Improving Nitrogen Use Efficiency in Crops for Sustainable Agriculture
Abstract
: In this review, we present the recent developments and future prospects of improving nitrogen use efficiency (NUE) in crops using various complementary approaches. These include conventional breeding and molecular genetics, in addition to alternative farming techniques based on no-till continuous cover cropping cultures and/or organic nitrogen (N) nutrition. Whatever the mode of N fertilization, an increased knowledge of the mechanisms controlling plant N economy is essential for improving NUE and for reducing excessive input of fertilizers, while maintaining an acceptable yield and sufficient profit margin for the farmers. Using plants grown under agronomic conditions, with different tillage conditions, in pure or associated cultures, at low and high N mineral fertilizer input, or using organic fertilization, it is now possible to develop further whole plant agronomic and physiological studies. These can be combined with gene, protein and metabolite profiling to build up a comprehensive picture depicting the different steps of N uptake, assimilation and recycling to produce either biomass in vegetative organs or proteins in storage organs. We provide a critical overview as to how our understanding of the agro-ecophysiological, physiological and molecular controls of N assimilation in crops, under varying environmental conditions, has been improved. We have used combined approaches, based on agronomic studies, whole plant physiology, quantitative genetics, forward and reverse genetics and the emerging systems biology. Long-term sustainability may require a gradual transition from synthetic N inputs to legume-based crop rotation, including continuous cover cropping systems, where these may be possible in certain areas of the world, depending on climatic conditions. Current knowledge and prospects for future agronomic development and application for breeding crops adapted to lower mineral fertilizer input and to alternative farming techniques are explored, whilst taking into account the constraints of both the current world economic situation and the environment.1. Introduction: Socioeconomic and Environmental Stakes
Today, the main method to maintain or restore soil nutrients and increase crop yields is the application of mineral fertilizers such as nitrogen (N). The N used in commercial fertilizers is particularly soluble for easy uptake and assimilation by plants. Because of the simplicity of its storage and handling, N can easily be applied when plants need it most. Mineral fertilizers are now the main source of nutrients applied to soils, even if the contribution of animal manure remains important, especially when there is densely populated livestock nearby. After World War II, N fertilizers have been used extensively to increase crop yield. The use of synthetic N fertilizers has eliminated a major elemental constraint with respect to enriching the soil stock of organic C and N originally managed by organic manure amendments, leguminous cultures and fallow periods. The formation of ammonia and thus synthetic N fertilizers by the Haber–Bosch process was one of the most important inventions of the 20th century, thus allowing the production of food for nearly half of the world population [1,2]. Consequently, a dramatic escalation has occurred in global consumption of synthetic N, from 11.6 million tonnes (Tg) in 1961 to 104 Tg in 2006 [3,4]. Over 40 years, the amount of mineral N fertilizers applied to agricultural crops increased by 7.4 fold, whereas the overall yield increase was only 2.4 fold [5]. This means that N use efficiency, (NUE) which may be defined as the yield obtained per unit of available N in the soil (supplied by the soil + N fertilizer) has declined sharply. This obviously implies that NUE is higher at reduced levels of crop production when the use of N fertilization is much lower. NUE is the product of absorption efficiency (amount of absorbed N/quantity of available N) and the utilization efficiency (yield/absorbed N). For a large number of crops, there is a genetic variability for both N absorption efficiency and for N utilization efficiency [6]. Moreover, the occurrence of interactions between the genotype and the level of N led to the conclusion that the best performing crop varieties at high N fertilization input are not necessarily the best ones when the supply of N is lower [7]. This is mainly because breeding for most crops has been conducted over the last 50 years in the presence of high mineral fertilization inputs, thus missing the opportunity to exploit genetic differences under a low level of mineral or organic N fertilization conditions [8].
In most intensive agricultural production systems, over 50% and up to 75% of the N applied to the field is not used by the plant and is lost by leaching into the soil [9-11]. Some microorganisms are able to improve soil fertility by metabolizing the N that is not absorbed by plants. It is however a lengthy process which involves a major risk because mineral N, especially nitrate (NO3-) and urea {CO(NH2)2} are very soluble and can run off into the surface water or flow into the groundwater. Water contaminated by nitrate is not potable and at high concentrations can be a serious risk for human health [12,13]. Moreover, the water industry must bear additional costs to remove nitrates from groundwater sources [14,15].
The detrimental impacts of nitrate loss from the soil have toxicological implications for animals and humans [16] and also on the environment leading to the eutrophication of freshwater [17] and marine ecosystems [18]. This phenomenon is manifested by a proliferation of green algae, reduced infiltration of light, oxygen depletion in surface water, disappearance of benthic invertebrates and the production of toxins harmful to fish, livestock and humans. Soils are also at risk from eutrophication, as excessive amounts of nutrients can cause oxygen depletion in the soil and thus prevent the proper functioning of natural microorganisms. This, in turn, affects soil fertility. Moreover, it has been reported that synthetic N fertilizers can promote microbial C utilization depleting both soil and sub-soil organic N content [4]. Eutrophic soils are the source for the emission of N2O (nitrous oxide), which can react with the stratospheric ozone [19], thus increasing the greenhouse effect and also the emission of toxic ammonia (NH3) into the atmosphere that can contribute to acidification [20-22]. The process of gaseous ammonia loss from plant foliage can range from 2 to 15kg N/ha/year released, depending on the crop examined or the location [23,24]. Additionally, when the plant does not take up urea fertilizers applied to the soil, up to 40% can also be lost in the form of ammonia [25,26].
Mineral N fertilizers produced by the Haber–Bosh process are very costly in energy production [1,27] and represent nowadays up to 50% of the operational cost for the farmer depending on the cultivated crop [28]. Thus, NUE and energy input are seen as important indicators for the environmental impact of the production of conventional crops but also of energy crops, since they have a large capacity to produce biomass with the minimal amount of N fertilizer [29]. Comparatively, the net energy cost of N2 fixation in leguminous species is lower than that necessary for an equivalent production of synthetic N fertilizers [30,31]. Therefore, it will be advantageous to the farmer to include more legumes both in crop rotations and in cover crops, whether the main cultivated crop is grown for grain or biomass.
Biological dinitrogen (N2) fixation is one of the most important sources of N in agricultural system, since it has been estimated to be around 122 Tg per year. The most important N-fixing agents are the symbiotic associations between crop and forage/fodder legumes and bacteria of the genus Rhizobia [31,32]. There are accurate estimations of annual inputs of symbiotically fixed N by legume crops. However, the amount of N fixed by other agricultural production systems involving non-symbiotic N2 fixing associations, such as rice, sugar cane and cereals is much more difficult to estimate (see [30,33,34] for reviews).
To feed the world population in 2050, which will probably reach 9 billion people, it will be necessary to increase agricultural production by 1.7 fold [6]. It is clear that even if this increase in production must be realized in developing countries that need it most, other countries that use intensive agriculture do not consider reducing their production of N fertilizers. As such, they will continue to produce as much or more mineral fertilizers, while at the same time protecting the environment will be essential to preserve the equilibrium of most earth ecosystems. The detrimental impact of the overuse of N fertilizers on the environment can be minimized if it is accompanied by sustainable agricultural practices, such as fertilizer use rationalization, crop rotation, establishment of ground cover and burial of crop residues. Rational fertilization means that the application of fertilizers both organic and inorganic is performed under the proper conditions required to prevent runoff at the appropriate growth stages of the plant and in the correct doses [6]. For example, fractionating N fertilization is currently being performed to grow wheat and other crops such as rice and oilseed rape. Such fertilization strategies have in 15–20 years decreased by 15–20%, the amount of N fertilizer applied to crops in the field [35]. Alternatively, cropping systems using carefully designed species mixtures may be a way to lower N fertilization input, while maintaining economic profitability [36].
Others strategies to improve NUE are to use genetic modification or to breed for new varieties that take up more organic or inorganic N from the soil N and utilize the absorbed N more efficiently [6,37].
Additionally, breeding for more efficient symbioses with Rhizobia and arbuscular micorrhizal (AM) fungi can be an interesting alternative for increasing plant productivity using the same amount of synthetic N fertilizer [38,39]. Conservation tillage using no till and continuous cover cropping cultures are also known to increase significantly the potentiality and diversity of plant colonization by AM fungi in comparison to conventional tillage [40-43]. Thus, these new alternative farming techniques could also be an attractive way to increase NUE for a number of crops through the beneficial action of AM.
Lastly, the occurrence of plant growth promoting bacteria (PGPB) and its relationship with the improvement of N nutrition needs to be considered. Through the release of hormones PGPB, can stimulate root development thus increasing nutrient acquisition including N (see [44,45] for reviews).
2. Nitrogen Fertilization in Agriculture
In the developed countries, mineral fertilizers are the main source of N applied to crops [46], followed closely by livestock manure [47]. There are also other sources of N to the soil: the major one being symbiotic N2 fixation in legume nodules and in the rhizosphere of a range of plants [34,45]. Minor ones include N deposition from the atmosphere, in the form of ammonia and various nitrogen oxides, and the recycling of sewage sludge, which can be applied to cultivated land despite the presence of toxic compounds [48,49]. The importance of these varies from one country to another [50,51].
The mineral commercial fertilizers commonly applied to cultivated soils are anhydrous ammonia, urea, ammonium sulfate and ammonium nitrate. They are particularly soluble for easy assimilation by crops. Both urea and ammonia are converted to nitrate at different rates depending on the nature of the soil and of the climatic conditions, thus leading to various loss mechanisms either by volatilization for ammonia or runoff for nitrate or urea after heavy rainfall and leaching into groundwater [52,53]. However, it appears that the functional diversity of the autotrophic nitrifiers, the ecology (abundance and bacterial community structure) and the nitrification kinetics performed by bacterial ammonia-oxidizers, leading to nitrite (NO2-) production and its further oxidation to nitrate by nitrite-oxidizing microorganisms are affected by tillage practices or cover cropping systems [54-56]. Therefore, the final inorganic N budget is strongly affected by the nitrification process occurring in the soil via the action of root-associated or free living microbes that alter rates of nutrient supply and the partitioning of resources between the crop and the soil flora [57].
Manures are the second in nutrient inputs to agricultural land. The nutrient content of manure varies from one country to another and from one region to another within the same country. It depends on the type of farming, grazing systems and nutrient content of different foods and fodder for livestock. There is evidence that at least 50% of manure is lost in storage and transport and another 25% of manure is lost after application [58,59]. An incubation study with composted poultry manure showed a gradual release of inorganic N, mineralizing 0.4 to 5.8% of the total N over 56 days compared to 25.4–39.8% of the total N in uncomposted poultry manure [60].
The application of manure with different level of humification, (i.e composted), has frequently been shown to increase soil fertility [61] and to stimulate soil microbial activity through the improvement of soil structure [62]. Additionally, it has been demonstrated that humic substances have auxin-like activity and positive effects on plant physiology by influencing nutrient uptake and root architecture [63,64]. Simultaneously, it has been shown that through the use of flow-through colorimetry that there is and adsorption of nitrate on to humic substances, thus improving N availability to the plant [65].
From information on N inputs to agricultural soils and estimates of N uptake by crops and grass, a calculation of the excess amounts of N applied to agricultural land can be established. This method of calculating the excess N is known as N balance at the surface [66]. The surface balance can be used as an indicator that highlights areas potentially threatened by N pollution under various environmental scenarios [67,68]. In addition, monitoring the evolution of these surpluses over several years can be used to evaluate the effectiveness of agri-environmental measures to avoid pollution by nitrates. The calculation of the surplus cannot however be immediately interpreted as an indicator of N loss in water. The balance between inputs and outputs for a system includes all potential losses described in the above sections, and inventory changes of N, mainly in the soil.
3. Nitrogen Fertilization Using Green Manure and Cover Crops
Green manure fertilization (see [69] for a review) aims to improve soil fertility and quality by incorporation into the soil of any field or forage crop while the cultivated plant is still at the green vegetative stage, or just after the flowering stage. Green manure can also be crushed or rolled before no-till seeding (Figure 1).
A cover crop is any crop grown to provide soil cover, regardless of whether it is later incorporated into the soil. Cover crops are grown primarily to prevent soil erosion by wind and water. Cover crops and green manures can be annual, biennial, or perennial herbaceous plants grown in a pure or mixed stand during all or part of the year (Figure 1). In addition to providing ground cover and, in the case of a symbiotic N2-fixing legume, they provide substantial amounts of N. They also help suppress weeds [70] via allelopathic legume cover and mulching species [71] and reduce insect pests and diseases [72-74]. When cover crops are planted to reduce nutrient leaching (N in particular) following a main crop, they are often termed “catch crops.” [75,76]. Moreover, growing green manures on site is a way to prevent the often inhibitive handling and transportation costs of other organic inputs [69]. There are a large variety of cover crop species that are appropriate for a farmer and a particular region. Details on the use of catch crops to prevent N leaching losses during the winter period and of N fertilization using green manures (including N fixing legumes), can be found in the review by Thorup-Kristensen et al. [77] and in the handbook: Managing Cover Crop Profitability [78].
