Concentration Levels of Imidacloprid and Dinotefuran in Five Tissue Types of Black Walnut, Juglans nigra
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Sites
2.2. Sampling
2.3. Extraction, Clean-Up and Quantification of Chemicals
2.4. Data Analysis
3. Results
3.1. Concentration Levels of Imidacloprid in Walnut Tissue Types
| Sampling Time | |||||
|---|---|---|---|---|---|
| 20 May 2011 | 20 July 2011 | 20 September 2011 | 20 December 2011 | 20 April 2011 | |
| Tissue Types | |||||
| Upper Twig | 11.98 ± 1.74 dE ** | 63.62 ± 2.55 dD | 87.30 ± 1.34 dC | 101.23 ± 2.34 dB | 123.11 ± 2.10 dA |
| Upper Leaf | 12.89 ± 1.25 cE | 68.85 ± 3.25 cD | 97.13 ± 1.78 cC | 123.09 ± 2.14.0 cB | 134.22 ± 1.09 cA |
| Upper Nutmeat | – | – | 72.19 ± 1.99 e | – | – |
| Upper Husk | – | – | 11.79 ± 1.13 h | – | – |
| Lower Twig | 24.79 ± 1.66 bE | 96.79 ± 2.06 bD | 112.97 ± 1.24 bC | 134.45 ± 2.67 bB | 157.43 ± 3.42 bA |
| Lower Leaf | 31.46 ± 1.89 aE | 99.45 ± 2.69 aD | 129.26 ± 1.44 aC | 157.32 ± 2.34 aB | 171.29 ± 1.31 aA |
| Lower Nutmeat | – | – | 84.02 ± 2.56 e | – | – |
| Lower Husk | – | – | 56.40 ± 2.09 f | – | – |
| Trunk Core | 1.25 ± 0.54 eC | 34.56 ± 2.13 eB | 45.05 ± 6.45 gA | 47.13 ± 5.39 eA | 49.56 ± 4.97 eA |
3.2. Concentration Levels of Dinotefuran in Walnut Tissue Types
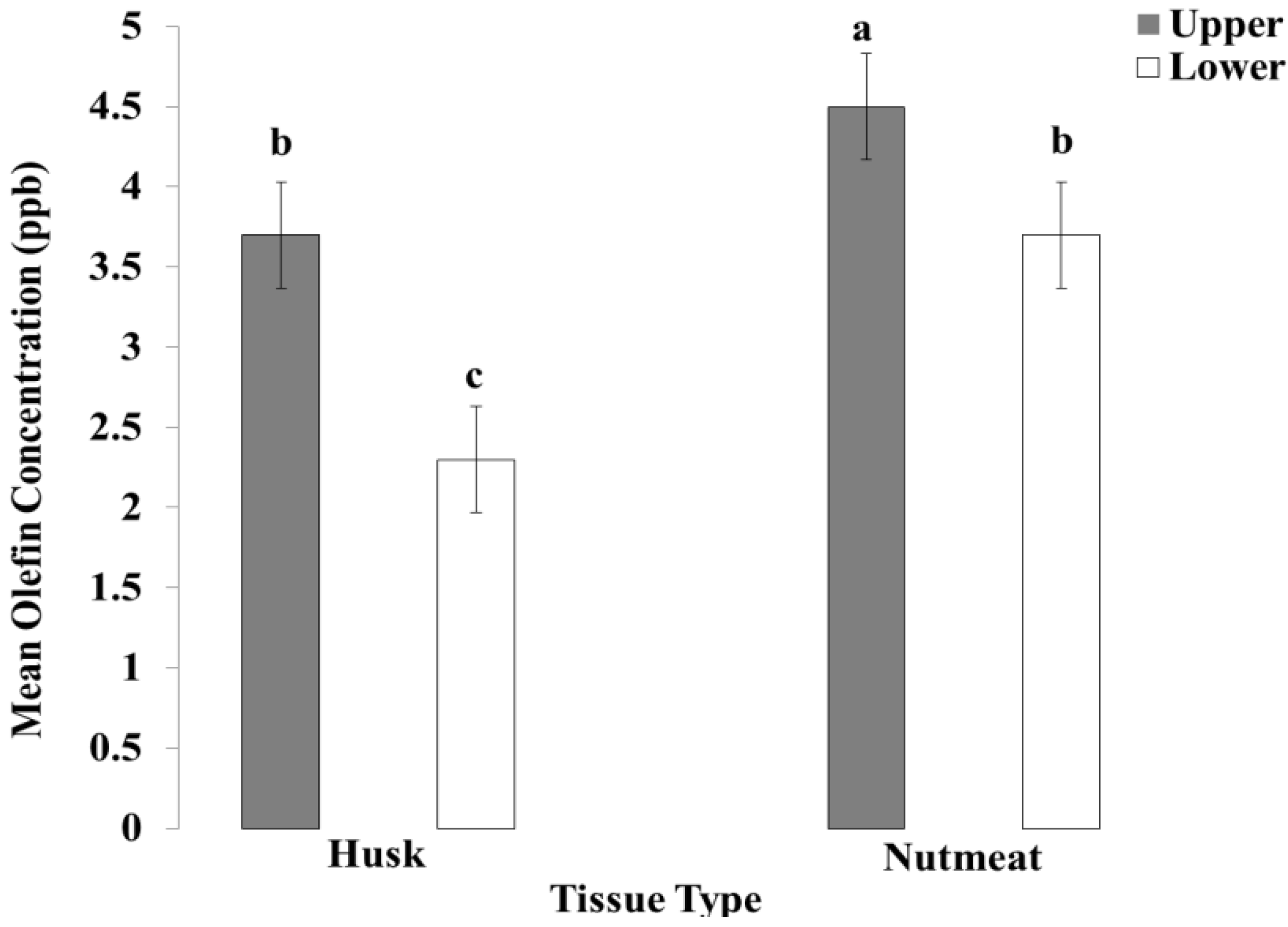
3.3. Concentration Levels of Olefin-Imidacloprid in Walnut Husk and Walnut Nutmeat
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Williams, R.D. Black Walnut. In Silvics of North America; Burns, R.M., Honkala, B.H., Eds.; United States Department of Agriculture: Washington, DC, USA, 1990; pp. 391–399. [Google Scholar]
- Harlow, W.; Harrar, E. Textbook of Dendrology, 5th ed; McGraw-Hill: New York, NY, USA, 1969; p. 213. [Google Scholar]
