Advancing the Use of Secondary Inputs in Geopolymer Binders for Sustainable Cementitious Composites: A Review
Abstract
: Because of concerns over the construction industry's heavy use of cement and the general dissatisfaction with the performance of building envelopes with respect to durability, there is a growing demand for a novel class of “green” binders. Geopolymer binders have re-emerged as binders that can be used as a replacement for Portland cement given their numerous advantages over the latter including lower carbon dioxide emissions, greater chemical and thermal resistance, combined with enhanced mechanical properties at both normal and extreme exposure conditions. The paper focuses on the use of geopolymer binders in building applications. It discusses the various options for starting materials and describes key engineering properties associated with geopolymer compositions that are ideal for structural applications. Specific properties, such as compressive strength, density, pore size distribution, cumulative water absorption, and acid resistance, are comparable to the specifications for structures incorporating conventional binders. This paper presents geopolymer binders, with their three dimensional microstructure, as material for structural elements that can be used to advance the realization of sustainable building systems.1. Introduction
The construction industry is under increasing pressure to adopt green practices in the delivery of built infrastructure. There are specific concerns over atmospheric CO2 concentrations, which at 390 ppm reached record breaking levels (U.N. Intergovernmental Panel on Climate Change [1]. Major CO2-producing sectors, such as power generation, transportation, oil refining, and manufacturing of steel and concrete are under pressure to adopt measures that would drastically reduce the global CO2 emission rate by 2030. Within the concrete industry, cement manufacturing is the main culprit [2,3]. One avenue that is expected to significantly reduce the concrete industry's carbon footprint is the use of blended cements [2,4,5]. In addition to being more sustainable, blended cements are also less vulnerable to cracking and more durable.
Examples of non-traditional materials used in blended cements include geopolymer binders derived from pozzolans. Such use of geopolymers as binders for building application is not a new concept— the use of pozzolans in construction had precedence in ancient civilization. It is reported that the Romans extracted volcanic ash from volcanic craters for use in applications such as buildings, ports, road ways, as well as irrigation systems that have aesthetic values but have also withstood the test of time [6]. Following industrialization, there was a shift in the market from pozzolans, triggered by the emergence of Portland cement which is now indisputably the most widely used binder in construction applications.
The renewed interest in new classes of binders for built environment applications has been largely fueled by: (1) concerns over greenhouse gas emissions associated with the cement production process; (2) the need for binders with enhanced durability performance [7]. The scope of this paper is restricted to construction applications of geopolymer binders incorporating secondary input materials (volcanic ash, fly ash and slags). The deployment of such products is particularly advantageous in buildings, where durability, strength and fired resistance are of primary concern.
Despite the existence of strong economic and environmental drivers, geopolymers are still not widely used throughout the world. For example, in Australia of the 13 Mt of coal fly ash produced annually, it is estimated that just 4 Mt is recycled. Key challenges impeding the widespread use of geopolymers as binders include lack of sufficient information, especially the kind that can be used to characterize such binders with respect to processing, chemical and physical behavior, and performance [8]. In general, construction professionals are slow to embrace products that they are not familiar with. To build confidence in the use of natural pozzolans, researchers and developers will need to generate sufficient empirical data attesting to the quality of the product. Because of regional variations, such tests have to be context specific. The resulting empirical data can also be instrumental in addressing the institutional barriers linked to the use of prescriptive building codes, a major obstacle to sustainable development of the concrete industry. It is worth noting that there have been some positive developments in some places where codes and standards have been modified to explicitly make provisions for the use of non-traditional binders. This is not however a universal practice.
Although some studies have been done to investigate the potential of using “pozzolanic” secondary input material in cement and ceramic products, significant knowledge gaps still exist. For example, an extensive literature search by the authors did not reveal that refined prediction models can be used to enhance the durability performance of the resulting structures through designing appropriate strategies. Although deterioration of structures is a normal and expected occurrence, as such assets require significant capital investments, their use must be coupled with adequate strategies for maintaining, repairing and rehabilitating them in a sustainable and scalable manner. Consequently, prediction models are indispensible. Additionally, the use of non-conventional binders (geopolymer cement in this case) introduces a new scientific challenge. The resulting combinations of materials, being different from that of “traditional” concrete, will interact in ways that are yet to be quantified. There is therefore need for further studies to be done to characterize the impact of using non-traditional materials on things such as the rate at which concrete develops fundamental properties (for example, strength), which will ultimately affect its resistance to deterioration.
A key goal for the research discussed in this paper is characterizing the microstructure of the cementitious systems incorporating geopolymer binders. In principle, the evolution of microstructure can be captured using electron microscopical techniques. However, these typically result in micrographs of fracture surfaces that identify the qualitative features of hydration. It is still difficult to quantitatively identify differences between two nominally similar systems [9]. In addition, the large amounts of non-cementitious components in the resulting cementitious systems can be expected to drown the signal from the cementitious component. This makes it difficult for one to accurate generate quantitative data on the evolution of microstructure during hydration, knowledge that is essential for designing and developing cementitious materials with specific properties. Such design efforts are further impeded by difficulties in linking any observed differences to either desirable or undesirable physio-mechanical properties that would be indicative of the materials' expected performance during service conditions. Clearly, there is a great need for additional research work with a specific focus on characterizing geopolymer binders and designing compositions that would optimize their performance in cementitious systems. The discussion in this paper is a contribution to efforts directed at doing just that.
