Interannual Variation in Phytoplankton Primary Production at A Global Scale
Abstract
:1. Introduction
2. Results and Discussion
2.1. Climatology of Primary Production and Comparison with VGPM
2.2. Interannual Variability
3. Experimental Section
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Behrenfeld, M.J.; Randerson, J.T.; McClain, C.R.; Feldman, G.C.; Los, S.O.; Tucker, C.J.; Falkowski, P.G.; Field, C.B.; Frouin, R.; Esaias, W.E. Biospheric primary production during an enso transition. Science 2001, 291, 2594–2597. [Google Scholar]
- Ciotti, A.M.; Lewis, M.R.; Cullen, J.J. Assessment of the relationships between dominant cell size in natural phytoplankton communities and the spectral shape of the absorption coefficient. Limnol. Oceanogr 2002, 47, 404–417. [Google Scholar]
- Mouw, C.B.; Yoder, J.A. Optical determination of phytoplankton size composition from global seawifs imagery. J. Geophys. Res 2006, 115, C12018. [Google Scholar]
- Alvain, S.; Moulin, C.; Dandonneau, Y.; Bréon, F.M. Remote sensing of phytoplankton groups in case 1 waters from global seawifs imagery. Deep Sea Res. Part I: Oceanogr. Res. Papers 2005, 52, 1989–2004. [Google Scholar]
- Uitz, J.; Claustre, H.; Morel, A.; Hooker, S.B. Vertical distribution of phytoplankton communities in open ocean: An assessment based on surface chlorophyll. J. Geophys. Res 2006, 111, C08005. [Google Scholar]
- Aiken, J.; Fishwick, J.R.; Lavender, S.; Barlow, R.; Moore, G.F.; Sessions, H.; Bernard, S.; Ras, J.; Hardman-Mountford, N.J. Validation of meris reflectance and chlorophyll during the bencal cruise october 2002: Preliminary validation of new demonstration products for phytoplankton functional types and photosynthetic parameters. Int. J. Remote Sens 2007, 28, 497–516. [Google Scholar]
- Dutkiewicz, S.; Follows, M.J.; Bragg, J.G. Modeling the coupling of ocean ecology and biogeochemistry. Glob. Biogeochem. Cy 2009, 23, GB4017. [Google Scholar]
- Le Quere, C.; Harrison, S.P.; Colin Prentice, I.; Buitenhuis, E.T.; Aumont, O.; Bopp, L.; Claustre, H.; Cotrim Da Cunha, L.; Geider, R.; Giraud, X.; et al. Ecosystem dynamics based on plankton functional types for global ocean biogeochemistry models. Glob. Chang. Biol 2005, 11, 2016–2040. [Google Scholar]
