Population Dynamics of Lepidoptera Pests in Eucalyptus urophylla Plantations in the Brazilian Amazonia
Abstract
:1. Introduction
2. Experimental Section
2.1. Study Sites
| Characteristics | Caracuru | Pacanari | Ponte Maria | Felipe |
|---|---|---|---|---|
| Latitude | 00°32' S | 00°36' S | 00°47' S | 00°54' S |
| Longitude | 52°51'W | 52°36' W | 52°47' W | 52°21' W |
| Altitude | 110 m | 126 m | 88 m | 164 m |
| Planting date of Eucalyptus | March 1990 | March 1992 | March 1991 | March 1990 |
| Annual rainfall | 1988.0 mm | 2066.5 mm | 2276.0 mm | 2276.0 mm |
| Annual average temperature | 28.0 °C | 27.5 °C | 27.3 °C | 27.5 °C |
| Annual average relative humidity | 86.6% | - | 84.0% | 84.0% |
2.2. Lepidoptera Sampling
2.3. Data Analysis
3. Results and Discussion
| Site | Ponte Maria | Pacanari | Caracuru | Felipe | GT | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Family/Year | I | II | III | IV | V | T | I | II | III | IV | V | T | I | II | III | IV | V | T | I | II | III | IV | V | T | |
| Arctiidae | |||||||||||||||||||||||||
| Eupseudosoma aberrans | 24 | 17 | 16 | 12 | 9 | 78 | 56 | 64 | 40 | 41 | 14 | 215 | 147 | 44 | 43 | 32 | 18 | 284 | 28 | 27 | 9 | 11 | 0 | 75 | 652 |
| Eupseudosoma involuta | 16 | 15 | 18 | 4 | 1 | 54 | 90 | 27 | 14 | 16 | 1 | 148 | 99 | 164 | 7 | 15 | 11 | 296 | 22 | 11 | 6 | 1 | 2 | 42 | 540 |
| Geometridae | |||||||||||||||||||||||||
| Glena sp. | 0 | 8 | 7 | 2 | 6 | 23 | 0 | 39 | 8 | 63 | 17 | 127 | 4 | 17 | 15 | 54 | 1 | 91 | 5 | 11 | 2 | 6 | 2 | 26 | 267 |
| Oxydia vesulia | 2 | 27 | 81 | 6 | 4 | 120 | 26 | 8 | 24 | 11 | 1 | 70 | 27 | 12 | 5 | 18 | 1 | 63 | 32 | 87 | 57 | 61 | 0 | 237 | 490 |
