Mapping and Modelling Spatial Variation in Soil Salinity in the Al Hassa Oasis Based on Remote Sensing Indicators and Regression Techniques
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Area
2.2. Field Sampling
2.3. Satellite Data Acquisition and Processing
2.4. Data Analysis, Model Generation and Selection
2.5. Model Validation
3. Results
3.1. Data Analysis
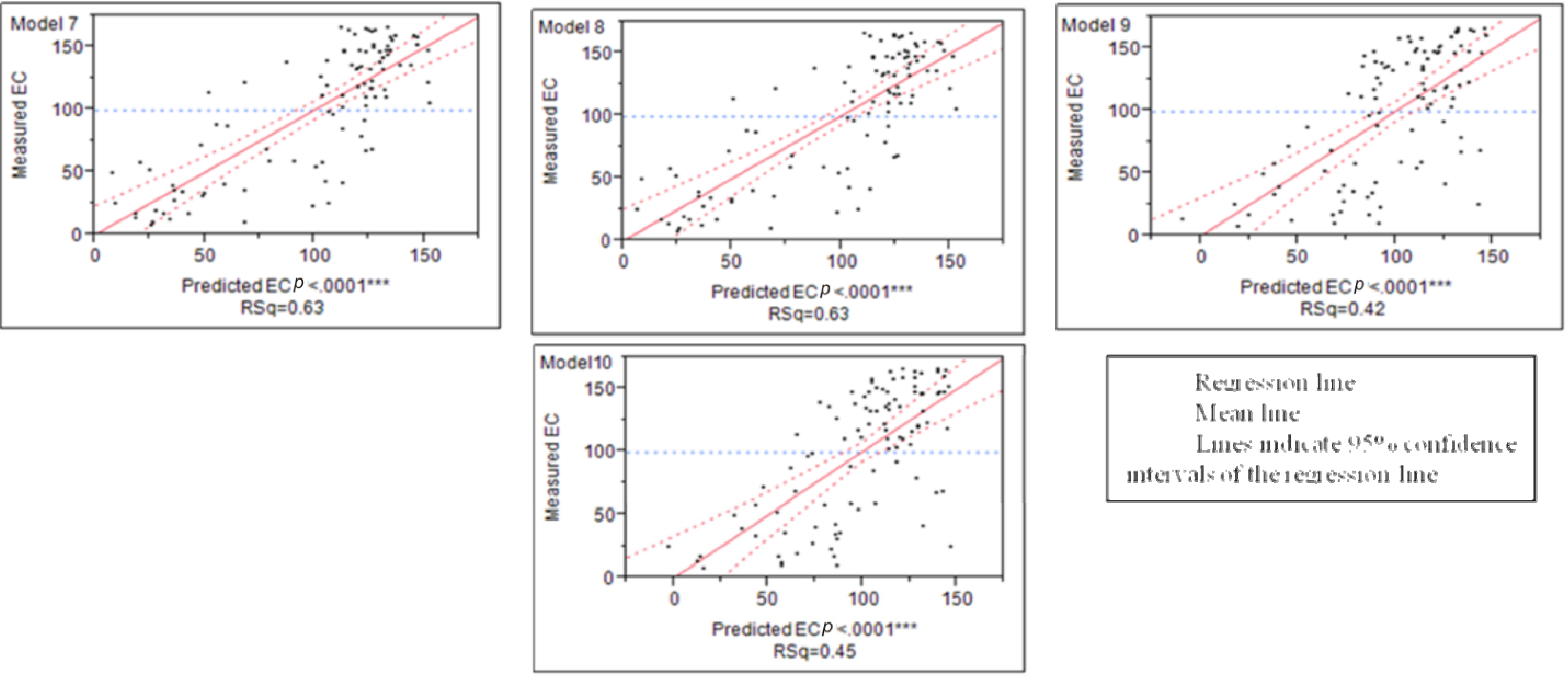
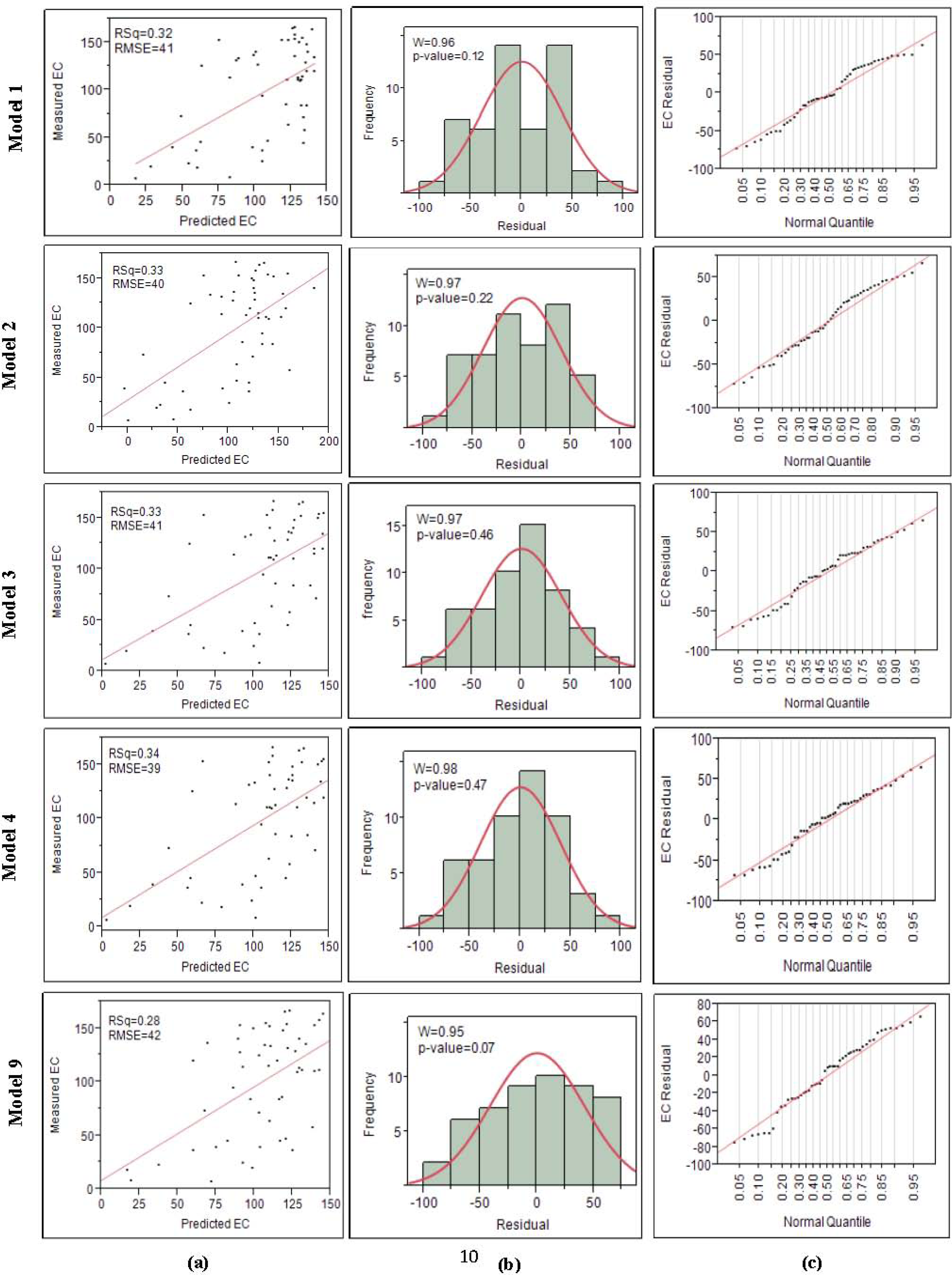
3.2. Models Development and Valuations
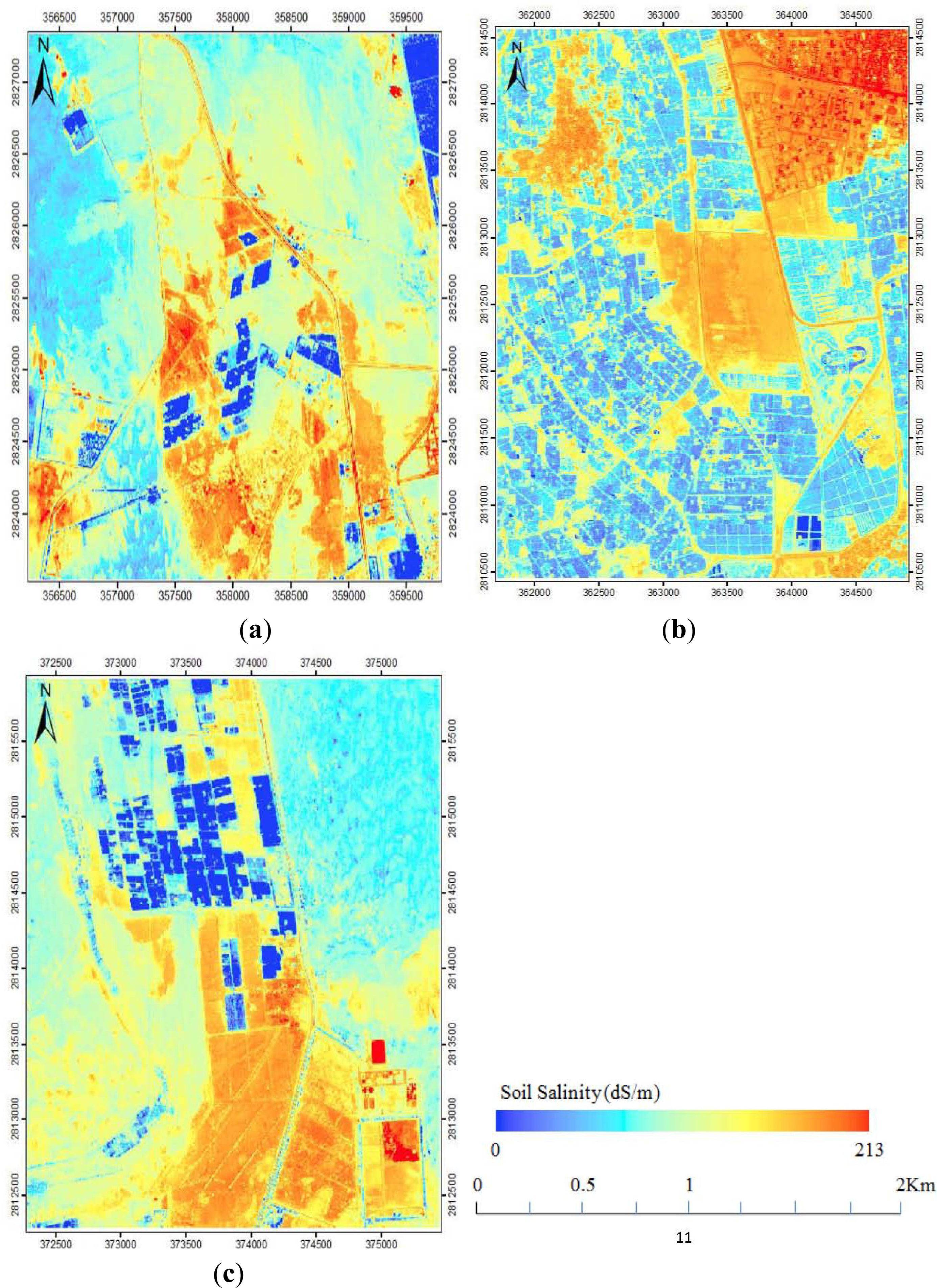
3.3. Spatial Variation in Soil Salinity Maps
4. Discussion
4.1. The Developed Regressions Models
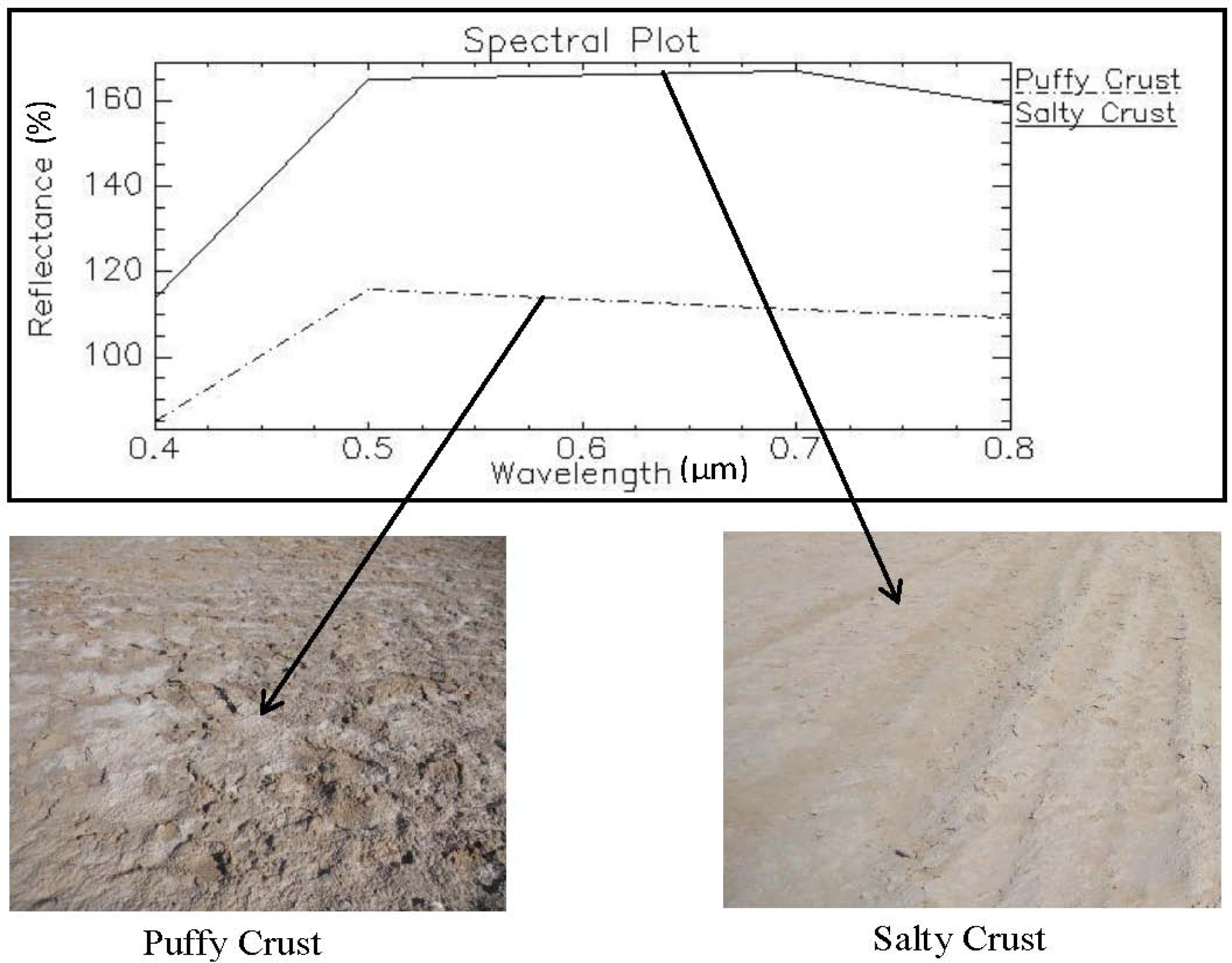
4.2. Mapping Spatial Variation in Soil Salinity
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Tanji, K. Salinity in the Soil Environment. In Salinity: Environment-Plants-Molecules; Lauchli, A., Luttge, U., Eds.; Kluwer Academic Publisher: Dordrecht, The Netherlands, 2004; pp. 21–51. [Google Scholar]
- Jordán, M.; Navarro-Pedreño, J.; García-Sánchez, E.; Mateu, J.; Juan, P. Spatial dynamics of soil salinity under arid and semi-arid conditions: Geological and environmental implications. Environ. Geol 2004, 45, 448–456. [Google Scholar]
- Douaik, A.; Meirvenne, M.; Toth, T. Stochastic Approaches for Space–Time Modeling and Interpolation of Soil Salinity. In Remote Sensing of Soil Salinization: Impact on Land Management; Metternicht, G., Zinck, J.A., Eds.; CRC Press: Boca Raton, FL, USA, 2008; pp. 273–289. [Google Scholar]
