New Elastomeric Materials Based on Natural Rubber Obtained by Electron Beam Irradiation for Food and Pharmaceutical Use
Abstract
:1. Introduction
2. Experimental
2.1. Materials
2.2. Blends Preparation
2.3. Electron Beam Irradiation
2.4. Sol-Gel Analysis
2.5. Crosslink Density
2.6. Fourier Transform Infrared Spectroscopy (FTIR)
2.7. Scanning Electron Microscopy (SEM)
2.8. Aqueous Solutions Absorption Test
3. Results and Discussion
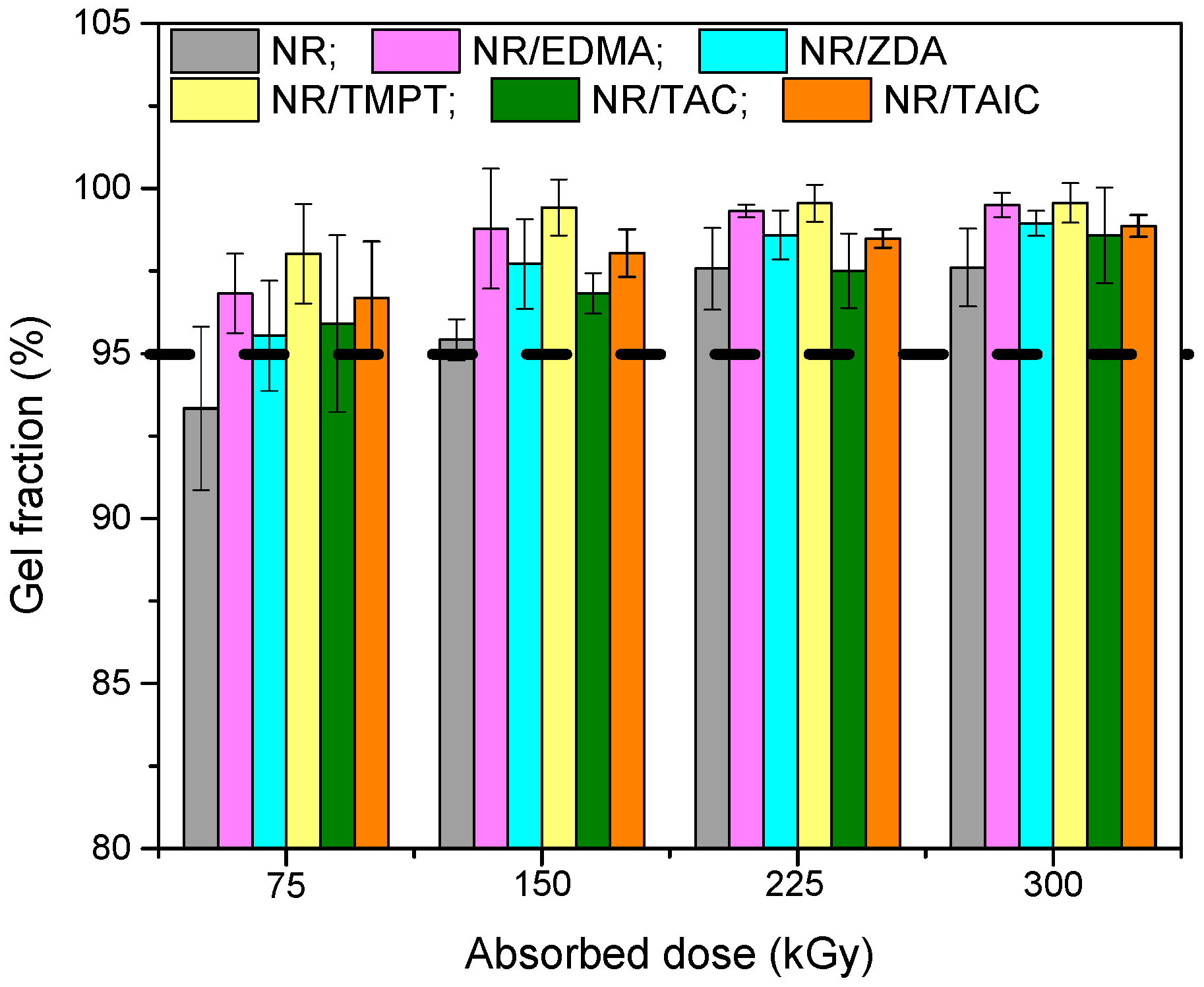
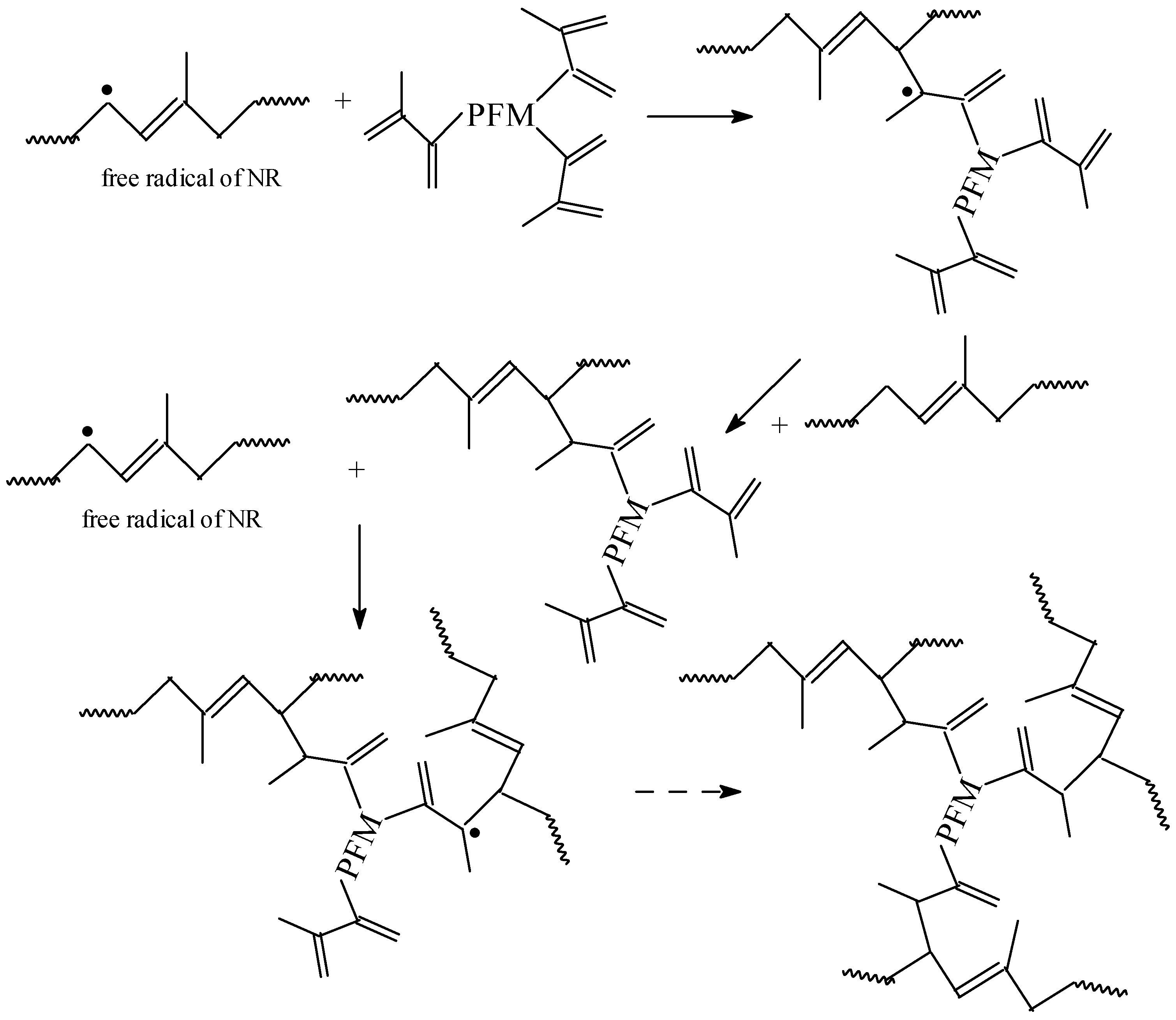
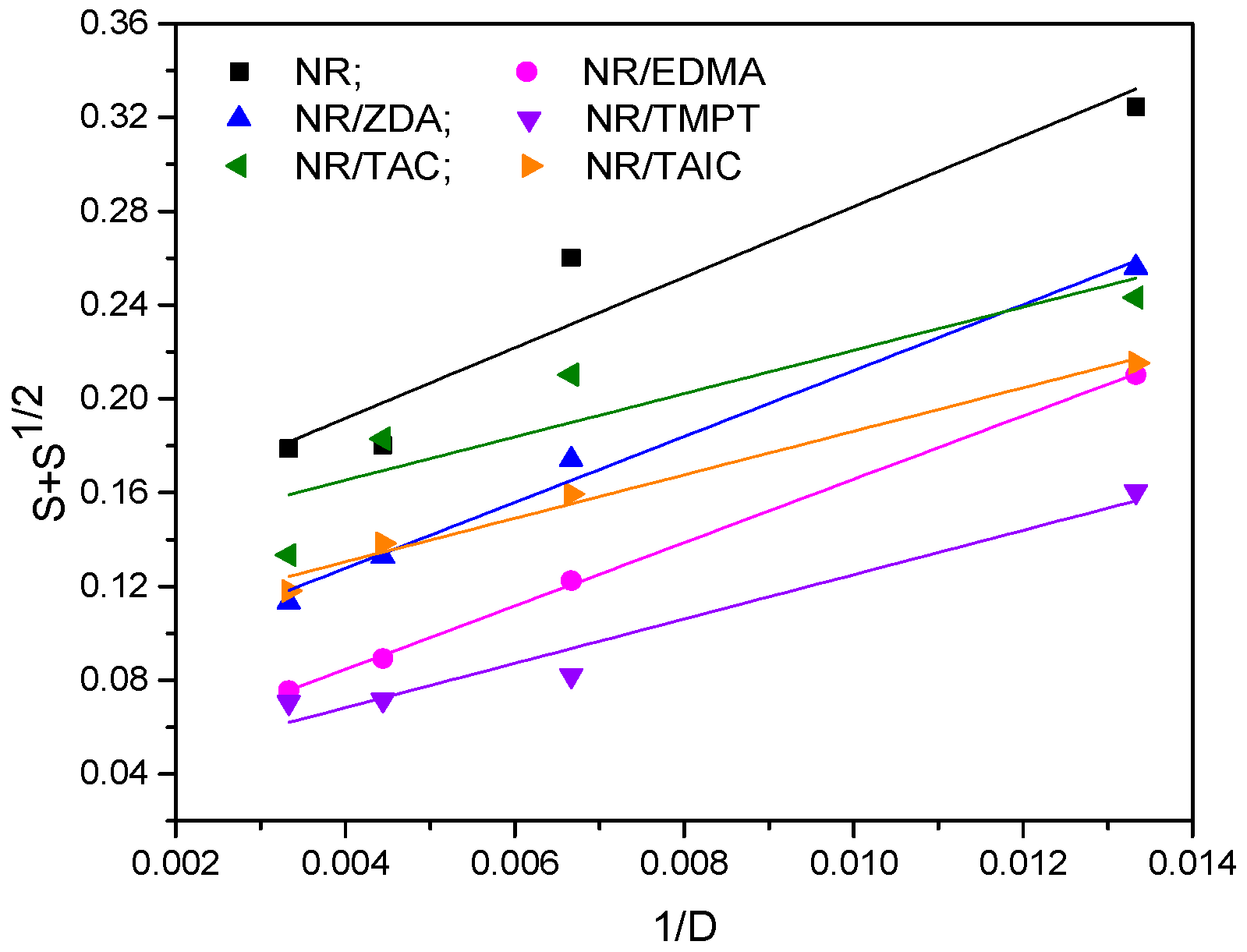