Legumes are widely used as cover crops since there is a large choice of different species suited to a particular environment (Figure 1). Legumes are defined by their unique flower structure, their pod, and the ability of 88% of the species examined so far to form atmospheric N2 fixing nodules [79,80]. Legumes are only of second importance after grasses to humans, by contributing significantly to grain, pasture and forage, and forestry production [33,81]. Since legumes are able to fix symbiotically atmospheric N2, they require minimal or even no inputs of N fertilizers. If part of this “free” N is made available to a following cultivated crop, the use of legumes in a rotation can allow a significant reduction in the use of N fertilizers. Additionally, legumes can also enhance both the colonization of crop roots by mycorrhizae [82] and the tripartite symbiosis between the host plant AM fungi and N-fixing bacteria thus finally affecting N uptake by the host plant [83]. The legumes used as cover crops or green manure can be classified into two categories: tropical and temperate. Warmer climates or warmer winter temperatures allow temperate species to persist during the winter, and tropical species are more adapted to the summer months. It is the intra- and inter-specific genetic variability that partly explains why some legumes grow more and accumulate more N than others. However, it is mainly the soil and climatic conditions that are the predominant factors that restrict the selection of the best performing legumes species. For example, Brandsaeter et al. [84] showed in a recent study that the biochemical quality of the plants differed between species and dates of harvesting, and that this was reflected in the dynamics of net N mineralization. A number of reviews have focused on selection criteria, breeding methods and genetic modification approaches and have covered future improvements in legume crops that will be beneficial not only to the environment and farmers but also to consumers in both developed and developing countries [85-87]. Studies using quantitative genetics approaches to improve NUE in legumes are scarce. However, it seems that both root and nodule traits are important for efficient N assimilation for further translocation to the seeds [88].
N production from legumes is a key benefit of growing cover crops and green manures. The amount of N available from legumes depends on the species of legume grown, the total biomass produced, and the percentage of N in the plant tissue. Cultural and environmental conditions that limit legume growth, such as a delayed planting date, poor stand establishment, and drought will reduce the amount of N produced. Conditions that encourage good N production include getting a good stand, optimum soil nutrient levels and soil pH, good nodulation, and adequate soil moisture. The portion of green-manure N available to a following crop is usually about 40% to 60% of the total amount contained in the legume [76]. Interestingly, it has been demonstrated that leguminous cover crops were also able to replace 60% of the chemical N fertilization for cotton production, although the quantity of available N derived from the cover crop was not synchronized with the requirements of the cotton plant [89]. In turn, one has to consider that NUE is strongly affected by the organic residues remaining from the preceding crop and the application rate of both synthetic N or organic fertilizers applied to the next crop [90].
Both raw and composted manures are useful in organic crop production (for a review see [91]). Used properly, with attention to balancing soil fertility, manures can supplant all or most needs for purchased N fertilizer, especially when combined with a whole system fertility plan that includes crop rotation and cover cropping with N-fixing legumes. However, there is often a lack of synchronization between the timing of N mineralization originating from the catch crop and the N requirement of the main crop, thus leading to a loss of part of the N initially saved by the catch crop. It is therefore necessary to improve estimates of the longer-term N effects of catch crops and to optimize crop sequences in order to estimate accurately the turnover of N retained in the soil by the nitrate catch crops [92,93]. Thus, the grower needs to monitor nutrients in the soil via soil testing, and learn the characteristics of the manure and/or compost to be used. The grower should adjust the rates and select additional fertilizers and amendments accordingly. Finally, development of viable green manure-based alternatives leading to applied crop synergisms will probably not occur without refinement of whole-systems approaches within which green manure secure multiple ecosystemic services [94], utilizing and conserving functional agro-biodiversity services [95].
In addition to legumes, commonly used cover crops include annual cereals (rye, wheat, barley oats), annual or perennial forage grasses such as ryegrass, warm season grasses such as sorgum-sundangrass hybrids and brassicas (Figure 1) including mustard (see [78], for details on their benefits and management).
If organic farming needs to use both classical and green manure to replace chemical N fertilization, it appears that plant genetic adaptations and breeding for these alternative farming techniques are needed to increase crop NUE, for example in wheat [96-99]. Additionally, the development of biomarkers for determining the potential of NUE and optimization of N inputs in crop plants under organic farming cultivation conditions will be required [100].
4. Nitrogen Assimilation by Plants
Nitrate is the principal N source for most wild and crop species, whatever the source of inorganic or organic N provided to the plant [101,102]. It is taken up by means of specific high and low affinity transporters located in the root cell membrane [103,104]. Nitrates are then reduced to nitrite through the reaction catalysed by the enzyme nitrate reductase (NR; EC 1.6.6.1), [105] followed by the reduction of nitrite to ammonia catalysed by the enzyme nitrite reductase (NiR; EC 1.7.7.1), [106]. Under particular environments, root ammonia transporters [107] can allow a direct uptake of ammonia when available in the soil, in rice paddy fields or in acidic forest habitats [101,108]. Ammonia can be generated inside the plant by a variety of metabolic pathways such as photorespiration, phenylpropanoid metabolism, utilization of N transport compounds and amino acids catabolism. Symbiotically fixed N is also an important source of ammonia readily available to herbaceous plants or woody species that are able to form a symbiotic relationship with N fixing microorganisms [87,109]; (Figure 2).
Several studies have shown that a wide variety of plant species are able to take up organic N compounds, especially under low N conditions [10,102,110-113]. However, the importance of this N source and the methods used to evaluate its contribution to plant N requirements has been questioned. A few studies have been done on the uptake of organic N by commercial crops: e.g., corn [114], agricultural grasses including species of clover [112] and wheat [96]. Despite these limited studies, they demonstrate the ability of plants to directly take up organic N, but have not established the importance and significance of organic N as a source of crop N, for example when they are grown under organic farming conditions.
In line with the finding that plants can take organic N up directly, there is also an interesting report in which it has been shown that herbaceous species can use protein as a N source without the assistance of other organisms. This indicates that the spectrum of N compounds that can be taken up by the roots is quite diverse, indicating that the relationships existing between the soil fauna and the plant for N capture is more complex than originally thought [115].
Urea is a low molecular weight organic molecule containing N that exists in natural systems and is also applied as a synthetic fertilizer in conventional agriculture. It is well known that urea is absorbed as an intact molecule by plant leaves and roots [116] by means of specific root transporters [117,118]. Although the use of urea is mainly as a source of N fertilizer, the contribution of plant urea uptake and metabolism in a physiological and agricultural context is still not investigated. However, plants possess urea transporters, and can hydrolyse and use urea very efficiently [119].
The importance of AM fungi for nutrient uptake by plants is well documented [120-122]. Several studies have shown that AM fungi-infected plants can take up organic N compounds [10,111,112]. Thus, AM fungi can be used as a source of biological fertilization, since they are able to develop symbiotic associations with most terrestrial plants. They are able to alleviate the effects of different stresses both on growth and yield, by significantly increasing the uptake of water and nutrients (including N) by the host plant [123-128]. In particular, it has been reported that the hyphae of AM are able to use inorganic N more efficiently, thus enabling the host plant to indirectly have access to soil N through its fungal partner [129]. However the quantitative contribution of AM fungi to the direct uptake of organic N by plants is still not well established [128], even though recent progress have been made in this field of research. Nevertheless, Tian et al. [130] showed that AM fungi were able to absorb both organic and inorganic N and synthesize organic N molecules such as arginine that are further released by the fungal hyphae and then absorbed by the host plant. Interestingly, the occurrence of a transfer of symbiotically fixed N to a crop such as maize via vesicular-AM hyphae has been demonstrated [131-133], indicating that associated or continuous cover cropping systems could be an alternative way to rationalize plant N nutrition by optimizing field conditions favourable to mycorrhizal colonization.
Ammonia, which is the ultimate form of inorganic N available to the plant, is then incorporated into the amino acid glutamate through the action of two enzymes. The first reaction catalyzed by enzyme glutamine synthetase (GS; EC 6.3.1.2) [134] is considered to be the major route facilitating the incorporation of inorganic N into organic molecules in conjunction with the second enzyme glutamate synthase (GOGAT; EC 1.4.7.1) [135], which recycles glutamate and incorporates C skeletons as a form of 2-oxoglutarate into the cycle. The amino acids glutamine and glutamate are then further used as amino group donors to all the other N-containing molecules notably other amino acids used for storage, transport and protein synthesis and to nucleotides used as basic molecules for RNA and DNA synthesis [134-136].
The two enzymes GS and GOGAT are present in the plant in several isoenzymic forms located in different cellular compartments and differentially expressed in a particular organ or cell type according to the developmental stage. The GS enzyme exists as a cytosolic form (GS1) present in a variety of organ and tissues such as roots, leaves, phloem cells and a plastidic form (GS2) localized in the chloroplasts of photosynthetic tissues and the plastids of roots and etiolated tissues. It has also been proposed that GS2 is located in the mitochondria [137]. However, in numerous previous studies using immunocytolocalization techniques, the presence of the enzyme in the mitochondria has never been reported [138]. The relative proportions of GS1 and GS2 vary within the organs of the same plant and between plant species, each GS isoform playing a specific role in a given metabolic process, such as photorespiratory ammonia assimilation, nitrate reduction, N translocation and recycling [134,139]. The enzyme GOGAT also exists as two forms that have specific roles during primary N assimilation or N recycling. A ferredoxin-dependent iseoenzyme (Fd-GOGAT) is mainly involved, in conjunction with GS2, in the reassimilation of photorespiratory ammonia and a pyridine nucleotide-dependent isoenzyme (NADH-GOGAT; EC 1.4.1.14) involved in the synthesis of glutamate both in photosynthetic and non-photosynthetic organs or tissues to sustain plant growth and development [134,136]. Moreover, by virtue of their differential mode of expression regulated either at the transcriptional and post transcriptional levels, both GS and GOGAT isoenzymes have been shown to play a specific role at particular stages of the plant life cycle and under particular environmental conditions related mainly to the mode of N nutrition [134,135,139].
The reversible reaction catalyzed by the enzyme glutamate dehydrogenase (GDH; EC 1.4.1.2) [134], which has theoretically the capacity to incorporate ammonia into 2-oxoglutarate to form glutamate, was originally thought to be the main enzyme involved in inorganic N assimilation in plants. Later on, a number of experiments using 15N labeling techniques and mutants deficient in GS and GOGAT have demonstrated that over 95% of the ammonia made available to the plant is assimilated via the GS/GOGAT pathway [134,140]. A number of 15N labeling experiments followed by GCMS or NMR-spectroscopy analysis have shown that GDH operates in the direction of glutamate deamination to provide organic acids notably when the cell is C-limited [141,142]. The finding that under certain physiological conditions GDH is able to assimilate ammonia also needs to be taken into consideration, although the rate of glutamate synthesis is probably far lower than that formed through the GS/GOGAT pathway [143]. Recently the hypothesis that GDH plays an important role in controlling glutamate homeostasis has been put forward [142]. This function, which may have a signaling role at the interface of C and N metabolism, may be of importance under certain phases of plant growth and development when there is an important release or accumulation of ammonia [144-146].
Over the last two decades, our knowledge of the various pathways involved in the synthesis of the twenty amino acids that are used to build up proteins, particularly those derived from glutamate and glutamine, has been increased through the use of mutant and transgenic plants in which amino acid biosynthesis has been altered. There are excellent reviews describing extensively our current knowledge on plant amino acid biosynthesis and its regulation [136,143]. Therefore, we will not cover this complex aspect of N assimilation in this review, even though it is of major importance for plant growth and productivity. However, there are some examples of genetic modification in crops in which these pathways have been altered particularly to increase the content of lysine and methionine, which are often the most limiting for both humans and animal nutrition [147-149].
Significant progress has been made during the last few years on the regulation of inorganic N metabolism and the relationships with C metabolism, both at the cellular and organ levels. In particular, attempts to integrate large transcriptomic and physiological data sets at the whole plant level have increased our understanding of the regulation of N assimilation not only under controlled growth conditions but also under the constantly changing environmental constraints usually occurring in field situations [6]. This integration is required, because in addition to regulating a range of cellular processes including N assimilation itself through the co-ordination of nitrate or ammonia uptake and use, nitrate and N metabolite levels in the cell can regulate directly or indirectly a number of closely related metabolic and developmental processes [150,151]. These processes, which may also be regulated through the action of hormones [152], include the synthesis and accumulation of amino acids and organic acids and the modification of plant development including the extent and form of root growth and the timing of flower induction. All these processes, acting either individually or synergistically, condition N allocation in newly developing tissues or in storage organs to finally ensure plant vegetative or sexual reproduction.
5. Improvement of Nitrogen Utilization Using Genetically Modified Crops
Nitrate reduction is rarely limiting for optimal grain yield or biomass production. In contrast, this is not the case for the ammonia assimilatory pathway [153]. For example the work of Fuentes et al. [154] showed that, in tobacco, overexpression of a gene encoding cytosolic glutamine synthetase (GS1) from alfalfa, causes an increase in photosynthesis and growth under a low N fertilization regime. These results suggest that the transgenic tobacco plants overexpresing GS1 are able to utilize N more efficiently under N stress conditions. Interestingly, Oliveira et al. [155] also showed that in tobacco, the overexpression of a gene encoding a pea GS1 lead to increased biomass production both under limiting and non-limiting N feeding conditions.
By overexpressing a pine GS1 gene in poplar, Jing et al. [156] and Man et al. [157] observed that the transgenic trees, which were older than five years exhibited a 41%, increase in growth rate, whereas the other phenotypic characteristics of the genetically modified plants remained similar.
