Comparative Structural Dynamics of the Janj Mixed Old-Growth Mountain Forest in Bosnia and Herzegovina: Are Conifers in a Long-Term Decline?
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Site
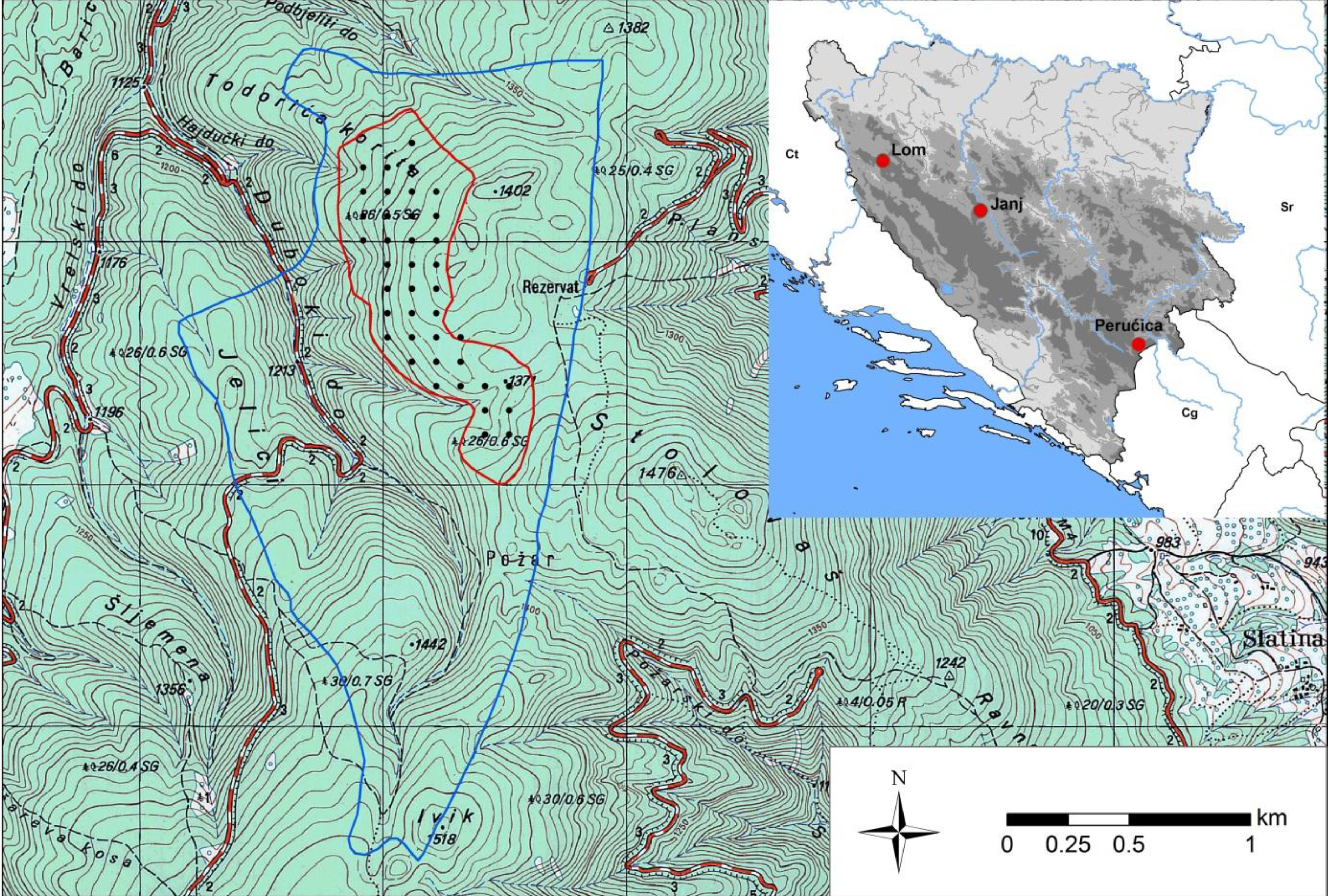
2.2. Field Measurements
2.3. Analyses
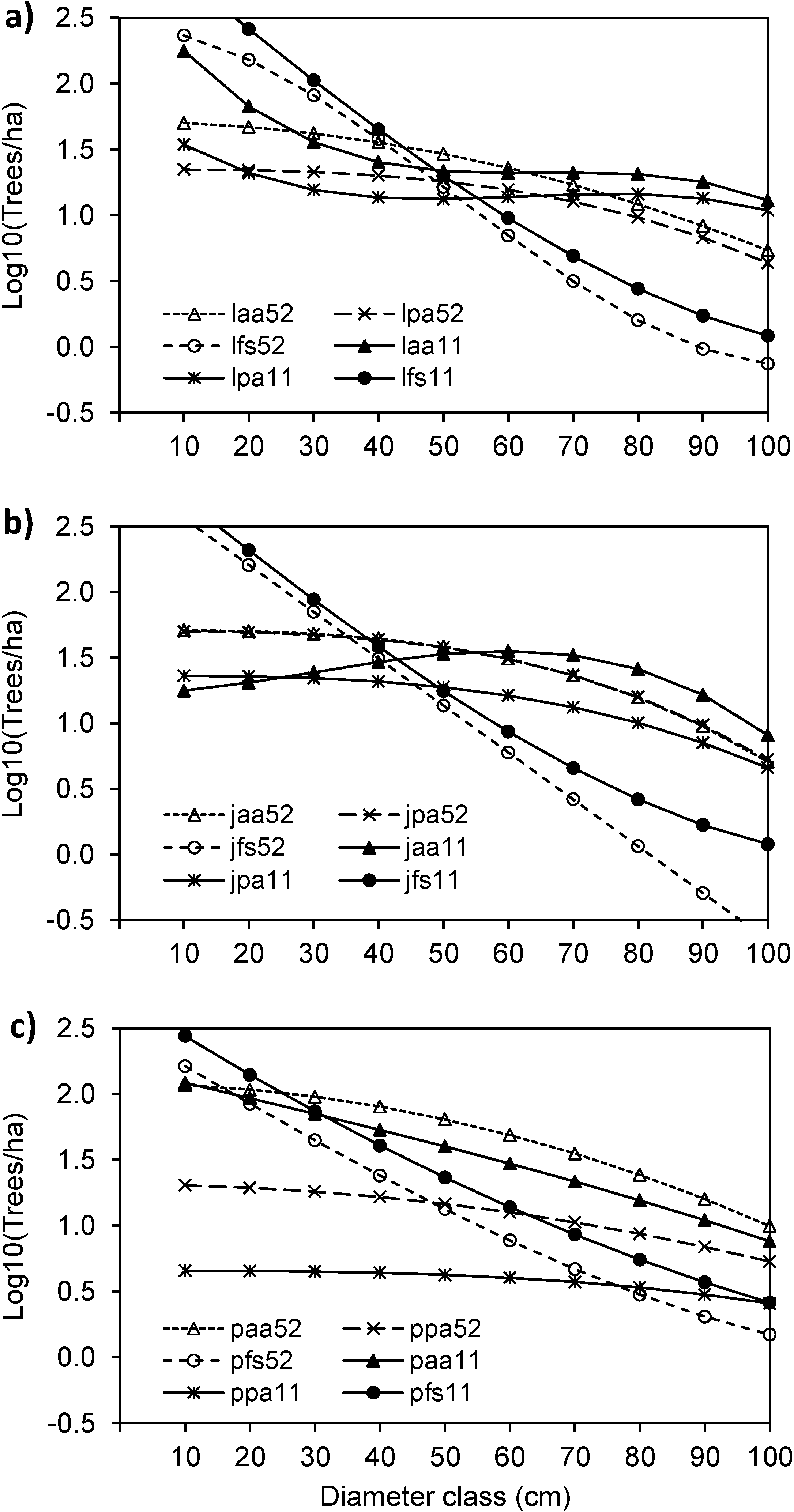
3. Results
3.1. Structure and Site Quality of the Janj Old-Growth Forest
| Mean diameter (cm) | Density (trees ha−1) | % | Basal area (m2∙ha−1) | % | Volume (m3∙ha−1) | |
|---|---|---|---|---|---|---|
| European beech | 22.5 | 327 | 63 | 11.7 | 18 | 192 |
| Silver fir | 60.6 | 117 | 23 | 34.7 | 52 | 648 |
| Norway spruce | 58.2 | 72 | 14 | 20.2 | 30 | 376 |
| Sycamore maple | – | <1 | <1 | – | <1 | – |
| Total | 46.7 | 516 | 66.7 | 1215 | ||
| Range | 9.0–100.1 | 243–995 | 26.1–102.5 | 419–1946 | ||
| St. deviation | 23.0 | 155 | 17.7 | 347 |

3.2. Coarse Woody Debris
| Snags (m3∙ha−1) | Logs (m3∙ha−1) | Stumps (m3∙ha−1) | Total (m3∙ha−1) | |
|---|---|---|---|---|
| Volume | 70.9 | 306.7 | 8.8 | 386.5 |
| Range | 0–510.2 | 103.8–582.5 | 0–26.1 | 162.1–892.6 |
| SD | 107.8 | 125.2 | 5.5 | 169.7 |
| Decay classes | Snags (m3∙ha−1) | Logs (m3∙ha−1) | Stumps (m3∙ha−1) |
|---|---|---|---|
| 1 | 20.1 | 2.2 | 0 |
| 2 | 16.2 | 30.3 | 0.5 |
| 3 | 18.8 | 64.2 | 3.0 |
| 4 | 15.8 | 88.8 | 5.3 |
| 5 | 0 | 121.2 | 0 |
3.3. Replacement Patterns and Neighbourhood Effects
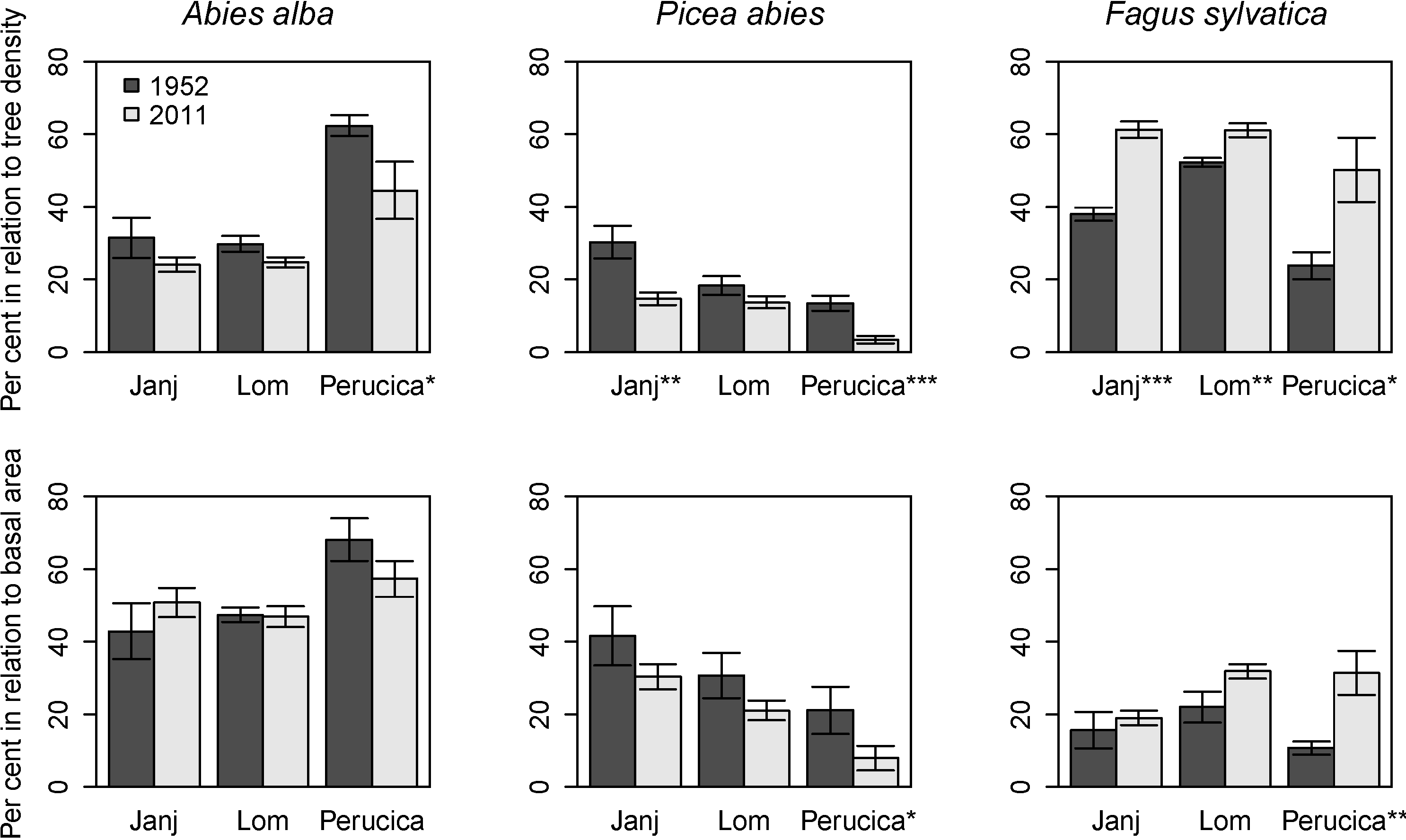
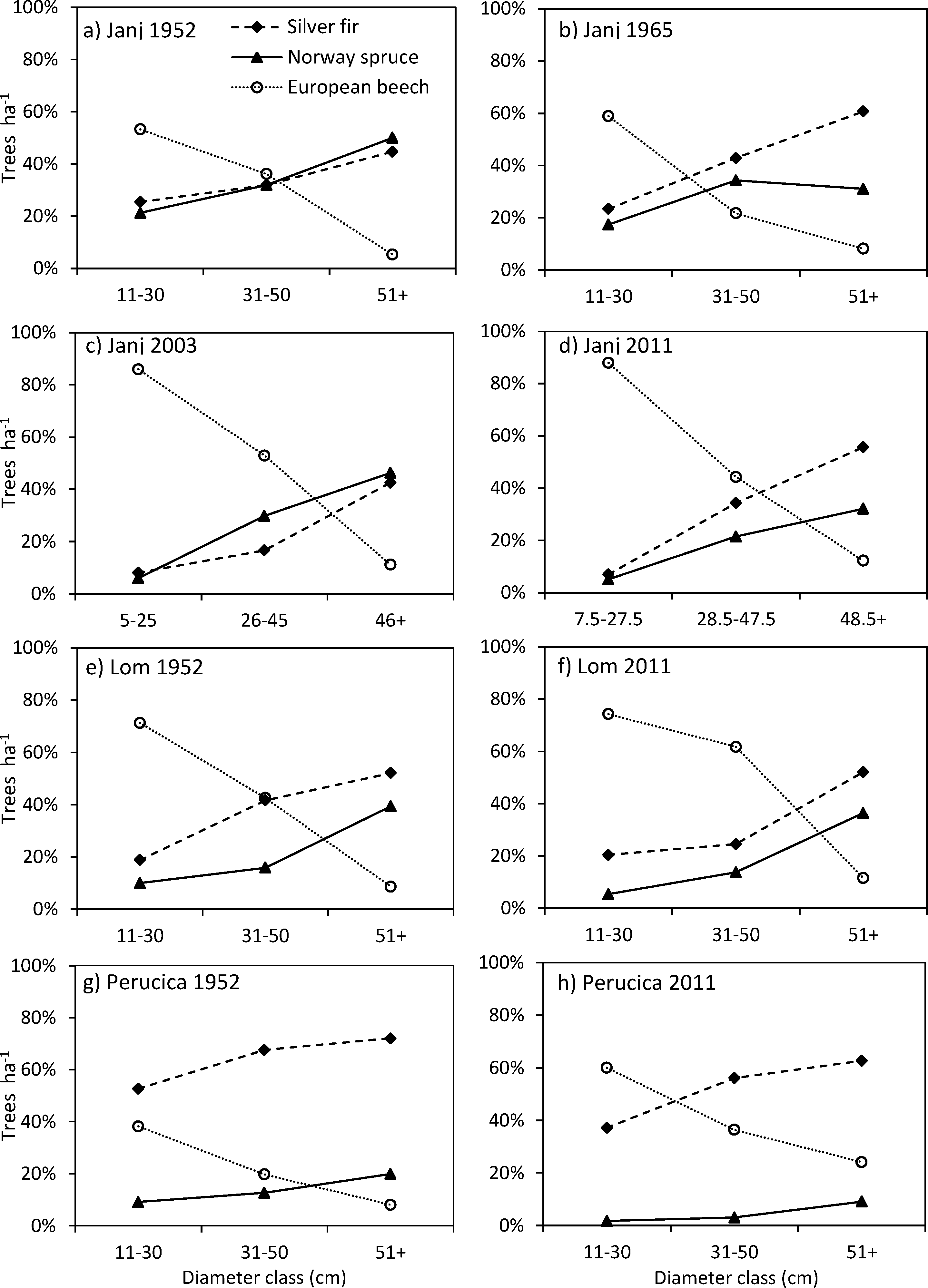
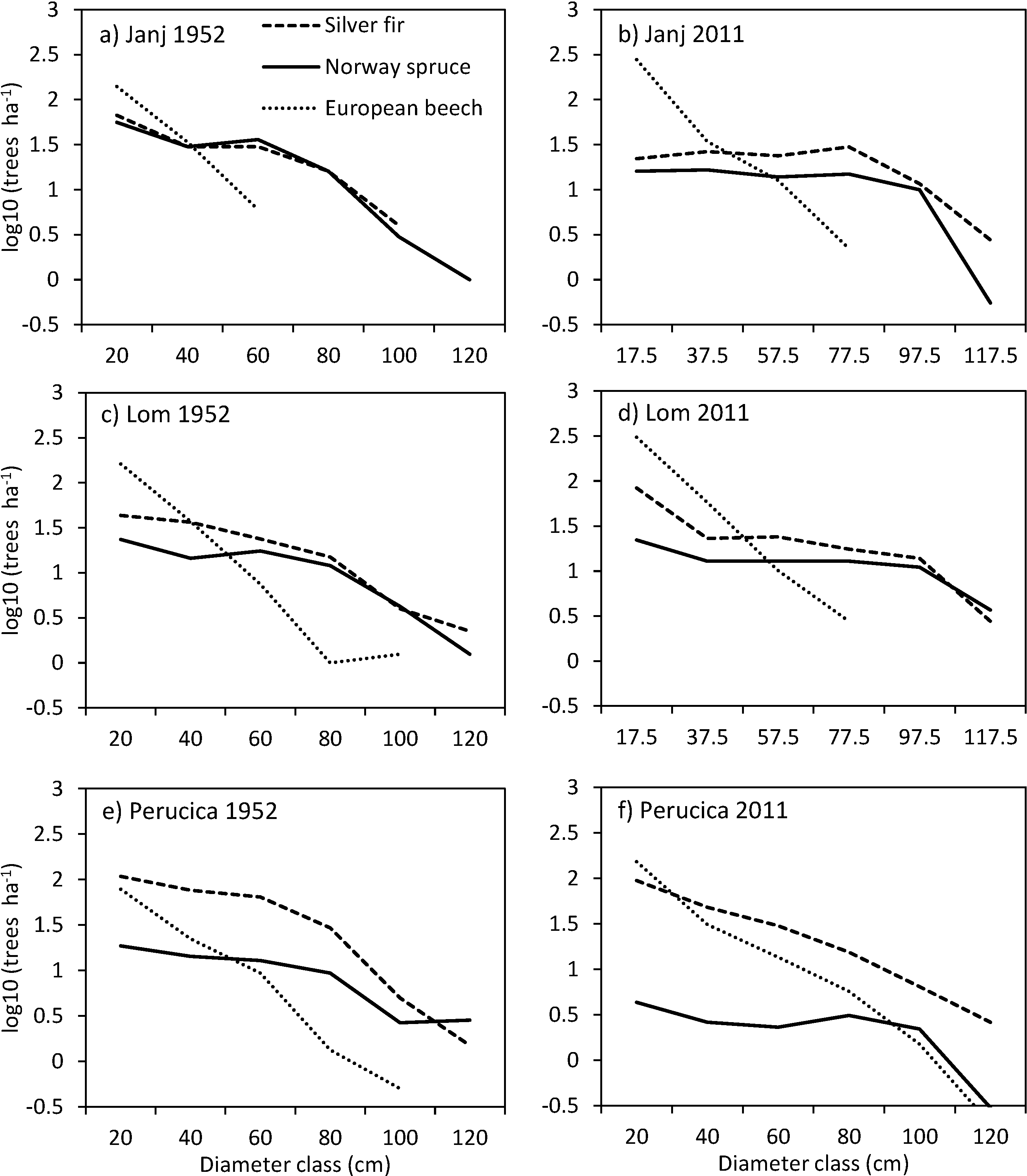
3.4. Structural Changes of the Janj, Lom and Perucica Old-Growth Forests over Six Decades
| Upper story | Relative | |||
|---|---|---|---|---|
| Understory | beech (0.24) | fir (0.48) | spruce (0.28) | abundances |
| beech (0.69) | 0.73 | 0.69 | 0.68 | 0.74 |
| fir (0.15) | 0.14 | 0.11 | 0.20 | 0.16 |
| spruce (0.05) | 0.07 | 0.09 | 0.05 | 0.10 |
| maple (0.11) | 0.06 | 0.11 | 0.06 | /* |
| Middle story | ||||
| Understory | beech (0.83) | fir (0.10) | spruce (0.07) | |
| beech | 0.63 | 0.75 | 0.68 | 0.69 |
| fir | 0.16 | 0.13 | 0.16 | 0.19 |
| spruce | 0.10 | 0.04 | 0.09 | 0.12 |
| maple | 0.11 | 0.08 | 0.07 | /* |
| Upper story | ||||
| Middle story | beech | fir | spruce | |
| beech | 0.75 | 0.83 | 0.83 | 0.77 |
| fir | 0.15 | 0.10 | 0.09 | 0.14 |
| spruce | 0.10 | 0.07 | 0.08 | 0.09 |
| Janj 1952 | Janj 2011 | |||||||
|---|---|---|---|---|---|---|---|---|
| Fir | Spruce | Beech | Cumulative | Fir | Spruce | Beech | Cumulative | |
| RMSE | 0.12 | 0.11 | 0.09 | 0.12 | 0.12 | 0.23 | 0.13 | 0.06 |
| Adj. R2 | 0.92 | 0.93 | 0.99 | 0.95 | 0.95 | 0.78 | 0.99 | 0.99 |
| N (dbh class) | 5 | 5 | 4 | 5 | 6 | 6 | 6 | 6 |
| Shape | IQ | IQ | NE | IQ | UNI | IQ | CO | RS |
| Lom 1952 | Lom 2011 | |||||||
