Swiss Needle Cast in Western Oregon Douglas-Fir Plantations: 20‐Year Monitoring Results
Abstract
:1. Introduction
2. Materials and Methods
2.1. Aerial Survey
2.2. Cascade Mountains Plots
3. Results
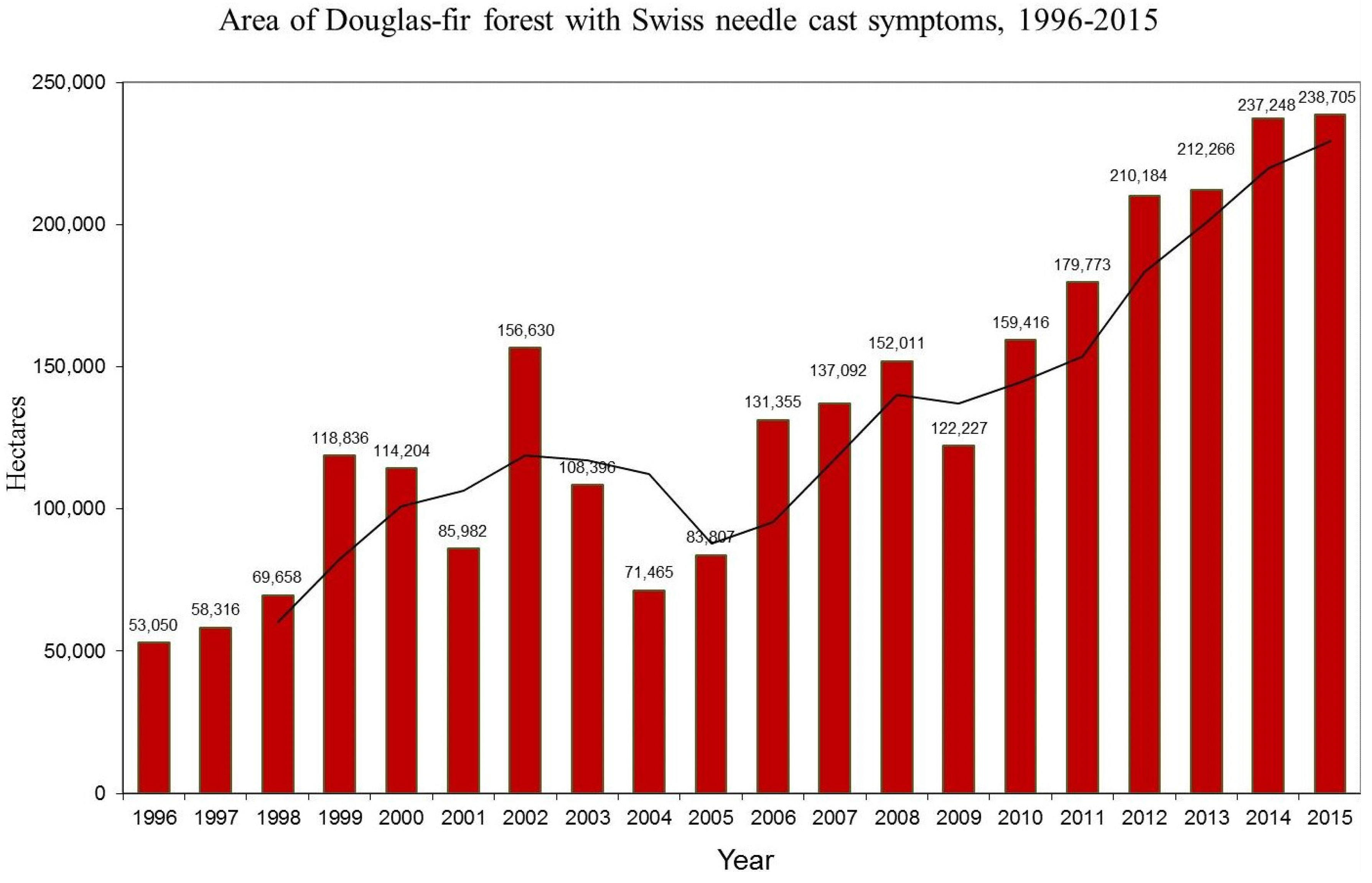
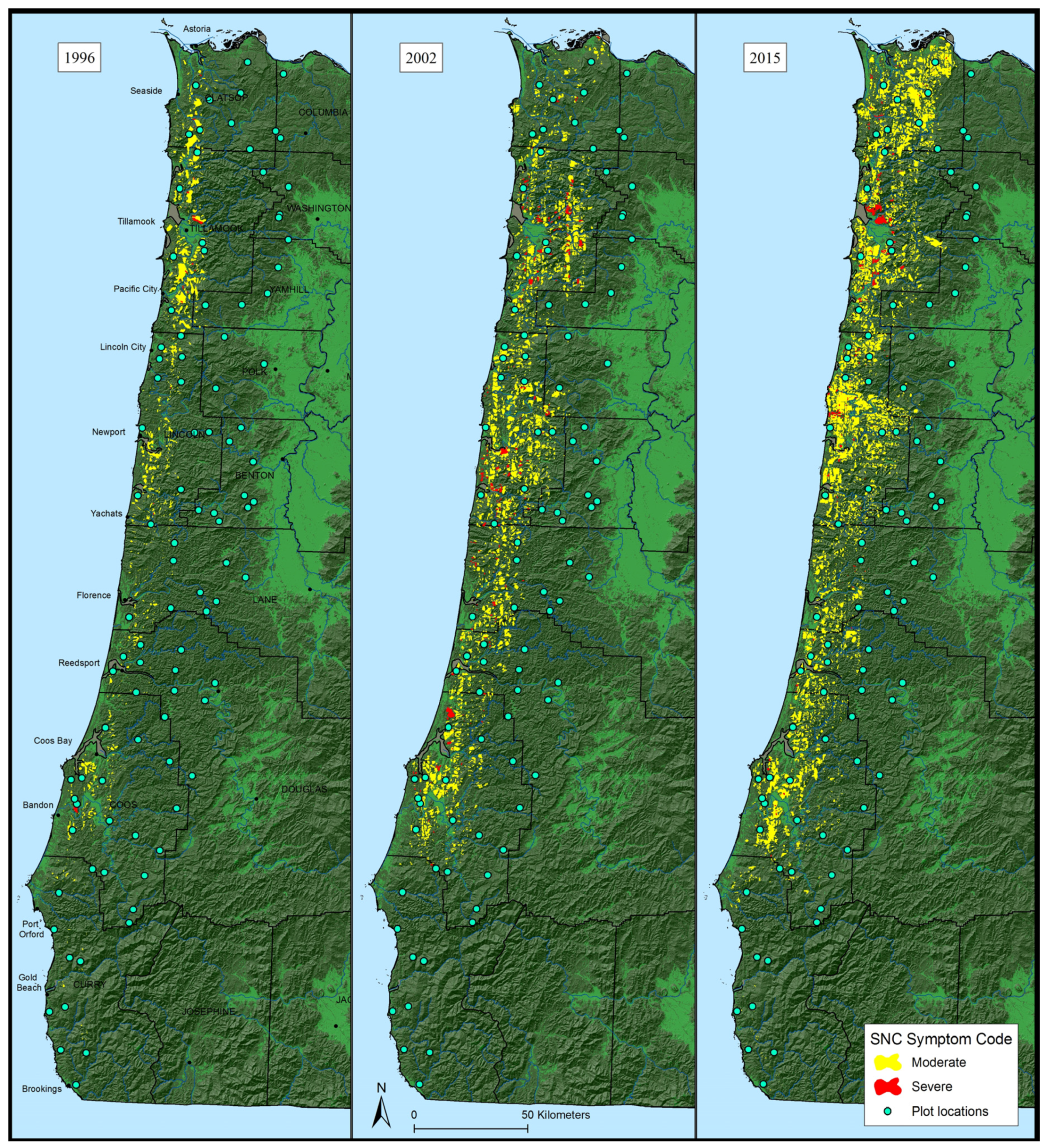
3.1. Aerial Survey
3.2. Cascade Mountains Plots
4. Discussion
Acknowledgments
Author Contributions
Conflict of Interest
Abbreviations
| SNCC | Swiss Needle Cast Cooperative |
| SNC | Swiss needle cast |
| PNW | Pacific Northwest |
References
- Hansen, E.M.; Stone, J.K.; Capitano, B.R.; Rosso, P.; Sutton, W.; Kanaskie, A.; McWilliams, M.G. Incidence and impact of Swiss needle cast in forest plantations of Douglas-fir in Coastal Oregon. Plant Dis. 2000, 84, 773–778. [Google Scholar] [CrossRef]
- Maguire, D.A.; Mainwaring, D.B.; Kanaskie, A. Ten-year growth and mortality in young Douglas-fir stands experiencing a range in Swiss needle cast severity. Can. J. For. Res. 2011, 41, 2064–2076. [Google Scholar] [CrossRef]