2. The Geopolymerization Process
2.1. The Chemistry of Geopolymers
Geopolymers are formed through mixing solid silicate-aluminate raw materials with alkali or alkali silicate solutions [7]. The primary input is usually thermally-activated/calcined clays such as kaolinite. It is common to include secondary raw materials, such as volcanic deposits, fly ash, broken down masonry, brick scrap, glass industry waste, ceramic waste materials (tiles sanitary porcelain and kiln lining material) and other sources of amorphous alumina silicates with SiO2 + Al2O3 ≥ 80 wt % [10-17]. The geopolymer structure consists of chain, sheet-like and three dimensional networks made of various Q units types of connected SiO4 (S) and AlO4 (A) tetrahedral [11]. When in contact with a high pH alkaline solution, the input materials (amorphous or semi crystalline aluminosilicates) dissolve progressively to form oligomers; geopolymers are then precipitated.
Ghukhovsky [12] reported the geological transformation of some volcanic rocks into zeolites during the formation of sedimentary rocks at low temperatures and pressure. It is therefore possible to model and develop cementitious systems through synthesis of alkaline aluminosilicates minerals. A direct synthesis of alkaline aluminosilicate minerals with the phase composition of such cementitious systems enhances the durability of the artificially formed stones. The amorphous nature of volcanic ash due to its high non-crystalline aluminosilicates content is indicative of the dissolution of this material in a highly concentrated alkaline solution [13]. It results in the formation of condensed polysialates that acquire strength with curing time. The rate of dissolution of aluminum and silicon species is directly linked to the amorphous nature of the input materials as well as the fineness of particles. Input materials with a smaller particle size can be expected to have higher reactivity and geopolymerization rates. During geopolymerization, volcanic ash, which is an amorphous aluminosilicate, can be expected to manifest behavior similarly to that of metakaolin, slags or fly ash. This is largely because all these materials have disordered silica and alumina randomly dispersed in amorphous matrices, which prompts dissolution in a highly alkaline media.
Among the evaluated primary inputs for geopolymerization, metakaolin has historically been regarded as the aluminosilicates material with the highest release per weight for both silicate and aluminate [18,19]. This property has been observed even after a few hours of immersion in an alkaline solution. This can be attributed to the purity of the material, which is essentially composed of SiO2 and Al2O3. By contrast, the high stability of the glassy phase of fly ash, slags, and others aluminosilicate wastes with high temperature thermal history, such as volcanic ash, explains their slow dissolution in a highly concentrated alkaline solution. Despite the relatively slow rate of dissolution of these waste products, several researchers have made a compelling case for their inclusion in the geopolymerization process [13,19,20].
On the basis of economic and ecological considerations, volcanic ash, fly ash and slags are all desirable starting materials [10]. The most ideal fly ash materials include ones with low calcium content, Class F fly ash (ASTM C618-05), and a general chemical composition comparable to that of calcined clays, bricks wastes or volcanic ash. Prominent producers of fly ash suitable for geopolymerization include countries such as South Africa (5 Mt), France (4.65 Mt), Hungary (4.09 Mt) and Canada (3.15 Mt). Depending on availability, volcanic ash can be a suitable starting material. The phrase “volcanic ash” as used in this paper refers to small, jagged pieces of rocks, minerals, volcanic glass (the size of sand) and silt (less than 2 mm in diameter) erupted through volcanic activity. Across the globe, volcanic ash exists in abundance in places such as areas around Mount St. Helens in the USA, Etna in Italy, Tinatubo in Phillipines, Tarawera in New Zealand, Santiaguito and Pacaya in Guatemala, Fuego in Costa Rica, Fako in Cameroon and Reykjavik in Iceland.
Volcanic ash, fly ash, slags, together with mineral wastes, metakaolin and many by-products from ceramic industries contain amorphous aluminosilicates regarded as glassy or disordered phases which can easily be observed through X-ray diffraction. The amorphous constituents can reach 90% in volcanic ash and slags due to the high temperature conditions prevailing at the materials' point of origin [6,18]. In addition to the amorphous phase, other crystalline phases can be observed. These include: (1) pyroxene groups associated with plagioclase in volcanic ash [10]; (2) quartz residue, and mullite are generally found in fly ash, and (3) melilites (sorosilicates) in slags [7].
The amorphous nature of these materials promotes their dissolution in highly concentrated alkalis solution (generally 6 to 10 M of sodium or potassium hydroxide) [7-10]. Usually a 2 to 5 wt % of alkali oxide in concentrated solution form can be used to activate 100 wt % of aluminosilicates described above (generally with SiO2/M2O (M = Na, K), … >2.5). The reaction of dissolution occurs at room temperature—this causes the alumina and silica oligomers to leach rapidly [18]. The condensation process in the systems of chains and rings of polysialates occurs in two stages: (a) quick condensation between aluminate and silicate species followed by; (b) a slow condensation stage involving just the silicate species [19].