3.1. Gel Fraction and Cross-Link Density of the Blends
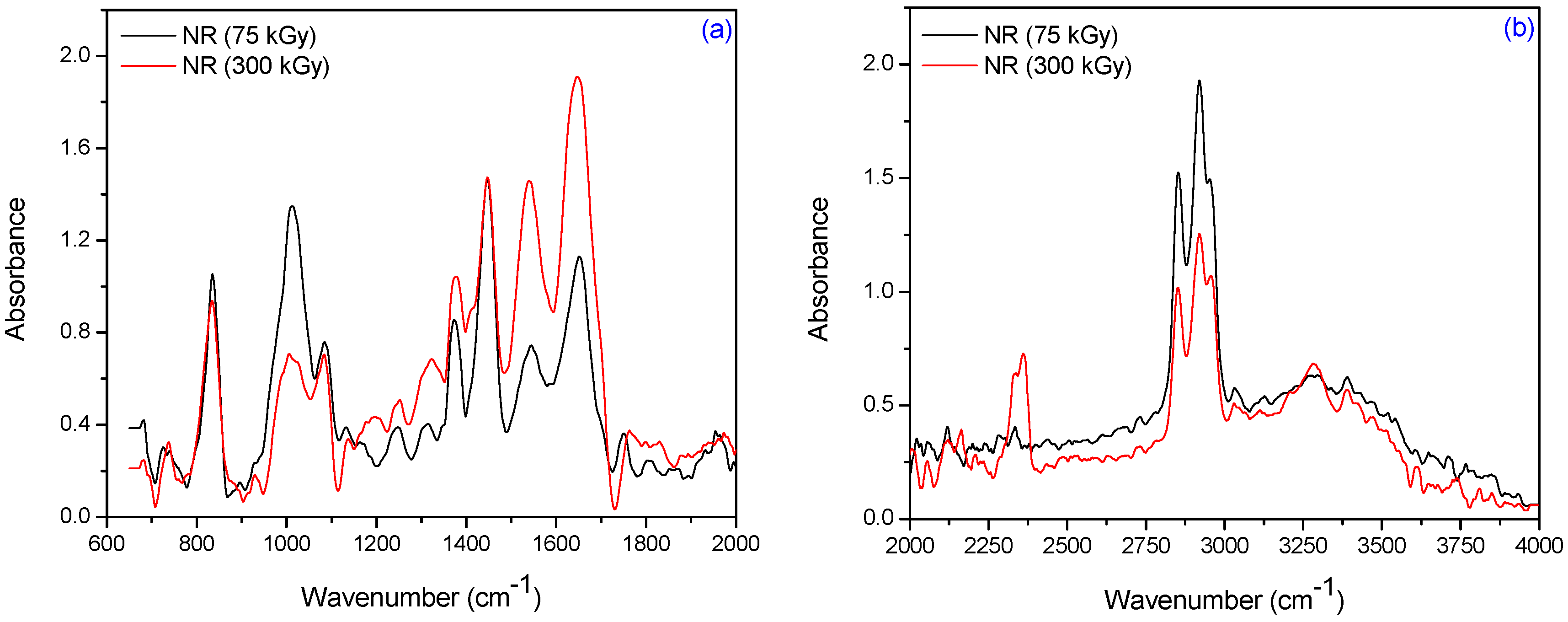
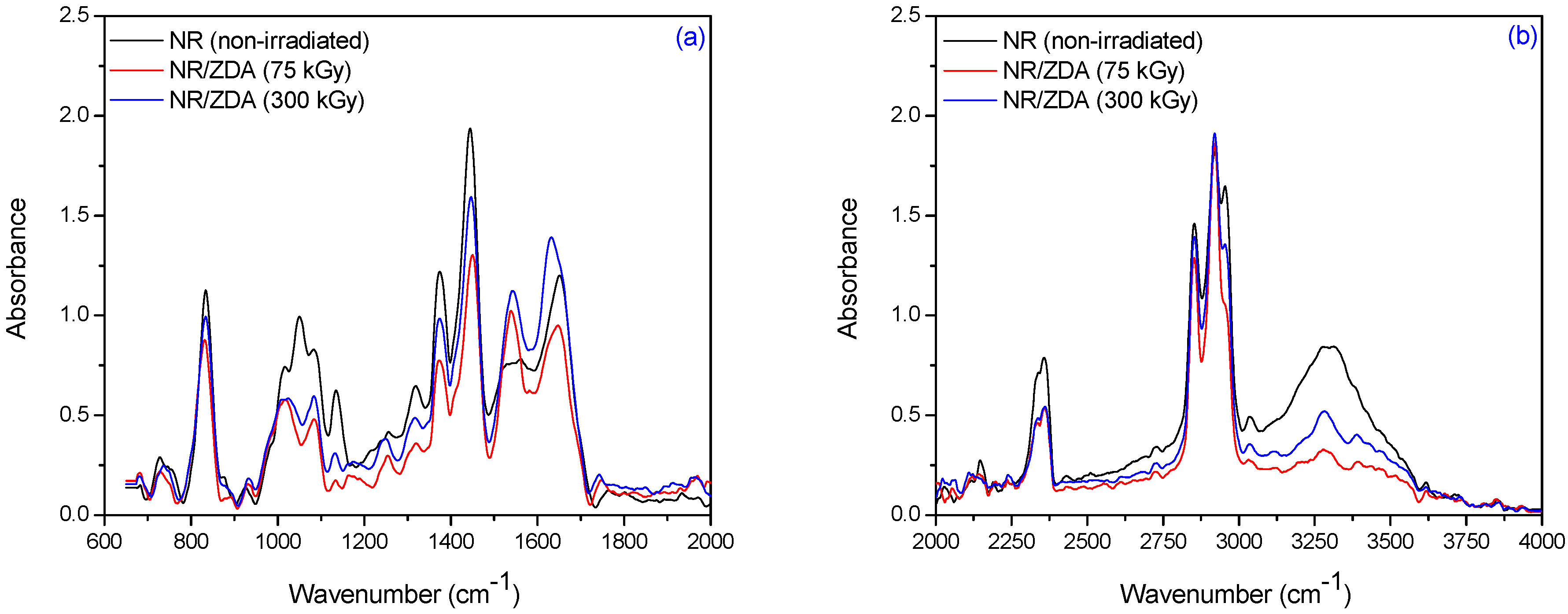
3.2. Fourier Transform Infrared Spectroscopy (FTIR)
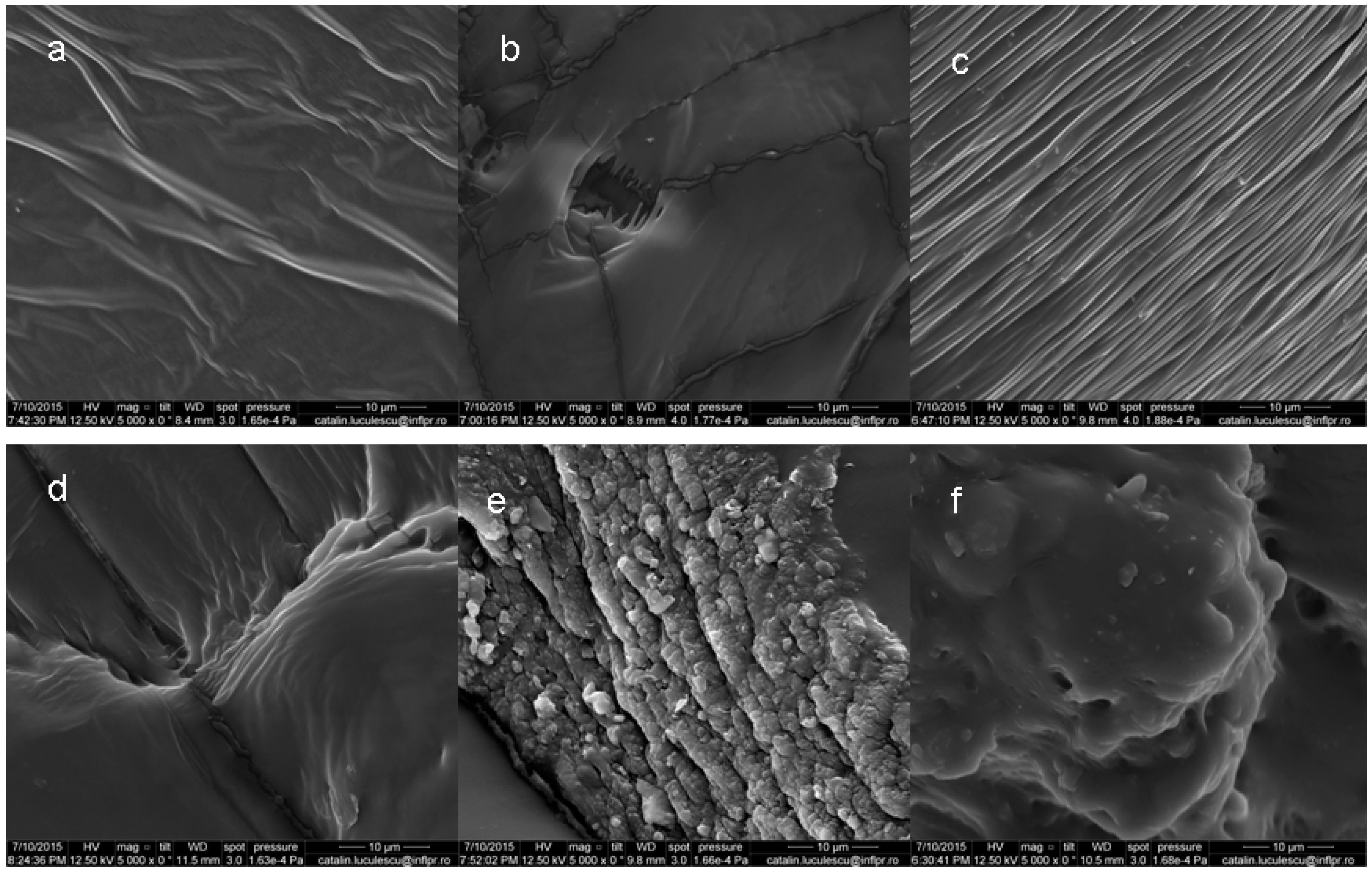
3.3. Scanning Electron Microscopy (SEM)
3.4. Absorption Tests on Different Aqueous Solutions
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- De Coster, N.; Magg, H. NBR in contact with food, potable water, pharmaceutical and cosmetic applications. Kautsch. Gummi Kunstst. 2003, 56, 405–411. [Google Scholar]