- Ben-Dor, E.; Goldshleger, N.; Eshel Mor, V. Combined Active and Passive Remote Sensing Methods for Assessing Soil Salinity: A Case Study from Jezre’el Valley, Northern Israel. In Remote Sensing of Soil Salinization: Impact on Land Management; Metternicht, G., Zinck, J.A., Eds.; CRC Press: Boca Raton, FL, USA, 2008; pp. 236–253. [Google Scholar]
- Goldshleger, N.; Chudnovsky, A.; Ben-Binyamin, R. Predicting salinity in tomato using soil reflectance spectra. Int. J. Remote Sens 2013, 34, 6079–6093. [Google Scholar]
- Metternicht, G.; Zinck, A. Remote Sensing of Soil Salinization: Impact on Land Management; CRC Press: Boca Raton, FL, USA, 2008; p. 377. [Google Scholar]
- Farifteh, J.; van der Meer, F.; Atzberger, C.; Carranza, E. Quantitative analysis of salt-affected soil reflectance spectra: A comparison of two adaptive methods (PLSR and ANN). Remote Sens. Environ 2007, 110, 59–78. [Google Scholar]
- Akramkhanov, A.; Vlek, P. The assessment of spatial distribution of soil salinity risk using neural network. Environ. Monit. Assess 2012, 184, 2475–2485. [Google Scholar]
- Patel, R.; Prasher, S.; God, P.; Bassi, R. Soil salinity prediction using artificial neural networks. J. Am. Water Resour. Assoc 2002, 38, 91–100. [Google Scholar]
- Fethi, B.; Magnus, P.; Ronny, B.; Akissa, B. Estimating soil salinity over a shallow saline water table in semiarid Tunisia. Open Hydrol. J 2010, 4, 91–101. [Google Scholar]