- Moore, J.K.; Doney, S.C.; Lindsay, K. Upper ocean ecosystem dynamics and iron cycling in a global three-dimensional model. Glob. Biogeochem. Cy 2004, 18, GB4028. [Google Scholar]
- Dunne, J.P.; Armstrong, R.A.; Gnanadesikan, A.; Sarmiento, J.L.; Slater, R.D. Empirical and mechanistic models for the particle export ratio. Glob. Biogeochem. Cy 2005, 19, GB4026. [Google Scholar]
- Doney, S.C.; Ducklow, H.W. A decade of synthesis and modeling in the us joint global ocean flux study. Deep Sea Res. Part II: Top. Stud. Oceanogr 2006, 53, 451–458. [Google Scholar]
- Hirata, T.; Hardman-Mountford, N.J.; Barlow, R.; Lamont, T.; Brewin, R.; Smyth, T.; Aiken, J. An inherent optical property approach to the estimation of size-specific photosynthetic rates in eastern boundary upwelling zones from satellite ocean colour: An initial assessment. Prog. Oceanogr 2009, 83, 393–397. [Google Scholar]
- Dandonneau, Y.; Deschamps, P.-Y.; Nicolas, J.-M.; Loisel, H.; Blanchot, J.; Montel, Y.; Thieuleux, F.; Bécu, G. Seasonal and interannual variability of ocean color and composition of phytoplankton communities in the north atlantic, equatorial pacific and south pacific. Deep Sea Res. Part II: Top. Stud. Oceanogr 2004, 51, 303–318. [Google Scholar]
- Masotti, I.; Moulin, C.; Alvain, S.; Bopp, L.; Tagliabue, A.; Antoine, D. Large-scale shifts in phytoplankton groups in the equatorial pacific during enso cycles. Biogeosciences 2011, 8, 539–550. [Google Scholar]
- Rousseaux, C.S.; Gregg, W.W. Climate variability and phytoplankton composition in the pacific ocean. J. Geophys. Res 2012, 117, C10006. [Google Scholar]
- Martinez, E.; Antoine, D.; D’Ortenzio, F.; Gentili, B. Climate-driven basin-scale decadal oscillations of oceanic phytoplankton. Science 2009, 326, 1253–1256. [Google Scholar]
- Uitz, J.; Claustre, H.; Gentili, B.; Stramski, D. Phytoplankton class-specific primary production in the world’s oceans: Seasonal and interannual variability from satellite observations. Glob. Biogeochem. Cy 2010, 24, GB3016. [Google Scholar]
- Kameda, T.; Ishizaka, J. Size-fractionated primary production estimated by a two-phytoplankton community model applicable to ocean color remote sensing. J. Oceanogr 2005, 61, 663–672. [Google Scholar]
- Brewin, R.J.; Lavender, S.J.; Hardman-Mountford, N.J. Mapping size-specific phytoplankton primary production on a global scale. J. Maps 2010, 6, 448–462. [Google Scholar]