- Basfar, A.A.; Abdel-Aziz1, M.M.; Mofti, S. Influence of dierent curing systems on the physico-mechanical properties and stability of SBR and NR rubbers. Radiat. Phys. Chem. 2002, 63, 81–87. [Google Scholar] [CrossRef]
- Milani, G.; Hanel, T.; Donetti, R.; Milani, F. A closed form solution for the vulcanization prediction of NR cured with sulphur and different accelerators. J. Math. Chem. 2015, 53, 975–997. [Google Scholar] [CrossRef]
- Fishbein, L. Chemicals used in the rubber industry: An overview. Scand. J. Work Environ. Health 1983, 9, 7–14. [Google Scholar] [PubMed]
- Alvarez Grima, M.M. Novel Co-Agents for Improved Properties in Peroxide Cure of Saturated Elastomers. Ph.D. Thesis, University of Twente, Enschede, The Netherlands, 2007. [Google Scholar]
- Dluzneski, P.R. Peroxide vulcanization of elastomers. Rubber Chem. Technol. 2001, 74, 451–492. [Google Scholar] [CrossRef]
- Pire, M.; Oikonomou, E.K.; Imbernon, L.; Lorthioir, C.; Iliopoulos, I.; Norvez, S. Crosslinking of epoxidized natural rubber by dicarboxylic acids: An alternative to standard vulcanization. Macromol. Symp. 2013, 331, 89–96. [Google Scholar] [CrossRef]
- Pire, M.; Norvez, S.; Iliopoulos, I.; Rossignol, B.; Leibler, L. Dicarboxylic acids may compete with standard vulcanisation processes for crosslinking epoxidised natural rubber. Compos. Interfaces 2014, 21, 45–50. [Google Scholar] [CrossRef]
- MGM Rubber Company-Research and Development, Electron Beam Radiation Technology for Curing. 2007. Available online: http://www.mgmrc.com/rd-news/latest-technologies (accessed on 5 July 2016).