| 0.09 | 0.09 | 0.15 | 0.10 | 0.08 | 0.02 | 0.09 | 0.04 | |
| Adj. R2 | 0.97 | 0.96 | 0.91 | 0.98 | 0.97 | 0.99 | 0.99 | 0.99 |
| N (dbh class) | 6 | 6 | 5 | 6 | 6 | 6 | 6 | 6 |
| Shape | IQ | IQ | VAR | NE | RS | RS | CO | RS |
| Perucica 1952 | Perucica 2011 | |||||||
| 0.18 | 0.09 | 0.08 | 0.14 | 0.03 | 0.10 | 0.08 | 0.09 | |
| Adj. R2 | 0.94 | 0.95 | 0.99 | 0.97 | 0.99 | 0.86 | 0.99 | 0.99 |
| N (dbh class) | 7 | 7 | 7 | 7 | 7 | 7 | 7 | 7 |
| Shape | IQ | IQ | CO | IQ | CO | IQ | CO | NE |
4. Discussion
4.1. Recent Stand Structure in Janj and Comparison with Similar Old-Growth Forests
4.2. Six Decades of Structural Changes in the Janj, Lom and Perucica Old-Growth Forests
4.3. Historical Evidence and Possible Causes of Beech Progression

5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Appendix
| Species | European beech | Silver fir | Norway spruce | |||
|---|---|---|---|---|---|---|
| dbh | height | dbh | height | dbh | height | |
| 1 | 48 | 31.2 | 72 | 38.6 | 68 | 37.9 |
| 2 | 48 | 30.4 | 73 | 40.3 | 70 | 36.3 |
| 3 | 51 | 31.5 | 74 | 34.7 | 70 | 35.5 |
| 4 | 53 | 30.5 | 74 | 38.4 | 71 | 37.5 |
| 5 | 54 | 40.8 | 75 | 41.0 | 74 | 37.4 |
| 6 | 56 | 33.4 | 80 | 42.3 | 75 | 40.1 |
| 7 | 57 | 32.1 | 81 | 45.0 | 75 | 41.8 |
| 8 | 58 | 30.0 | 85 | 42.0 | 75 | 43.0 |
| 9 | 58 | 33.0 | 86 | 35.3 | 77 | 36.3 |
| 10 | 58 | 39.0 | 94 | 39.4 | 77 | 34.6 |
| 11 | 59 | 36.0 | 96 | 49.5 | 78 | 39.3 |
| 12 | 59 | 37.2 | 98 | 39.0 | 83 | 46.2 |
| 13 | 60 | 32.5 | 99 | 44.5 | 85 | 43.5 |
| 14 | 60 | 39.2 | 108 | 50.5 | 91 | 41.9 |
| 15 | 69 | 35.6 | 110 | 48.5 | 92 | 44.9 |
| 16 | 70 | 35.6 | 110 | 46.2 | 95 | 44.5 |
| 17 | 72 | 35.5 | 117 | 47.5 | 96 | 43.4 |
| 18 | 79 | 35.4 | 117 | 44.8 | 99 | 44.3 |
| 19 | 83 | 44.7 | 120 | 46.5 | 100 | 45.5 |
| 20 | 92 | 41.6 | 124 | 44.8 | 110 | 45.0 |
| Mean | 62.2 | 35.3 | 94.7 | 42.9 | 83.1 | 41.0 |
References
- Bohn, U. General Map of Natural Vegetation of Europe; German Federal Agency for Natural Conservation: Bonn, Germany, 1996. [Google Scholar]
- Ellenberg, H. Vegetation Ecology of Central Europe; Cambridge University Press: Cambridge, UK, 1988. [Google Scholar]
- Peterken, G.F. Natural Woodland: Ecology and Conservation in Northern Temperate Regions; Cambridge University Press: Cambridge, UK, 1996. [Google Scholar]
- Safar, J. Ugibanje i Obnavljanje Jele u Prebornim Šumama Gorskog Kotara. Sumarski List 1951, 75, 299–303. [Google Scholar]
- Leibundgut, H. Europäische Urwälder der Bergstufe; Haupt Verlag AG: Bern, Switzerland, 1982. [Google Scholar]
- Korpel, S. Die Urwälder der Westkarpaten; Gustav Fischer Verlag: Stuttgart, Germany, 1995. [Google Scholar]