- Taghizadeh-Mehrjardi, R.; Minasny, B.; Sarmadian, F.; Malone, B. Digital mapping of soil salinity in Ardakan region, central Iran. Geoderma 2014, 213, 15–28. [Google Scholar]
- Tóth, T.; Pasztor, L.; Kabos, S.; Kuti, L. Statistical prediction of the presence of salt-affected soils by using digitalized hydrogeological maps. Arid Land Res. Manag 2002, 16, 55–68. [Google Scholar]
- Malins, D.; Metternicht, G. Assessing the spatial extent of dryland salinity through fuzzy modeling. Ecol. Model 2006, 193, 387–411. [Google Scholar]
- Douaik, A.; van Meirvenne, M.; Toth, T.; Serre, M. Space-time mapping of soil salinity using probabilistic bayesian maximum entropy. Stoch. Environ. Res. Risk Assess 2004, 18, 219–227. [Google Scholar]
- Triantafilis, J.; Odeh, I.; McBratney, A. Five geostatistical models to predict soil salinity from electromagnetic induction data across irrigated cotton. Soil Sci. Soc. Am. J 2001, 65, 869–878. [Google Scholar]
- Tajgardan, T.; Ayoubi, S.; Shataee, S.; Sahrawat, K.L.; Gorgan, I. Soil surface salinity prediction using ASTER data: Comparing statistical and geostatistical models. Aust. J. Basic Appl. Sci 2010, 4, 457–467. [Google Scholar]
- Eldeiry, A.A.; García, L.A. Using Deterministic and Geostatistical Techniques to Estimate Soil Salinity at the Sub-Basin Scale and the Field Scale. Proceedings of the 31th Annual Hydrology Days, Fort Collins, CO, USA, 21–23 March 2011.
- Douaoui, A.E.K.; Nicolas, H.; Walter, C. Detecting salinity hazards within a semiarid context by means of combining soil and remote-sensing data. Geoderma 2006, 134, 217–230. [Google Scholar]