- Morel, A. Light and marine photosynthesis: A spectral model with geochemical and climatological implications. Prog. Oceanogr 1991, 26, 263–306. [Google Scholar]
- Friedrichs, M.A.M.; Carr, M.E.; Barber, R.T.; Scardi, M.; Antoine, D.; Armstrong, R.A.; Asanuma, I.; Behrenfeld, M.J.; Buitenhuis, E.T.; Chai, F.; et al. Assessing the uncertainties of model estimates of primary productivity in the tropical pacific ocean. J. Mar. Syst 2009, 76, 113–133. [Google Scholar]
- Carr, M.E.; Friedrichs, M.A.M.; Schmeltz, M.; Noguchi Aita, M.; Antoine, D.; Arrigo, K.R.; Asanuma, I.; Aumont, O.; Barber, R.; Behrenfeld, M.; et al. A comparison of global estimates of marine primary production from ocean color. Deep Sea Res. Part II: Top. Stud. Oceanogr 2006, 53, 741–770. [Google Scholar] [Green Version]
- Saba, V.S.; Friedrichs, M.A.M.; Carr, M.E.; Antoine, D.; Armstrong, R.A.; Asanuma, I.; Aumont, O.; Bates, N.R.; Behrenfeld, M.J.; Bennington, V.; et al. Challenges of modeling depth-integrated marine primary productivity over multiple decades: A case study at bats and hot. Glob. Biogeochem. Cy 2010, 24, GB3020. [Google Scholar]
- Behrenfeld, M.J.; Falkowski, P.G. Photosynthetic rates derived from satellite-based chlorophyll concentration. Limnol. Oceanogr 1997, 42, 1–20. [Google Scholar]
- Behrenfeld, M.J.; Boss, E.; Siegel, D.A.; Shea, D.M. Carbon-based ocean productivity and phytoplankton physiology from space. Glob. Biogeochem. Cy 2005, 19, 14. [Google Scholar]
- Field, C.B.; Behrenfeld, M.J.; Randerson, J.T.; Falkowski, P. Primary production of the biosphere: Integrating terrestrial and oceanic components. Science 1998, 281, 237. [Google Scholar]
- Gregg, W.W.; Casey, N.W. Sampling biases in modis and seawifs ocean chlorophyll data. Remote Sens. Environ 2007, 111, 25–35. [Google Scholar]
- Chavez, F.P.; Messié, M.; Pennington, J.T. Marine primary production in relation to climate variability and change. Annu. Rev. Mar. Sci 2011, 3, 227–260. [Google Scholar]
- Chavez, F.P.; Strutton, P.G.; Friederich, G.E.; Feely, R.A.; Feldman, G.C.; Foley, D.G.; McPhaden, M.J. Biological and chemical response of the equatorial pacific ocean to the 1997–98 el niño. Science 1999, 286, 2126–2131. [Google Scholar]