- Vrska, T.; Adam, D.; Hort, L.; Kolár, T.; Janík, D. European Beech (Fagus sylvatica L.) and Silver Fir (Abies alba Mill.) Rotation in the Carpathians—A Developmental Cycle or a Linear Trend Induced by Man? For. Ecol. Manag. 2009, 258, 347–356. [Google Scholar] [CrossRef]
- Malek, J. Jedle v Pralesovitých Rezervacích a Vybernych Lesich. Lesnicka Prace 1981, 60, 255–259. [Google Scholar]
- Matic, S. Utjecaj Ekoloskih i Strukturnih Cinilaca na Prirodno Pomladivanje Prebornih Suma Jele i Bukve u Gorskom Kotaru. Glasnik za Sumske Pokuse 1983, 21, 223–400. [Google Scholar]
- Ficko, A.; Poljanec, A.; Boncina, A. Do changes in Spatial Distribution, Structure and Abundance of Silver Fir (Abies alba Mill.) Indicate Its Decline? For. Ecol. Manag. 2011, 261, 844–854. [Google Scholar] [CrossRef]
- Boncina, A.; Cavlovic, J.; Curovic, M.; Govedar, Z.; Klopcic, M.; Medarevic, M. A Comparative Analysis of Recent Changes in Dinaric Uneven–Aged Forests of the NW Balkans. Forestry 2014, 87, 71–84. [Google Scholar] [CrossRef]
- Safar, J. Problemi Izmjene Vrsta u Šumama. Sumarski List 1952, 76, 89–100. [Google Scholar]
- Paluch, J. The Spatial Pattern of a Natural European Beech (Fagus sylvatica L.)–Silver Fir (Abies alba Mill.) Forest: A Patch-Mosaic Perspective. For. Ecol. Manag. 2007, 253, 161–170. [Google Scholar] [CrossRef]
- Klopcic, M.; Jerina, K.; Boncina, A. Long-Term Changes of Structure and Tree Species Composition in Dinaric Uneven-Aged Forests: Are Red Deer an Important Factor? Eur. J. For. Res. 2010, 129, 277–288. [Google Scholar] [CrossRef]
- Diaci, J.; Rozenbergar, D.; Anic, I.; Mikac, S.; Saniga, M.; Kucbel, S.; Visnjic, C.; Ballian, D. Structural Dynamics and Synchronous Silver Fir Decline in Mixed Old-Growth Mountain Forests in Eastern and Southeastern Europe. Forestry 2011, 5, 479–491. [Google Scholar]
- Ott, E.; Frehner, M.; Frey, H.-U.; Lüscher, P. Gebirgsnadelwälder: Praxisorientierter Leitfaden Für Eine Standortgerechte Waldbehandlung; Verlag Paul Haupt: Bern, Switzerland, 1997. [Google Scholar]
- Mlinsek, D. Pomlajevanje in Nekatere Razvojne Znacilnosti Bukovega in Jelovega Mladovja v Pragozdu na Rogu. Zbornik Biotehniske Fakultete 1967, 15, 7–32. [Google Scholar]
- Wraber, M. Topografski, Ekoloski in Socioloski Podatki o Slovenskih Pragozdovih. In Juznoevropske Prasume i Visokoplaninska Flora i Vegetacija Istocnoalpsko-Dinarskog Prostora; Fukarek, P., Ed.; Academy of Sciences Bosnia and Herzegovina: Sarajevo, Bosnia and Herzegovina, 1970; pp. 91–109. [Google Scholar]
- Nagel, T.A.; Svoboda, M.; Rugani, T.; Diaci, J. Gap Regeneration and Replacement Patterns in an Old-Growth Fagus-Abies Forest of Bosnia and Herzegovina. Plant Ecol. 2010, 208, 307–318. [Google Scholar] [CrossRef]