- Eldeiry, A.A.; Garcia, L.A. Detecting soil salinity in alfalfa fields using spatial modeling and remote sensing. Soil Sci. Soc. Am. J 2008, 72, 201–211. [Google Scholar]
- Judkins, G.; Myint, S. Spatial variation of soil salinity in the Mexicali valley, Mexico: Application of a practical method for agricultural monitoring. Environ. Manag 2012, 50, 478–489. [Google Scholar]
- Fan, X.; Pedroli, B.; Liu, G.; Liu, Q.; Liu, H.; Shu, L. Soil salinity development in the yellow river delta in relation to groundwater dynamics. Land Degrad. Dev 2012, 23, 175–189. [Google Scholar]
- McBratney, A.; Santos, M.d.L.M.; Minasny, B. On digital soil mapping. Geoderma 2003, 117, 3–52. [Google Scholar]
- Scull, P.; Franklin, J.; Chadwick, O.; McArthur, D. Predictive soil mapping: A review. Progr. Phys. Geogr 2003, 27, 171–197. [Google Scholar]
- Lesch, S.M.; Strauss, D.J.; Rhoades, J.D. Spatial prediction of soil salinity using electromagnetic induction techniques: 1. Statistical prediction models: A comparison of multiple linear regression and cokriging. Water Resour. Res 1995, 31, 373–386. [Google Scholar]
- Yonghua, Q.; Siongb, J.; Xudonga, L. A Partial Least Square Regression Method to Quantitatively Retrieve Soil Salinity using Hyper-Spectral Reflectance Data. Proceedings of the SPIE 7147, Geoinformatics 2008 and Joint Conference on GIS and Built Environment: Classification of Remote Sensing Images, Guangzhou, China, 31 October 2008.