- Wang, X.; Christian, J.R.; Murtugudde, R.; Busalacchi, A.J. Ecosystem dynamics and export production in the central and eastern equatorial pacific: A modeling study of impact of ENSO. Geophys. Res. Lett 2005, 32, L02608. [Google Scholar]
- Strutton, P.G.; Chavez, F.P. Primary productivity in the equatorial pacific during the 1997–1998 el niño. J. Geophys. Res 2000, 105, 20089–26101. [Google Scholar]
- Feely, R.A.; Boutin, J.; Cosca, C.E.; Dandonneau, Y.; Etcheto, J.; Inoue, H.Y.; Ishii, M.; Quéré, C.L.; Mackey, D.J.; McPhaden, M.; et al. Seasonal and interannual variability of co2 in the equatorial pacific. Deep Sea Res. Part II: Top. Stud. Oceanogr 2002, 49, 2443–2469. [Google Scholar]
- Wolter, K.; Timlin, M.S. Measuring the strength of enso events: How does 1997/98 rank? Weather 1998, 53, 315–324. [Google Scholar]
- Matsumoto, K.; Furuya, K. Variations in phytoplankton dynamics and primary production associated with enso cycle in the western and central equatorial pacific during 1994–2003. J. Geophys. Res 2011, 116, C12042. [Google Scholar]
- Villanoy, C.L.; Cabrera, O.C.; Yniguez, A.; Camoying, M.; de Guzman, A.; David, L.T.; Flament, P. Monsoon-driven coastal upwelling off zamboanga peninsula, philippines. Oceanography 2011, 24, 156–165. [Google Scholar]
- Dave, A.C.; Lozier, M.S. Local stratification control of marine productivity in the subtropical north pacific. J. Geophys. Res 2010, 115, C12032. [Google Scholar]
- Kostadinov, T.S.; Siegel, D.A.; Maritorena, S. Global variability of phytoplankton functional types from space: Assessment via the particle size distribution. Biogeosci. Discuss 2010, 7, 4295–4340. [Google Scholar]
- Behrenfeld, M.J.; O’Malley, R.T.; Siegel, D.A.; McClain, C.R.; Sarmiento, J.L.; Feldman, G.C.; Milligan, A.J.; Falkowski, P.G.; Letelier, R.M.; Boss, E.S. Climate-driven trends in contemporary ocean productivity. Nature 2006, 444, 752–755. [Google Scholar]
- Dave, A.C.; Lozier, M.S. Examining the global record of interannual variability in stratification and marine productivity in the low-latitude and mid-latitude ocean. J. Geophys. Res 2013, 118, 3114–3127. [Google Scholar]
- Follows, M.J.; Dutkiewicz, S.W. Meteorological modulation of the north atlantic spring bloom. Deep Sea Res. Part II: Top. Stud. Oceanogr 2002, 49, 321–344. [Google Scholar]