Relatively low concentrations of alkaline solution (∼5 M) have been known to activate slags with mostly calcium-silicate-hydrates instead of polysialates in the final phases. This is similar to what happens with Portland cement and it can be attributed to the calcium (around 40 wt % of CaO) and alumina content.
2.2. Microstructure of Geopolymers
The degree of aluminosilicate polymerization in the matrix depends on the concentration and type of alkali solution used (either sodium or potassium silicate), the starting materials crystallinity and Si/Al ratio (Figures 1 and 2). The selective dissolution of Al will result in heterogeneity in the distribution of phases; the initial phases formed will be highly alumina-rich while the latter phases will be silica-rich.
Figure 3 shows micrographs of fly ash-based geopolymer materials as produced, and after exposure to hydriochloric acid solution [21]. EDS results confirm that the material's high resistance to acid attack and its stability can be linked to the relative high Si/Al ratio and the dense structure of the matrix.
3. Engineering Properties
3.1. Mechanical Properties
Most properties of geopolymer materials can be attributed to the M-A-S-H (M = Na, K) gel; a gel that is commonly called polysialates [14]. This gel behaves like a binder—it acts as a cementitious material with its amorphous structure gluing together particles and elements in an equilibrated chemical system. The nanostructure of the phases makes characterization through XRD difficult. They are usually characterized using TIR analysis. The main spectrum of the M-A-S-H gel is a band at 980 cm−1. This band characterizes sorosilicates [S2O7]−6 with partial substitution of Si4+ by Al3+ in tetrahedral positions. This constitutes the main vibration band of geopolymer network formation [22-24]. The shift of the vibration band towards lower wave numbers is indicative of reactivity with continuous destruction of amorphous aluminosilicates and a replacement with polysialates systems. The exact position of the band (in the regions 1,100–950 cm−1), which characterizes the asymmetric stretch of the Si-O-T (T = Si or Al) and T-O-M (M = K+ or Na+), is sensitive to both the length and angle of Si-O-Si bond. This bond is affected by the local network and thus the next-nearest neighbor [22]. Q4(nAl) (where n = 1, 2, 3, 4) can be found in the gel, influenced by the Si/Al, Na/Al and Si/Na molar ratios and water content.
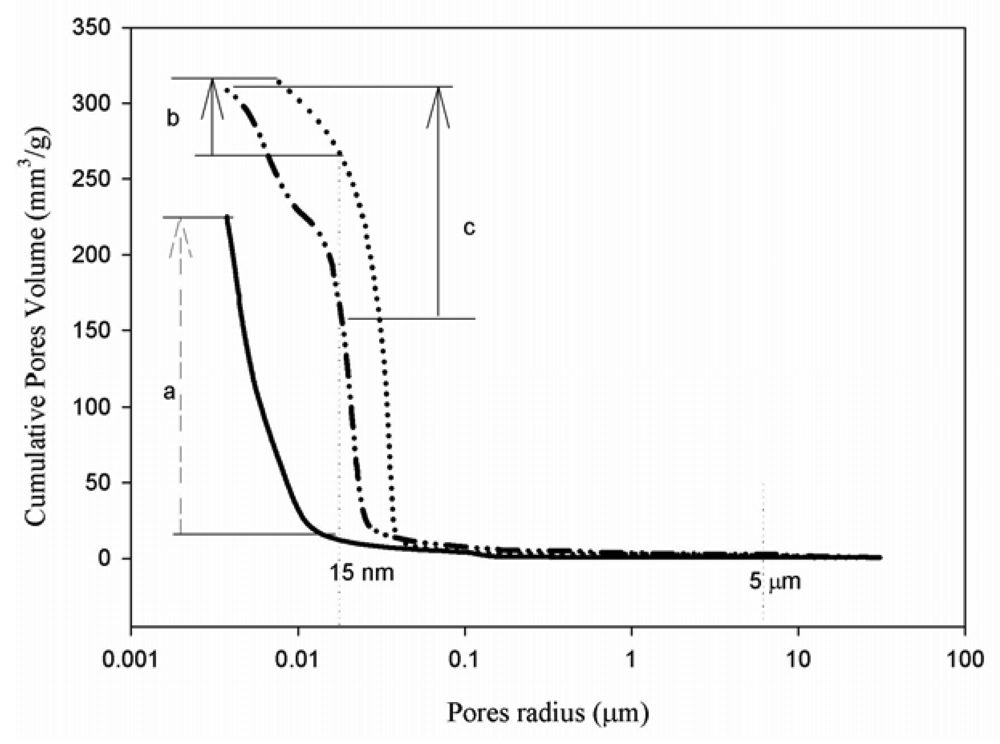
The thermal history of the aluminosilicates used will also affect the rate of dissolution and polymerization, time taken to achieve a stable matrix and the volume fraction of effective geopolymerization. It can therefore be inferred that this property will directly influence on porosity, pore size distribution, permeability and engineering properties (Figures 1, 2, 3). These are important features of the geopolymeric microstructure, which directly influence engineering properties [25,26]. It is generally accepted that the strength of any given mortar is fundamentally a function of the form and distribution of voids space and porosity. This strength is influenced primarily by the pore size distribution rather than by the cumulative pore volume (Figure 3). Gel pores diameters that are less than 15 nm are too small to initiate cracking under relatively low stress and therefore do not detract from the strength of the mortar [20]. The cumulative pore volume and pore size distribution are directly affected by Si/Al, and especially by alumina content (Figure 2). A minimum Al2O3 content of 10 to 15 wt % in raw materials that are essentially aluminosilicate 3D matrix formation assure good strength values, sometimes exceeding 30 MPa [27-29]. The average pore size is usually below 15 nm—his is linked to gel pores that are greater than 50%. With an increased amount of alumina content, the compressive strength can increase by as much as 22 wt %. Compressive strength values can exceed 120 MPa when optimized composition and synthesis technologies are applied.