In wheat, the overexpression of a gene for GS1 from French bean led to an increase in grain yield (grain weight in particular) and therefore of NUE, which was estimated to be about 20% [158]. However, to our knowledge there has been no further development of this interesting study, either because of the difficulty of field testing in Europe or because this testing is currently being performed in the private sector. Similar work was conducted in maize consisting in the overexpression of a native gene encoding GS1 (Gln1-3) of maize. Grain yield (mainly grain number) of the maize transgenic plants grown under greenhouse conditions was increased by about 30%. However, grain N content and biomass production of the transgenic plants were not modified at maturity [159]. More recently, transgenic rice lines overexpressing GS1 showed improved harvest index, N harvest index and N utilization efficiency. However, these lines did not exhibit higher NUE under N-limiting conditions compared to non-limiting N conditions [160].
In other species, the overexpression of GS1 had a rather negative impact on growth and yield of the plant. For example, overexpression of a GS1gene from tobacco in the legume birds foot trefoil (Lotus corniculatus L.) grown on nitrate led to an acceleration of senescence, which was apparently detrimental to the overall plant developmental process [161]. When the transgenic L. corniculatus plants were grown under symbiotic N-fixing conditions an increase in plant biomass production was unexpectedly observed. However, the physiological mechanisms involved in this increase remain unknown [86].
In rape (canola), the overexpression of a gene encoding the enzyme alanine aminotransferase (AlaAT) from barley, directed by a rape root-specific promoter, led to a dramatic increase in biomass production and seed yield [162]. Improvement of plant productivity was only observed under low N fertilization conditions and was attributed to a higher flux of nitrate, associated or induced by a decrease in the content of glutamine and glutamate in the stem. In the field when the applied N fertilizer rate was reduced by 40%, the agronomic performance of the transgenic rapeseed plants overexpressing AlaAT was similar to that of untransformed control plants grown under higher optimal N fertilizer rates.
Overexpression of the same gene in rice led to increased biomass production and N content of stems [163]. Unlike in rapeseed, there was an increase of glutamine and asparagine content both in the stems and in the roots. The genetically modified rice plants had a finer, denser and more branched root system, which was presumably more favorable for the absorption of N. This result indicates that genetic modification targeted to improve N utilization efficiency also had an impact on plant development, although the effect of AlaAT overexpression was variable from one species to another in terms of both plant growth and metabolic activity.
There are a few other examples of successful genetic modification of N metabolism using either structural or putative regulatory genes. When the bacterial enzyme glutamate dehydrogenase (GDH A) from E. coli was constitutively overexpressed in tobacco, biomass production of the transgenic plants was increased by about 10–15%. In addition to the increase in biomass production GDHA overexpressors had more leaves and their free amino acid content was higher, suggesting that both N metabolism and C metabolism were modified [164]. The transgenic tobacco plants were also more tolerant to water stress.
In rice, overexpression of a gene of unknown function OsENOD93-1, a N-responsive gene identified following genome-wide gene expression profiling, led to an increase in grain yield, of 13–14% and 19–23% under limiting and non-limiting N nutrition conditions respectively [165]. When a gene encoding NAD(H)-dependent GOGAT from alfalfa was constitutively expressed in tobacco, a significant increase in biomass production was observed [166]. Overexpression of the native NAD(H)-dependent GOGAT in rice led to an increase in grain weight [167,168]. These results suggest that the GOGAT enzyme plays a major role with respect to organic N management and is used either for growth or for grain production depending on the species examined.
There are fewer studies in which the importance of regulatory genes has been clearly demonstrated [169]. When a Dof1 gene encoding a transcription factor from maize was overexpressed in Arabidopsis (Arabidopsis thaliana L.), an increase in amino acid content and of N uptake was observed, especially when plants were grown at a low level of N supply. In addition, the transgenic plants produced more biomass under low N supply and they did not exhibit symptoms of N deficiency in comparison to the untransformed control plants, which developed much earlier symptoms of senescence. When the Dof 1 gene was overexpressed in potato, transgenic plants accumulated more amino acids especially glutamine and glutamate [169]. These two sets of experiments suggest that this gene could be used to improve the uptake and utilization of N in several species. Thus, overexpressing regulatory genes rather than structural genes, such as genes encoding GS, GOGAT or AlaAT appears to be an interesting alternative to improve plant NUE and overall plant growth and development in a more stable and balanced way across species.
When vegetable crops such as lettuce or spinach are grown under greenhouse conditions they can accumulate substantial amounts of nitrate in the leaf cell vacuoles. The threshold of nitrate accumulation often exceeds the limits permitted by law, even when N fertilization is reduced because mineralization of soil organic matter always provides a surplus of nitrate to the plant [170]. In human food, when nitrate is absorbed in excess, its reduction to nitrite during digestion can oxidize hemoglobin, causing a kind of anemia. Moreover, nitrites can be converted to carcinogenic nitrosamines [12,13]. Conventional methods of selection have led to the development of varieties able to reduce the absorbed nitrate more efficiently instead of storing it, but these varieties are not able to completely eliminate any risk of toxic accumulation. Studies were therefore undertaken to limit nitrate accumulation by increasing the capacity of a plant to reduce nitrate by increasing nitrate reductase (NR) activity in genetically modified plants, by overexpressing a gene that allows the deregulation of the synthesis of the enzyme [171]. In tobacco a 50% reduction in leaf nitrate content was observed after introduction of the native structural NR gene (Nia2) placed under the control of the 35S strong constitutive promoter. Using the same approach, encouraging results were obtained in a variety of potato [172] that showed a 95% decrease in the amount nitrate in the tubers. In another variety of potato, the transgenic plants showed a marked improvement in biomass production, especially in tubers, with still lower amounts of nitrate. The more effective reduction of nitrate probably allowed a better allocation of N to the photosynthetic apparatus and to enzymes involved in C metabolism, which was demonstrated by higher leaf chlorophyll content in the transgenic potato plants [173].
In lettuce transformed with the same 35S-Nia2 construct, a problem of post-transcriptional regulation of the NR enzyme was encountered [174]. The transgenic lettuce accumulated 21% less nitrate after 22 days. However, the nitrate content was only 4% lower in 84 days-old transgenic plants. The hypothesis that the strength of the 35S promoter decreases during plant ageing was put forward, suggesting that a way to maintain NR activity at a high level regardless of plant age needs to be found. Such a strategy to reduce the nitrate content in vegetable crops requires further research before the use of the Nia2 transgene can be efficiently mastered.
Although we do not have any clear information from the private sector about the recent development and commercialization of transgenic plants modified for NUE, it seems to be likely that crops overexpressing the enzymes AlaAT and GS1 will be commercially released within the next five years, following extensive validation of their function under different field trial conditions and using different genetic backgrounds.
6. Deciphering the Genetic Basis of Nitrogen Use Efficiency in Crops
There have been an increasing number of studies only performed on the model species Arabidopsis, in an attempt to link plant physiology to whole genome expression in order to obtain an integrated view on how the expression of genes can affect overall plant functioning [151]. When a structural or regulatory gene putatively involved in the control of a metabolic pathway or a developmental process or both is identified, information can then be obtained by producing overexpressors or selecting deficient mutants of the gene in question. By studying the impact of the genetic modification or the mutation on the phenotype or the physiology of the plant, it is often possible to determine whether the expression of this specific gene is a limiting step in the development of a particular organ or of a metabolic pathway. In general, this targeted approach, which allows the identification of a single limiting reaction, or a co-limiting/non-limiting reaction does not adequately take into account the variation in complex traits such as those controlling NUE, which involves multiple genes and thus multiple enzyme reactions and regulatory factors.
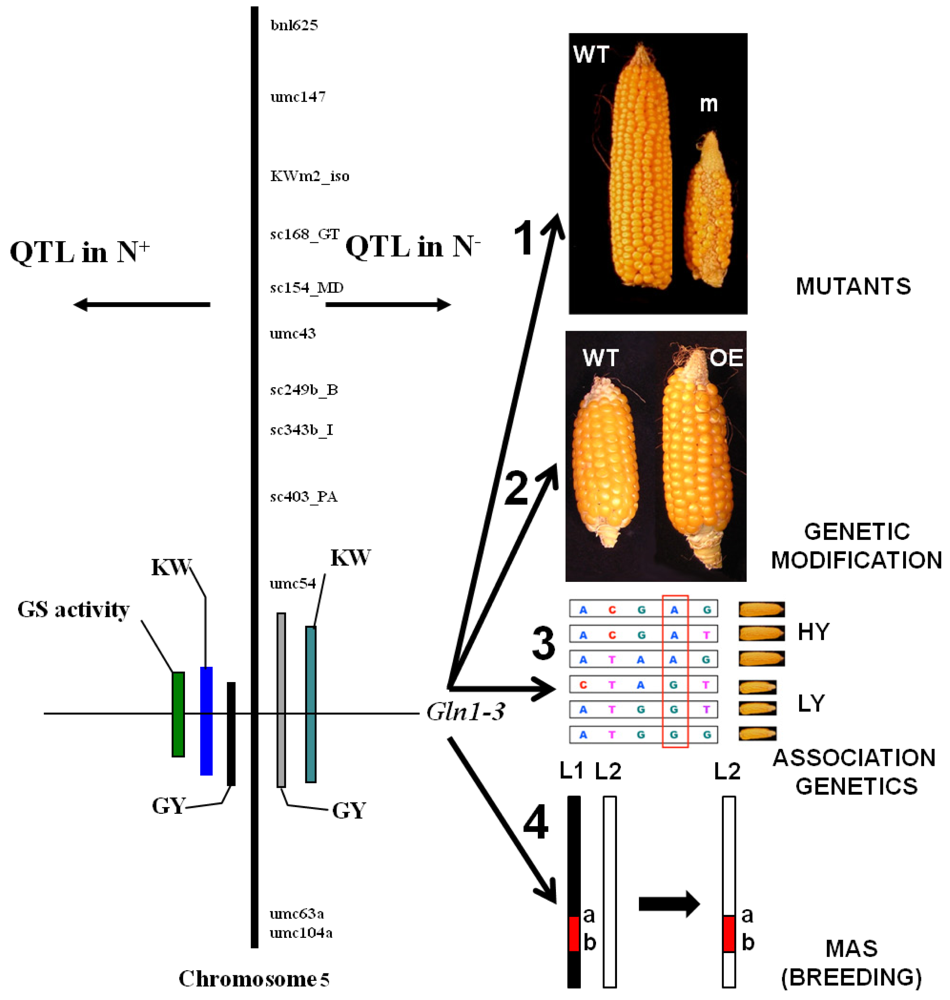
Over the last ten years, quantitative genetics, through the detection of quantitative trait loci (QTL), has become an important approach for identifying key regulatory or structural genes involved in the expression of complex physiological and agronomic traits in an integrated manner and for the study of plant responses to environmental constraints [175]. When QTLs for agronomic and phenotypic traits are located on a genetic map, it is possible to look for their genetic significance by establishing the co-location of QTLs for physiological or biochemical traits with genes putatively involved in the control of the trait of interest (candidate genes). Validation of candidate genes can then be undertaken using transgenic technologies (forward genetics) or mutagenesis (reverse genetics) or by studying the relationship between allelic polymorphism and the trait of interest (association genetics; Figure 3) either at a single gene or genome-wide level [176]. Positional cloning is another alternative strategy that can be used to focus on the chromosomal region controlling the trait of interest and that ultimately allows access to a single gene [177].
Therefore, quantitative genetic approaches were developed first in maize for which recombinant inbred lines (RIL) populations were used to build-up genetic maps and then study QTLs. The aim of such studies was to identify chromosomal regions involved in the control of yield and its components at high or low N fertilization input, and to determine whether or not some of these regions were specific for one of the two nutrition regimes. In one study, a limited number of QTLs for yield was detected only at low N-input [178]. In another study, it was found that most of the chromosomal regions for grain composition and traits related to NUE detected at low N-input, corresponded to QTLs detected at high N-input [179]. These contrasting results suggest that depending on the RIL population, the response of yield to various levels of N fertilization could be different and thus controlled by a different set of genes.
In a more detailed investigation by Bertin and Gallais [179] using maize RILs, agronomic traits, NUE and physiological traits were associated with DNA markers [180,181]. Interestingly, coincidences were detected between QTLs for yield and two genes encoding cytosolic GS (Gln1-3 and Gln1-4) and whole leaf enzyme activity. As a result of which, the hypothesis that cytosolic GS activity could be a major element controlling grain yield was put forward. [180]. Since a QTL for a thousand kernel weight was coincident with the Gln1-4 locus and QTLs for a thousand kernel weight and yield were coincident with the Gln1-3 locus (Figure 3), further work was undertaken to validate the function of these two putative candidate genes. In another study also performed in maize, fine QTL mapping of C and N metabolism enzymes activities was performed on a different RIL population. These QTLs did not colocalize with those reported by other authors [180], which indicates that there are large differences in diversity traits in maize [182].
The impact of the knockout mutations gln1-3 and gln1-4 on kernel yield and its components were examined in plants grown under controlled conditions [159]. The phenotype of the two mutant lines was characterized by a reduction of kernel size in the gln1-4 mutant and by a reduction of kernel number in the gln1-3 mutant. In the gln1-3/1-4 double mutant, a cumulative effect of the two mutations was observed. In transgenic plants overexpressing Gln1-3 constitutively in the leaves, there was an increase in kernel number, thus providing further evidence that the cytosolic GS isoenzyme GS1-3 plays a major role in controlling kernel yield [159]; Figure 3). The hypothesis that GS is one of the key steps in the control of cereal productivity was strengthened by a study performed on rice, in which a co-localization of a QTL for the GS1;1 locus and a QTL for one-spikelet weight was identified [183]. As a confirmation, a strong reduction in growth rate and grain yield was observed in rice GS1;1 deficient mutants [184].