| Stenalcidia grosica | 15 | 39 | 65 | 137 | 61 | 317 | 15 | 82 | 46 | 1091 | 50 | 1284 | 75 | 50 | 27 | 1385 | 129 | 1666 | 54 | 14 | 33 | 181 | 20 | 302 | 3,569 |
| Thyrinteina arnobia | 19 | 12 | 5 | 13 | 264 | 313 | 11 | 247 | 44 | 266 | 802 | 1370 | 27 | 17 | 15 | 411 | 4950 | 5420 | 3 | 1 | 4 | 36 | 128 | 172 | 7,275 |
| Lymantriidae | |||||||||||||||||||||||||
| Sarsina violascens | 8 | 14 | 1 | 18 | 9 | 50 | 6 | 3 | 4 | 18 | 12 | 43 | 23 | 2 | 17 | 189 | 18 | 249 | 37 | 21 | 4 | 26 | 10 | 98 | 440 |
| Notodontidae | |||||||||||||||||||||||||
| Misogada blerula | 11 | 8 | 3 | 3 | 2 | 27 | 20 | 52 | 1 | 15 | 3 | 91 | 76 | 17 | 1 | 46 | 0 | 140 | 3 | 13 | 0 | 7 | 0 | 23 | 281 |
| Nystalea nyseus | 10 | 3 | 2 | 2 | 10 | 27 | 52 | 1 | 1 | 3 | 52 | 109 | 3 | 148 | 7 | 3 | 3 | 164 | 11 | 3 | 4 | 5 | 11 | 34 | 334 |
| Psorocampa denticulata | 1 | 1 | 1 | 3 | 0 | 6 | 0 | 1 | 1 | 0 | 0 | 2 | 0 | 8 | 0 | 0 | 0 | 8 | 0 | 0 | 0 | 1 | 0 | 1 | 17 |
| Total | 1015 | 202 | 312 | 58 | 13,865 | ||||||||||||||||||||
| Site | Ponte Maria | Pacanari | Caracuru | Felipe | GT | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Family/Year | I | II | III | IV | V | T | I | II | III | IV | V | T | I | II | III | IV | V | T | I | II | III | IV | V | T | |
| Amatidae | |||||||||||||||||||||||||
| Cosmosoma auge | 1 | 5 | 3 | 4 | 1 | 14 | 2 | 1 | 0 | 2 | 0 | 5 | 3 | 0 | 3 | 0 | 0 | 6 | 0 | 0 | 3 | 1 | 0 | 4 | 29 |
| Arctiidae | |||||||||||||||||||||||||
| Idalus admirabilis | 9 | 34 | 30 | 17 | 21 | 111 | 9 | 41 | 26 | 53 | 9 | 138 | 45 | 299 | 38 | 48 | 5 | 435 | 40 | 67 | 45 | 16 | 2 | 170 | 854 |