There have been some improvement in mechanical properties geopolymers incorporating some waste material inputs that can be attributed to the concurrent development of Na(K)-A-S-H and C-S-H, which, despite the relatively high cumulative pore volume, contributes to strengthening matrices [30-32]. The flexural and compressive strength values are both higher in specimens with high alumina content and a high alkali concentration. Decreasing the alumina content reduces the amount of alkali required for the activation of the solid amorphous aluminosilicate.
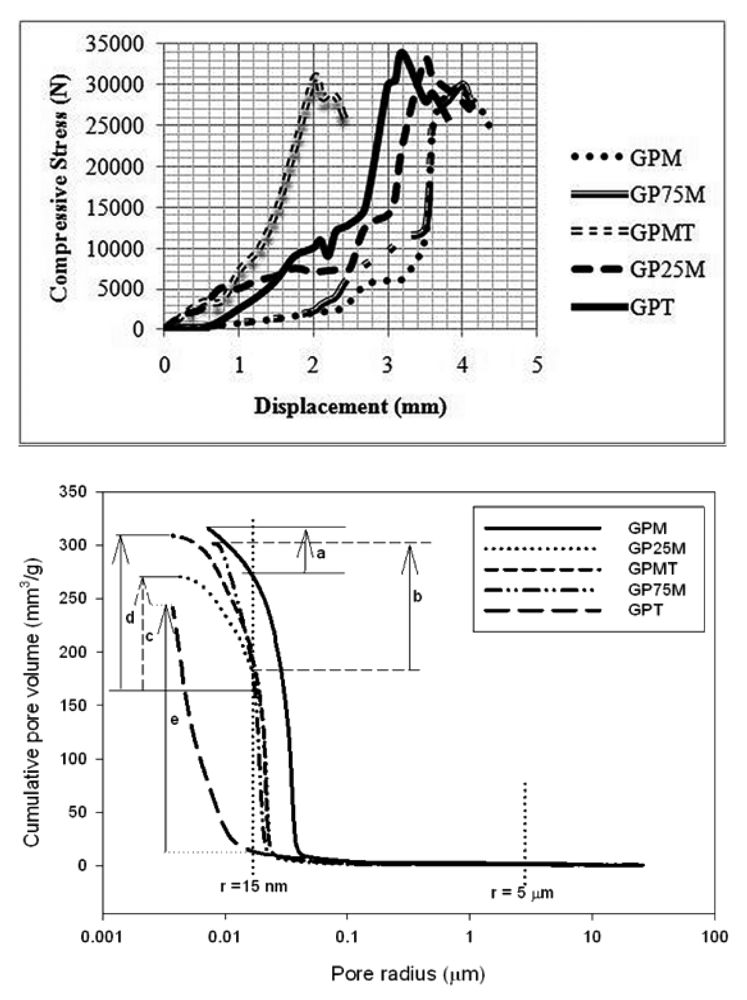
Despite the relatively high cumulative pore volume, gel pores present in the microstructure strengthen the matrices (Figure 4) [29]. Al-rich based geopolymer materials (GPM, in Figure 4) have matrices with larger pore size and high porosity that can be controlled by the introduction into the system of Si-rich metakaolin (GPT) as substitute to Al-rich clay with the final effects of reducing pore size and development of more dense structure.
Fernandez et al. [32] demonstrated that for a given composition of slags, the amount of alkali required can vary depending on the differences in specific surface area. Some proportions of the main elements (Si, Al and Ca) can be embedded in the crystalline structures. The nucleids can also be dispersed in the amorphous matrix of the waste. Consequently, such materials are not as effective as volcanic ash when used as reactive substrates [10]. It is important to control use of the reactive elements as excess alkali in the matrix that could catalyze the development of carbonates and reduce the durability of the products in service.
From the foregoing, it is evident that the porosity, flexural and compressive strength as well as fracture toughness of geopolymers can be optimized through designing specific chemical compositions for the required homogeneous or relatively coarse microstructure.
3.2. Resistance to Mechanical and Environmental Stresses
When a geopolymer matrix is subjected to high applied stress, failure occurs in a relatively short time—this behavior is similar to that of brittle materials. However, if the load is reduced, the time to failure increases.