The role of the GS enzyme and other N-related physiological traits in the control of agronomic performance in wheat still remains to be clearly established. Using a quantitative genetics approach, Fontaine et al. [185] found only a co-localization between a QTL for GS activity and GSe, a structural gene encoding cytosolic GS, but no obvious colocalization with a QTL for yield, in agreement with previous work published by Habash et al. [158]. In contrast, in recent work, physical mapping, sequencing, annotation and candidate gene validation of an NUE QTL on wheat chromosome 3B suggested that the NADH-dependent GOGAT enzymes contribute to NUE in wheat and other cereals [186] in agreement with work previously performed on rice [167].
Interestingly, in a woody species such as maritime pine that is far away from cereals on an evolutionary point of view, a protein QTL for GS co-localized with a GS gene and a QTL for biomass [187]. Functional validation of the pine GS gene in transgenic poplars (see above), which can be considered as a crop for wood production, shows once again that quantitative genetics represent one of the most powerful approaches for identifying NUE candidate genes that may be involved in the control of plant productivity.
To date, there are only a few reports reporting specific breeding for organic input systems and especially N [188]. A question that could be addressed is whether the genetic control of NUE under organic or conventional fertilization conditions is similar or if there are specific genes or combinations of genes that are more adapted to one mode of fertilization compared to the other, taking into account that organic material can be directly taken up by the plant [189]. Moreover its appears that using appropriate selection environments is important for breeding crops adapted to organic farming systems [190].
Further work is necessary to identify whether other root and shoot enzymes or regulatory proteins could play a specific role under low or high N availability, whatever the type of N fertilization conditions (organic or mineral). Such proteins include those directly involved in N metabolism or those positioned at the interface between C and N metabolism during plant growth and development [150,191,192]. It will be necessary therefore to identify new N-responsive genes through detailed analyses of transcriptomic data sets [189], including using systems biology approaches [109]. The analyses will be targeted specifically to N uptake, assimilation and recycling in vegetative [165 and reproductive organs [193] at various stages of plant development, using plants grown under different levels of N fertilization. Systems biology consists in taking advantage of various ‘omics’ data sets including transcriptomics, proteomics and metabolomics that can be further analysed in an integrated manner through the utilization of various mathematical, bioinformatic and computational tools [192]. Ultimately, such integrated analyses may allow the identification of the key individual or common regulatory elements involved in the control of a given biological process [157]. Such an approach, originally developed for the model plant Arabidopsis by virtue of the wealth of information available at the transcriptome level, when transferred to crops, may help in identifying key master genes involved in the control of NUE. In parallel, metabolomic studies are becoming more and more extensively used for the high throughput phenotyping necessary for large scale molecular and quantitative genetic studies aimed at identifying new candidate genes involved in the control of plant productivity [194,195]. This has prompted a number of groups, to focus their research efforts on developing data integration tools for metabolic reactions that complement gene expression studies. Encouragingly, on the modeling side, an increasing number of genome-scale metabolic models of plants have recently been released [196,197]. Such metabolic models should help to unravel key reactions and thus limit the steps required for the control of NUE, taking into account both tissue-specificities and environmental constraints.
Using the knowledge gained from these various systems biology approaches, it should then be possible to map the newly identified genes encoding regulatory proteins or enzymes, taking advantage of the recent progress in crop genomics through the availability of both physical and genetic high density maps and QTL or Meta-QTL genetic map positions generated by the plant science community [186,198]. Comparative genomics and synteny approaches similar to those of Quraishi et al. [186] can complete such analyses by linking the genetic maps of maize, rice, barley and wheat harboring N related QTLs, thus allowing the reinforcement of the weight of selected putative candidate genes.
Ultimately, following the functional validation of candidate genes using all the available approaches offered by mutagenesis, genetic modification and association genetics, marker-assisted selection (MAS) can be then undertaken (Figure 3). However, there are still a number of technical and scientific challenges that remain to be resolved before MAS can be routinely used in breeding for complex traits such as NUE. This is mainly due to the number of interactions that govern the expression of such traits both at the genetic and environmental levels [199], whether we are dealing with conventional or organic farming growth conditions.
7. Conclusion and Perspectives
A large number of studies have been carried out over the last two decades to identify by means of agronomic, physiological and genetic studies, the rate limiting steps of NUE both in model and crop species, as a function of environmental conditions. For abiotic stress improvement in crops, NUE has become the second priority after drought both in the private and in the public sector. To decipher the genetic and physiological basis of NUE, many tools are available for most crops and for cereals in particular. They include mutant collections, wide genetic diversity, recombinant inbred lines (RILs) or Doubled Haploid Line populations (DHLs), straightforward transformation protocols and physiological, biochemical and genomic data for systems biology development [6,200]. In addition, the commercial crop research effort is paralleled by research in the public sector, notably with the release of the genome sequences for rice [201] and maize [202] and the current development of sequencing projects for wheat [203], barley [204] and a number of other crops.
Cereal grains such as rice, wheat and maize provide 60% of the world's nutrition, the rest being represented by barley, coarse grains of legumes along with root crops. These crops account for the majority of end products used for human diets [205] and it is likely that they will still contribute either directly in the human diet or indirectly as animal feed in the next century [200]. Thus, considering both the economical and environmental challenge represented by reducing both the cost and application of N fertilizers, all major maize seed breeding companies such as Monsanto, DuPont-Pioneer and Syngenta are investing in genomic research for improving NUE. Moreover, improvement in yield for most crops over the last 50 years has been estimated to be 40%, due to improvements in cultural practices and 60% due to genetic gains, thus indicating that breeding for improved NUE is still possible [206]. However, to our knowledge, improving NUE either through genetic engineering or marker assisted breeding is still at the stage of proof of concept. Therefore, very little information is currently released from both the private and public sector in consideration of the potential economic value of crop NUE improvement.
However, both on the genetic and physiological side, the identification of key steps involved in the control of NUE from gene expression to metabolic activity remains incomplete. It is likely because the regulatory mechanisms involved in the control of the two components of NUE (N uptake and utilization efficiencies) are species-specific [6]. Moreover, they are subjected to changes or adaptation in a constantly changing soil and aerial environment during plant growth and development that require the taking into account the various genotypic/environment interactions [207].
NUE is controlled by a complex array of physiological, developmental and environmental interactions that are organ and tissue-specific and which are specific to the genotype of a given species. It is therefore essential that a much more extensive survey of a wide range of genotypes covering the genetic diversity of a crop should be performed. This can be achieved using the various available “omics” techniques, combined with agronomic and physiological approaches in order to identify both common and specific elements controlling NUE and plant productivity of plants grown in the field under organic or mineral N fertilizer conditions [208].
Over the last two decades, the construction of cereals that can fix atmospheric N has always been a challenge for plant scientists, in order to reduce the need for mineral N fertilization. Although, the signaling pathway for recognition of N-fixing bacteria is present in cereals, complex genetic modification will be necessary to allow bacterial colonization and nodule organogenesis [209].
At the field level, only agronomic predictive models using the appropriate biogical and environmental parameters [210] should be able to take into account interactions between plants and their environment to obtain an integrated view of the various inputs or outputs, influencing crop NUE [211,212]. One of the main challenges in the future will be to develop reliable decision support systems with the help of sensors [213,214] and biological diagnostic tools in precision agriculture, in order to optimize the application of N under organic or conventional conditions in a more sustainable manner. Moreover, the establishment of such models will need to be scaled up at the ecological level [44], in order to obtain a better understanding as to how N cycling is occurring from organisms to the whole ecosystem [57].
A proposed strategy for integrating multidisciplinary approaches for improving crop NUE is summarized in Figure 4. This strategy highlights the necessity to develop an integrated approach between the public and private sectors to improve our understanding and control of the biological and agronomic basis of NUE in crops of major economical importance. However, the nature of an agronomic trait such as NUE is complex, due to the intervention of multiple elements interacting with each other as a function of both plant development and environmental constraints. Moreover, the interaction between these elements appears to be not only species-specific but also specific to a given genetic background. Therefore, improvement of this understanding will require the development of a multi-disciplinary approach, integrating expertise from fundamental and more applied studies in crop developmental biology, physiology, genomics, genetics, physiology, modeling, agronomy and breeding [212]. In addition, taking advantage of the genetic variability that already exists or that can be created, will provide a valuable contribution to the genetic and physiological dissection of NUE under mineral and organic N nutrition conditions and an evaluation of the genes or group of genes involved. The major breakthrough expected from this multidisciplinary approach will be to provide 1) useful alleles or gene-based markers to breeders for the production of genetically modified plants or for marker assisted selection (MAS) ; 2) predictive biological markers for breeders to improve selection for higher NUE by conventional breeding; 3) tools for farmers to monitor and adjust mineral and/or organic N fertilization for obtaining optimal yields compatible with a strategy for sustainability of the agricultural practices needed to feed the world population, while preserving the environment.



References and Notes
- Erisman, J.W.; Sutton, M.A.; Galloway, J.N.; Klimont, Z.; Winiwarter, W. How a century of ammonia synthesis changed the world. Nat. Geosci. 2008, 1, 636–639. [Google Scholar]
- Galloway, J.N.; Townsend, A.R.; Erisman, J.W.; Bekunda, M.; Cai, Z.; Freney, J.R.; Martinelli, L.A.; Seitzinger, S.P.; Sutton, M.A. Transformation of the nitrogen cycle: Recent trends, questions, and potential solutions. Science 2008, 320, 889–892. [Google Scholar]
- Hoang, V.N.; Alauddin, M. Assessing the eco-environmental performance of agricultural production in OECD countries: The use of nitrogen flows and balance. Nutr. Cycl. Agroecosys. 2010, 87, 353–36. [Google Scholar]
- Mulvaney, R.L.; Khan, S.A.; Ellsworth, T.R. Synthetic nitrogen depleted soil nitrogen: A global dilemma for sustainable cereal production. J. Environ. Qual. 2009, 38, 2295–2314. [Google Scholar]
- Tilman, D.; Cassman, K.G.; Matson, P.A.; Naylor, R.; Polasky, S. Agricultural sustainability and intensive production pratices. Nature 2002, 418, 671–677. [Google Scholar]
- Hirel, B.; Le Gouis, J.; Ney, B.; Gallais, A. The challenge of improving nitrogen use efficiency in crop plants: Towards a more central role for genetic variability and quantitative genetics within integrated approaches. J. Exp. Bot. 2007, 58, 2369–2387. [Google Scholar]
- Gallais, A.; Coque, M. Genetic variation and selection for nitrogen use efficiency in maize: A synthesis. Maydica 2005, 50, 531–537. [Google Scholar]
- Ceccarelli, S. Adaptation to low/high input cultivation. Euphytica 1995, 92, 203–204. [Google Scholar]
- Raun, W.R.; Johnson, G.V. Improving nitrogen use efficiency for cereal production. Agron. J. 1999, 91, 357–363. [Google Scholar]
- Hodge, A.; Robinson, D.; Fitter, A. Are microorganisms more effective than plants at competing for nitrogen? Trends Plant Sci. 2000, 5, 304–308. [Google Scholar]
- Asghari, H.R.; Cavagnaro, T.R. Arbuscular mycorrhizas enhance plant interception of leached nutrients. Funct. Plant Biol. 2011, 38, 219–226. [Google Scholar]
- Al-Redhaiman, K.N. Nitrate accumulation in plants and hazards to man and livestock: A review. J. King Saud Univ. 2000, 12, 143–156. [Google Scholar]
- Umar, A.S.; Iqbal, M. Nitrate accumulation in plants, factors affecting the process, and human health implications. A review. Agron. Sustain. Dev. 2007, 27, 45–57. [Google Scholar]
- Harris, R.C.; Skinner, A.C. Controlling diffuse pollution of groundwater from agriculture and industry. Water Environ. J. 1992, 6, 569–574. [Google Scholar]
- Cameron, S.G.; Schipper, L.A. Nitrate removal and hydraulic performance of organic carbon for use in denitrification beds. Ecol. Eng. 2010, 36, 1588–1595. [Google Scholar]
- Camarguo, J.A.; Alonso, A. Ecological and toxicological effects of inorganic nitrogen pollution in aquatic ecosystems: A global assessment. Environ. Int. 2006, 32, 831–849. [Google Scholar]
- London, J.G. Nitrogen study fertilizes fears of pollution. Nature 2005, 433, 791. [Google Scholar]
- Beman, J.M.; Arrigo, K.; Matson, P.M. Agricultural runoff fuels large phytoplankton blooms in vulnerable areas of the ocean. Nature 2005, 434, 211–214. [Google Scholar]
- Sutton, M.; Howard, C.M.; Erisman, J.W.; Billen, G.; Bleeker, A.; Grennfelt, P.; van Grinsven, H.; Grizetti, B. Assessing our nitrogen inheritance. In The European Nitrogen Assessment. Sources, Effects and Policy Perspectives; Sutton, M.A., Howard, C.M., Erisman, J.W., Billen, G., Bleeker, A., Grennfelt, P., van Grinsven, H., Grizetti, B., Eds.; Cambridge University Press: Cambridge UK, 2011; pp. 1–6. [Google Scholar]
- Ramos, C. Effect of agricultural practices on the nitrogen losses to the environment. Fertilizer Res. 1996, 43, 183–189. [Google Scholar]
- Stulen, I.; Perez-Soba, M.; De Kok, L.J.; Van Der Eerden, L. Impact of gaseous nitrogen deposition on plant functioning. New Phytol. 1998, 139, 61–70. [Google Scholar]
- David, M.; Loubet, B.; Cellier, P.; Mattson, M.; Schjoerring, J.K.; Nemitz, E.; Roche, R.; Riedo, M.; Sutton, M.A. Ammonia sources and sinks in an intensively managed grassland canopy. Biogeosciences 2009, 6, 1903–1915. [Google Scholar]
- Ammonia emission from mineral fertilizers and fertilized crops. Adv. Agron. 2004, 82, 557–622.