| Eucleidae | |||||||||||||||||||||||||
| Phobethon hypparchia | 1 | 5 | 2 | 3 | 2 | 13 | 0 | 1 | 6 | 4 | 2 | 13 | 0 | 15 | 3 | 11 | 0 | 29 | 10 | 2 | 0 | 1 | 1 | 14 | 69 |
| Megalopygidae | |||||||||||||||||||||||||
| Megalopyge albicollis | 1 | 6 | 24 | 3 | 3 | 37 | 2 | 4 | 4 | 4 | 1 | 15 | 2 | 52 | 1 | 0 | 0 | 55 | 4 | 15 | 9 | 0 | 0 | 28 | 135 |
| Mimallonidae | |||||||||||||||||||||||||
| Mimallo amilia | 2 | 7 | 8 | 1 | 1 | 19 | 6 | 1 | 6 | 0 | 0 | 13 | 4 | 38 | 6 | 2 | 0 | 50 | 4 | 15 | 0 | 0 | 0 | 19 | 101 |
| Saturniidae | |||||||||||||||||||||||||
| Eacles ducalis | 5 | 14 | 32 | 16 | 10 | 77 | 10 | 13 | 7 | 17 | 3 | 50 | 11 | 18 | 1 | 0 | 6 | 36 | 4 | 15 | 11 | 9 | 9 | 48 | 211 |
| Automeris illustris | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 2 | 1 | 0 | 4 | 1 | 3 | 0 | 2 | 0 | 6 | 0 | 1 | 4 | 5 | 1 | 11 | 21 |
| Eacles imperialis magnifica | 24 | 37 | 3 | 9 | 0 | 73 | 7 | 3 | 0 | 0 | 0 | 10 | 5 | 0 | 1 | 0 | 0 | 6 | 13 | 7 | 1 | 0 | 0 | 21 | 110 |
| Dirphia sp. | 4 | 3 | 0 | 0 | 0 | 7 | 0 | 1 | 0 | 0 | 0 | 1 | 8 | 4 | 6 | 23 | 2 | 43 | 5 | 1 | 1 | 2 | 0 | 9 | 60 |
| Total | 157 | 249 | 91 | 324 | 1590 | ||||||||||||||||||||

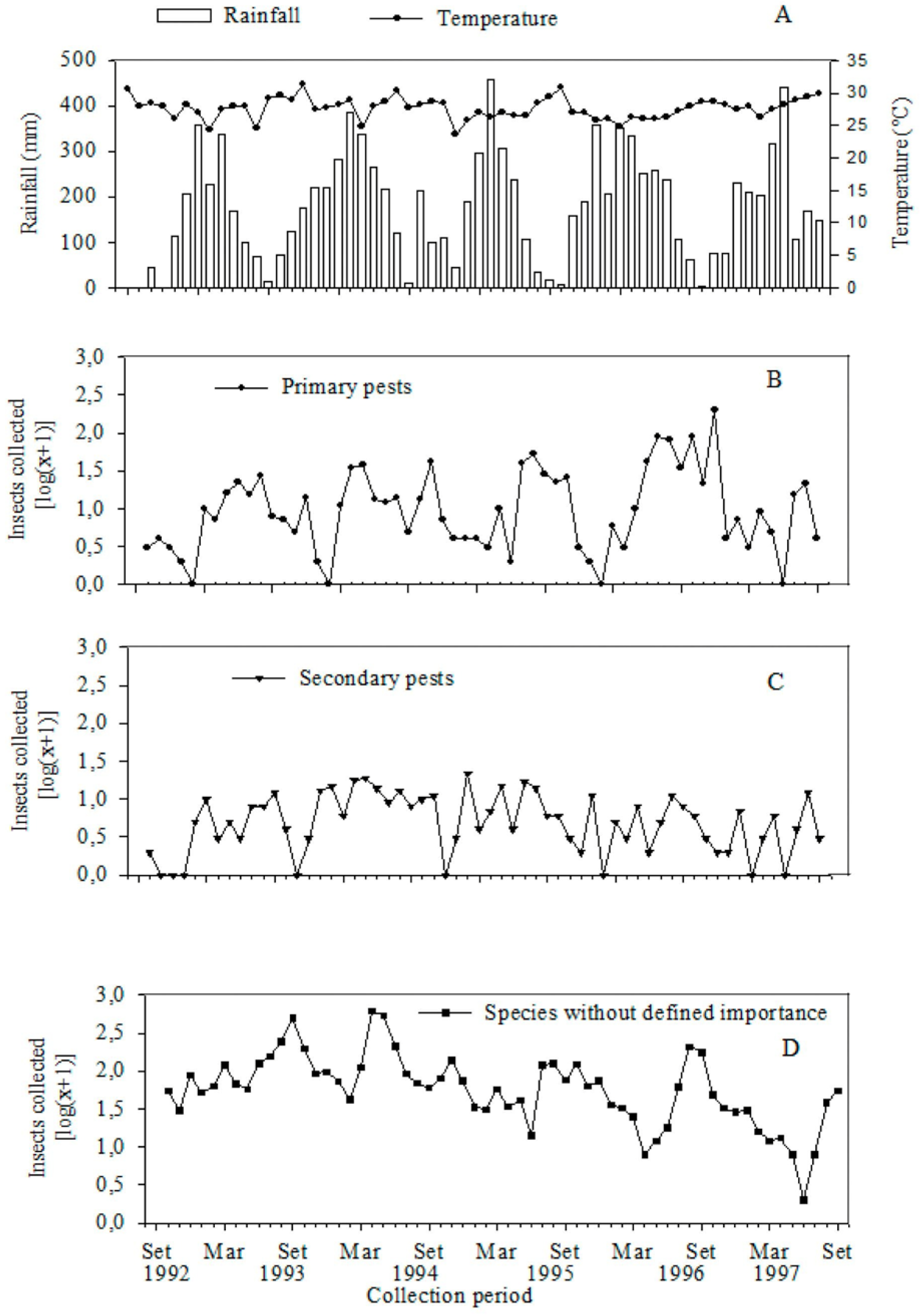

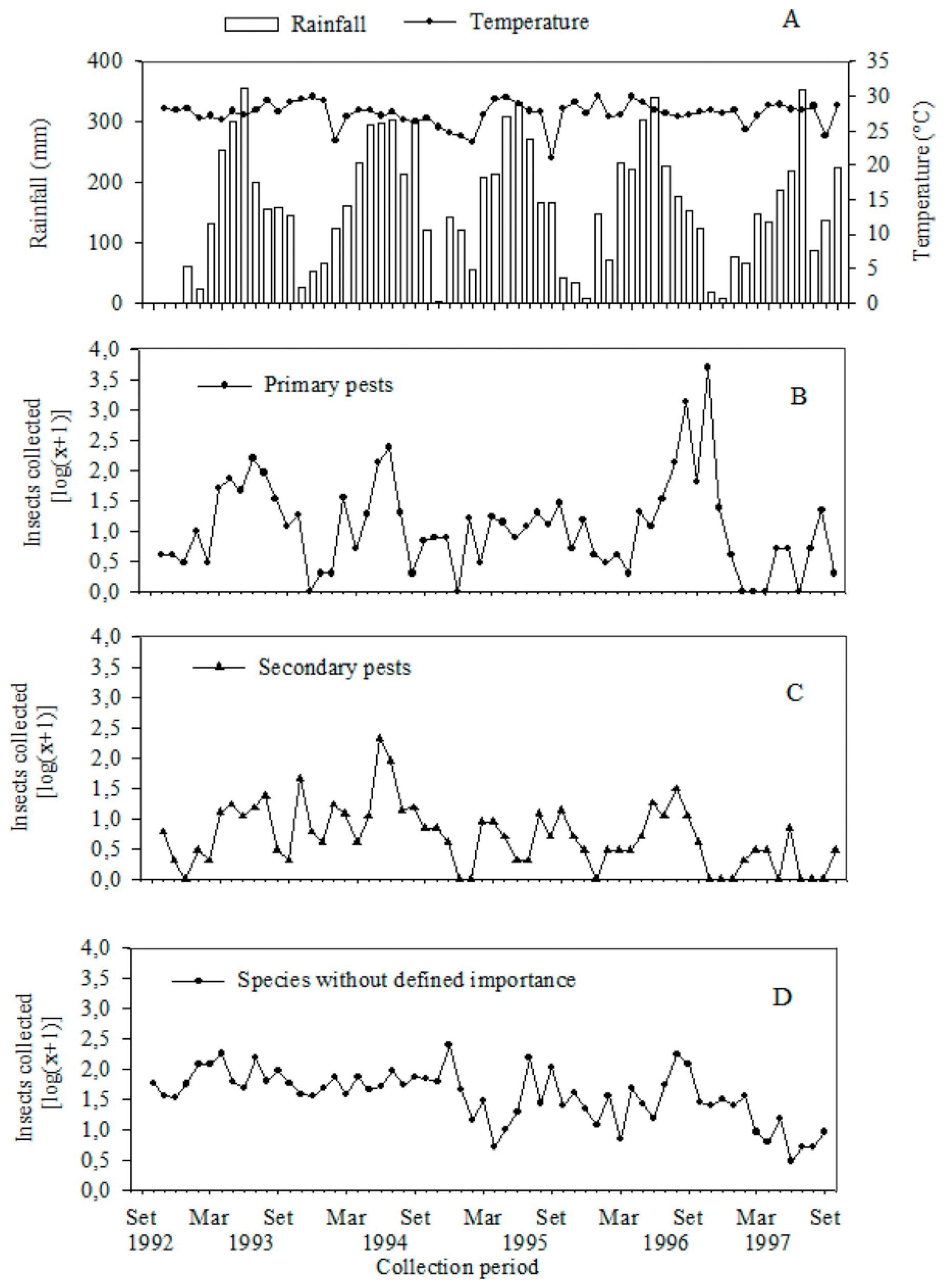
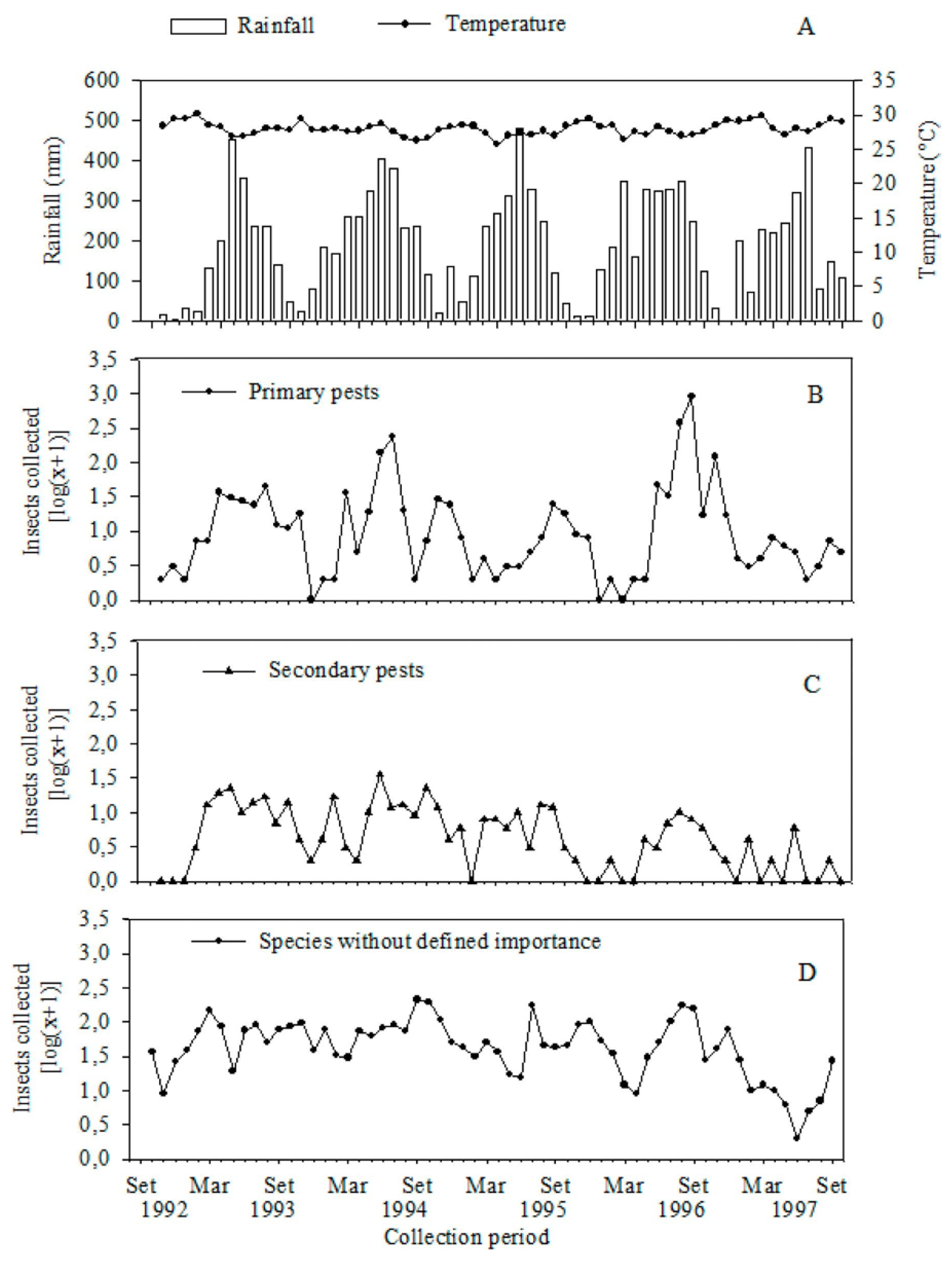
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Luiz de Queiroz, D.; Zanol, K.M.R.; Oliveira, E.B.; Anjos, N.; Majer, J. Feeding and oviposition preferences of Ctenarytaina spatulata Taylor (Hemiptera, Psyllidae) for Eucalyptus spp. and other Myrtaceae in Brazil. Revista Bras. Entomol. 2010, 54, 149–153. [Google Scholar] [CrossRef]