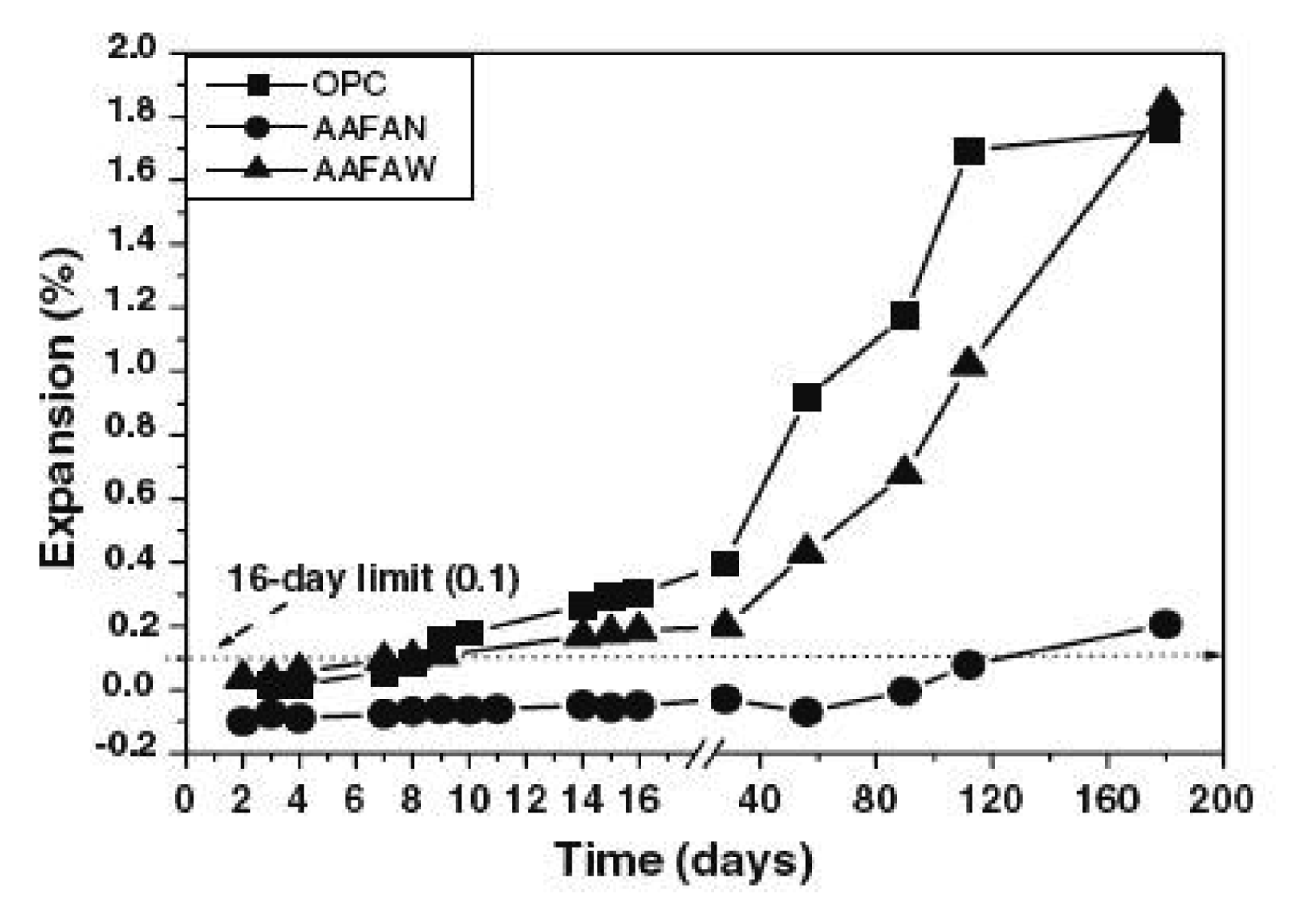
A key advantage of using geopolymers is the desirable impact they have on the durability performance of the resulting cementitious composite. This is linked to their dimensional stability, especially in geopolymer compositions with very low C-S-H (Figure 5) [29]. In fly ash-based and other amorphous aluminosilicates materials, such as metakaolin and volcanic ash with low Calcium content, the primary reaction is the alkali activation of the amorphous aluminolisicates. This essentially leads to the formation of polysialates (M-A-S-H). The secondary reaction is the attack of aggregates by the alkali, but in the absence of calcium, this will not be significant.
If a matrix with Si/Al ∼1 is exposed to damaging atmospheric conditions or weathering agents, debonding will occur. This is triggered either by high levels of alkalis or unreacted aluminosilicates. CO2 and H2O penetrate the matrix, cracking the nucleate through reacting with the various components that are poorly bound to structure. The growth of cracks will result in delayed failure of the materials. With Si/Al molar ratio between 2 and 3, the residue of amorphous silica and incongruently dissolved silica will perpetuate the “pozzolanic” reactivity, increasing strength over time. Crack bridging, as well as crack deflection phenomenon, will be more apparent in a coarser microstructure of high Si/Al compositions [33].