- Motta, R. Impact of Wild Ungulates on Forest Regeneration and Tree Composition of Mountain Forests in the Western Italian Alps. For. Ecol. Manag. 1996, 88, 93–98. [Google Scholar]
- Elling, W.; Dittmar, C.; Pfaffelmoser, K.; Rötzer, T. Dendroecological Assessment of the Complex Causes of Decline and Recovery of the Growth of Silver Fir (Abies alba Mill.) in Southern Germany. For. Ecol. Manag. 2009, 257, 1175–1187. [Google Scholar] [CrossRef]
- Drobyshev, I.; Overgaard, R.; Saygin, I.; Niklasson, M.; Hickler, T.; Karlsson, M.; Sykes, M.T. Masting Behaviour and Dendrochronology of European Beech (Fagus sylvatica L.) in Southern Sweden. For. Ecol. Manag. 2010, 259, 2160–2171. [Google Scholar] [CrossRef]
- Kellner, O.; Redbo-Torstensson, P. Effects of Elevated Nitrogen Deposition on the Field-Layer Vegetation in Coniferous Forests. Ecol. Bull. 1995, 227–237. [Google Scholar]
- Frelich, L.E.; Reich, P.B. Neighborhood Effects, Disturbance Severity and Community Stability in Forests. Ecosystems 1999, 2, 151–166. [Google Scholar] [CrossRef]
- Canham, C.D.; Denslow, J.S.; Platt, W.J.; Runkle, J.R.; Spies, T.A.; White, P.S. Light Regimes Beneath Closed Canopies and Tree-Fall Gaps in Temperate and Tropical Forests. Can. J. For. Res. 1990, 20, 620–631. [Google Scholar] [CrossRef]
- Zinke, P.J. The pattern of Influence of Individual Forest Trees on Soil Properties. Ecology 1962, 43, 130–133. [Google Scholar] [CrossRef]
- Mladenoff, D.J. Dynamics of Nitrogen Mineralization and Nitrification in Hemlock and Hardwood Treefall Gaps. Ecology 1987, 68, 1171–1180. [Google Scholar] [CrossRef]
- Finzi, A.C.; Canham, C.D.; van Breemen, N. Canopy Tree Soil Interactions within Temperate Forests: Species Effects on pH and Cations. Ecol. Appl. 1998, 8, 447–454. [Google Scholar]
- Packer, A.; Clay, K. Soil Pathogens and Spatial Patterns of Seedling Mortality in a Temperate Tree. Nature 2000, 404, 278–281. [Google Scholar] [CrossRef]
- Fox, J.F. Alternation and Coexistence of Tree Species. Am. Nat. 1977, 111, 69–89. [Google Scholar]
- Woods, K.D. Patterns of Tree Replacement—Canopy Effects on Understory Pattern in Hemlock—Northern Hardwood Forests. Vegetatio 1984, 56, 87–107. [Google Scholar]
- Tregubov, V. Les Forets Vierges Montagnardes des Alps Dinariques. Massif de Klekovatcha—Guermetch. Ph.D. Thesis, Montpellier University, Montpellier, France, 1941. [Google Scholar]
- Golubovic, U. Da li je Peručica Prašuma? Šumarski List 1963, 11/12, 484–486. [Google Scholar]
- Uotila, A.; Kouki, J.; Kontkanen, H.; Pulkkinen, P. Assessing the Naturalness of Boreal Forests in Eastern Fennoscandia. For. Ecol. Manag. 2002, 161, 257–277. [Google Scholar] [CrossRef]