- Henson, S.A.; Robinson, I.; Allen, J.T.; Waniek, J.J. Effect of meteorological conditions on interannual variability in timing and magnitude of the spring bloom in the irminger basin, north atlantic. Deep Sea Res. Part I: Oceanogr. Res. Papers 2006, 53, 1601–1615. [Google Scholar]
- Shutler, J.; Land, P.; Brown, C.; Findlay, H.; Donlon, C.; Medland, M.; Snooke, R.; Blackford, J. Coccolithophore surface distributions in the north atlantic and their modulation of the air-sea flux of CO2 from 10 years of satellite earth observation data. Biogeosciences 2013, 10, 2699–2709. [Google Scholar]
- Raitsos, D.E.; Lavender, S.J.; Pradhan, Y.; Tyrrell, T.; Reid, P.C.; Edwards, M. Coccolithophore bloom size variation in response to the regional environment of the subarctic north atlantic. Limnol. Oceanogr 2006, 51, 2122–2130. [Google Scholar]
- Lovenduski, N.S.; Gruber, N. Impact of the southern annular mode on southern ocean circulation and biology. Geophys. Res. Lett 2005, 32, L11603. [Google Scholar]
- Arrigo, K.R.; van Dijken, G.L.; Bushinsky, S. Primary production in the southern ocean, 1997–2006. J. Geophys. Res 2008, 113, L19603. [Google Scholar]
- Hurrell, J.W. Decadal trends in the north atlantic oscillation. Science 1995, 269, 676–679. [Google Scholar]
- Weyhenmeyer, G.A.; Blenckner, T.; Petterson, K. Changes of the plankton spring outburst related to the north atlantic oscillation. Limnol. Oceanogr 1999, 44, 1788–1792. [Google Scholar]
- Irigoien, X.; Harris, R.P.; Head, R.N.; Harbour, D. North atlantic oscillation and spring bloom phytoplankton composition in the english channel. J. Plankton Res 2000, 22, 2367–2371. [Google Scholar]
- Reid, P.C.; Edwards, M.; Hunt, H.G.; Warner, A.J. Phytoplankton change in the north atlantic. Nature 1998, 391, 546–546. [Google Scholar]
- Sameoto, D. Decadal changes in phytoplankton color index and selected calanoid copepods in continuous plankton recorder data from the scotian shelf. Can. J. Fish. Aquat. Sci 2001, 58, 749–761. [Google Scholar]
- Henson, S.; Lampitt, R.; Johns, D. Variability in phytoplankton community structure in response to the north atlantic oscillation and implications for organic carbon flux. Limnol. Oceanogr 2012, 57, 1591–1601. [Google Scholar]
- Bates, N.R. Interannual variability of oceanic co2 and biogeochemical properties in the western North Atlantic subtropical gyre. Deep Sea Res. Part II: Top. Stud. Oceanogr 2001, 48, 1507–1528. [Google Scholar]