- Elaissi, A.; Salah, K.H.; Mabrouk, S.; Larbi, K.M.; Chemli, R.; Harzallah-Skhiri, F. Antibacterial activity and chemical composition of 20 Eucalyptus species’ essential oils. Food Chem. 2011, 129, 1427–1434. [Google Scholar] [CrossRef]
- Brissola, S.H.; Demarco, D. Stem anatomical analysis of Eucalyptus grandis, E. urophylla and E. grandis × E. urophylla: wood development and its industrial importance. Scientia Forestalis 2011, 39, 317–330. [Google Scholar]
- Zanuncio, A.J.V.; Pastori, P.L.; Kirkendall, L.R.; Lino-Neto, J.; Serrão, J.E.; Zanuncio, J.C. Megaplatypus mutatus (Chapuis) (Coleoptera: Curculionidae: Platypodinae) attacking hybrid Eucalyptus clones in southern Espírito Santo, Brazil. Coleopt. Bull. 2010, 64, 81–83. [Google Scholar] [CrossRef]
- Potts, B.M.; Dungey, H.S. Interspecific hybridization of Eucalyptus: Key issues for breeders and geneticists. New For. 2004, 27, 115–138. [Google Scholar]
- Zanuncio, J.C.; Zanuncio, T.V.; Freitas, F.A.; Pratissoli, D. Population density of Lepidoptera in a plantation of Eucalyptus urophylla in the State of Minas Gerais, Brazil. Anim. Biol. 2003, 53, 17–26. [Google Scholar] [CrossRef]
- Freitas, F.A.; Zanuncio, T.V.; Zanuncio, J.C.; Conceição, P.M.; Fialho, M.C.Q.; Bernardino, A.S. Effect of plant age, temperature and rainfall on Lepidoptera insect pests collected with light traps in a Eucalyptus grandis plantation in Brazil. Ann. For. Sci. 2005, 62, 85–90. [Google Scholar] [CrossRef]