- Anonymous. Census of Game Density in Hunting Area Sipovo, Sume Republike Srpske. Unpublished Report. Ministry of Agriculture, Forestry and Water Resources of the Republika Srpska: Banja Luka, Bosnia and Herzegovina, 2013. [Google Scholar]
- Berge, E.; Bartnicki, J.; Olendrzynski, K.; Tsyro, S.G. Long-Term Trends in Emissions and Transboundary Transport of Acidifying Air Pollution in Europe. J. Environ. Manag. 1999, 57, 31–50. [Google Scholar] [CrossRef]
- Bambulovic, P. Susenje Cetinjavih Suma u Bosni. Sumarski List 1930, 54, 446–450. [Google Scholar]
- Batic, J. Susenje Jelovih i Smrekovih Suma u Lici. Sumarski List 1930, 54, 345–347. [Google Scholar]
- Popovic, J. Susenje Cetinara Zbog Napada Potkornjaka u Šumama Drinske i Vrbaske Banovine; Radovi fitopatoloskog zavoda u Sarajevu: Sarajevo, Bosnia and Herzegovina, 1931. [Google Scholar]
- Frölich, J. Urwaldpraxis; Neumann Verlag: Radebeul/Berlin, Germany, 1954. [Google Scholar]
- Eic, N. Nase presume. Narodni Sumar 1951, 2/3, 56–67. [Google Scholar]
- Schütz, J.-P. Etude des Phénomenes de la Croissance en Hauteur et en Diametre du Sapin (Abies alba Mill.) et de l’épicéa (Picea abies Karst.) Dans Deux Peuplements Jardinés et une Foret Vierge. Ph.D. Thesis, ETH, Zurich, Switzerland, 1969. [Google Scholar]
- Maunaga, Z.; Burlica, C.; Pavlovic, B.; Bozalo, G.; Koprivica, M.; Lazarev, V.; Mataruga, M.; Rapajic, Z.; Govedar, Z.; Stanivukovic, Z.; et al. Studija Prasume Janj i Lom; Sumarski Fakultet Univerziteta u Banjaluci: Banjaluka, Bosnia and Herzegovina, 2005. [Google Scholar]
- Drinic, P. Taksacioni Elementi Sastojina Jele, Smrce i Bukve Prasumskog Tipa u Bosni. Radovi Poljoprivredno-Sumarskog Fakulteta u Sarajevu 1956, 1, 107–160. [Google Scholar]
- Fukarek, P. Sumske Zajednice Prasumskog Rezervata Perucice u Bosni. In Juznoevropske Prasume i Visokoplaninska Flora i Vegetacija Istocnoalpsko-Dinarskog Prostora; Fukarek, P., Ed.; Academy of Sciences Bosnia and Herzegovina: Sarajevo, Bosnia and Herzegovina, 1970; pp. 157–262. [Google Scholar]
- Pintaric, K. Urwald Perucica als Naturliches Forschungslaboratorium. Algebr. For. Z. 1978, 33, 691–693. [Google Scholar]
- Motta, R.; Berretti, R.; Castagneri, D.; Dukic, V.; Garbarino, M.; Govedar, Z.; Lingua, E.; Maunaga, Z.; Meloni, F. Toward a Definition of the Range of Variability of Central European Mixed Fagus-Abies Forests: The Nearly Steady-State Forest of Lom (Bosnia-Herzegovina). Can. J. For. Res. 2011, 41, 1871–1884. [Google Scholar] [CrossRef]
- Bottero, A.; Garbarino, M.; Dukic, V.; Govedar, Z.; Lingua, E.; Nagel, T.A.; Motta, R. Gap-Phase Dynamics in the Old-Growth Forest of Lom, Bosnia and Herzegovina. Silva Fenn. 2011, 45, 875–887. [Google Scholar]