- Racault, M.-F.; Le Quéré, C.; Buitenhuis, E.; Sathyendranath, S.; Platt, T. Phytoplankton phenology in the global ocean. Ecol. Indic 2012, 14, 152–163. [Google Scholar]
- Leterme, S.C.; Edwards, M.; Seuront, L.; Attrill, M.; Reid, P.; John, A. Decadal basin-scale changes in diatoms, dinoflagellates, and phytoplankton color across the north atlantic. Limnol. Oceanogr 2005, 50, 1244–1253. [Google Scholar]
- Hu, A.; Rooth, C.; Bleck, R.; Deser, C. Nao influence on sea ice extent in the Eurasian coastal region. Geophys. Res. Lett 2002, 29, 2053–2056. [Google Scholar]
- Henson, S.A.; Dunne, J.P.; Sarmiento, J.L. Decadal variability in North Atlantic phytoplankton blooms. J. Geophys. Res 2009, 114, C04013. [Google Scholar]
- Mantua, N.J.; Hare, S.R. The pacific decadal oscillation. J. Oceanogr 2002, 58, 35–44. [Google Scholar]
- Mantua, N.J.; Hare, S.R.; Zhang, Y.; Wallace, J.M.; Francis, R.C. A pacific interdecadal climate oscillation with impacts on salmon production. Bull. Am. Meteorol. Soc 1997, 78, 1069–1079. [Google Scholar]
- Corno, G.; Karl, D.M.; Church, M.J.; Letelier, R.M.; Lukas, R.; Bidigare, R.R.; Abbott, M.R. Impact of climate forcing on ecosystem processes in the North Pacific subtropical gyre. J. Geophys. Res 2007, 112, C04021. [Google Scholar]
- Thomas, A.C.; Brickley, P.; Weatherbee, R. Interannual variability in chlorophyll concentrations in the humboldt and california current systems. Prog. Oceanogr 2009, 83, 386–392. [Google Scholar]
- Karl, D.M.; Bidigare, R.R.; Letelier, R.B. Sustained and aperiodic variability in organic matter production and phototrophic microbial community structure in the north pacific subtropical gyre. In Phytoplankton Productivity: Carbon Assimilation in Marine and Freshwater Ecosystems; Le B. Williams, P.J., Thomas, D.N., Reynolds, C.S., Eds.; Blackwell Science: Hoboken, NJ, USA, 2002; pp. 222–264. [Google Scholar]
- Karl, D.M.; Bidigare, R.R.; Letelier, R.M. Long-term changes in plankton community structure and productivity in the North Pacific subtropical gyre: The domain shift hypothesis. Deep Sea Res. Part II: Top. Stud. Oceanogr 2001, 48, 1449–1470. [Google Scholar]