- Wang, H.; Wang, J.; Liu, G. Spatial Regression Analysis on the Variation of Soil Salinity in the Yellow River Delta. Proceedings of the SPIE 6753, Geoinformatics 2007: Geospatial Information Science, Nanjing, China, 10 June 2007.
- Shrestha, R. Relating soil electrical conductivity to remote sensing and other soil properties for assessing soil salinity in northeast Thailand. Land Degrad. Dev 2006, 17, 677–689. [Google Scholar]
- Pakparvar, M.; Gabriels, D.; Aarabi, K.; Edraki, M.; Raes, D.; Cornelis, W. Incorporating legacy soil data to minimize errors in salinity change detection: A case study of Darab Plain, Iran. Int. J. Remote Sens 2012, 33, 6215–6238. [Google Scholar]
- Noroozi, A.A.; Homaee, M.; Farshad, A. Integrated application of remote sensing and spatial statistical models to the identification of soil salinity: A case study from Garmsar Plain, Iran. Environ. Sci 2012, 9, 59–74. [Google Scholar]
- Mehrjardi, R.T.; Mahmoodi, S.H.; Taze, M.; Sahebjalal, E. Accuracy assessment of soil salinity map in Yazd-Ardakan Plain, Central Iran, based on Landsat ETM+ imagery. Am.-Eurasian J. Agric. Environ. Sci 2008, 3, 708–712. [Google Scholar]
- Fernandez-Buces, N.; Siebe, C.; Cram, S.; Palacio, J. Mapping soil salinity using a combined spectral response index for bare soil and vegetation: A case study in the former lake Texcoco, Mexico. J. Arid Environ 2006, 65, 644–667. [Google Scholar]
- Eldeiry, A.A.; Garcia, L.A. Spatial modeling using remote sensing, GIS, and field data to assess crop yield and soil salinity. Hydrol. Days 2004, 7, 55–66. [Google Scholar]
- Afework, M. Analysis and Mapping of Soil Salinity Levels in Metehara Sugarcane Estate Irrigation Farm Using Different Models. Addis Ababa University, Ethiopia, Addis Ababa, 2009. [Google Scholar]
- Weng, Y.; Gong, P.; Zhu, Z. Reflectance spectroscopy for the assessment of soil salt content in soils of the Yellow River Delta of China. Int. J. Remote Sens 2008, 29, 5511–5531. [Google Scholar]
- Shamsi, F.R.S.; Sanaz, Z.; Abtahi, A.S. Soil salinity characteristics using moderate resolution imaging spectroradiometer (MODIS) images and statistical analysis. Arch. Agron. Soil Sci 2013, 59, 471–489. [Google Scholar]
- Tajgardan, T.; Shataee, S.; Ayoubi, S. In Spatial Prediction of Soil Salinity in the Arid Zones Using ASTER Data, Case study: North of Ag Ghala, Golestan Province, Iran. Proceedings of Asian Conference on Remote Sensing (ACRS), Kuala Lumpur, Malaysia, 12–16 November 2007.
- Bouaziz, M.; Matschullat, J.; Gloaguen, R. Improved remote sensing detection of soil salinity from a semi-arid climate in Northeast Brazil. Comptes Rendus Geosci 2011, 343, 795–803. [Google Scholar]
- Al-Abdoulhadi, I.A.; Dinar, H.A.; Ebert, G.; Bttner, C. Effect of salinity on leaf growth, leaf injury and biomass production in date palm (Phoenix dactylifera L.) cultivars. Indian J. Sci. Technol 2011, 4, 1542–1546. [Google Scholar]
- Al-Dakheel, Y.; Massoud, M. Towards Sustainable Development for Groundwater in Al-Hassa and the Role of Geographic Information Systems. Proceeidngs of the The 2nd International Conference on Water Resources & Arid Environment, Riyadh, Saudi Arabia, 26–29 November 2006.
- Hussain, G.; Al-Zarah, A.I.; Latif, M.S. Influence of groundwater irrigation on chemical properties of soils in the vicinity of wastewater drainage canals in Al-Alisa Oasis. Res. J. Environ. Toxicol. (RJET) 2012, 7, 1–17. [Google Scholar]
- Al-Naeem, A.A. Evaluation of groundwater of al-hassa oasis, eastern region Saudi Arabia. Res. J. Environ. Sci 2011, 5, 624–642. [Google Scholar]
- Al Tokhais, A.S.; Rausch, R. In The Hydrogeology of Al Hassa Springs. Proceedings the 3rd International Conference on Water Resources and Arid Environments and the 1 Arab Water Forum, st Riyadh, Saudi Arabia, 16–19 November 2008; pp. 16–19.