Some studies [17,34,35] have established that geopolymers are highly resistant to conventional sulfate attack and sea water. This is largely due to the absence of high-calcium phases. The fluctuation in flexural strength that has been observed in some metakaolin-based geopolymers is linked to the high alumina content that does not support the formation of stable matrices. While designing aluminosilicates for geopolymerization, it is ideal for alumina content not to exceed 22 wt % even when adhering to the conventional SiO2 + Al2O3 ≥ 80 wt %. To realize aluminosilicates that are well designed with respect to bulk chemical composition, care should be taken when incorporating the alkali that is necessary for the dissolution as well as the alkali silicate required for the development of the most condensed polysialates structure. High percentage of unreacted particles will over time result in an unstable matrix while excess of alkalis in the matrix will develop concentrated stresses, which will trigger delayed failure. Bakharev [35] established that the stability of alkali-activated fly ash pastes in aggressive environments depended on the intrinsic ordering of the components within the aluminosilicates gel. There were some fluctuations in strength that can be linked to the migration of alkalis from the geopolymers to the solution.
3.3. Global Warming Mitigation
It is generally accepted that one of the most efficient approaches to sustainable construction involves incorporating waste products from industrial and natural activities as secondary input materials in the material production process. As indicated in a previous section, geopolymer binders can reduce the adverse environmental impact of the construction industry as they are produced through environmentally friendly and energy efficient processes. The use of wastes products in construction processes is not just a sustainable practice but a necessary one in countries such as Holland and Japan which lack adequate stocks of virgin materials [6]. Another aspect of sustainability that is increasingly receiving great research attention is the notion of durability. The discussion in the preceding section has outlined specific ways through which geopolymers attain enhanced durability performance.
Due to their lightweight, geopolymer-based materials are suitable for use in noise, fire, and heat resistant products. It is also a well known fact that pore size distribution and capillary pores contribute for a rapid absorption and very slow release of humidity. The water retention properties of porous geopolymers make them ideal for use in cooling applications to counter the so called “heat island” [36] (Table 1). Such materials can be used in temperature regulation strategies to counter the increase in temperature expected in cities.
4. Discussion and Conclusions
In response to calls for sustainable production processes, many industrial sectors have shifted from the “extract and dump” mode of operation to a more cyclical-based use of resources. Strategies that have been explored to attain “green” production processes include ones that enhance the sustainable management of waste through promoting the use of secondary (recycled) resources. In recent years, many countries have developed strict regulations controlling the disposal of industrial byproducts in a bid to minimize the impact of exploitation of virgin materials on the environment and improve the management of wastes. Such actions have promoted research and development in the area of waste recovery and recycling. There are some global initiatives championing such actions at a much larger scale. An example of efforts focusing on greening the concrete industry is the World Board of Commerce for Sustainable Development's (WBCSD) sustainable cement initiative [13]. WBCSD has included the use of geopolymer binders in its definition of appropriate strategies for reducing greenhouse and volatile gas emissions associated with cement production.
The discussion in this paper has reviewed the use of natural (volcanic ash) and industrial wastes as secondary inputs for geopolymer binders. The composition of the described materials makes their performance comparable to that of conventional binders based on the use of Portland cement in several respects. In addition, their use also results in cementitious composites that are highly resistant to destructive agents. Such materials are suitable for use in various structural applications and often result in building elements with impressive durability and functional performance. Through carefully designing the porosity and pore size distribution, it is possible to develop structural elements with high strength values and chemical resistance performance (hence enhanced durability). From the authors experiments, they recommend limiting the Si/Al molar ratio to 2–3, then dissolving the resulting, carefully controlled, ground powder in an optimized alkaline solution, M/Al ≤ 1 (M = Na or K).
Despite existence of strong economic benefits, environmental drivers and structural performance advantages, the use of blended cement based on the use of geopolymer binders is still not a widespread practice throughout the world. A primary impediment has been the lack of empirical data on the performance of geopolymer-based building materials. In addition to perpetuating the lack of confidence in the material among professionals, the lack of sufficient knowledge also makes it difficult for many regulatory bodies to make provisions for the use of non-conventional geopolymer binders in building codes and specifications.
In their review, the authors established that a significant proportion of the work done to characterize the performance of geopolymers in building applications was for fly ash-based materials. Although it has been embraced by some stakeholders and incorporated in design guidelines by some regulatory bodies, the use of fly ash remains a controversial issue with strong arguments for and against its inclusion in cementitious composites based on health and ecological concerns. With the generation of additional empirical data, the authors are confident that such concerns will eventually be sufficiently addressed. However, as long as there is some pessimism among some would be users, the concerns will continue to impede the widespread adoption of geopolymer binders and the required changes in building codes and regulations can also be expected to occur very slowly. There is, therefore a need for other options. This can be achieved through further assessing the performance of geopolymer binder incorporating natural pozzolans, such as metakaolin and volcanic ash. Naturally occurring secondary input materials are presented in this paper as being particularly desirable for use in efforts directed at reducing the built environment's reliance on cement. Depending on the location, their inclusion can be cheaper than industrial waste products which would have to be transported.
From the discussion in this paper, it is clear that the use of geopolymer binders can result in cementitious systems that are not only cheaper than ones erected using conventional materials, but that are also more ecologically benign and more durable. However, many potential applications of such binders remain unexplored largely because the research and development community still has a limited understanding of the properties of the material. With time, this is expected to change and these materials can then be expected to find novel applications within the construction industry.