- Govedar, Z. Nacini Prirodnog Obnavljanja Mjesovitih Suma Jele i Smrce (Abieti-Piceetum illyricum) na Podrucju Zapadnog Djela Republike Srpske. Ph.D. thesis, Belgrade University, Belgrade, Serbia, 2005. [Google Scholar]
- Lucic, R. Structural and Productivity Characteristics of Forests with Old-Growth Character in the National Park Sutjeska. Master Thesis, Belgrade University, Belgrade, Serbia, 2012. [Google Scholar]
- Van Wagner, C.E. The Line Intersect Method in Forest Fuel Sampling. For. Sci. 1968, 14, 20–26. [Google Scholar]
- Goedhart, P.W.; van Hees, A.F.M.; van der Werf, B.; Kramer, K. Biodiversity on Coarse Woody Debris, Model Description and Parameterisation. In EU 5.OP Project Nat Man Working Report; Wageningen UR: Wageningen, The Netherlands, 2004. [Google Scholar]
- Matic, V.; Drinic, P.; Stefanovic, V.; Ciric, M. Stanje Šuma u SR Bosni i Hercegovini Prema Inventuri Šuma na Velikim Površinama u 1964–1968. Godini, Šumarski Fakultet i Institut za Sumarstvo u Sarajevu 1971, 7, 1–639. [Google Scholar]
- Matic, V. Prirast i Prinos Suma; Faculty of Forestry: Sarajevo, Bosnia and Herzegovina, 1980. [Google Scholar]
- Motta, R.; Berretti, R.; Lingua, E.; Piussi, P. Coarse Woody Debris, Forest Structure and Regeneration in the Valbona Forest Reserve, Paneveggio, Italian Alps. For. Ecol. Manag. 2006, 235, 155–163. [Google Scholar] [CrossRef]
- Leak, W.A. Long-Term Structural Change in Uneven-Aged Northern Hardwoods. For. Sci. 1996, 42, 160–165. [Google Scholar]
- Janowiak, M.K.; Nagel, L.M.; Webster, C.R. Spatial Scale and Stand Structure in Northern Hardwood Forests: Implications for Quantifying Diameter Distributions. For. Sci. 2008, 54, 497–506. [Google Scholar]
- White, P.S.; Mackenzie, M.D.; Busing, R.T. A Critique on Overstory Understory Comparisons Based on Transition-Probability Analysis of an Old Growth Spruce-Fir Stand in the Appalachians. Vegetatio 1985, 64, 37–45. [Google Scholar] [CrossRef]
- Stevens, M.H.H. A Primer of Ecology with R; Springer: Dordrecht, The Netherlands; New York, NY, USA, 2009. [Google Scholar]
- Anic, I.; Mikac, S. Struktura, Tekstura i Pomlađivanje Dinarske Bukovo-Jelove Prasume Čorkova uvala. Sumarski List 2008, 11–12, 505–515. [Google Scholar]
- Firm, D.; Nagel, T.A.; Diaci, J. Disturbance History and Dynamics of an Old-Growth Mixed Species Mountain Forest in the Slovenian Alps. For. Ecol. Manag. 2008, 257, 1893–1901. [Google Scholar] [CrossRef]
- Horvat, I.; Glavac, V.; Ellenberg, H. Vegetation Sudosteuropas; G. Fischer: Stuttgart, Germany, 1974. [Google Scholar]
- Motta, R.; Garbarino, M.; Berretti, R.; Meloni, F.; Nosenzo, A.; Vacchiano, G. Development of Old-Growth Characteristics in Uneven-Aged Forests of the Italian Alps. Eur. J. For. Res. 2014. submitted. [Google Scholar]