- AI-Barrak, S.A. Characteristics of some soils under date palm in AI-Hassa eastern oasis, Saudi Arabia. J. King Saud Univ. Agric. Sci 1990, 2, 115–130. [Google Scholar]
- Elprince, A.M. Model for the soil solution composition of an oasis. Soil Sci. Soc. Am. J 1985, 49, 1121–1128. [Google Scholar]
- Bouaziz, M.; Gloaguena, R.; Samirb, B. Remote mapping of susceptible areas to soil salinity, based on hyperspectral data and geochemical, in the Southern Part of Tunisia. Proc. SPIE 2011, 8174. [Google Scholar] [CrossRef]
- Richards, L. Determination of the Properties of Saline and Alkali Soils. In Diagnosis and Improvement of Saline and Alkali Soils, Agriculture Handbook No. 60; US Regional Salinity Laboratory: Riverside, CA, USA, 1954; pp. 7–33. [Google Scholar]
- Dial, G.; Bowen, H.; Gerlach, F.; Grodecki, J.; Oleszczuk, R. IKONOS satellite, imagery, and products. Remote Sens. Environ 2003, 88, 23–36. [Google Scholar]
- Chavez, P.S., Jr. Image-based atmospheric corrections-revisited and improved. Photogramm. Eng. Remote Sens 1996, 62, 1025–1035. [Google Scholar]
- Tripathi, N.K.; Rai, B.K.; Dwivedi, P. Spatial modelling of soil alkalinity in GIS environment using IRS data. Proceedings of the 18th Asian Conference in Remote Sensing, ACRS Kuala Lumpur, Malaysia, 20–25 October 1997; pp. 81–86.
- Royston, P.; Sauerbrei, W. Multivariable Model-Building: A Pragmatic Approach to Regression Anaylsis Based on Fractional Polynomials for Modelling Continuous Variables; John Wiley & Sons: Chichester, UK, 2008; p. 322. [Google Scholar]
- Moriasi, D.; Arnold, J.; van Liew, M.; Bingner, R.; Harmel, R.; Veith, T. Model evaluation guidelines for systematic quantification of accuracy in watershed simulations. Trans. ASABE 2007, 50, 885–900. [Google Scholar]
- Field, A.; Miles, J.; Field, Z. Discovering Statistics Using R; SAGE Publications: London, UK, 2012; p. 992. [Google Scholar]
- Gao, J. Digital Analysis of Remotely Sensed Imagery, 1st ed; McGraw Hill Professional: New York, NY, USA, 2008; p. 674. [Google Scholar]
- Odeh, I.O.; Onus, A. Spatial analysis of soil salinity and soil structural stability in a semiarid region of New South Wales, Australia. Environ. Manag 2008, 42, 265–278. [Google Scholar]
- Iqbal, F. Detection of salt affected soil in rice-wheat area using satellite image. Afr. J. Agric. Res 2011, 6, 4973–4982. [Google Scholar]
- Khan, N.M.; Rastoskuev, V.V.; Sato, Y.; Shiozawa, S. Assessment of hydrosaline land degradation by using a simple approach of remote sensing indicators. Agric. Water Manag 2005, 77, 96–109. [Google Scholar]
- Lobell, D.; Lesch, S.; Corwin, D.; Ulmer, M.; Anderson, K.; Potts, D.; Doolittle, J.; Matos, M.; Baltes, M. Regional-scale assessment of soil salinity in the Red River Valley using multi-year MODIS EVI and NDVI. J. Environ. Qual 2010, 39, 35–41. [Google Scholar]
- Hamzeh, S.; Naseri, A.A.; AlaviPanah, S.K.; Mojaradi, B.; Bartholomeus, H.M.; Clevers, J.; Behzad, M. Estimating salinity stress in sugarcane fields with spaceborne hyperspectral vegetation indices. Int. J. Appl. Earth Obs. Geoinformation 2012, 21, 282–290. [Google Scholar]
- Naumann, J.C.; Young, D.R.; Anderson, J.E. Spatial variations in salinity stress across a coastal landscape using vegetation indices derived from hyperspectral imagery. Plant Ecol 2009, 202, 285–297. [Google Scholar]
- Arasteh, P.D. Soil Salinity Change Detection in Irrigated Area Under Gazvin Plain Irrigation Network Using Satellite Imagery. Proceedings of the 9th International Drainage Symposium, Québec City, QC, Canada, 13–16 June 2010; pp. 1–9.
- Mariappan, V.E.N. Soil salinity assessment using geospatial technology, perspectives, approaches and sctrategies. Indian Cartogr 2010, 30, 25–30. [Google Scholar]
- Metternicht, G.; Zinck, J.A. Spectral Behavior of Salt Types. In Remote Sensing of Soil Salinization: Impact on Land Management; Metternicht, G., Zinck, J.A., Eds.; CRC Press: Boca Raton, FL, USA, 2008; pp. 21–36. [Google Scholar]
- Goldshleger, N.; Livne, I.; Chudnovsky, A.; Ben-Dor, E. New results in integrating passive and active remote sensing methods to assess soil salinity: A case study from Jezre’el Valley, Israel. Soil Sci 2012, 177, 392–401. [Google Scholar]
- Panah, S.K.A.; Goossens, R.; Matinfar, H.R.; Mohamadi, H.; Ghadiri, M.; Irannegad, H.; Asl, M.A. The efficiency of Landsat TM and ETM+ thermal data for extracting soil information in arid regions. J. Agric. Sci. Technol 2008, 10, 439–460. [Google Scholar]
- De Jong, S.M.; Addink, E.A.; van Beek, L.P.H.; Duijsings, D. Physical characterization, spectral response and remotely sensed mapping of Mediterranean soil surface crusts. Catena 2011, 86, 24–35. [Google Scholar]