- Chiba, S.; Batten, S.; Sasaoka, K.; Sasai, Y.; Sugisaki, H. Influence of the pacific decadal oscillation on phytoplankton phenology and community structure in the western north pacific. Geophys. Res. Lett 2012, 39, L15603. [Google Scholar]
- Karl, D.M. Minireviews: A sea of change: Biogeochemical variability in the north pacific subtropical gyre. Ecosystems 1999, 2, 181–214. [Google Scholar]
- Gregg, W.W.; Casey, N.W. Modeling coccolithophores in the global oceans. Deep Sea Res. Part II: Top. Stud. Oceanogr 2007, 54, 447–477. [Google Scholar]
- Gregg, W.W. Assimilation of seawifs ocean chlorophyll data into a three-dimensional global ocean model. J. Mar. Syst 2008, 69, 205–225. [Google Scholar]
- Gregg, W.W.; Casey, N.W.; O’Reilly, J.E.; Esaias, W.E. An empirical approach to ocean color data: Reducing bias and the need for post-launch radiometric re-calibration. Remote Sens. Environ 2009, 113, 1598–1612. [Google Scholar]
- Gregg, W.W.; Casey, N.W. Improving the consistency of ocean color data: A step toward climate data records. Geophys. Res. Lett 2010, 37, L04605. [Google Scholar]
- Gregg, W.W.; Casey, N.W. Skill assessment of a spectral ocean-atmosphere radiative model. J. Mar. Syst 2009, 76, 49–63. [Google Scholar]
- Aas, E. Two-stream irradiance model for deep waters. Appl. Opt 1987, 26, 2095–2101. [Google Scholar]
- Ackleson, S.G.; Balch, W.M.; Holligan, P.M. Response of water-leaving radiance to particulate calcite and chlorophyll a concentrations: A model for gulf of maine coccolithophore blooms. J. Geophys. Res 1994, 99, 7483–7499. [Google Scholar]
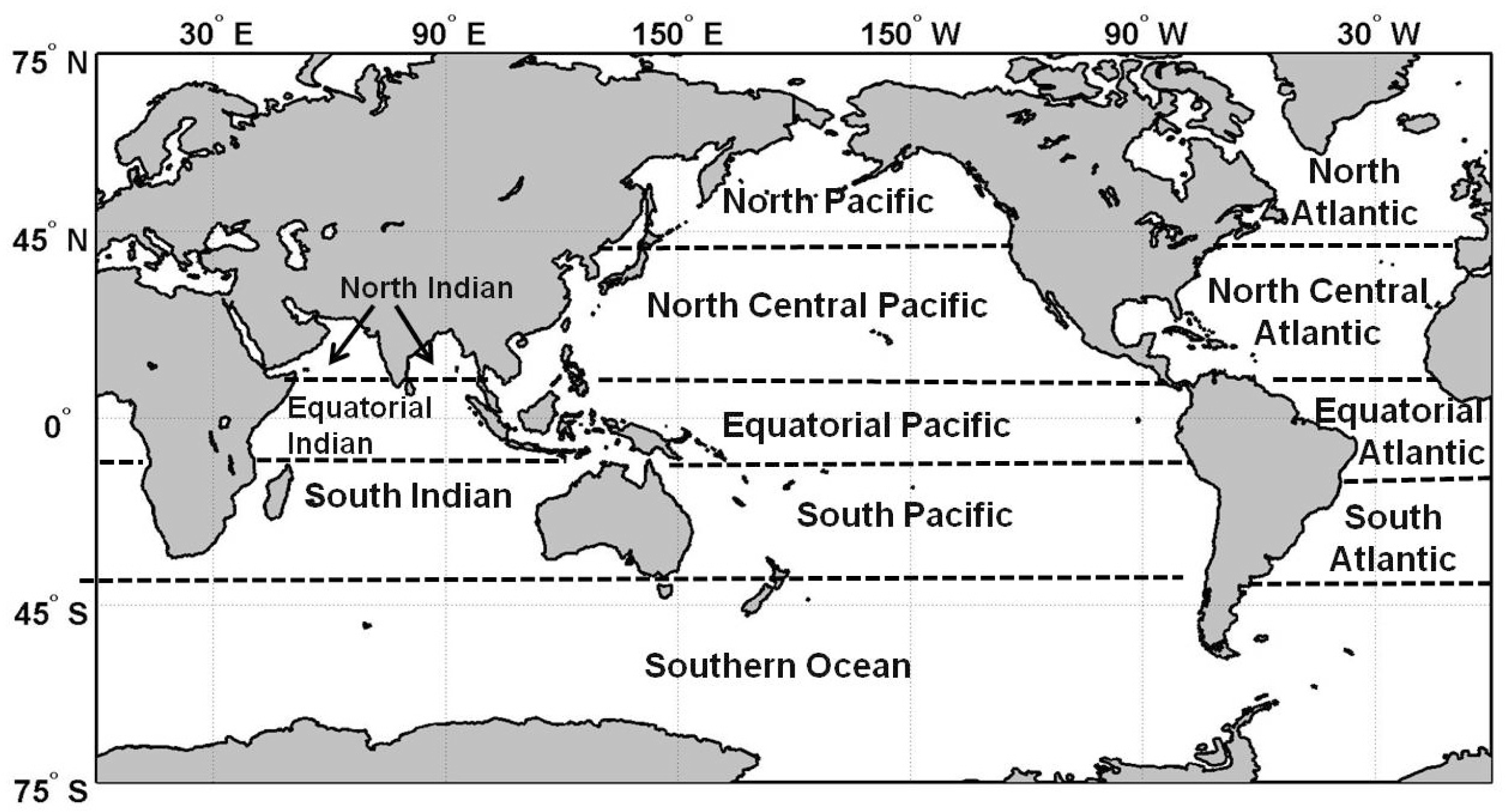
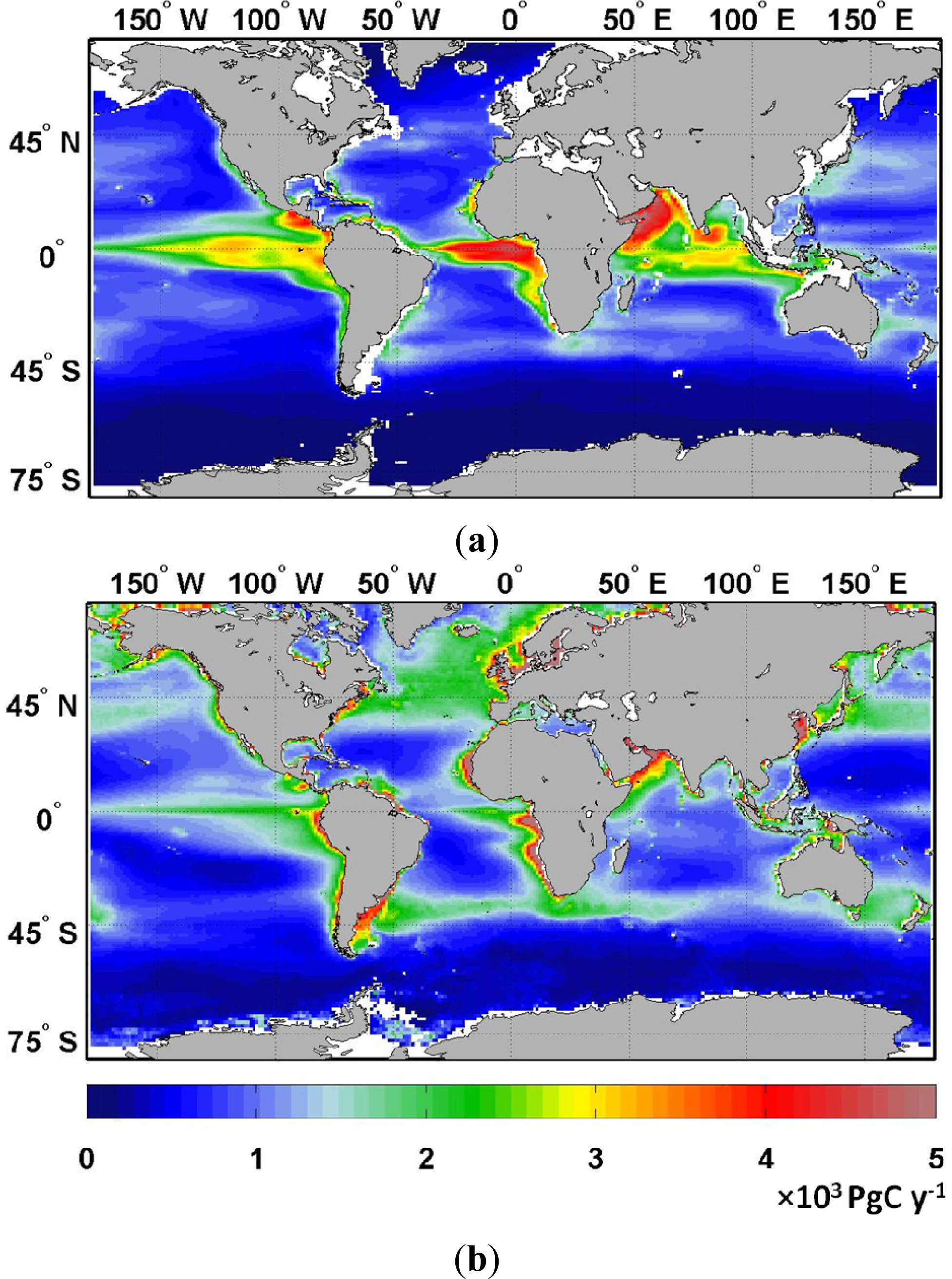

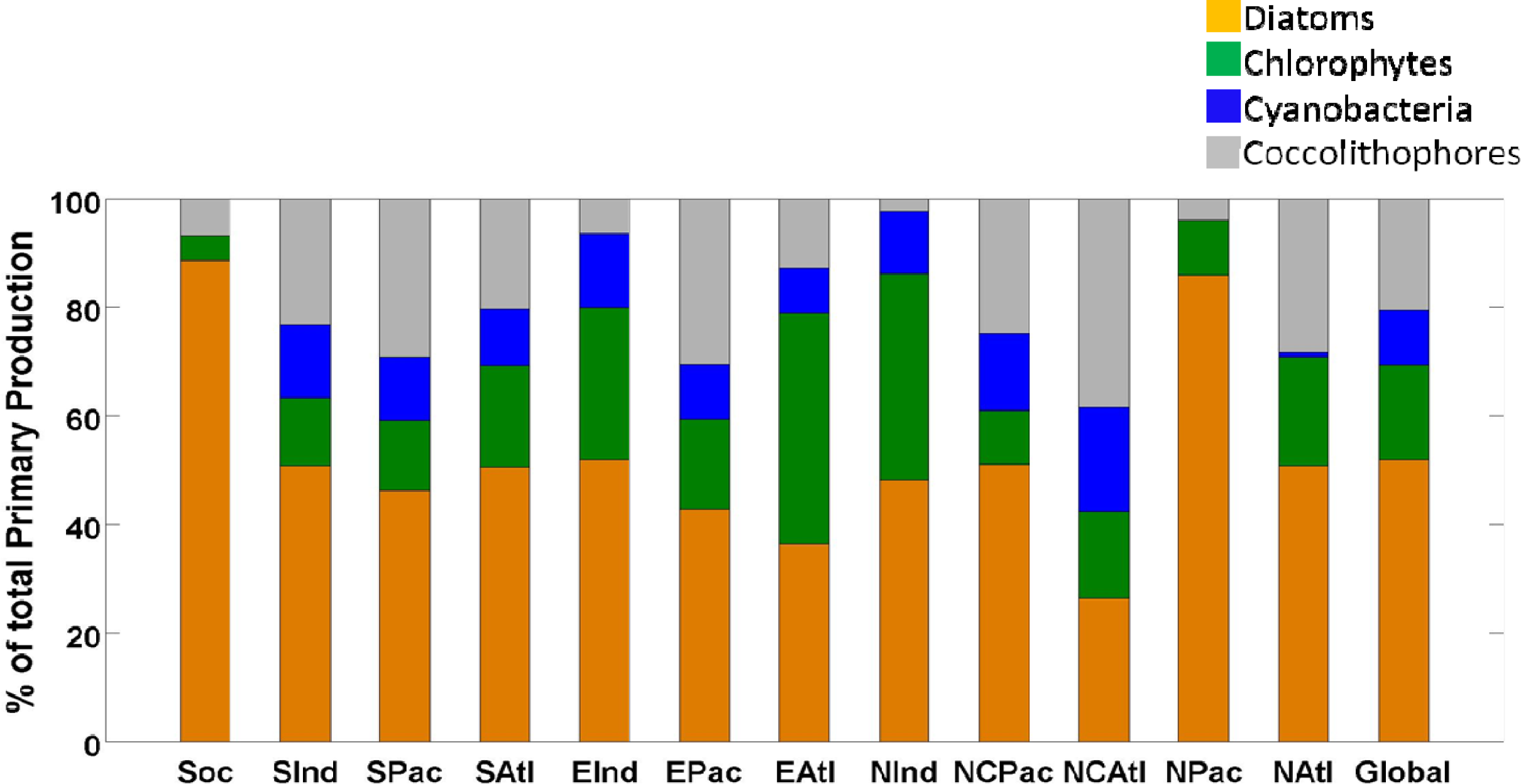


| Diatoms | Chlorophytes | Cyanobacteria | Coccolithophores | Total | |||||
|---|---|---|---|---|---|---|---|---|---|
| PgC·y−1 | % | PgC·y−1 | % | PgC·y−1 | % | PgC·y−1 | % | PgC·y−1 | |
| Southern Ocean (SOC) | 4.0 | 89 | 0.2 | 5 | 0.0 | 0 | 0.3 | 7 | 4.5 |
| South Indian (SIND) | 1.7 | 51 | 0.4 | 13 | 0.5 | 13 | 0.8 | 23 | 3.4 |
| South Pacific (SPAC) | 2.2 | 46 | 0.6 | 13 | 0.6 | 12 | 1.4 | 29 | 4.7 |
| South Atlantic (SATL) | 1.2 | 51 | 0.4 | 19 | 0.2 | 10 | 0.5 | 20 | 2.3 |
| Equatorial Indian (EIND) | 1.9 | 52 | 1.0 | 28 | 0.5 | 14 | 0.2 | 6 | 3.7 |
| Equatorial Pacific (EPAC) | 2.8 | 43 | 1.1 | 16 | 0.7 | 10 | 2.0 | 31 | 6.5 |