- Shaw, D.C.; Filip, G.M.; Kanaskie, A.; Littke, W. Managing an epidemic of Swiss needle cast in the Douglas-fir region of Oregon: The role of the Swiss Needle Cast Cooperative. J. For. 2011, 109, 109–119. [Google Scholar]
- Boyce, J.S. A needle-cast of Douglas-fir associated with Adelopus gaeumannii. Phytopathology 1940, 30, 649–659. [Google Scholar]
- Swiss Needle Cast Cooperative. Available online: http://sncc.forestry.oregonstate.edu/ (accessed on 19 July 2016).
- Maguire, D.; Kanaskie, A.; Voelker, W.; Johnson, R.; Johnson, G. Growth of young Douglas-fir plantations across a gradient in Swiss needle cast severity. West. J. Appl. For. 2002, 17, 86–95. [Google Scholar]
- Stone, J.K.; Capitano, B.R.; Kerrigan, J.L. The histopathology of Phaeocryptopus gaeumannii on Douglas-fir needles. Mycologia 2008, 100, 431–444. [Google Scholar] [CrossRef] [PubMed]
- Manter, D.K.; Bond, B.J.; Kavanagh, K.L.; Rosso, P.H.; Filip, G.M. Pseudothecia of Swiss needle cast fungus, Phaeocryptopus gaeumannii, physically block stomata of Douglas fir, reducing CO2 assimilation. New Phytol. 2000, 148, 481–491. [Google Scholar]
- Manter, D.K.; Kavanagh, K.L. Stomatal regulation in Douglas-fir following a fungal-mediated chronic reduction in leaf area. Trees 2003, 17, 485–491. [Google Scholar] [CrossRef]
- Cannell, M.G.R.; Morgan, J. Theoretical study of variables affecting the export of assimilates from branches of Picea. Tree Physiol. 1990, 6, 275–266. [Google Scholar] [CrossRef]
- Manter, D.K.; Reeser, P.W.; Stone, J.K. A climate-based model for predicting geographic variation in Swiss needle cast severity in the Oregon Coast Range. Phytopathology 2005, 95, 1256–1265. [Google Scholar] [CrossRef] [PubMed]
- Temel, F.; Johnson, G.R.; Stone, J.K. The relationship between Swiss needle cast symptom severity and level of Phaeocryptopus gaeumannii colonization in coastal Douglas-fir (Pseudotsuga menziesii var. menziesii). For. Pathol. 2004, 34, 383–394. [Google Scholar] [CrossRef]
- Aerial Detection Surveys information. Available online: http://www.fs.usda.gov/detail/r6/forest-grasslandhealth/insects-diseases/?cid=stelprdb5286951 (accessed on 19 July 2016).