- Christensen, M.; Hahn, K.; Mountford, P.E.; Odor, P.; Standovar, T.; Rozenbergar, D.; Diaci, J.; Wijdeven, S.; Meyer, P.; Winter, S.; et al. Dead Wood in European Beech (Fagus sylvatica) Forest Reserves. For. Ecol. Manag. 2005, 210, 267–282. [Google Scholar] [CrossRef]
- Harmon, M.E.; Franklin, J.F.; Swanson, F.J.; Sollins, P.; Gregory, S.V.; Lattin, J.D.; Anderson, N.H.; Cline, S.P.; Aumen, N.G.; Sedell, J.R.; et al. Ecology of Coarse Woody Debris in Temperate Ecosystems. Adv. Ecol. Res. 1986, 15, 133–302. [Google Scholar] [CrossRef]
- Simak, M. Untersuchungen Über den Natürlichen Baumartenwechsel in Schweizerischen Plenterwäldern. Mitteilungen Schweiz. Anst. Forstl. Vers. 1951, 27, 406–468. [Google Scholar]
- Battipaglia, G.; Saurer, M.; Cherubini, P.; Siegwolf, R.T.W.; Cotrufo, M.F. Tree Rings Indicate Different Drought Resistance of Native (Abies alba Mill.) and a Nonnative (Picea abies (L.) Karst.) Species Co-occurring at a Dry Site in Southern Italy. For. Ecol. Manag. 2009, 257, 820–828. [Google Scholar] [CrossRef]
- Shimano, K. A Power Function for Forest Structure and Regeneration Pattern of Pioneer and Climax Species in Patch Mosaic Forests. Plant Ecol. 2000, 146, 207–220. [Google Scholar] [CrossRef]
- Ducey, J.M.; Gunn, S.J.; Whitman, A.A. Late-Successional and Old-Growth Forests in the Northeastern United States: Structure, Dynamics, and Prospects for Restoration. Forests 2013, 4, 1055–1086. [Google Scholar] [CrossRef]
- Diaci, J.; Rozenbergar, D.; Boncina, A. Stand Dynamics of Dinaric Old-Growth Forest in Slovenia: Are Indirect Human Influences Relevant? Plant Biosyst. 2010, 144, 194–201. [Google Scholar] [CrossRef]
- Nowacki, G.J.; Abrams, M.D. Forest Composition, Structure, and Disturbance History of the Alan-Seeger-Natural-Area, Huntington County, Pennsylvania. Bull. Torrey Bot. Club 1994, 121, 277–291. [Google Scholar]
- Mlinsek, D. Rast in Sposobnost Reagiranja Pragozdne Bukve. Zbornik Biotehniške Fakultete 1967, 15, 63–79. [Google Scholar]
- Sercelj, A. The Origins and Development of Forests in Slovenia; Academia Scientiarum et Artium Slovenica, Classis IV: Historia Naturalis, Ljubljana, Slovenia, 1996. [Google Scholar]
- Mann, M.E.; Bradley, R.S.; Hughes, M.K. Northern Hemisphere Temperatures During the Past Millennium: Inferences, Uncertainties, and Limitations. Geophys. Res. Lett. 1999, 26, 759–762. [Google Scholar]
- Buntgen, U.; Kyncl, T.; Ginzler, C.; Jacks, D.S.; Esper, J.; Tegel, W.; Heussner, K.U.; Kyncl, J. Filling the Eastern European Gap in Millennium-Long Temperature Reconstructions. Proc. Natl. Acad. Sci. USA 2013, 110, 1773–1778. [Google Scholar]