- Smith, C.C.; Follmer, D. Food preferences of squirrels. Ecology 1972, 53, 82–91. [Google Scholar] [CrossRef]
- Walnut Tree Quarantine in Jefferson County due to Thousand Cankers Disease, 2012. Tennessee Government. Available online: http://news.tn.gov/node/10016 (accessed on 15 April 2013).
- Newton, L.P.; Fowler, G.; Neely, A.D.; Schall, R.A.; Takeuchi, Y. Pathway Assessment: Geosmithia sp. and Pityophthorus juglandis Blackman Movement from the Western into the Eastern United States, 2009. USDA Animal and Plant Health Inspection Service. Available online: http://mda.mo.gov/plants/pdf/tc_pathwayanalysis.pdf (accessed on 20 April 2012).
- Reid, W.; Coggeshall, M.V.; Hunt, K.L. Cultivar Evaluation and Development for Black Walnut Orchards. In Black Walnut in a New Century, Proceedings of the 6th Walnut Council Research symposium, Lafayette, IN, USA, 25–28 July 2004; pp. 18–24.
- Weber, B.C.; Anderson, R.L.; Hoffard, W.H. How to Diagnose Black Walnut Damage, General Technical Report NC-57; United States Department of Agriculture Forest Service: St. Paul, MN, USA, 1992; p. 20. [Google Scholar]
- Grant, J.F.; Windham, W.T.; Walker, H.G.; Wiggins, G.J.; Lambdin, P.L. Initial assessment of thousand cankers disease on black walnut, Juglans nigra, in eastern Tennesse. Forests 2011, 2, 741–748. [Google Scholar] [CrossRef]
- Leslie, C.A.; Seybold, S.J.; Graves, A.D.; Cranshaw, W.; Tisserat, N. Potential impacts of thousand cankers disease on commercial walnut production and walnut germplasm conservation. Acta Hortic. 2010, 861, 431–434. [Google Scholar]
- Seybold, S.; Haugen, D.; O’Brien, J.; Graves, A. Pest Alert: Thousand Cankers Disease, 2010. United States Forest Service. Available online: http://na.fs.fed.us/pubs/palerts/cankers_disease/thousand_cankers_disease_screen_res.pdf (accessed on 22 February 2013).