- Wang, L.; Xu, Y-C.; Schjoerring, J. K. Seasonal variation in ammonia compensation point and nitrogen pools in beech leaves (Fagus sylvatica). Plant Soil 2011, 343, 51–66. [Google Scholar]
- Fowler, D.B.; Brydon, J. No-till winter wheat production in the Canadian prairies: Timing of nitrogen fixation. Agron. J. 1989, 81, 817–825. [Google Scholar]
- San Francisco, S.; Urrutia, O.; Martin, V.; Peristeropoulos, A.; Garcia-Mina, J.M. Efficiency of urease and nitrification inhibitors in reducing ammonia volatilization from diverse nitrogen fertilizers applied to different soil types and wheat straw mulching. J. Sci. Food. Agr. 2011, 91, 1569–1575. [Google Scholar]
- Olson, R.A. Fertilizers for food production vs energy needs and environmental quality. Ecotox. Environ. Safe. 1977, 1, 311–26. [Google Scholar]
- Reganold, J.P.; Papendick, R.I.; Parr, F.F. Sustainable agriculture. Sci. Am. 1990, 262, 112–120. [Google Scholar]
- Lewandowski, I.; Schmidt, U. Nitrogen, energy and land use efficiencies of miscanthus, reed canary grass and triticale as determined by the boundary line approach. Agr. Ecosyst. Environ. 2006, 112, 335–346. [Google Scholar]
- Andrews, M.; Lea, P.J.; Raven, J.A.; Azevedo, R.A. Nitrogen use efficiency. 3. Nitrogen fixation: Genes and costs. Ann. Appl. Biol. 2009, 155, 1–13. [Google Scholar]
- Fustec, J.; Lesuffleur, F.; Mahieu, S.; Cliquet, J.B. Nitrogen rhizodeposition of legumes. A review. Agron. Sustain. Dev. 2010, 30, 57–66. [Google Scholar]
- Liu, Y.Y.; Wu, L.H.; Baddeley, J.A.; Watson, C.A. Models of biological nitrogen fixation of legumes. A review. Agron. Sustain. Dev. 2011, 31, 155–172. [Google Scholar]
- Herridge, D.F.; People, M.B.; Boddey, R.M. Global inputs of biological nitrogen fixation in agricultural systems. Plant Soil 2008, 311, 1–18. [Google Scholar]
- Andrews, M.; Hodge, S.; Raven, J.A. Positive plant microbial reactions. Ann. Appl. Biol. 2010, 157, 317–320. [Google Scholar]
- Meynard, J.; Sebillotte, M.M. L'élaboration du rendement du blé, base pour l'étude des autres céréales à talles. In Elaboration du Rendement des Principales Cultures Annuelles; Combe, L., Picard, D., Eds.; INRA: Paris, France, 1994; pp. 31–51. [Google Scholar]
- Malézieux, E.; Crozat, Y.; Dupraz, C.; Laurans, M.; Makowski, D.; Ozier-Lafontaine, H.; Rapidel, B.; de Tourdonnet, S.; Valentin-Morison, M. Mixing plant species in cropping systems: Concepts, tools and models. A review. Agron. Sustain. Dev. 2009, 29, 43–62. [Google Scholar]
- Good, A.G.; Shrawat, A.K.; Muench, D.G. Can less yield more? Is reducing nutrient input into the environment compatible with maintaining crop production? Trends Plant Sci. 2004, 9, 597–605. [Google Scholar]
- Rengel, Z. Breeding for better symbiosis. Plant Soil 2002, 245, 147–162. [Google Scholar]
- Raviv, M. The use of mycorrhiza in organically-grown crops under semi arid conditions: A review of benefits, constraints and future challenges. Symbiosis 2010, 52, 65–74. [Google Scholar]
- Kabir, Z.; O'Halloran, I.P.; Hamel, C. Seasonal changes of arbuscular mycorrhizal fungi as affected by tillage practices and fertilization. Plant Soil 1997, 192, 285–293. [Google Scholar]
- Kabir, Z.; O'Halloran, I.P.; Hamel, C. Overwinter survival of arbuscular mycorrhizal hyphae is favored by attachment to roots but diminished by disturbance. Mycorrhiza 1997, 7, 197–200. [Google Scholar]
- Kabir, Z.; Rhamoun, M.; Lazicki, P.; Horwath, W. Cover crops and conservation tillage increase mycorrhizal colonization of corn and tomato roots. Sustainable Agriculture Farming System Project; Volume 9, No. 1. University of California: Davis CA, USA, 2008. Available online: http://safs.ucdavis.edu/newsletter/v09n1/page3.htm (accessed on 23 August 2011). [Google Scholar]
- Aggarwal, N.A.; Gaur, A.; Bhalla, E.; Gupta, S.R. Soil aggregate carbon and diversity of mycorrhiza as affected by tillage practices in a rice-wheat cropping system in northern India. Int. J. Ecol. Environ. Sci. 2010, 36, 233–243. [Google Scholar]
- Kraiser, T.; Gras, D.; Gutièrrez, A.G.; Gonzalez, B.; Gutièrrez, A.R. A holistic view of nitrogen acquisition in plants. J. Exp. Bot. 2011, 62, 1455–1466. [Google Scholar]
- Tikhonovich, I.A.; Provorov, N.A. Microbiology is the basis of sustainable agriculture: An opinion. Ann. Appl. Biol. 2011, 159, 155–168. [Google Scholar]
- Robertson, G.P.; Vitousek, P.M. Nitrogen in Agriculture: Balancing the cost of an essential resource. Annu. Rev. Envir. Resour. 2009, 34, 97–125. [Google Scholar]
- Hooda, P.S.; Edwards, A.C.; Anderson, H.A.; Miller, A. A review of water quality concerns in livestock farming areas. Sci. Total. Environ. 2000, 250, 143–167. [Google Scholar]
- Smith, S.R. Organic contaminants in sewage sludge (biosolids) and their significance for agricultural recycling. Phil. Trans. R. Soc. B. 2009, 367, 4005–4041. [Google Scholar]
- Giller, K.E.; Witter, E.; McGrath, S.P. Heavy metals and soil microbes. Soil Biol. Biochem. 2009, 41, 2031–2037. [Google Scholar]
- Billen, G.; Beusen, A.; Bouwman, L.; Garnier, J. Anthropogenic nitrogen autotrophy and heterotrophy of the world's watersheds: Past, present, and future trends. Global. Biogeochem. Cy. 2010, 24, GB0A11. [Google Scholar]
- Spiertz, J.H.J. Nitrogen, sustainable agriculture and food security. A review. Agron. Sustain. Develop. 2010, 30, 43–55. [Google Scholar]
- Vitosh, M. L.; Johnson, J. W.; Mengel, D. B. Tri-State Fertilizer Recommendations for Corn, Soybean, Wheat and Alfalfa. Extension Bulletin; Ohio State University: Columbus Ohio, USA, 1995; p. E-2567. Available online http://ohioline.osu.edu/e2567/ (accessed 23 August 2011). [Google Scholar]
- Jarvis, S.; Hutchings, N.; Brentrup, F.; Olesen, J.E.; van de Hoek, K.W. Nitrogen flows in farming systems across Europe. In The European Nitrogen Assessment. Sources, Effects and Policy Perspectives; Sutton, M.A., Howard, C.M., Erisman, J.W., Billen, G., Bleeker, A., Grennfelt, P., van Grinsven, H., Grizetti, B., Eds.; Cambridge University Press: Cambridge, UK, 2011; pp. 21–28. [Google Scholar]
- Webster, G.; Embley, T.M.; Freitag, T.E.; Smith, Z.; Prosser, J.I. Links between ammonia oxidizer species composition, functional diversity and nitrification kinetics in grassland soils. Environ. Microbiol. 2005, 7, 676–684. [Google Scholar]
- Le Roux, X.; Poly, F.; Currey, P.; Commeaux, C.; Hai, B.; Nicol, G.W.; Prosser, I.; Schloter, M.; Attard, E.; Klumpp, K. Effect of aboveground grazing on coupling among nitrifier activity, abundance and community structure. ISME J. 2008, 2, 221–232. [Google Scholar]
- Attard, E.; Poly, F.; Commeaux, C.; Laurent, F.; Terada, A.; Smets, B.F.; Recous, S.; Le Roux, X. Shifts between Nitrospira- and Nitrobacter-like nitrite oxidizers underlie the response of soil potential nitrite oxidation to changes in tillage practices. Environ. Microbiol. 2010, 12, 315–326. [Google Scholar]
- Van der Heidjen, M.G.A.; Bardgett, R.D.; van Straalen, N.M. The unseen majority: soil microbes as drivers of plant diversity and productivity in terrestrial ecosystems. Ecol. Lett. 2008, 11, 296–310. [Google Scholar]
- Bouldin, D.R.; Klausner, S.D.; Reid, S.D. Use of nitrogen from manure. In Nitrogen in Crop Production; Hauck, R.D., Ed.; ASA-CSSA-SSSA: Madison, WI, USA, 1984; pp. 221–245. [Google Scholar]
- Maguire, R.O.; Kleinman, P.J.A.; Beegle, D.B. Novel manure management technologies in no-till and forage systems: Introduction to the special series. J. Environ. Qual. 2011, 40, 287–291. [Google Scholar]
- Tyson, S.C.; Cabrera, M.L. Nitrogen mineralization in soils amended with composted and uncomposted poultry litter. Commun. Soil Sci. Plant Anal. 1993, 24, 2361–2374. [Google Scholar]
- Glaser, B.; Lehmann, J.; Zech, W. Ameliorating physical and chemical properties of highly watered soils in the tropics with charcoal—A review. Biol. Fert. Soils 2002, 35, 219–230. [Google Scholar]
- Watson, C.A.; Atkinson, D.; Gosling, P.; Jackson, L.R.; Rayns, F.W. Managing soil fertility in organic farming systems. Soil Use Manag. 2002, 18, 239–247. [Google Scholar]
- Canellas, L.P.; Teixeira Junior, L.R.; Teixeira Junior, L.R.L.; Dobbss, L.B.; Silva, C.A.; Medici, L.O.; Zandonadi, D.B.; Façanha, A.R. Humic acid crossinteractions with root and organic acids. Ann. Appl. Biol. 2008, 153, 157–166. [Google Scholar]
- Trevisan, S.; Francioso, O.; Quaggiotti, S.; Nardi, S. Humic substances biological activity at the plant-soil interface; From environmental aspects to molecular factors. Plant Signal. Behav. 2010, 5, 635–643. [Google Scholar]
- Klucakova, M. Adsorption of nitrate on humic acids studied by flow-through coulometry. Environ. Chem. Lett. 2010, 8, 145–148. [Google Scholar]
- Goodwin, D.C.; Singh, U. Nitrogen balance and crop response to nitrogen in upland and lowland cropping systems. In Understanding Options for Agricultural Production; Tsuji, G.Y., Hoogenboom, G., Thornton, P.K., Eds.; Kluwer: Dordrecht, The Netherlands, 1998; Volume 7, pp. 55–78. [Google Scholar]
- Eulenstein, F.; Werner, A.; Willms, M.; Juszczak, R.; Schlindwein, S.L.; Chijjnicki, B.H.; Olenik, J. Model based scenario studies to optimize the regional nitrogen balance and reduce leaching of nitrate and sulfate of an agricultural water catchment. Nutr. Cycl. Agroecosys. 2008, 82, 33–49. [Google Scholar]
- Bechini, L.; Castoldi, N. Calculating the soil surface nitrogen balance at regional scale: Example application and critical evaluation of tools and data. Ital. J. Agron. 2006, 1, 665–676. [Google Scholar]
- Cherr, C.M.; Scholberg, J.M.S.; McSorley, R. Green manure approaches to crop production: A synthesis. Agron. J. 2006, 98, 302–319. [Google Scholar]
- Burgos, N.R.; Talberg, T.E. Weed control and sweet corn (Zea mays, var. Rugosa) response in no-till system with cover crops. Weed Sci. 1996, 44, 355–361. [Google Scholar]
- Caamal-Maldonado, J.A.; Jimenez-Osornio, J.J.; Torres-Barragan, A.; Anaya, A.L. The use of allelopathic legume cover and mulch species for weed control in cropping systems. Agron. J. 2001, 93, 27–36. [Google Scholar]
- Caswell, E.P.; Defranck, J.; Apt, W.J.; Tang, C.S. Influence of non-host plants on population decline of Rotylenchus reniformis. J. Nematol. 1991, 23, 91–98. [Google Scholar]
- Stevenson, F.C.; Van Kessel, C. The nitrogen and non-nitrogen rotation benefits of pea to succeeding crops. Can. J. Plant Sci. 1996, 76, 735–734. [Google Scholar]
- Unkovich, M.J.; Pate, J.S.; Sanford, P. Nitrogen fixation by annual legumes in Australian Mediterranean agriculture. Aust. J. Agr. Res. 1997, 48, 267–293. [Google Scholar]
- Sullivan, P.S. Overview of cover crops and green manures. National Center for Appropriate Technology Sustainable Agricultural Project; National Center for Appropriate Technology: Butte, MT, USA, 2003; p. IP024. Available online: https://attra.ncat.org/attra-pub/summaries/summary.php?pub=288 (accessed on 23 August 2011). [Google Scholar]
- Rinnofner, T.; Friedel, J.K.; de Kruiff, R.; Freyer, G.P. Effect of catch crops on N dynamics and following crops in organic farming. Agron. Sustain. Dev. 2008, 28, 551–558. [Google Scholar]
- Thorup-Kristensen, K.; Magrid, J.; Stoumann Jesen, L. Catch crops and green manures as biological tools in nitrogen management in temperate zone. Adv. Agron. 2003, 79, 227–302. [Google Scholar]
- Managing Cover Crops Profitability., Third Ed. ed; Handbook Series 9; Sustainable Agriculture Research and Education: Beltsville, MD, USA, 2007; p. 224.