- Pereira, A.I.A.; Curvêlo, C.R.S.; Guerra, A.M.N.M.; Andrade, G.S.; Zanuncio, J.C. Eucalyptus cloeziana as a new host to Hylesia paulex (Lepidoptera: Saturniidae) in Southeast Brazil. Revista Caatinga 2009, 22, 1–5. [Google Scholar]
- Zanuncio, J.C.; Guedes, R.N.C.; Zanuncio, T.V.; Fabres, A.S. Species richness and abundance of defoliating Lepidoptera associated with Eucalyptus grandis in Brazil and their response to plant age. Austral Ecol. 2001, 26, 582–589. [Google Scholar] [CrossRef]
- Zanuncio, T.V.; Zanuncio, J.C.; Miranda, M.M.M.; Medeiros, A.G.B. Effect of plantation age on diversity and population fluctuation of Lepidoptera collected in Eucalyptus plantations in Brazil. For. Ecol. Manag. 1998, 108, 91–98. [Google Scholar] [CrossRef]
- Zanuncio, T.V.; Zanuncio, J.C.; Freitas, F.A.; Pratissoli, D.; Sediyama, C.A.Z.; Maffia, V.P. Main lepidopteran pest species from an eucalyptus plantation in Minas Gerais, Brazil. Revista Biol. Trop. 2006, 54, 553–560. [Google Scholar]
- Pereira, J.M.M.; Zanuncio, T.V.; Zanuncio, J.C.; Pratissoli, D. Lepidoptera pests collected in Eucalyptus urophylla plantations during five years in Três Marias, State of Minas Gerais, Brazil. Revista Biol. Trop. 2001, 49, 997–1006. [Google Scholar]
- Salati, E.; dos Santos, Â.A.; Klabin, I. Relevant environmental issues. Estud. Av. 2006, 20, 107–127. [Google Scholar] [CrossRef]
- Kitching, R.L.; Orr, A.G.; Thalib, L.; Mitchell, H.; Hopkins, M.S.; Graham, A.W. Moth assemblages as indicators of environmental quality in remnants of upland Australian rain forest. J. Appl. Ecol. 2000, 37, 284–297. [Google Scholar]