- Freeman, F. Swiss needle cast monitoring transects in the Oregon Cascades. In Swiss Needle Cast Cooperative Annual Report; Filip, G., Ed.; College of Forestry, Oregon State University: Corvallis, OR, USA, 2001; pp. 11–13. [Google Scholar]
- Filip, G.M.; Kanaskie, A.; Kavanagh, K.; Johnson, G.; Johnson, R.; Maguire, D. Silviculture and Swiss Needle Cast: Research and Recommendations; Forestry Publications Office, Oregon State University: Corvallis, OR, USA, 2000. [Google Scholar]
- Ritokova, G.D.; Shaw, D.; Maguire, D.; Mainwaring, J.; Browning, M.; Gourley, G.F.; Kanaskie, A. SNCC Research and Monitoring Plot Network. In Swiss Needle Cast Annual Report 2014; Ritokova, G., Shaw, D., Eds.; Oregon State University: Corvallis, OR, USA, 2014; pp. 19–23. [Google Scholar]
- Rosso, P.H.; Hansen, E.M. Predicting Swiss needle cast disease distribution and severity in young Douglas-fir plantations in coastal Oregon. Phytopathology 2003, 93, 790–798. [Google Scholar] [CrossRef] [PubMed]
- Shaw, D.C.; Woolley, T.; Kanaskie, A. Vertical foliage retention in Douglas-fir across environmental gradients of the western Oregon Coast Range influenced by Swiss needle cast. Northwest Sci. 2014, 88, 23–32. [Google Scholar] [CrossRef]
- Temel, F.; Johnson, G.R.; Adams, W.T. Early genetic testing of coastal Douglas-fir for Swiss needle cast tolerance. Can. J. For. Res. 2005, 35, 521–529. [Google Scholar] [CrossRef]
- Black, B.A.; Shaw, D.C.; Stone, J.K. Impacts of Swiss needle cast on overstory Douglas-fir forests of western Oregon Coast Range. For. Ecol. Manag. 2010, 259, 1673–1680. [Google Scholar] [CrossRef]
- Zhao, J.; Mainwaring, D.B.; Maguire, D.A.; Kanaskie, A. Regional and annual trends in Douglas-fir foliage retention: Correlations with climatic variables. For. Ecol. Manag. 2011, 262, 1872–1886. [Google Scholar] [CrossRef]
- Lee, E.H.; Beedlow, P.A.; Waschmann, R.S.; Burdick, C.A.; Shaw, D.C. Tree-ring analysis of the fungal disease Swiss needle cast in Western Oregon coastal forests. Can. J. For. Res. 2013, 43, 677–690. [Google Scholar] [CrossRef]
- Coop, L.; Stone, J. Prediction maps of Swiss needle cast needle retention based on climatic factors. In Swiss Needle Cast Annual Report 2007; Ritokova, G., Shaw, D., Eds.; Oregon State University: Corvallis, OR, USA; Available online: http://sncc.forestry.oregonstate.edu/sites/default/files/sncc07.pdf (accessed on 19 July 2016).
- Zhao, J.; Maguire, D.A.; Mainwaring, D.B.; Kanaskie, A. Climatic influences on needle cohort survival mediated by Swiss needle cast in coastal Douglas-fir. Trees 2012, 26, 1361–1371. [Google Scholar] [CrossRef]
- Latta, G.; Adams, D.; Shaw, D. Mapping western Oregon Douglas-fir foliage retention with a simultaneous autoregressive model. In Swiss Needle Cast Annual Report 2009; Ritokova, G., Shaw, D., Eds.; Oregon State University: Corvallis, OR, USA; Available online: http://sncc.forestry.oregonstate.edu/sites/default/files/sncc09.pdf (accessed on 19 July 2016).