- De Faria, S.M.; Lewis, G.P.; Sprent, J.I.; Sutherland, J.M. Occurrence of nodulation in the leguminosae. New Phytol. 1989, 111, 607–619. [Google Scholar]
- Novak, K. On the efficiency of legume supernodulating mutants. Ann. Appl. Biol. 2010, 157, 321–342. [Google Scholar]
- Graham, P.H.; Vance, C.P. Legumes: Importance and constraints to greater use. Plant Physiol. 2003, 131, 872–877. [Google Scholar]
- Hoffman, C.A.; Carroll, C.R. Can we sustain the biological basis of agriculture? Annu. Rev. Ecol. Syst. 1995, 26, 69–92. [Google Scholar]
- Wang, X.; Pan, Q.; Chen, F.; Yan, X.; Liao, H. Effects of co-inoculation with arbuscular mycorrhizal fungi and rhizobia on soybean growth as related to root architecture and availability of N and P. Mycorrhiza 2011, 21, 173–181. [Google Scholar]
- Brandsaeter, L.O.; Heggen, H.; Riley, H.; Stubhaug, E.; Henriksen, T.M. Winter survival, biomass accumulation and N mineralization of winter annual and biennial legumes sown at various times of the year in northern temperate regions. Eur. J. Agron. 2008, 28, 437–448. [Google Scholar]
- Ranalli, P. Breeding methodologies for the improvement of grain legumes. In Improvement strategies for Leguminosae Biotechnology; Jaiwal, P.K., Singh, R.P., Eds.; Kluwer: Dordrecht, The Netherlands, 2003; pp. 3–21. [Google Scholar]
- Hirel, B.; Harrison, J.; Limami, A. Improvement of Nitrogen Utilization. In Improvement strategies for Leguminosae Biotechnology; Jaiwal, P.K., Singh, R.P., Eds.; Kluwer: Dordecht, The Netherlands, 2003; pp. 201–220. [Google Scholar]
- Valentine, A.J.; Vagner, A.; Benedito, A.; Kandy, Y. Legume nitrogen and soil abiotic stress: From physiology to genomics and beyond. Valentine. In Annual Plant Reviews, Nitrogen Metabolism in Plants in the Post-genomic Era; Foyer, C.H., Zhang, H., Eds.; Wiley-Blackwell: Chichester, UK, 2011; Volume 42, pp. 207–248. [Google Scholar]
- Bourion, V.; Hasan Risvi, S.M.; Fournier, S.; de Lambergue, H.; Galmiche, F.; Marget, P.; Duc, G.; Burstin, J. Genetic dissection of nitrogen nutrition in pea through a QTL approach of root, nodule, and shoot variability. Theor. Appl. Genet. 2010, 212, 71–86. [Google Scholar]
- Zablotowicz, R.M.; Reddy, K.N.; Krutz, L.J.; Gordon, R.E.; Jackson, R.E.; Price, L.D. Can leguminous cover crops partially replace nitrogen fertilization in Mississipi delta cotton production? Int. J. Agron. 2011. [Google Scholar] [CrossRef]
- Rahimizadeh, M.; Kashani, A.; Zare-Feizabadi, A.; Koocheki, A.R.; Nassiri-Mahallati, M. Nitrogen use efficiency of wheat as affected by preceding crop, application rate of nitrogen and crop residues. Aust. J. Crop Sci. 2010, 4, 363–368. [Google Scholar]
- Kuepper, G. Manures for organic crop production. National Center for Appropriate Technology Sustainable Agriculture Project; National Center for Appropriate Technology: Butte, MT, USA, 2003. Available online: http://attra.ncat.org/attra-pub/manures.html (accessed on 23August 2011). [Google Scholar]
- Thomsen, I.K.; Christensen, B.T. Nitrogen conserving potential successive ryegrass catch crops in continuous spring barley. Soil Use Manage. 1999, 15, 195–200. [Google Scholar]
- Möller, K.; Stinner, W.; Leithold, G. Growth, composition, biological N2 fixation and nutrient uptake of a leguminous cover crop mixture and the effect of their removal on field nitrogen balance and nitrate leaching risk. Nutr. Cycl. Agroecosys. 2008, 82, 233–249. [Google Scholar]
- Anderson, R.L. Synergism: A rotation effect of improved growth efficiency. Adv. Agron. 2011, 112, 205–226. [Google Scholar]
- Jackson, L.E.; Pascual, U.; Hodgkin, T. Utilizing and conserving agrobiodiversity in agriculture landscapes. Agr. Ecosys. Environ. 2007, 121, 196–210. [Google Scholar]
- Näsholm, T.; Huss-Danell, K.; Högberg, P. Uptake of glycine by field grown wheat. New Phytol. 2001, 150, 59–63. [Google Scholar]
- Baresel, J.P.; Zimmerman, G.; Reents, H.J. Effect on genotype and environment on N uptake and N partition in organically grown winter wheat (Triticum aestivum L.) in Germany. Euphytica 2008, 163, 347–354. [Google Scholar]
- Loschenberger, F.; Fleck, A.; Grausgruber, H.; Hetzendorfer, H.; Hof, G.; Lafferty, J.; Marn, M.; Neumayer, A.; Pfaffinger, G.; Birschitzsky, J. Breeding for organic agriculture: the example of winter wheat in Austria. Euphytica 2008, 163, 469–480. [Google Scholar]
- Reeve, J.R.; Smith, J.L.; Carpenter-Boggs, L.; Reganold, J.P. Glycine, nitrate and ammonium uptake by classic and modern wheat varieties in a short-term microcosm study. Biol. Fertil. Soils 2009, 45, 723–732. [Google Scholar]
- Kumar, A.; Gupta, N.; Gupta, A.K.; Gaur, V.K. Identification of biomarkers for determining genotypic potential of nitrogen-use-efficiency and optimization of the nitrogen inputs in crop plants. J. Crop Sci. Biotech. 2009, 12, 183–194. [Google Scholar]
- Salsac, L.; Chaillou, S.; Morot-Gaudry, J.F.; Lesaint, C.; Jolivet, E. Nitrate and ammonium nutrition in plants. Plant Physiol. Biochem. 1987, 25, 805–812. [Google Scholar]
- Näsholm, T.; Kielland, K.; Ganeteg, U. Uptake of organic nitrogen by plants. New Phytol. 2009, 182, 31–48. [Google Scholar]
- Miller, A.J.; Fan, X.; Orsel, M.; Smith, S.J.; Wells, D.M. Nitrate transport and signaling. J. Exp. Bot. 2007, 58, 2297–2306. [Google Scholar]
- Dechorgnat, J.; Nguyen, C.T.; Armengaud, P.; Jossier, M.J.; Diatloff, E.; Filleur, S.; Daniel-Vedele, F. From the soil to the seeds: The long journey of nitrate in plants. J. Exp. Bot. 2011, 62, 1349–1359. [Google Scholar]
- Kaiser, W.M.; Planchet, E.; Rümer, S. Nitrate reductase and nitric oxide. In Annual Plant Reviews, Nitrogen Metabolism in Plants in the Post-genomic Era; Foyer, C.H., Zhang, H., Eds.; Wiley-Blackwell: Chichester, UK, 2011; Volume 42, pp. 127–146. [Google Scholar]
- Sétif, P.; Hirasawa, M.; Cassan, N.; Lagoutte, B.; Tripathy, J.N.; Knaff, D.B. New insights into the catalytic cycle of plant nitrite reductase. Electron transfer kinetics and charge storage. Biochemistry 2009, 48, 2828–2838. [Google Scholar]
- Ludewig, U.; Neuhäuser, B.; Dynowski, M. Molecular mechanisms of ammonium transport and accumulation in plants. FEBS Lett. 2007, 581, 2301–2308. [Google Scholar]
- Mae, T. Physiological nitrogen efficiency in rice: Nitrogen utilization, photosynthesis and yield. In Plant Nutrition for Sustainable Food Production and Environment; Ando, T., Fujita, K., Mae, T., Matsumoto, H., Mori, S., Sekiya, J., Eds.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 1997; pp. 51–60. [Google Scholar]
- Hirel, B.; Lea, P.J. The molecular genetics of nitrogen use efficiency in crops. In The Molecular and Physiological Basis of Nutrient Use Efficiency in Crops; Hawkesford, M.J., Barraclough, P.B., Eds.; Wiley-Blackwell: Chichester, UK, 2011; pp. 139–164. [Google Scholar]
- Schimel, J.P.; Chapin, F.S. Tundra plant uptake of amino acid and NH4+ nitrogen in situ: Plants compete well for amino acid N. Ecology 1996, 77, 2142–2147. [Google Scholar]
- Näsholm, T.; Ekblad, A.; Nordin, R.; Giesler, M.; Hogberg, M.; Hogberg, P. Boreal forest plants take up organic nitrogen. Nature 1998, 392, 914–916. [Google Scholar]
- Näsholm, T.; Huss-Danell, K.; Högberg, P. Uptake of organic nitrogen in the field by four agriculturally important plant species. Ecology 2000, 81, 1155–1161. [Google Scholar]
- Harrison, K.A.; Bol, R.; Bardgett, R.D. Do plant species with different growth strategies vary in their ability to compete with soil microbes for chemical forms of nitrogen? Soil Biol. Biochem. 2008, 40, 228–237. [Google Scholar]
- Biernath, C.; Fischer, H.; Kuzyakov, Y. Root uptake of N-containing and N-free low molecular weight organic substances by maize. A 14C/15N tracer study. Soil Biol. Biochem. 2008, 40, 2237–2245. [Google Scholar]
- Paugfoo-Lonhienne, C.; Lonhienne, T.G.A.; Rentch, D.; Robinson, N.; Christie, M.; Webb, R.I.; Gamage, H.K.; Caroll, B.J.; Schenk, P.M.; Schmidt, S. Plants can use protein as a nitrogen source without assistance from other organisms. Proc. Natl. Acad. Sci. USA 2007, 105, 4524–4529. [Google Scholar]
- Tan, X.W.; Ikeda, H.; Oda, M. The absorption, translocation, and assimilation of urea, nitrate or ammonium in tomato plants at different plant growth stages in hydroponic culture. Sci. Hortic. Amsterdam 2000, 84, 275–283. [Google Scholar]
- Kojima, S.; Bohner, A.; von Wirén, N. Molecular mechanisms of urea transport in plants. J. Membrane. Biol. 2006, 212, 83–91. [Google Scholar]
- Kojima, S.; Bohner, A.; Gassert, B.; Yuan, L.; von Wirén, N. AtDUR3 represents the major transporter for high-affinity urea transport across the plasma membrane of nitrogen-deficient Arabidopsis roots. Plant J. 2007, 52, 30–40. [Google Scholar]
- Witte, C.P. Urea metabolism in plants. Plant Sci. 2010, 180, 431–438. [Google Scholar]
- Smith, S.E.; Read, D.J. Mycorrhizal Symbiosis, 3rd ed.; Academic Press: London, UK, 2008; p. 800. [Google Scholar]
- Hodge, A.; Helgason, T.; Fitter, A.H. Nutritional ecology of arbuscular mycorrhizal fungi. Fungal Ecol. 2010, 3, 267–273. [Google Scholar]
- Peay, K.G.; Bidartondo, M.I.; Arnold, A.E. Not every fungus is everywhere: Scaling to the biogeography of fungal-plant interactions across roots, shoots and ecosystems. New Phytol. 2010, 185, 878–882. [Google Scholar]
- Tanaka, Y.; Yano, K. Nitrogen delivery to maize via myccorhizal hyphae depends on the form of N supplies. Plant Cell Environ. 2005, 28, 1247–1254. [Google Scholar]
- Jackson, L.E.; Burger, M.; Cavagnaro, T.R. Nitrogen transformation and ecosystem services. Annu. Rev. Plant. Biol. 2008, 59, 341–363. [Google Scholar]
- Miransari, M.; Bahrami, H.A.; Rejali, F.; Malakouti, M.J. Using arbuscular mycorrhiza to reduce the stressful effects of soil compaction on wheat (Triticum aestivum L.) growth. Soil. Biol. Biochem. 2008, 40, 1197–1206. [Google Scholar]
- Miransari, M.; Rejali, F.; Bahrami, H.A.; Malakouti, M.J. Effect of soil compaction and arbuscular mycorrhiza on corn (Zea mays L.) nutrient uptake. Soil. Till. Res. 2009, 103, 282–290. [Google Scholar]
- Daei, G.; Ardakani, M.; Rejali, F.; Teimuri, S.; Miransari, M. Alleviation of salinity on wheat yield, yield components, and nutrient uptake using arbuscular myccorhizal fungi under field condition. J. Plant Physiol. 2009, 166, 617–625. [Google Scholar]
- Miransari, M. Arbuscular mycorrhizal fungi and nitrogen uptake. Arch. Microbiol. 2011, 193, 77–81. [Google Scholar]
