 | 2.3 | 20.3 | 10.9 | 6.1 | 12.0 | 17.2 | 14.6 | 8.6 | 17.3 | 11.4 | 5.0 | 19.3 | 20.3 | 19.8 | 0.5 | |
| Photic Zone & | Z1% = ~Zc (Chlamydomonas) | m | 3.8 | 7.1 | 5.2 | 1.1 | 2.9 | 5.3 | 4.0 | 0.8 | 3.6 | 3.9 | 3.8 | 2.3 | 2.9 | 2.4 | 0.4 | 1.6 | 1.8 | 1.7 | 0.1 |
| Zm/Zc | - | 0.4 | 2.5 | 1.5 | 0.7 | 1.0 | 2.1 | 1.5 | 0.4 | 1.3 | 1.8 | 1.5 | 1.8 | 2.3 | 2.1 | 0.3 | 1.2 | 1.3 | 1.2 | 0.1 | |
| depth Zm | m | 3.1 | 15.3 | 7.4 | 3.8 | 4.9 | 7.2 | 5.6 | 0.9 | 5.0 | 6.5 | 5.8 | 5.1 | 5.1 | 5.1 | 0.1 | 2.1 | 2.0 | 2.1 | 0.1 | |
| IOPs | Suspended Solids | Pigm. | Meas. AOP | |
|---|---|---|---|---|
| TSS | POC | chl-a | Kd (PAR) | |
| ap (440) | 0.71 * | 0.80 * | 0.02 | 0.88 * |
| bp (660) | 0.99 * | 0.94 * | −0.27 | 0.94 * |
| bbp (660) | 0.99 * | 0.94 * | −0.32 | 0.95 * |
| Measured AOPS | ||||
|---|---|---|---|---|
| Kd (440) | Kd (560) | Kd (660) | Kd (PAR) | |
| TSS | 0.84 * | 0.94 * | 0.96 * | 0.91 * |
| POC | 0.89 * | 0.95 * | 0.97 * | 0.93 * |
| aCDOM | −0.48 | −0.34 | −0.25 | −0.37 |
| chl-a | −0.20 | −0.20 | −0.21 | −0.19 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lobo, F.D.L.; Costa, M.; Novo, E.M.L.D.M.; Telmer, K. Effects of Small-Scale Gold Mining Tailings on the Underwater Light Field in the Tapajós River Basin, Brazilian Amazon. Remote Sens. 2017, 9, 861. https://doi.org/10.3390/rs9080861
Lobo FDL, Costa M, Novo EMLDM, Telmer K. Effects of Small-Scale Gold Mining Tailings on the Underwater Light Field in the Tapajós River Basin, Brazilian Amazon. Remote Sensing. 2017; 9(8):861. https://doi.org/10.3390/rs9080861
Chicago/Turabian StyleLobo, Felipe De Lucia, Maycira Costa, Evlyn Márcia Leão De Moraes Novo, and Kevin Telmer. 2017. "Effects of Small-Scale Gold Mining Tailings on the Underwater Light Field in the Tapajós River Basin, Brazilian Amazon" Remote Sensing 9, no. 8: 861. https://doi.org/10.3390/rs9080861
APA StyleLobo, F. D. L., Costa, M., Novo, E. M. L. D. M., & Telmer, K. (2017). Effects of Small-Scale Gold Mining Tailings on the Underwater Light Field in the Tapajós River Basin, Brazilian Amazon. Remote Sensing, 9(8), 861. https://doi.org/10.3390/rs9080861