- Manaila, E.; Stelescu, M.D.; Craciun, G. Aspects regarding radiation crosslinking of elastomers. In Advanced Elastomers—Technology, Properties and Applications; Boczkowska, A., Ed.; InTech: Rijeka, Croatia, 2012; pp. 3–34. [Google Scholar]
- Gehman, S.D. Network chain distribution and strength of vulcanizates. Rubber Chem. Technol. 1969, 42, 659–665. [Google Scholar] [CrossRef]
- Tobolsky, A.V.; Lyons, P.F. Tensile strength of rubbers. J. Polym. Sci. Part A-2 Polym. Phys. 1968, 6, 1561–1566. [Google Scholar] [CrossRef]
- Manaila, E.; Craciun, G.; Stelescu, M.D.; Ighigeanu, D.; Ficai, M. Radiation vulcanization of natural rubber with polyfunctional monomers. Polym. Bull. 2014, 71, 57–82. [Google Scholar] [CrossRef]
- Youm, J.S.; Kim, J.C.; Yang, K.S. Elastic property of polyolefin elastomer film cross linked by electron beam irradiation. Fibers Polym. 2012, 13, 1165–1169. [Google Scholar] [CrossRef]
- Drobny, J.G. Ionizing Radiation and Polymers: Principles, Technology and Applications, 1st ed.; Elsevier/William Andrew: Norwich, NY, USA, 2012; pp. 88–91. [Google Scholar]
- Endstra, W.C. Application of co-agents for peroxide cross-linking. Kautsch. Gummi Kunstst. 1990, 43, 790–793. [Google Scholar]
- Dikland, H.G.; Ruardy, T.; Van der Does, L.; Bantjes, A. New coagents in peroxide vulcanization of EPM. Rubber Chem. Technol. 1993, 66, 693–711. [Google Scholar] [CrossRef]
- Chaiear, N. Health and Safety in the Rubber Industry; SMITHERS RAPRA: Shropshire, UK, 2001; pp. 102–103. [Google Scholar]
- Smoke, T.; Smoking, I. IARC monographs on the evaluation of carcinogenic risks to humans. In Silica, Some Silicates, Coal Dust and Para—Aramid Fibrils; World Health Organization, International Agency for Research on Cancer (IARC): Lyon, France, 1997; Volume 68, pp. 380–385. Available online: https://monographs.iarc.fr/ENG/Monographs/vol68/mono68.pdf (accessed on 18 September 2014).
- Beliczky, L.S.; Fajen, J. Rubber industry. General profile. In Encyclopaedia of Occupational Health and Safety, 4th ed.; Stellman, J.M., Ed.; International Labor Organization: Geneva, Switzerland, 1998; Volume 3, pp. 80.1–80.3. [Google Scholar]
- Marshall, R. Cleland, Industrial applications of electron accelerators—Ion beam applications. In Proceedings of the CERN Accelerator School/Small Accelerator Course, Zeegse, The Netherlands, 24 May–2 June 2005.
- Manaila, E.; Stelescu, M.D.; Craciun, G.; Ighigeanu, D. Wood sawdust/natural rubber ecocomposites cross-linked by electron beam irradiation. Materials 2016, 9, 503. [Google Scholar] [CrossRef]
- Lopez-Manchado, M.A.; Herrero, B.; Arroyo, M. Preparation and characterization of organoclay nanocomposites based on natural rubber. Polym. Int. 2003, 52, 1070–1077. [Google Scholar] [CrossRef]
- Chenal, J.M.; Chazeau, L.; Guy, L.; Bomal, Y.; Gauthier, C. Molecular weight between physical entanglements in natural rubber: A critical parameter during strain-induced crystallization. Polymer 2007, 48, 1042–1046. [Google Scholar] [CrossRef]
- Stelescu, M.D. Characteristics of silicone rubber blends. Leather Footwear J. 2010, 10, 51–58. [Google Scholar]
- Ratnam, C.T.; Nasir, M.; Baharin, A.; Zaman, K. Electron beam irradiation of epoxidized natural rubber. Nucl. Instrum. Methods B 2000, 171, 455–464. [Google Scholar] [CrossRef]
- Muhammad, N.H.; Abdullah, I.; Mohd, D.H. Effect of electron beam irradiation on natural rubber/linear low density polyethylene blends with m-phenylenebismaleimide. Sains Malays. 2011, 40, 685–689. [Google Scholar]
- Yasin, T.; Ahmed, S.; Ahmed, M.; Yoshii, F. Effect of concentration of polyfunctional monomers on physical properties of acrylonitrile-butadiene rubber under-electron beam irradiation. Radiat. Phys. Chem. 2005, 73, 155–158. [Google Scholar] [CrossRef]
- Henning, S.K.; Boye, W.M. Fundamentals of curing elastomers with peroxides and coagents II: Understanding the relationship between coagent and elastomer. Rubber World 2009, 240, 31–39. [Google Scholar]
- Henning, S.K.; Costin, R. Fundamentals of curing elastomers with peroxides and coagents I: Coagent structure—Property relationships. In Proceedings of the Technical Meeting of the Rubber Division, American Chemical Society, San Antonio, TX, USA, 16–18 May 2005; pp. 1–18.
- Henning, S.K. The use of coagents in the radical cure of elastomers. In Proceedings of the 56th International Wire & Cable Symposium, Lake Buena Vista, FL, USA, 11–14 November 2007; pp. 587–593.