- Perakis, S.S.; Maguire, D.A.; Bullen, T.D.; Cromack, K.; Waring, R.H.; Boyle, J.R. Coupled nitrogen and calcium cycles in forests of the Oregon Coast Range. Ecosystems 2006, 8, 1–12. [Google Scholar] [CrossRef]
- Mulvey, R.L.; Shaw, D.C.; Maguire, D.A. Fertilization impacts on Swiss needle cast disease severity in Douglas-fir. For. Ecol. Manag. 2013, 287, 147–158. [Google Scholar] [CrossRef]
- Abatzoglou, J.T.; Rupp, D.E.; Mote, P.W. Seasonal Climate Variability and Change in the Pacific Northwest of the United Sates. Am. Meteorol. Soc. 2014, 27, 2125–2142. [Google Scholar]
- Lee, E.H.; Beedlow, P.A.; Waschmann, R.S.; Tingey, D.T.; Wickham, C.; Cline, S.T. Regional effects of Swiss needle cast disease and climate on growth of Douglas-fir in western Oregon. In Swiss Needle Cast Annual Report 2014; Ritokova, G., Shaw, D., Eds.; Oregon State University: Corvallis, OR, USA; Available online: http://sncc.forestry.oregonstate.edu/sites/default/files/2014AnnualReport.pdf (accessed on 19 July 2016).
- Jayawickrama, K.J.S.; Shaw, D.; Ye, T.Z. Genetic Selection in Coastal Douglas-fir for Tolerance to Swiss Needle Cast Disease. Swiss Needle Cast Coop. Annu. Rep. 2012, 29, 256–261. [Google Scholar]
- Mulvey, R.L.; Shaw, D.C.; Filip, G.M.; Chastagner, G.A. Swiss Needle Cast of Douglas-fir. In Forest Insect and Disease Leaflet (FIDL) 181; USDA Forest Service: Washington, DC, USA. Available online: http://www.fs.usda.gov/Internet/FSE_DOCUMENTS/stelprdb5426973.pdf (accessed on 19 July 2016).
- Zhao, J.; Maguire, D.A.; Mainwaring, D.; Wehage, J.; Kanaskie, A. Thinning mixed-species stands of Douglas-fir and western hemlock in the presence of Swiss needle cast: Guidelines based on relative basal area growth of individual trees. For. Sci. 2014, 60, 191–199. [Google Scholar] [CrossRef]
- Mainwaring, D.B.; Maguire, D.A.; Kanaskie, A.; Brandt, J. Growth responses to commercial thinning in Douglas-fir stands with varying severity of Swiss needle cast in Oregon, USA. Can. J. For. Res. 2005, 35, 2394–2402. [Google Scholar] [CrossRef]
- Kastner, W.; Dutton, S.; and Roche, D. Effects of Swiss needle cast on three Douglas-fir seed sources on a low-elevation site in the northern Oregon Coast Range: Results after five growing seasons. West. J. Appl. For. 2001, 16, 31–34. [Google Scholar]




© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ritóková, G.; Shaw, D.C.; Filip, G.; Kanaskie, A.; Browning, J.; Norlander, D. Swiss Needle Cast in Western Oregon Douglas-Fir Plantations: 20‐Year Monitoring Results. Forests 2016, 7, 155. https://doi.org/10.3390/f7080155
Ritóková G, Shaw DC, Filip G, Kanaskie A, Browning J, Norlander D. Swiss Needle Cast in Western Oregon Douglas-Fir Plantations: 20‐Year Monitoring Results. Forests. 2016; 7(8):155. https://doi.org/10.3390/f7080155
Chicago/Turabian StyleRitóková, Gabriela, David C. Shaw, Greg Filip, Alan Kanaskie, John Browning, and Danny Norlander. 2016. "Swiss Needle Cast in Western Oregon Douglas-Fir Plantations: 20‐Year Monitoring Results" Forests 7, no. 8: 155. https://doi.org/10.3390/f7080155