| Geopolymers and other alkali-activated materials | Commercial bricks used for construction | |
|---|---|---|
| Compressive Strength (MPa) | 21–120 | 21–110 |
| Modulus of rupture (MPa) | 3.8–30 | 4.8–27.6 |
| Modulus of Elasticity (GPa) | 4–35 | 9.7–34.5 |
| Bulk Density (g/cm3) | 1.3–2.5 | 1.65–2.08 |
| Water Absorption (%) | 10.00–30.00 | 0.5–10 |
| Thermal Expansion (%) | 0.1 | 4.5–9.0 |
| Thermal Conductivity (W/m.K) | 0.15–1 | 0.43–1.44 |
| Moisture Expansion (%) | 0 | 0.02–0.09 |
| Shrinkage in Service (%) | 0 | 0 |
| Corrosion Resistance | Resistant to conventional chemical environments | Resistant to all alkali and acids, except hydrofluoric |
References
- IPCC, Climate Change 2007: Mitigation. Contribution of Working Group III to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change; Metz, B., Davidson, O.R., Bosch, P.R., Dave, R., Meyer, L.A., Eds.; Cambridge University Press: Cambridge, UK, 2007.
- Mehta, P.K.; Walters, M. Roadmap to a Sustainable Concrete Construction Industry. Constr. Specifier 2008, 61, 48–57. [Google Scholar]
- Activity Report 2007; The European Cement Association: Brussels, Belgium, 2008; p. 44.
- Malhotra, V.M.; Mehta, P.K. High-Performance, High-Volume Fly Ash Concrete; Supplementary Cementing Materials for Sustainable Development, Inc.: Ottawa, ON, Canada, 2008. [Google Scholar]
- Manmohan, D.; Mehta, P.K. Heavily Reinforced Shear Walls and Mass Foundations Built with ‘Green’ Concrete. Concr. Int. 2002, 24, 64–70. [Google Scholar]
- Barker, L. Sustainable Construction Practiques. In Proceedings of the Joint International Conference Sustainable Building 2000 & Green Building Challenge 2000, Maastricht, The Netherlands, 23–25 October 2000; Aeneas Technical Publishers: Boxtel, The Netherlands, 2000. [Google Scholar]
- Davidovits, J. Geopolymer: Man-made rocks geosynthesis and the resulting development of very early high strength cement. J. Mat. Educ. 1994, 16, 91–139. [Google Scholar]
- Juenger, M.C.G.; Winnefeld, F.; Provis, J.L.; Ideker, J.H. Advances in alternative cementitious binders. Cement Concr. Res. 2010. in press. [Google Scholar]
- Scrivener, K.L. Backscattered electron imaging of cementitious microstructures: Understanding and quantification. Cement Concr. Compos. 2004, 26, 935–945. [Google Scholar]
- Weil, M.; Jeske, U.; Dombrowski, K.; Buchwald, A. Sustainable Design of Geopolymers–– Evaluation of Raw Materials by the Integration of Economic and Environmental Aspects in the Early Phases of Material Development. Advances in Life Cycle Engineering for Sustainable Manufacturing Businesses: Proceedings of the 14th CIRP Conference on Life Cycle Engineering, Waseda University, Tokyo, Japan, 11–13 June 2007; pp. 279–283.
- Singh, P.S.; Bastow, T.; Trigg, M. Structural studies of geopolymers by 29Si and 27Al MAS-NMR. J. Mater. Sci. 2005, 40, 3951–3961. [Google Scholar]
- Glukhovsky, V.D. Soil Silicates: Their Properties, Technology and Manufacturing and Fields of Application. Ph.D. Thesis, Civil Engineering Institute, Kiev, Ukraine, 1965. [Google Scholar]
- Kamseu, E.; Leonelli, C.; Perera, D.; S Melo, U.C.; Lemougna, P.N. Investigation of Volcanic ash based geopolymers as Potential Building Materials. Interceram 2009, 58, 136–140. [Google Scholar]
- Davidovits, J. Geopolymers: Inorganic polymeric new materials. J. Therm. Anal. 1991, 37, 1633–1656. [Google Scholar]
- Duxson, P.; Mallicoat, S.W.; Lukey, G.C.; Kriven, W.M.; van Deventer, J.S.J. Understanding the relationship between geopolymer composition, microstructure and mechanical properties. Colloid. Surface. A 2005, 269, 47–58. [Google Scholar]
- Duxson, P.; Fernandez-Jimenez, A.; Provis, J.L.; Lukey, G.C.; Palomo, A.; van Deventer, J.S.J. Geopolymer technology: The current state of the art. J. Mater. Sci. 2007, 42, 2917–2933. [Google Scholar]
- Gasteiger, H.A.; Frederick, W.J.; Streisel, R.C. Solubility of aluminosilicates in alkaline solutions and a thermodynamic equilibrium model. Ind. Eng. Chem. Res. 1992, 31, 1183–1190. [Google Scholar]
- Xu, H.; van Deventer, J.S.J. The geopolymerisation of alumino-silicate minerals. Int. J. Miner. Process. 2000, 59, 247–266. [Google Scholar]
- Komnitsas, K.; Zaharaki, D. Geopolymerisation: A review and prospects for the minerals industry. Miner. Eng. 2007, 20, 1261–1277. [Google Scholar]