- Tobar, R.; Azcon, R.; Barea, J.M. Improved nitrogen uptake and transport from 15N-labelled nitrate by external hyphae of arbuscular mycorrhiza under water stressed conditions. New Phytol. 1994, 126, 119–122. [Google Scholar]
- Tian, C.; Kasiborski, B.; Koul, R.; Mammers, P.J.; Bucking, H.; Shachar-Hill, Y. Regulation of the nitrogen transfer pathway in the arbuscular mycorrhizal symbiosis: Gene characterization and the coordination of expression with nitrogen flux. Plant Physiol. 2010, 153, 1175–1187. [Google Scholar]
- Frey, B.; Schüpp, H. Transfer of symbiotically fixed nitrogen from berseem (Trifolium alexandrinum to maize via vesicular arbuscular mychorrhizal hyphae. New Phytol. 1992, 122, 447–454. [Google Scholar]
- Bonfante, P.; Anca, I.A. Plants, mycorrhizal fungi, and bacteria: A network of interactions. Annu. Rev. Microbiol. 2009, 63, 363–383. [Google Scholar]
- Bonfante, P.; Genre, A. Mechanisms underlying beneficial plant-fungus interactions in mycorrhizal symbiosis. Nat. Commun. 2010, 1, 1–11. [Google Scholar]
- Lea, P.J.; Miflin, B.J. Nitrogen assimilation and its relevance to crop improvement. In Annual Plant Reviews, Nitrogen Metabolism in Plants in the Post-genomic Era; Foyer, C.H., Zhang, H., Eds.; Wiley-Blackwell: Chichester, UK, 2011; Volume 42, pp. 1–40. [Google Scholar]
- Suzuki, A.; Knaff, D.B. Glutamate synthase: Structural, mechanistic and regulatory properties, and role in the amino acid metabolism. Photosynth. Res. 2005, 83, 191–217. [Google Scholar]
- Hirel, B.; Lea, P.J. Amino acid metabolism. In Plant Nitrogen; Lea, P.J., Morot-Gaudry, J.F., Eds.; INRA, Springer-Verlag: Berlin, Germany, 2001; pp. 79–99. [Google Scholar]
- Taira, M.; Valtersson, U.; Burkhardt, B.; Ludwig, R.A. Arabidopis thaliana GLN2-encoded glutamine synthetase is dual targeted to leaf mitochondria and chloroplasts. Plant Cell 2004, 16, 2048–2058. [Google Scholar]
- Dubois, F.; Brugière, N.; Sangwan, R.S.; Hirel, B. Localization of tobacco cytosolic glutamine synthetase enzymes and the corresponding transcripts shows organ- and cell-specific patterns of protein synthesis and gene expression. Plant Mol. Biol. 1996, 31, 803–817. [Google Scholar]
- Cren, M.; Hirel, B. Glutamine synthetase in higher plants: Regulation of gene and protein expression from the organ to the cell. Plant Cell Physiol. 1999, 40, 1187–1193. [Google Scholar]
- Lea, P.J.; Ireland, R.J. Nitrogen metabolism in higher plants. In Plant Amino Acids; Singh, B.K., Ed.; Dekker M.: New York, NY, USA, 1999; pp. 1–47. [Google Scholar]
- Aubert, S.; Bligny, R.; Douce, R.; Ratcliffe, R.G.; Roberts, J.K.M. Contribution of glutamate dehydrogenase to mitochondrial metabolism studied by 13C and 31P nuclear magnetic resonance. J. Exp. Bot. 2001, 52, 37–45. [Google Scholar]
- Labboun, S.; Tercé-Laforgue, T.; Roscher, A.; Bedu, M.; Restivo, F.M.; Velanis, C.N.; Skopelitis, D.S.; Moshou, P.N.; Roubelakis-Angelakis, K.A.; Suzuki, A. Resolving the role of plant glutamate dehydrogenase: I. In vivo real time nuclear magnetic resonance spectroscopy experiments. Plant Cell Physiol. 2009, 50, 1761–1773. [Google Scholar]
- Skopelitis, D.S.; Paranychiankis, N.V.; Paschalidis, K.A.; Plianokis, E.D.; Delis, I.D.; Yakoumakis, D.I.; Kouvarakis, A.; Papadakis, E.D.; Stephanou, E.G.; Roubelakis-Angelakis, K.A. Abiotic stress generates ROS that signal expression of anionic glutamate dehydrogenase to form glutamate for proline synthesis in tobacco and grapevine. Plant Cell 2006, 18, 2767–2781. [Google Scholar]
- Masclaux, C.; Quilleré, I.; Gallais, A.; Hirel, B. The challenge of remobilisation in plant nitrogen economy. A survey of physio-agronomic and molecular approaches. Ann. Appl. Biol. 2001, 138, 69–81. [Google Scholar]
- Stitt, M.; Müller, C.; Matt, P.; Gibon, Y.; Carillo, P.; Morcuende, R.; Sheible, W.R.; Krapp, A. Steps towards an integrated view of nitrogen metabolism. J. Exp. Bot. 2002, 53, 959–970. [Google Scholar]
- Tercé-Laforgue, T.; Dubois, F.; Ferrario-Mery, S.; Pou de Crecenzo, M.A.; Sangwan, R.; Hirel, B. Glutamate dehydrogenase of tobacco (Nicotiana tabacum L.) is mainly induced in the cytosol of phloem companion cells when ammonia is provided either externally or released during photorespiration. Plant Physiol. 2004, 136, 4308–4317. [Google Scholar]
- Morot Gaudry, J.F.; Job, D.; Lea, P.J. Amino acid metabolism. In Plant Nitrogen; Lea, P.J., Morot Gaudry, J.F., Eds.; INRA Springer-Verlag: Berlin, Germany, 2001; pp. 167–211. [Google Scholar]
- Lea, P.J.; Azevedo, R.A. Nitrogen use efficiency. 2. Amino acid metabolism. Ann. Appl. Biol. 2007, 151, 269–275. [Google Scholar]
- Galili, S.; Amir, R.; Galili, G. Genetic engineering of amino acids in plants. Adv. Plant Biochem. Mol. Biol. 2008, 1, 49–80. [Google Scholar]
- Gutiérrez, R.A.; Lejay, L.V.; Dean, A.; Chiaromonte, F.; Shasha, D.E.; Coruzzi, G.M. Qualitative network models and genome-wide expression data define carbon/nitrogen-responsive molecular machines in Arabidopsis. Genome Biol. 2007, 8, R7. [Google Scholar]
- Coruzzi, G.M.; Burga, A.R.; Katari, M.S.; Gutiérrez, R.A. Systems biology: Principles and applications in plant research. In Annual Plant Reviews, Plant Systems Biology; Coruzzi, G.M., Guttiérez, R.A., Eds.; Wiley-Blackwell: Chichester, UK, 2009; Volume 35, pp. 3–40. [Google Scholar]
- Thum, K.E.; Shin, M.J.; Gutiérrez, R.A.; Mukherjee, I.; Katari, M.S.; Nero, D.; Shasha, D.; Coruzzi, G.M. An integrated genetic, genomic and systems approach defines gene networks regulated by the interaction of light and carbon signalling pathways in Arabidopsis. BMC Syst. Biol. 2008, 2, 31. [Google Scholar]
- Andrews, M.; Lea, P.J.; Raven, J.A.; Lindsey, K. Can genetic manipulation of plant nitrogen assimilation enzymes result in increased crop yield and greater N-use efficiency? An assessment. Ann. Appl. Biol. 2004, 145, 25–40. [Google Scholar]
- Fuentes, S.I.; Alen, D.J.; Ortiz-Lopez, A.; Hernandez, G. Overexpression of cytosolic glutamine synthetase increases photosynthesis and growth at low nitrogen concentrations. J. Exp. Bot. 2001, 52, 1071–1081. [Google Scholar]
- Oliveira, I.C.; Brears, T.; Knight, T.J.; Clark, A.; Coruzzi, G.M. Overexpression of cytosolic glutamine synthetase. Relation to nitrogen, light, and photorespiration. Plant Physiol. 2002, 129, 1170–1180. [Google Scholar]
- Jing, Z.P.; Gallardo, F.; Pascual, M.B.; Sampalo, R.; Romero, J.; Torres de Vavarra, A.; Canovas, F.M. Improved growth in a field trial of transgenic hybrid poplar overexpressing glutamine synthetase. New Phytol. 2004, 164, 137–145. [Google Scholar]
- Man, H.M.; Boriel, R.; El-Khatib, R.; Kirby, E.G. Characterization of transgenic poplar with ectopic expression of pine cytosolic glutamine synthetase under conditions of varying nitrogen ability. New Phytol. 2005, 167, 31–39. [Google Scholar]
- Habash, D.Z.; Massiah, A.J.; Rong, H.L.; Wallsgrove, R.M.; Leigh, R.A. The role of cytosolic glutamine synthetase in wheat. Ann. Appl. Biol. 2001, 138, 83–89. [Google Scholar]
- Martin, A.; Lee, J.; Kichey, T.; Gerentes, D.; Zivy, M.; Tatou, C.; Balliau, T.; Valot, B.; Davanture, M.; Dubois, F.; et al. Two cytosolic glutamine synthetase isoforms of maize (Zea mays L.) are specifically involved in the control of grain production. Plant Cell 2006, 18, 3252–3274. [Google Scholar]
- Brauer, E.K.; Rochon, A.; Bi, Y.M.; Bozzo, G.G.; Rothstein, S.J.; Shelp, B. Reappraisal of nitrogen use efficiency in rice overexpressing glutamine synthetase 1. Physiol. Plantarum. 2011, 141, 361–372. [Google Scholar]
- Vincent, R.; Fraisier, V.; Chaillou, S.; Limami, M.A.; Deléens, E.; Phillipson, B.; Douat, C.; Boutin, J.P.; Hirel, B. Overexpression of a soybean gene encoding cytosolic glutamine synthetase in shoots of transgenic Lotus corniculatus L. plants triggers changes in ammonium assimilation and plant development. Planta 1997, 201, 424–433. [Google Scholar]
- Good, A.G.; Johnson, S.J.; De Pauw, M.; Carroll, R.T.; Savodiv, N.; Vidmar, J.; Lu, Z.; Taylor, G.; Stroeher, V. Engineering nitrogen use efficiency with alanine aminotransferase. Can. J. Bot. 2007, 85, 252–262. [Google Scholar]
- Shrawat, A.K.; Carroll, R.T.; DePauw, M.; Taylor, G.J.; Good, A.G. Genetic engineering of improved nitrogen use efficiency in rice by the tissue specific expression of alanine amino-transferase. Plant Biotechnol. J. 2008, 6, 722–732. [Google Scholar]
- Ameziane, R.; Bernhard, K.; Lightfoot, D. Expression of the bacterial gdhA gene encoding a NADPH-glutamate dehydrogenase in tobacco affects plant growth and development. Plant Soil 2000, 221, 47–57. [Google Scholar]
- Bi, Y.M.; Kant, S.; Clark, J.; Gidda, S.; Ming, F.; Xu, J.; Rochon, A.; Shelp, B.J.; Hao, L.; Zhao, R.; et al. Increased nitrogen use efficiency in transgenic rice plants over-expressing a nitrogen-responsive early nodulin gene identified from rice expression profiling. Plant Cell Environ. 2009, 32, 1749–1760. [Google Scholar]
- Chichkova, S.; Arellano, J.; Vance, C.P.; Hernandez, G. Transgenic tobacco plants that overexpress alfalfa NADH-glutamate synthase have higher carbon and nitrogen content. J. Exp. Bot. 2001, 52, 2079–2087. [Google Scholar]
- Yamaya, T.; Obara, M.; Nakajima, H.; Sasaki, S.; Hayakawa, T.; Sato, T. Genetic manipulation and quantitative trait loci mapping for nitrogen recycling in rice. J. Exp. Bot. 2002, 53, 917–925. [Google Scholar]
- Tabuchi, M.; Abiko, T.; Yamaya, T. Assimilation of ammonium ions and reutilization of nitrogen in rice (O. sativa L.). J. Exp. Bot. 2007, 58, 2319–2327. [Google Scholar]
- Yanagisawa, S.; Akiyama, A.; Kisaka, H.; Uchimiya, H.; Miwa, T. Metabolic engineering with Dof1 transcription factor in plants: Improved nitrogen assimilation and growth under low-nitrogen conditions. Proc. Natl. Acad. Sci. USA 2004, 101, 7833–7838. [Google Scholar]
- Fonder, N.; Heens, B.; Xanthoulis, D. Optimisation de la fertlisation azotée de cultures industrielles légumières sous irrigation. Biotechnol. Agron. Soc. Environ. 2010, 14, 103–111. [Google Scholar]
- Quilléré, I.; Dufossé, C.; Roux, Y.; Foyer, C.H.; Caboche, M.; Morot-Gaudry, J.F. The effects of deregulation of NR gene expression on growth and nitrogen metabolism of Nicotiana plumbaginifolia plants. J. Exp. Bot. 1994, 278, 1205–1211. [Google Scholar]
- Djennane, S.; Chauvin, J.E.; Quilleré, I.; Meyer, C.; Chupeau, Y. Introduction and expression of a deregulated tobacco nitrate reductase gene in potato lead to highly reduced nitrate levels in transgenic tubers. Transgenic Res. 2002, 11, 175–184. [Google Scholar]