- Ben-Dor, E.; Goldlshleger, N.; Benyamini, Y.; Agassi, M.R.; Blumberg, D.G. The spectral reflectance properties of soil structural crusts in the 1.2-to 2.5-μm spectral region. Soil Sci. Soc. Am. J 2003, 67, 289–299. [Google Scholar]
- Agassi, M.; Morin, J.; Shainberg, I. Effect of raindrop impact energy and water salinity on infiltration rates of sodic soils. Soil Sci. Soc. Am. J 1985, 49, 186–190. [Google Scholar]
- Metternicht, G.I.; Zinck, J.A. Remote sensing of soil salinity: Potentials and constraints. Remote Sens. Environ 2003, 85, 1–20. [Google Scholar]
- Elnaggar, A.A.; Noller, J.S. Application of remote-sensing data and decision-tree analysis to mapping salt-affected soils over large areas. Remote Sens 2010, 2, 151–165. [Google Scholar]
- Goldshleger, N.; Ben-Dor, E.; Benyamini, Y.; Agassi, M. Soil reflectance as a tool for assessing physical crust arrangement of four typical soils in Israel. Soil Sci 2004, 169, 677–687. [Google Scholar]
- Metternicht, G.; Zinck, J. Spatial discrimination of salt-and sodium-affected soil surfaces. Int. J. Remote Sens 1997, 18, 2571–2586. [Google Scholar]
- Schmid, T.; Koch, M.; Gumuzzio, J. Applications of Hyper-Spectral Imagery to Soil Salinity Mapping. In Remote Sensing of Soil Salinization: Impact on Land Management; Metternicht, G., Zinck, J., Eds.; CRC Press: Boca Raton, FL, USA, 2009; pp. 113–140. [Google Scholar]
- Bilgili, A.V. Spatial assessment of soil salinity in the Harran Plain using multiple kriging techniques. Environ. Monit. Assess 2013, 185, 777–795. [Google Scholar]
- Keddy, P.A. Wetland Ecology: Principles and Conservation; Cambridge University Press: Cambridge, UK, 2010; p. 497. [Google Scholar]
- Ashraf, M.; Öztürk, M.A.; Athar, H.R. Salinity and Water Stress: Improving Crop Efficiency; Springer: Dordrecht, The Netherlands, 2009; p. 260. [Google Scholar]
- Jardine, A.; Speldewinde, P.; Carver, S.; Weinstein, P. Dryland salinity and ecosystem distress syndrome: Human health implications. EcoHealth 2007, 4, 10–17. [Google Scholar]
- Hussain, N.; Al-Rawahy, A.S.; Rabee, J.; Al-Amri, M. Causes, origin, genesis and extent of soil salinity in the Sultanate of Oman. Pak. J. Agric. Sci 2006, 43, 1–2. [Google Scholar]
- Thiruchelvam, S.; Pathmarajah, S. An Economic Analysis of Salinity Problems in the Mahaweli River System H Irrigation Scheme in Sri Lanka; Economy and Environment Program for Southeast Asia (EEPSEA): Ottawa, Canada, 1999; p. 39. [Google Scholar]
- Cetin, M.; Kirda, C. Spatial and temporal changes of soil salinity in a cotton field irrigated with low-quality water. J. Hydrol 2003, 272, 238–249. [Google Scholar]
- Zheng, Z.; Zhang, F.; Ma, F.; Chai, X.; Zhu, Z.; Shi, J.; Zhang, S. Spatiotemporal changes in soil salinity in a drip-irrigated field. Geoderma 2009, 149, 243–248. [Google Scholar]
- Sakadevan, K.; Nguyen, M.-L. Extent, impact, and response to soil and water salinity in arid and semiarid regions. Adv. Agron 2010, 109, 55–74. [Google Scholar]
- Alhammadi, M.; Glenn, E. Detecting date palm trees health and vegetation greenness change on the eastern coast of the United Arab Emirates using SAVI. Int. J. Remote Sens 2008, 29, 1745–1765. [Google Scholar]
- Ramoliya, P.; Pandey, A. Soil salinity and water status affect growth of Phoenix dactylifera seedlings. N. Z. J. Crop Hortic. Sci 2003, 31, 345–353. [Google Scholar]
- Alhammadi, M.S.; Edward, G.P. Effect of salinity on growth of twelve cultivars of the United Arab Emirates date palm. Commun. Soil Sci. Plant Anal 2009, 40, 2372–2388. [Google Scholar]
- Hussein, A.; El-Desouki, M.; El-Kased, F.; Nour, G.; Abd-El-Hamid, N. Effect of salinity on date palm seeds germination and early seedling growth. J. Agric. Sci. Mansoura Univ 1993, 18, 1306–1314. [Google Scholar]
- Verma, K.; Saxena, R.; Barthwal, A.; Deshmukh, S. Remote sensing technique for mapping salt affected soils. Int. J. Remote Sens 1994, 15, 1901–1914. [Google Scholar]
- Alavipanah, S.K.; Goossens, R. Relationship between the Landsat TM, MSS data and soil salinity. J. Agric. Sci. Technol 2001, 3, 101–111. [Google Scholar]
- Weng, Y.; Gong, P.; Zhu, Z. Soil salt content estimation in the Yellow River delta with satellite hyperspectral data. Can. J. Remote Sens 2008, 34, 259–270. [Google Scholar]
- Lu, N.; Zhang, Z.; Gao, Y. Recognition and Mapping of Soil Salinization in Arid Environment with Hyperspectral Data. Proceedings of the IGARSS’05. IEEE International Geoscience and Remote Sensing Symposium, Seoul, Korea, 25–29 July 2005; pp. 4520–4523.