- Manshaie, R.; Khorasani, S.N.; Veshare, S.J.; Abadchi, M.R. Effect of electron beam irradiation on the properties of natural rubber (NR)/styrene-butadiene rubber (SBR) blend. Radiat. Phys. Chem. 2011, 80, 100–106. [Google Scholar] [CrossRef]
- Dikland, H.G.; Vanderdoes, L.; Bantjes, A. FT-IR spectroscopy, a major tool for the analysis of peroxide vulcanization processes in the presence of coagents. I. mechanism of EPM peroxide vulcanization with aromatic bis(Allyl)esters as coagents. Rubber Chem. Technol. 1993, 66, 196–212. [Google Scholar] [CrossRef]
- Murgic, Z.H.; Jelenic, J.; Murgic, L. The mechanism of triallylcyanurate as a coagent in EPDM peroxide vulcanization. Polym. Eng. Sci. 1998, 38, 689–692. [Google Scholar] [CrossRef]
- Tikku, V.K.; Biswas, G.; Despande, R.S.; Majali, A.B.; Chaki, T.K.; Bhowmich, A.K. Electron beam initiated grafting of trimethylolpropane trimethacrylate onto polyethylene—Structure and properties. Radiat. Phys. Chem. 1995, 45, 829–833. [Google Scholar] [CrossRef]
- Van Duin, M. Chemistry of EPDM cross-linking. Kautch. Gummi Kunstst. 2002, 55, 150–156. [Google Scholar]
- Shyichuk, A.; Tokaryk, G. A comparison of methods to determination of macromolecule crosslinking yield from gel fraction data. Polimery 2005, 50, 219–221. [Google Scholar]
- Charlesby, A.; Pinner, S.H. Analysis of the solubility behaviour of irradiated polyethylene and other polymers. Proc. R Soc. A Math. Phys. Eng. Sci. 1959, 249, 367–386. [Google Scholar] [CrossRef]
- Ali, A.M.M.; Subban, R.H.Y.; Bahron, H.; Winie, T.; Latif, F.; Yahya, M.Z.A. Grafted natural rubber-based polymer electrolytes: ATR-FTIR and conductivity studies. Ionics 2008, 14, 491–500. [Google Scholar] [CrossRef]
- Aik-Hwee, E.; Tanaka, Y.; Seng-Neon, G. FTIR studies on amino groups in purified Hevea rubber (short communication). J. Nat. Rubber Res. 1992, 7, 152–155. [Google Scholar]
- Chaikumpollert, O.; Yamamoto, Y.; Suchiva, K.; Kawahara, S. Protein-free natural rubber. Colloid Polym. Sci. 2012, 290, 331–338. [Google Scholar] [CrossRef]
- Watcharakul, N.; Poompradub, S.; Prasassarakich, P. In situ silica reinforcement of methyl methacrylate grafted natural rubber by sol–gel process. J. Sol-Gel. Sci. Technol. 2011, 58, 407–418. [Google Scholar] [CrossRef]
- Rajan, R.; Varghese, S.; George, K.E. Role of coagents in peroxide vulcanization of natural rubber. Rubber Chem. Technol. 2013, 86, 488–502. [Google Scholar] [CrossRef]
- Shmakova, N.A.; Feldman, V.I.; Sukhov, F.F. IR spectroscopic study of chemical transformations upon irradiation of the poly(vinyl chloride)–triallyl cyanurate system. High Energy Chem. 2001, 35, 224–228. [Google Scholar] [CrossRef]
- Pichayakorn, W.; Suksaeree, J.; Boonme, P. Chapter 13: Blends and IPNs of natural rubber with acrylic plastics. In Natural Rubber Materials, 2013 ed.; Thomas, S., Rajisha, K.R., Maria, H.J., Chan, C.H., Pothen, L.A., Eds.; Royal Society of Chemistry Publications: Cambridge, UK, 2014; Volume 1, p. 312. [Google Scholar]
- Mateev, M.; Karageorgiev, S. Electron microscopy analysis of the effect of EB irradiation on the macromolecular structure and crosslinking parameters in LDPE films. Radiat. Phys. Chem. 1996, 48, 443–448. [Google Scholar] [CrossRef]
- Dahlan, H.M.; Khairul Zamana, M.D.; Ibrahim, A. Liquid natural rubber (LNR) as a compatibiliser in NR/LLDPE blendsFII: The eects of electron-beam (EB) irradiation. Radiat. Phys. Chem. 2002, 64, 429–436. [Google Scholar] [CrossRef]
- Ratnam, C.T.; Mohamad, Z.; Siddiqui, M.K. Chapter 12: Radiation processing of natural rubber with vinyl plastics. In Natural Rubber Materials, 2013 ed.; Thomas, S., Rajisha, K.R., Maria, H.J., Chan, C.H., Pothen, L.A., Eds.; Royal Society of Chemistry Publications: Cambridge, UK, 2014; Volume 1, p. 296. [Google Scholar]
- Ng, M.X.; Ong, S.P.; Chin, N.L.; Chuah, L.A.; Law, C.L. Review of food toxicological issues associated in rubber products. In Proceedings of the 3rd International Symposium on Processing of Foods, Vegetables and Fruits (ISPFVF 2014), Kuala Lumpur, Malaysia, 11–13 August 2014; pp. 1–7.