| Equatorial Atlantic (EATL) | 1.1 | 36 | 1.2 | 42 | 0.2 | 8 | 0.4 | 13 | 2.9 |
| North Indian (NIND) | 0.7 | 48 | 0.6 | 38 | 0.2 | 12 | 0.0 | 2 | 1.5 |
| North Central Pacific (NCPAC) | 2.4 | 51 | 0.5 | 10 | 0.7 | 14 | 1.2 | 25 | 4.7 |
| North Central Atlantic (NCATL) | 0.6 | 26 | 0.4 | 16 | 0.5 | 19 | 0.9 | 39 | 2.4 |
| North Pacific (NPAC) | 1.1 | 86 | 0.1 | 10 | 0.0 | 0 | 0.1 | 4 | 1.3 |
| North Atlantic (NATL) | 0.6 | 51 | 0.2 | 20 | 0.0 | 1 | 0.3 | 28 | 1.1 |
| Global | 20.3 | 52 | 6.8 | 17 | 4.0 | 10 | 8.0 | 21 | 39.0 |
| SOC | SIND | SPAC | SATL | EIND | EPAC | EATL | NIND | NCPAC | NCATL | NPAC | NATL | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Diatoms | −0.12 | −0.14 | −0.10 | −0.17* | −0.46* | −0.68* | −0.05 | −0.09* | 0.21* | 0.10* | 0.13 | 0.18* |
| Chlorophytes | −0.13 | −0.09 | −0.06 | −0.17* | −0.38* | −0.28* | −0.14 | −0.35* | −0.18* | 0.06* | 0.01 | 0.00* |
| Cyanobacteria | −0.05 | 0.00 | −0.07 | 0.33* | 0.13* | 0.61* | 0.10 | 0.18* | 0.03* | −0.17* | 0.01 | −0.17* |
| Coccolithophores | 0.01 | −0.13 | −0.06 | −0.14* | 0.09* | 0.12* | −0.14 | 0.18* | 0.08* | 0.23* | 0.12 | 0.15* |
| Diatoms | Chlorophytes | Cyanobacteria | Coccolithophores | ||
|---|---|---|---|---|---|
| AAO | Southern Ocean | 0.09 | 0.16* | 0.08 | 0.14 |
| NAO | North Atlantic | −0.11* | −0.22* | −0.09 | −0.25* |
| North Central Atlantic | −0.17* | −0.05 | 0.21* | −0.12 | |
| Equatorial Atlantic | −0.06 | −0.05 | 0.00 | 0.06 | |
| South Atlantic | −0.02 | 0.12 | −0.22* | 0.14 | |
| PDO | North Pacific | 0.02 | −0.26* | −0.19* | −0.07 |
| North Central Pacific | 0.40* | −0.25* | 0.03 | −0.07 | |
| Equatorial Pacific | −0.24* | −0.13 | 0.45* | 0.17* | |
| South Pacific | −0.20* | 0.04 | 0.09 | 0.00 |
© 2014 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Rousseaux, C.S.; Gregg, W.W. Interannual Variation in Phytoplankton Primary Production at A Global Scale. Remote Sens. 2014, 6, 1-19. https://doi.org/10.3390/rs6010001
Rousseaux CS, Gregg WW. Interannual Variation in Phytoplankton Primary Production at A Global Scale. Remote Sensing. 2014; 6(1):1-19. https://doi.org/10.3390/rs6010001
Chicago/Turabian StyleRousseaux, Cecile S., and Watson W. Gregg. 2014. "Interannual Variation in Phytoplankton Primary Production at A Global Scale" Remote Sensing 6, no. 1: 1-19. https://doi.org/10.3390/rs6010001