- Osaki, K.; Sayama, K.; Ueda, A.; Ito, M.; Tabuchi, K.; Hironaga, T. Short-term, efficient sampling strategies for estimating forest moth diversity using light traps. Ann. Entomol. Soc. Am. 2011, 104, 739–748. [Google Scholar] [CrossRef]
- Hawes, J.; Motta, C.S.; Overal, W.L.; Barlow, J.; Gardner, T.A.; Peres, C.A. Diversity and composition of Amazonian moths in primary, secondary and plantation forests. J. Trop. Ecol. 2009, 25, 281–300. [Google Scholar] [CrossRef]
- Zanuncio, J.C.; Zanuncio, T.V.; Lopes, E.T.; Ramalho, F. Temporal variation of Lepidoptera collected in an Eucalyptus plantation in the state of Goiás, Brazil. Neth. J. Zool. 2000, 50, 435–443. [Google Scholar]
- Wallner, W.E. Factors affecting insect population dynamics: Differences between outbreak and non-outbreak species. Ann. Rev. Entomol. 1987, 32, 317–340. [Google Scholar] [CrossRef]
- Oliveira, H.N.; Zanuncio, J.C.; Pedruzzi, E.P.; Espindula, M.C. Rearing of Thyrinteina arnobia (Lepidoptera: Geometridae) on guava and eucalyptus in laboratory. Braz. Arch. Biol. Technol. 2005, 48, 801–806. [Google Scholar]
- Zanuncio, J.C.; Alves, J.B.; Santos, G.P.; Campos, W.O. Levantamento e flutuação populacional de lepidópteros associados à eucaliptocultura: VI- Região de Belo Oriente, Minas Gerais. Pesqui. Agropecu. Bras. 1993, 28, 1121–1127. [Google Scholar]
- Paine, T.D.; Hanlon, C.C. Integration of tactics for management of Eucalyptus herbivores: influence of moisture and nitrogen fertilization on red gum lerp psyllid colonization. Entomol. Exp. Appl. 2010, 137, 290–295. [Google Scholar] [CrossRef]