- Ben-Dor, E.; Patkin, K.; Banin, A.; Karnieli, A. Mapping of several soil properties using DAIS-7915 hyperspectral scanner data-a case study over clayey soils in Israel. Int. J. Remote Sens 2002, 23, 1043–1062. [Google Scholar]
- Zhang, T.T.; Zeng, S.L.; Gao, Y.; Ouyang, Z.T.; Li, B.; Fang, C.M.; Zhao, B. Using hyperspectral vegetation indices as a proxy to monitor soil salinity. Ecol. Indic 2011, 11, 1552–1562. [Google Scholar]
- Hamzeh, S.; Naseria, A.A.; Panah, S.K.A.; Mojaradic, B.; Bartholomeus, H.M.; Herold, M. Mapping salinity stress in sugarcane fields with hyperspecteral satellite imagery. Proc. SPIE 2012. [Google Scholar] [CrossRef]


| Function Name | Equation | Equation Number |
|---|---|---|
| Coefficient of determination | ||
| Root mean square error | ||
| Mean | Max | Min | SD | CV (%) | |
|---|---|---|---|---|---|
| EC | 73.37 | 202 | 1.43 | 62.65 | 85.39 |
| Variables | B1 | B2 | B3 | B4 | SI |
|---|---|---|---|---|---|
| EC | 0.41 *** | 0.42 *** | 0.45 *** | 0.06 ns | 0.70 *** |
| Model | Variable | Regression Coefficient | Standard Error | p-Value |
|---|---|---|---|---|
| 1 | Intercept | −239.49 | 29.82 | <0.0001 *** |
| SI | 3.52 | 0.31 | <0.0001 *** | |
| R = 0.76***, R2 = 0.58 *** | ||||
| 2 | Intercept | −205.25 | 29.95 | <0.0001 *** |
| SI | −1.77 | 0.511 | 0.0008 *** | |
| B1 | 4.83 | 0.48 | <0.0001 *** | |
| R = 0.79***, R2 = 0.62 *** | ||||
| 3 | Intercept | −239.60 | 28.31 | <0.0001 *** |
| SI | 4.83 | 0.48 | <0.0001 *** | |
| B2 | −1.011254 | 0.30 | 0.001 ** | |
| R = 0.79***, R2 = 0.62*** | ||||
| 4 | Intercept | −269.13 | 29.99 | <0.0001 *** |
| SI | 4.87 | 0.52 | <0.0001 *** | |
| B3 | −0.83 | 0.26 | 0.002 ** | |
| R = 0.81***, R2 = 0.65*** | ||||
| 5 | Intercept | −193.18 | 83.48 | 0.02 * |
| SI | 4.81 | 0.49 | <0.0001 *** | |
| B1 | −2.39 | 4.04 | 0.5559 | |
| B2 | 0.36 | 2.35 | 0.8771 | |
| R = 0.79***, R2 = 0.62*** | ||||
| 6 | Intercept | −136.12 | 93.24 | 0.1476 |
| SI | 4.64 | 0.54 | <0.0001 *** | |
| B1 | −3.59 | 2.38 | 0.1356 | |
| B3 | 0.95 | 1.21 | 0.4355 | |
| R = 0.79***, R2 = 0.63*** | ||||
| 7 | Intercept | −115.70 | 84.33 | 0.1734 |
| SI | 4.31 | 0.59 | <0.0001 *** | |
| B2 | −4.97 | 2.56 | 0.0551 | |
| B3 | 3.49 | 2.24 | 0.1226 | |
| R = 0.79***, R2 = 0.63 *** | ||||
| 8 | Intercept | −130.65 | 93.04 | 0.1636 |
| SI | 4.23 | 0.62 | <0.0001 *** | |
| B1 | 1.93 | 4.95 | 0.6983 | |
| B2 | −6.80 | 5.36 | 0.2077 | |
| B3 | 4.12 | 2.78 | 0.1412 | |
| R = 0.79 ***, R2 = 0.63 *** | ||||
| 9 | Intercept | 320.95 | 74.94 | <0.0001 *** |
| B2 | −14.25 | 2.78 | <0.0001 *** | |
| B3 | 12.914 | 2.3 | <0.0001 *** | |
| R = 0.65 ***, R2 = 0.42 *** | ||||
| 10 | Intercept | 165.52 | 100.03 | 0.1013 |
| B1 | 13 | 5.69 | 0.02 * | |
| B2 | −25.44 | 5.61 | <0.0001 *** | |
| B3 | 16.02 | 2.62 | <0.0001 *** | |
| R = 0.67 ***, R2 = 0.45 *** | ||||
© 2014 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Allbed, A.; Kumar, L.; Sinha, P. Mapping and Modelling Spatial Variation in Soil Salinity in the Al Hassa Oasis Based on Remote Sensing Indicators and Regression Techniques. Remote Sens. 2014, 6, 1137-1157. https://doi.org/10.3390/rs6021137
Allbed A, Kumar L, Sinha P. Mapping and Modelling Spatial Variation in Soil Salinity in the Al Hassa Oasis Based on Remote Sensing Indicators and Regression Techniques. Remote Sensing. 2014; 6(2):1137-1157. https://doi.org/10.3390/rs6021137
Chicago/Turabian StyleAllbed, Amal, Lalit Kumar, and Priyakant Sinha. 2014. "Mapping and Modelling Spatial Variation in Soil Salinity in the Al Hassa Oasis Based on Remote Sensing Indicators and Regression Techniques" Remote Sensing 6, no. 2: 1137-1157. https://doi.org/10.3390/rs6021137
APA StyleAllbed, A., Kumar, L., & Sinha, P. (2014). Mapping and Modelling Spatial Variation in Soil Salinity in the Al Hassa Oasis Based on Remote Sensing Indicators and Regression Techniques. Remote Sensing, 6(2), 1137-1157. https://doi.org/10.3390/rs6021137