- Brunetti, M.; Maugeri, M.; Monti, F.; Nanni, T. Temperature and Precipitation Variability in Italy in the Last Two Centuries from Homogenised Instrumental Time Series. Int. J. Climatol. 2006, 26, 345–381. [Google Scholar] [CrossRef]
- Mlinsek, D. From Clear-Cutting to a Close-to-Nature Silvicultural System. IUFRO News 1996, 25, 6–8. [Google Scholar]
- Prpic, B. Propadanje Suma u SR Hrvatskoj i Jugoslaviji. Sumarksi List 1989, 113, 235–243. [Google Scholar]
- Sebesta, J.; Samonil, P.; Lacina, J.; Oulehle, F.; Houska, J.; Bucek, A. Acidification of Primeval Forests in the Ukraine Carpathians: Vegetation and Soil Changes over Six Decades. For. Ecol. Manag. 2011, 262, 1265–1279. [Google Scholar] [CrossRef]
- Böhm, R.; Auer, I.; Schöner, W.; Ganekind, M.; Gruber, C.; Jurkovic, A.; Orlik, A.; Ungersböck, M. Eine neue Webseite mit Instrumentellen Qualitäts-Klimadaten für den Grossraum Alpen zurück bis 1760. Wiener Mitteilungen 2009, 216, 7–20. [Google Scholar]
- Kress, A.; Saurer, M.; Siegwolf, R.T.; Frank, D.C.; Esper, J.; Bugmann, H. A 350 Year Drought Reconstruction from Alpine Tree Ring Stable Isotopes. Glob. Biogeochem. Cycles 2010, 24, 1–16. [Google Scholar]
- Omanovic, S. Izvjestaji—Pregled Radova na Suzbijanju Potkornjaka na Podrucju Direkcije sSuma u Sarajevu. Sumarski List 1932, 6, 374–380. [Google Scholar]
- Kantorowicz, W. Half a Century of Seed Years in Major Tree Species of Poland. Silvae Genetica 2000, 49, 245–249. [Google Scholar]
- Piovesan, G.; Adams, J.M. Masting Behaviour in Beech: Linking Reproduction and Climatic Variation. Can. J. Bot.-Rev. Can. Bot. 2001, 79, 1039–1047. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Keren, S.; Motta, R.; Govedar, Z.; Lucic, R.; Medarevic, M.; Diaci, J. Comparative Structural Dynamics of the Janj Mixed Old-Growth Mountain Forest in Bosnia and Herzegovina: Are Conifers in a Long-Term Decline? Forests 2014, 5, 1243-1266. https://doi.org/10.3390/f5061243
Keren S, Motta R, Govedar Z, Lucic R, Medarevic M, Diaci J. Comparative Structural Dynamics of the Janj Mixed Old-Growth Mountain Forest in Bosnia and Herzegovina: Are Conifers in a Long-Term Decline? Forests. 2014; 5(6):1243-1266. https://doi.org/10.3390/f5061243
Chicago/Turabian StyleKeren, Srdjan, Renzo Motta, Zoran Govedar, Radovan Lucic, Milan Medarevic, and Jurij Diaci. 2014. "Comparative Structural Dynamics of the Janj Mixed Old-Growth Mountain Forest in Bosnia and Herzegovina: Are Conifers in a Long-Term Decline?" Forests 5, no. 6: 1243-1266. https://doi.org/10.3390/f5061243
APA StyleKeren, S., Motta, R., Govedar, Z., Lucic, R., Medarevic, M., & Diaci, J. (2014). Comparative Structural Dynamics of the Janj Mixed Old-Growth Mountain Forest in Bosnia and Herzegovina: Are Conifers in a Long-Term Decline? Forests, 5(6), 1243-1266. https://doi.org/10.3390/f5061243