- Tisserat, N.; Cranshaw, W.; Leatherman, D.; Utley, C.; Alexander, K. Black walnut mortality in Colorado caused by the walnut twig beetle and thousand cankers disease. Plant Health Prog. 2009. [Google Scholar] [CrossRef]
- Thousand Cankers Disease, 2010. Tennessee Department of Agriculture. Available online: http://tennessee.gov/agriculture/regulatory/tcd.html (accessed on 17 November 2010).
- McCullough, D.G.; Smitley, D.R. Evaluation of insecticides to reduce maturation feeding by Tomicus piniperda (Coleoptera: Scolytidae) in Scotch pine. J. Econ. Entomol. 1995, 88, 693–699. [Google Scholar]
- Poland, T.M.; Haack, R.A.; Petrice, T.R.; Miller, D.L.; Bauer, L.S.; Gao, R. Field evaluations of systemic insecticides for control of Anoplophora glabripennis (Coleoptera: Cerambycidae) in China. J. Econ. Entomol. 2006, 99, 383–392. [Google Scholar]
- Russell, C.; Ugine, T.A.; Hajek, A.E. The effect of imidacloprid on Metarhizium anisopliae treated Asian longhorned beetle (Anoplophora glabripennis (Motschulsky)) (Coleoptera: Cerambycidae) survival, feeding and conidia production from mycosed individuals. J. Invertebr. Pathol. 2010, 105, 305–311. [Google Scholar] [CrossRef]
- Ugine, T.A.; Gardescu, S.; Lewis, P.A.; Hajek, A.E. Efficacy of imidacloprid, trunk-injected into Acer platanoides, for control of adult Asian longhorned beetle (Coleoptera: Cerambycidae). J. Econ. Entomol. 2012, 105, 2015–2028. [Google Scholar] [CrossRef]
- Herms, D.A.; McCullough, D.G.; Smitley, D.R.; Sadof, C.S.; Williamson, R.C.; Nixon, P.L. Insecticide Options for Protecting Ash Trees from Emerald Ash Borer; National IPM Center: Illinois, IL, USA, 2009; p. 12. [Google Scholar]
- Smitley, D.R.; Doccola, J.J.; Cox, D.L. Multiple-year protection of ash trees from emerald ash borer with a single trunk injection of emamectin benzoate, and single-year protection with an imidacloprid basal drench. Arboric. Urban For. 2010, 36, 206–211. [Google Scholar]
- Leicht, W. Imidacloprid, a chloronicotinyl insecticide: Biological activity and agricultural significance. Pflanzenschutz-Nachrichten Bayer 1996, 42, 71–84. [Google Scholar]
- Tolerances and Exemptions for Pesticide Chemical Residues in Food; Subpart C—Specific Tolerances—180.603 Dinotefuran; Tolerances for Residues, 2005. Office of the Federal Register. Available online: http://www.gpo.gov/fdsys/pkg/CFR-2010-title40-vol23/pdf/CFR-2010-title40-vol23-sec180-603.pdf (accessed on 1 March 2013).
- Protection of Environment; Part 180—Tolerances and Exemptions for Pesticide Chemical Residues in Food; Subpart C-Specific Tolerances—180.472 Imidacloprid; Tolerances for Residues, 2010 . Office of the Federal Register. Available online: http://www.gpo.gov/fdsys/pkg/CFR-2010-title40-vol23/pdf/CFR-2010-title40-vol23-sec180-472.pdf (accessed on 1 March 2013).