- Djennane, S.; Quilleré, I.; Leydecker, M.-T.; Meyer, C.; Chauvin, J.E. Expression of a deregulated tobacco nitrate reductase gene in potato increases biomass production and decreases nitrate concentration in all organs. Planta 2004, 219, 884–893. [Google Scholar]
- Curtis, I.S.; Power, J.B.; de Laat, A.M.M.; Caboche, M.; Davey, M.R. Expression of a chimeric nitrate reductase gene in transgenic lettuce reduces nitrate in leaves. Plant Cell Rep. 1999, 18, 889–896. [Google Scholar]
- Xu, Y. Quantitative trait loci: Separating, pyramiding, and cloning. Plant Breed. Rev. 1997, 15, 85–139. [Google Scholar]
- Yu, J.; Buckler, E.S. Genetic association mapping and genome organization of maize. Cur. Opin. Biotech. 2006, 17, 155–160. [Google Scholar]
- Salvi, S.; Tuberosa, R. Cloning QTLs in Plants. In Genomics-Assisted Crop Improvement; Varshney, R.K., Tuberosa, R., Eds.; Springer: Dordrecht, The Netherlands, 2007; Volume 1, pp. 207–226. [Google Scholar]
- Agrama, H.A.S.; Zacharia, A.G.; Said, F.B.; Tuinstra, M. Identification of quantitative trait loci for nitrogen use efficiency in maize. Mol. Breed. 1999, 5, 187–195. [Google Scholar]
- Bertin, P.; Gallais, A. Physiological and genetic basis of nitrogen use efficiency in maize. II. QTL detection and coincidences. Maydica 2001, 46, 53–68. [Google Scholar]
- Hirel, B.; Gallais, A.; Bertin, P.; Quillere, I.; Bourdoncle, W.; Attagan, C.; Dellay, C.; Gouy, A.; Cadiou, S.; Retaillau, C.; et al. Towards a better understanding of the genetic and physiological basis for nitrogen use efficiency in maize. Plant Physiol. 2001, 125, 1258–1270. [Google Scholar]
- Gallais, A.; Hirel, B. An approach of the genetics of nitrogen use efficiency in maize. J. Exp. Bot. 2004, 55, 295–306. [Google Scholar]
- Zhang, N.; Gibon, Y.; Gur, A.; Chen, C.; Lepak, N.; Höne, M.; Zhang, Z.; Kroon, D.; Tschoep, H.; Stitt, M.; et al. Fine quantitative trait loci mapping of carbon and nitrogen metabolism enzyme activities and seedling biomass in the maize IBM mapping population. Plant Physiol. 2010, 154, 1753–1765. [Google Scholar]
- Obara, M.; Kajiura, M.; Fukuta, Y.; Yano, M.; Hayashi, M.; Yamaya, T.; Sato, T. Mapping of QTLs associated with cytosolic glutamine synthetase and NADH-glutamate synthase in rice (Oryza sativa L.). J. Exp. Bot. 2001, 52, 1209–1217. [Google Scholar]
- Tabuchi, M.; Sugiyama, T.; Ishiyama, K.; Inoue, E.; Sato, T.; Takahashi, H.; Yamaya, T. Severe reduction in growth and grain filling of rice mutants lacking OsGS1;1, a cytosolic glutamine synthetase 1;1. Plant J. 2005, 42, 641–655. [Google Scholar]
- Fontaine, J.X.; Ravel, C.; Pageau, K.; Heumez, E.; Dubois, F.; Hirel, B.; Le Gouis, J. A quantitative genetic study for elucidating the contribution of glutamine synthetase, glutamate dehydrogenase and other nitrogen-related physiological traits to the agronomic performance of common wheat. Theor. Appl. Genet. 2009, 119, 645–662. [Google Scholar]
- Quraishi, U.M.; Abrouk, M.; Murat, F.; Pont, C.; Foucrier, S.; Demaizieres, G.; Confolent, C.; Rivière, N.; Charmet, G.; Paux, E.; et al. Cross-genome map based dissection of a nitrogen use efficiency ortho-meta QTL in bread wheat unravels concerted cereal genome evolution. Plant J. 2011, 65, 745–756. [Google Scholar]
- Plomion, C.; Bahrmann, N.; Costa, P.; Frigério, J.M.; Gerber, S.; Gion, J.M.; Lalanne, C.; Madur, D.; Pionneau, C. Proteomics for genetic and physiological studies in forest trees: Application in maritime pine. In Molecular Genetics and Breeding of Forest Trees; Kumar, S., Fladung, M., Eds.; Haworth Press: New York, NY, USA, 2004; pp. 53–80. [Google Scholar]
- Lammerts van Buren, E.T.; Jones, S.S.; Tamn, L.; Murphy, K.M.; Myers, J.R.; Leifert, C.; Mesmer, M.M. The need to breed crop varieties suitable for organic farming, using wheat, tomato and broccoli as examples: A review. NJAS Wageningen J. Life Sci. 2010. [Google Scholar] [CrossRef]
- Hawkesford, M.J.; Howarth, J.R. Transcriptional profiling approaches for studying nitrogen use efficiency. In Annual Plant Reviews, Nitrogen Metabolism in Plants in the Post-genomic Era; Foyer, C.H., Zhang, H., Eds.; Wiley-Blackwell: Chichester, UK, 2011; Volume 42, pp. 41–62. [Google Scholar]
- Messmer, M.M.; Burger, H.; Schmidt, W.; Geiger, H.H. Importance of appropriate selection environments for breeding maize adapted to organic farming systems. Tagung der Vereinigung der Pflanzenzücher und Saatgutkaufleute Österreichs 2009, 1–3. [Google Scholar]
- Krapp, A.; Truong, H.N. C/N interaction in model plant species. In Enhancing the Efficiency of Nitrogen Utilisation in Plants; Goyal, S.S., Tischner, R., Basra, A.S., Eds.; Haworth Press: Binghampton, NY, USA, 2005; pp. 127–173. [Google Scholar]
- Gutiérrez, R.A.; Gifford, M.L.; Poultney, C.; Wang, R.; Shasha, D.E.; Coruzzi, G.M.; Crawford, N.M. Insights into the genomic nitrate response using genetics and the sungear software system. J. Exp. Bot. 2007, 58, 2359–2367. [Google Scholar]
- Cañas, R.A.; Quilleré, I.; Christ, A.; Hirel, B. Nitrogen metabolism in the developing ear of maize (Zea mays L.): Analysis of two lines contrasting in their mode of nitrogen managementX. J. Exp. Bot. 2009, 184, 340–352. [Google Scholar]
- Meyer, R.C.; Steinfath, M.; Lisec, J.; Becher, M.; Witucha-Wall, H.; Törjék, O.; Fienh, O.; Eckardt, A.; Willmitzer, L.; Selbig, J.; et al. The metabolic signature related to high plant growth rate in Arabidopsis thaliana. Proc. Nat. Acad. Sci. USA 2007, 104, 4759–4664. [Google Scholar]
- Lisec, J.; Meyer, R.C.; Steinfath, M.; Redestig, H.; Becher, M.; Witucka-Wall, H.; Fienh, O.; Törjék, O.; Selbig, J.; Altman, T.; et al. Identification of metabolic and biomass QTL in Arabidopsis thaliana in a parallel analysis of RIL and Il populations. Plant J. 2008, 53, 960–972. [Google Scholar]
- Radrich, K.; Tsuruoka, Y.; Dobson, P.; Gevorgyan, A.; Swaitson, N.; Schwartz, J.M. Integration of metabolic databases for the reconstruction of genome-scale metabolic networks. BMC Syst. Biol. 2010, 4, 1–16. [Google Scholar] [CrossRef]
- De Oliveira Dal'Molin, C.G.; Quek, L.E.; Palfreyman, R.W.; Brumbley, S.M.; Nielsen, L.K. AraGEM, a genome-scale reconstruction of the primary metabolic network in Arabidopsis. Plant Physiol. 2010, 152, 579–589. [Google Scholar]
- Coque, M.; Martin, A.; Veyrieras, J.B.; Hirel, B.; Gallais, A. Genetic variation for N-remobilization and postsilking N-uptake in a set of maize recombinant inbred lines. 3. QTL detection and coincidences. Theor. Appl. Genet. 2008, 117, 729–747. [Google Scholar]
- Xu, Y.; Crouch, J.H. Marker-assisted selection in plant breeding: From publication to practice. Crop Sci. 2008, 48, 391–407. [Google Scholar]
- Hirel, B.; Le Gouis, J.; Bernard, M.; Perez, P.; Falque, M.; Quétier, F.; Joets, J.; Montalent, P.; Rogwoski, P.; Murigneux, A.; et al. Genomics and plant breeding: Maize and wheat. In Functional Plant Genomics; Morot-Gaudry, J.-F., Lea, P.J., Briat, J.-F., Eds.; Science Publishers: Enfield, NH, USA, 2007; pp. 614–635. [Google Scholar]
- Goff, S.A.; Ricke, D.; Lan, T.H.; Presting, G.; Wang, R.; Dunn, M.; Glazebrook, J.; Session, A.; Oeller, P.; Varma, H.; et al. A draft sequence of the rice genome (Oryza sativa L. ssp. japonica). Science 2002, 296, 92–100. [Google Scholar]
- Schnable, P.S.; Ware, D.; Fulton, R.S.; Stein, J.C.; Wei, F.; Pasternak, S.; Liang, C.; Khang, J.; Fulton, L.; Graves, T.A.; et al. The B73 maize genome: Complexity, diversity and dynamics. Science 2009, 326, 1112–1115. [Google Scholar]
- International Wheat Genome Organization. Available online: http://www.wheatgenome.org/ (accessed on 1 September 2011).
- International Barley Sequencing Consortium. Available online: http://barleygenome.org/ (accessed on 1 September 2011).
- Food and Agriculture Organization. Available online: http://www.fao.org/worldfoodsituation/wfs-home/csdb/en/(accessed on 1 September 2011).
- Edgerton, M.D. Increasing crop productivity to meet global needs for feed, food and fuel. Plant Physiol. 2009, 149, 7–13. [Google Scholar]
- Pilbeam, D.J. The utilization of nitrogen by plants: A whole plant perspective. In Annual Plant Reviews, Nitrogen Metabolism in Plants in the Post-genomic Era; Foyer, C.H., Zhang, H., Eds.; Wiley-Blackwell: Chichester, UK, 2011; Volume 42, pp. 305–352. [Google Scholar]
- Cañas, R.A.; Amiour, N.; Quilleré, I.; Hirel, B. An integrated statistical analysis of the genetic variability of nitrogen metabolism in the ear of three maize inbred lines (Zea mays L.). J. Exp. Bot. 2010, 62, 2309–2318. [Google Scholar]
- Charpentier, M.; Oldroyd, G. How close are we to nitrogen-fixing cereals? Curr. Opin. Plant Biol. 2010, 13, 556–564. [Google Scholar]
- Tremblay, M.; Wallach, D. Comparison of parameter estimation methods for crop models. Agronomie 2004, 24, 351–365. [Google Scholar]
- McCown, R.L.; Hammer, G.L.; Hargreaves, J.N.G.; Holzworth, D.P.; Freebairn, D.M. APSIM: A novel software system for model development, model testing and simulation in agricultural systems research. Agr. Syst. 1996, 50, 255–271. [Google Scholar]
- Stark, C.H.; Richards, K.G. The continuing challenge of agricultural nitrogen loss to the environment in the context of global change and advancing research. Dyn. Soil. Dyn. Plant 2008, 2, 1–12. [Google Scholar]
- Hammer, G.L.; Kropff, M.J.; Sinclair, T.R.; Porter, J.R. Future contributions of crop modeling—from heuristics and supporting decision-making to understanding genetic regulation and aiding crop improvement. Eur. J. Agron. 2002, 18, 15–31. [Google Scholar]
- Samborski, S.M.; Tremblay, N.; Fallon, E. Strategies to make use of plant sensors-based diagnostic information for nitrogen recommendations. Agron. J. 2009, 101, 800–816. [Google Scholar]
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Hirel, B.; Tétu, T.; Lea, P.J.; Dubois, F. Improving Nitrogen Use Efficiency in Crops for Sustainable Agriculture. Sustainability 2011, 3, 1452-1485. https://doi.org/10.3390/su3091452
Hirel B, Tétu T, Lea PJ, Dubois F. Improving Nitrogen Use Efficiency in Crops for Sustainable Agriculture. Sustainability. 2011; 3(9):1452-1485. https://doi.org/10.3390/su3091452
Chicago/Turabian StyleHirel, Bertrand, Thierry Tétu, Peter J. Lea, and Frédéric Dubois. 2011. "Improving Nitrogen Use Efficiency in Crops for Sustainable Agriculture" Sustainability 3, no. 9: 1452-1485. https://doi.org/10.3390/su3091452
APA StyleHirel, B., Tétu, T., Lea, P. J., & Dubois, F. (2011). Improving Nitrogen Use Efficiency in Crops for Sustainable Agriculture. Sustainability, 3(9), 1452-1485. https://doi.org/10.3390/su3091452