- van Deventer, J.S.J.; Provis, J.L.; Duxson, P.; Luckey, G.C. Reaction mechanisms in the geopolymeric conversion of inorganic waste to useful products. J. Hazard. Mater. 2007, A139, 506–513. [Google Scholar]
- Mozgawa, W.; Deja, J. Spetroscopic studies of alkaline activated slag geopolymers. J. Mol. Struct. 2009, 924-926, 434–441. [Google Scholar]
- Rees, C.A.; Provis, J.L.; Lukey, G.C.; van Deventer, J.S.J. The mechanism of geopolymer gel formation investigated through seeded nucleation. Colloid. Surface. A 2008, 312, 97–105. [Google Scholar]
- Rees, C.A.; Provis, J.L.; Lukey, G.C.; van Deventer, J.S.J. ATR-FTIR analysis of fly ash geopolymer gel ageing. Langmuir 2007, 23, 8170–8179. [Google Scholar]
- Rees, C.A.; Provis, J.L.; Lukey, G.C.; van Deventer, J.S.J. In situ ATR-FTR study of the early stages of fly ash geopolymer gel formation. Langmuir 2007, 23, 9076–9082. [Google Scholar]
- Sagoe-Crentsil, K.; Weng, L. Dissolution processes, hydrolysis and condensation reactions during geopolymer synthesis: Part II. High Si/Al ratio systems. J. Mater. Sci. 2007, 42, 3007–3014. [Google Scholar]
- Kendall, K.; Howard, A.J.; Birchall, J.D. The relation between porosity, microstructure and strength, and approach to advanced cement-based materials. Philos. Trans. R. Soc. Lond. 1983, A310, 139–154. [Google Scholar]
- Fernandez-Jimenez, A.; Garcia-Lodeiro, I.; Palomo, A. Durability of alkali-activated fly ash cementitious materials. J. Mater. Sci. 2007, 42, 3055–3065. [Google Scholar]
- Rubner, K.; Hoffmann, D. Characterization of Mineral Building Materials by Mercury-Intrusion Porosimetry. Part. Part. Syst. Charact. 2006, 23, 20–28. [Google Scholar]
- Oh, J.E.; Monteiro, P.J.M.; Jun, S.S.; Choi, S.; Clark, S.M. The evolution of strength and crystalline phases for alkali-activated ground blast furnace slag and fly ash-based geopolymers. Cement Concr. Res. 2010, 40, 189–196. [Google Scholar]
- Krizan, D.; Zivanovic, B. Effects of dosage and modulus of water glass on early hydration of alkali-slag cements. Cement Concr. Res. 2002, 32, 1181–1188. [Google Scholar]
- Collins, F.; Sanjayan, J.G. Effects of ultra-fine materials on workability and strength of concrete containing alkali-activated slag as binder. Cement Concr. Res. 1999, 29, 459–462. [Google Scholar]
- Fernandez-Jimenez, A.; Palomo, J.G.; Puertas, F. Alkali-activated slag mortars mechanical strength behavior. Cement Concr. Res. 1999, 29, 1313–1321. [Google Scholar]
- Kamseu, E.; Leonelli, C. Synthesis of polysialate matrixes from Al-rich and Si-rich metakaolins: Polycondensation and physico-chemical properties. Proceedings of 34th International Conference and Exposition on Advanced Ceramics and Composites (ICACC), Daytona Beach, FL, USA, 24–29 January 2010.
- Palomo, A.; Glasser, F.P. Chemically-bonded cementitious material based on metakaolin. Br. Ceram. Trans. 1992, 9, 1107–1329. [Google Scholar]
- Bakharev, T. Durability of geopolymer materials in sodium and magnesium sulfate solutions. Cement Concr. Res. 2005, 35, 1233–1246. [Google Scholar]
- Okada, K.; Ooyama, A.; Isobe, T.; Kameshima, Y.; Nakajima, A.; Makenzie, K.J.D. Water retention properties of porous geopolymers for use in cooling applications. J. Eur. Ceram. Soc. 2009, 29, 1917–1923. [Google Scholar]
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Obonyo, E.; Kamseu, E.; Melo, U.C.; Leonelli, C. Advancing the Use of Secondary Inputs in Geopolymer Binders for Sustainable Cementitious Composites: A Review. Sustainability 2011, 3, 410-423. https://doi.org/10.3390/su3020410
Obonyo E, Kamseu E, Melo UC, Leonelli C. Advancing the Use of Secondary Inputs in Geopolymer Binders for Sustainable Cementitious Composites: A Review. Sustainability. 2011; 3(2):410-423. https://doi.org/10.3390/su3020410
Chicago/Turabian StyleObonyo, Esther, Elie Kamseu, Uphie C. Melo, and Cristina Leonelli. 2011. "Advancing the Use of Secondary Inputs in Geopolymer Binders for Sustainable Cementitious Composites: A Review" Sustainability 3, no. 2: 410-423. https://doi.org/10.3390/su3020410
APA StyleObonyo, E., Kamseu, E., Melo, U. C., & Leonelli, C. (2011). Advancing the Use of Secondary Inputs in Geopolymer Binders for Sustainable Cementitious Composites: A Review. Sustainability, 3(2), 410-423. https://doi.org/10.3390/su3020410