- Tanis, S.R.; Cregg, B.M.; Mota-Sanchez, D.; McCullough, D.G.; Poland, T.M. Spatial and temporal distribution of trunk-injected 14C-imidacloprid in Fraxinus trees. Pest Manag. Sci. 2011, 68, 529–536. [Google Scholar]
- Mota-Sanchez, D.; Cregg, B.M.; McCullough, D.G.; Poland, T.M.; Hollingworth, R.M. Distribution of trunk-injected 14C-imidacloprid in ash trees and effects on emerald ash borer (Coleoptera: Buprestidae) adults. Crop Prot. 2009, 28, 655–661. [Google Scholar] [CrossRef]
- McCullough, D.G.; Poland, T.M.; Anulewicz, A.C.; Lewis, P.; Cappaert, D. Evaluation of Agrilus planipennis (Coleoptera: Buprestidae) control provided by emamectin benzoate and two neonicotinoid insecticides, one and two seasons after treatment. J. Econ. Entomol. 2011, 104, 1599–1612. [Google Scholar] [CrossRef]
- Coots, C.I. Spatial, Temporal, and Tri-Trophic Distribution and Persistence of Imidacloprid, Olefin-Imidacloprid and 5-Hydroxy in Eastern Hemlocks on Hemlock Woolly Adelgid in the Southern Appalachians. Ph.D. Thesis, University of Tennessee, Knoxville, TN, USA, December 2012. [Google Scholar]
- Dilling, C.I.; Lambdin, P.L.; Grant, J.F.; Rhea, J.R. Spatial and temporal distribution of imidacloprid in eastern hemlock in the Southern Appalachians. J. Econ. Entomol. 2010, 103, 368–373. [Google Scholar] [CrossRef]
- Schöning, R.; Schmuck, R. Analytical determination of imidacloprid and relevant metabolite residues by LC/MS/MS. Bull. Insectol. 2003, 56, 41–50. [Google Scholar]
- Kamel, A. Refined methodology for the determination of neonicotinoid pesticides and their metabolites in honey bees and bee products by liquid chromatography—Tandem mass spectrometry (LC-MS/MS). J. Agric. Food Chem. 2010, 58, 5926–5931. [Google Scholar] [CrossRef]
- SAS Institute. SAS User’s Guide; SAS Institute: Cary, NC, USA, 2005; p. 276. [Google Scholar]
- Dilling, C.I.; Lambdin, P.L.; Grant, J.F.; Rhea, J.R. Community response of insects associated with eastern hemlock to imidacloprid and horticultural oil treatments. J. Environ. Entomol. 2009, 38, 53–66. [Google Scholar] [CrossRef]
- Eisenback, B.M.; Salom, S.M.; Kok, L.T.; Lagalante, A.F. Lethal and sublethal effects of imidacloprid on hemlock woolly adelgid (Hemiptera: Adelgidae) and two introduced predator species. J. Econ. Entomol. 2010, 103, 1222–1234. [Google Scholar] [CrossRef]
- Luxmoore, R.J.; Grizzard, T.; Strand, R.H. Nutrient translocation in the outer canopy and understory of an eastern deciduous forest. Forest Sci. 1981, 27, 505–518. [Google Scholar]
- McCullough, D.G.; Poland, T.; Lewis, P.; Molongoski, J. Evaluation of Neo-nicotinoid Insecticides Applied as Trunk Sprays. In Proceedings of the Emerald Ash Borer and Asian Longhorned Beetle Research and Technology Development Meeting, Cincinnati, OH, USA, 29 October 2006–2 November 2006; USDA Forest Service, Forest Health Technology Enterprise Team: Morgantown, WV, USA, 2007; pp. 52–54. [Google Scholar]
- Nauen, R.; Tietjen, K.; Wagner, K.; Elbert, E. Efficacy of plant metabolites of imidacloprid against Myzus persicae and Aphis gossypii (Homoptera: Aphididae). Pest. Sci. 1998, 52, 53–57. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Nix, K.; Lambdin, P.; Grant, J.; Coots, C.; Merten, P. Concentration Levels of Imidacloprid and Dinotefuran in Five Tissue Types of Black Walnut, Juglans nigra. Forests 2013, 4, 887-897. https://doi.org/10.3390/f4040887
Nix K, Lambdin P, Grant J, Coots C, Merten P. Concentration Levels of Imidacloprid and Dinotefuran in Five Tissue Types of Black Walnut, Juglans nigra. Forests. 2013; 4(4):887-897. https://doi.org/10.3390/f4040887
Chicago/Turabian StyleNix, Katheryne, Paris Lambdin, Jerome Grant, Carla Coots, and Paul Merten. 2013. "Concentration Levels of Imidacloprid and Dinotefuran in Five Tissue Types of Black Walnut, Juglans nigra" Forests 4, no. 4: 887-897. https://doi.org/10.3390/f4040887
APA StyleNix, K., Lambdin, P., Grant, J., Coots, C., & Merten, P. (2013). Concentration Levels of Imidacloprid and Dinotefuran in Five Tissue Types of Black Walnut, Juglans nigra. Forests, 4(4), 887-897. https://doi.org/10.3390/f4040887