- Zanuncio, J.C.; Mezzomo, J.A.; Guedes, R.N.C.; Oliveira, A.C. Influence of strips of native vegetation on Lepidoptera associated with Eucalyptus cloeziana in Brazil. For. Ecol. Manag. 1998, 108, 85–90. [Google Scholar] [CrossRef]
- Thomas, J.A.; Bourn, N.A.D.; Clarke, R.T.; Stewart, K.E.; Simcox, D.J.; Pearman, G.S.; Curtis, R.; Goodger, B. The quality and isolation of habitat patches both determine where butterflies persist in fragmented landscapes. Proc. Biol. Sci. 2001, 268, 1791–1796. [Google Scholar] [CrossRef]
- Teja, T.; Roland, B. Plant-insect interactions in fragmented landscapes. Ann. Rev. Entomol. 2004, 49, 405–430. [Google Scholar] [CrossRef]
- Watt, A.D.; Stork, N.E.; Mcbeath, C.; Lawson, G.L. Impact of forest management on insect abundance and damage in a lowland tropical forest in southern Cameroon. J. Appl. Ecol. 1997, 34, 985–998. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zanuncio, J.C.; Lemes, P.G.; Santos, G.P.; Soares, M.A.; Wilcken, C.F.; Serrão, J.E. Population Dynamics of Lepidoptera Pests in Eucalyptus urophylla Plantations in the Brazilian Amazonia. Forests 2014, 5, 72-87. https://doi.org/10.3390/f5010072
Zanuncio JC, Lemes PG, Santos GP, Soares MA, Wilcken CF, Serrão JE. Population Dynamics of Lepidoptera Pests in Eucalyptus urophylla Plantations in the Brazilian Amazonia. Forests. 2014; 5(1):72-87. https://doi.org/10.3390/f5010072
Chicago/Turabian StyleZanuncio, José Cola, Pedro Guilherme Lemes, Germi Porto Santos, Marcus Alvarenga Soares, Carlos Frederico Wilcken, and José Eduardo Serrão. 2014. "Population Dynamics of Lepidoptera Pests in Eucalyptus urophylla Plantations in the Brazilian Amazonia" Forests 5, no. 1: 72-87. https://doi.org/10.3390/f5010072