- Kuempel, E.D.; Sorahan, T. Identification of Research Needs to Resolve the Carcinogenicity of High-Priority IARC Carcinogens; IARC Technical Publication No. 42; International Agency for Research on Cancer: Lyon, France, 2010; pp. 61–72. Available online: https://monographs.iarc.fr/ENG/Publications/techrep42/TR42-Full.pdf (accessed on 10 September 2016).
- Cao, X.L. Phthalate esters in foods: Sources, occurrence, and analytical methods. Compr. Rev. Food Sci. Food Saf. 2010, 9, 21–43. [Google Scholar] [CrossRef]
- Lahimer, M.C.; Ayed, N.; Horriche, J.; Belgaie, S. Characterization of plastic packaging additives: Food contact, stability and toxicity. Arab. J. Chem. 2013, 50, 1–17. [Google Scholar]









| Polyfunctional Monomer | Type/Functionality | Chemical Characteristics | Chemical Structure |
|---|---|---|---|
| Trimethylolpropane-trimethacrylate (TMPT) | I/3 |
|  |
| Zinc-diacrylate (ZDA) | I/2 |
|  |
| Ethylene glycol dimethacrylate (EDMA) | I/2 |
|  |
| Triallylcyanurate (TAC) | II/3 |
|  |
| Triallylisocyanurate (TAIC) | II/3 |
|  |
| Aqueous Solutions | Chemical Formula | Molar Mass (g·mol−1) | Range of Absorption |
|---|---|---|---|
| distilled water |  | 18.01 | |
| sodium hydroxide (NaOH, 1%) |  | 39.99 | −2%–+4% |
| ethylic alcohol (96%) |  | 46.07 | −2%–+7% |
| physiological serum (NaCl, 0.9%) |  | 58.44 | −2%–+4% |
| acetic acid (10%) |  | 60.05 | −2%–+6% |
| glucose (glucose monohydrate, 10%) |  | 180.16 | −2%–+4% |
| Samples | Type and Functionality of PFMs | p0/q0 |
|---|---|---|
| NR | 0.1315 | |
| NR/TAC | II/3 | 0.1283 |
| NR/TAIC | II/3 | 0.0934 |
| NR/ZDA | I/2 | 0.0715 |
| NR/EDMA | I/2 | 0.0306 |
| NR/TMPT | I/3 | 0.0305 |
| Sample Type | Water | Sodium Hydroxide (1%) | Ethylic Alcohol (96%) | Acetic Acid (10%) | Sodium Chloride (0.9%) | Glucose Monohydrate (10%) |
|---|---|---|---|---|---|---|
| NR 0 | ||||||
| 75 kGy | −0.141 ± 0.018 | 0.735 ± 0.084 | −0.561 ± 0.087 | −0.119 ± 0.011 | −0.194 ± 0.018 | −0.149 ± 0.013 |
| 150 kGy | −0.137 ± 0.015 | 0.643 ± 0.134 | −0.457 ± 0.031 | −0.11 ± 0.014 | −0.136 ± 0.017 | −0.093 ± 0.014 |
| 225 kGy | −0.13 ± 0.014 | 0.222 ± 0.042 | −0.43 ± 0.033 | −0.082 ± 0.021 | −0.111 ± 0.011 | −0.085 ± 0.017 |
| 300 kGy | −0.106 ± 0.012 | 0.212 ± 0.054 | −0.432 ± 0.013 | −0.078 ± 0.015 | −0.103 ± 0.011 | −0.073 ± 0.011 |
| NR + TMPT | ||||||
| 75 kGy | −0.189 ± 0.021 | −0.16 ± 0.010 | −0.451 ± 0.057 | −0.091 ± 0.011 | −0.243 ± 0.020 | −0.077 ± 0.059 |
| 150 kGy | −0.108 ± 0.026 | −0.137 ± 0.015 | −0.262 ± 0.017 | −0.053 ± 0.012 | −0.133 ± 0.010 | −0.034 ± 0.014 |
| 225 kGy | 0.103 ± 0.013 | −0.104 ± 0.015 | −0.255 ± 0.029 | −0.033 ± 0.012 | −0.103 ± 0.013 | −0.026 ± 0.010 |
| 300 kGy | −0.104 ± 0.010 | 0.075 ± 0.028 | −0.242 ± 0.010 | −0.023 ± 0.011 | −0.093 ± 0.021 | −0.033 ± 0.011 |
| NR + EDMA | ||||||
| 75 kGy | −0.305 ± 0.073 | 0.357 ± 0.014 | −0.52 ± 0.025 | −0.077 ± 0.048 | −0.288 ± 0.051 | −0.208 ± 0.018 |
| 150 kGy | −0.216 ± 0.032 | 0.324 ± 0.324 | −0.352 ± 0.040 | −0.072 ± 0.035 | −0.217 ± 0.026 | −0.176 ± 0.025 |
| 225 kGy | −0.197 ± 0.037 | 0.376 ± 0.073 | −0.286 ± 0.033 | −0.043 ± 0.065 | −0.165 ± 0.015 | −0.171 ± 0.048 |
| 300 kGy | −0.138 ± 0.052 | 0.622 ± 0.104 | −0.284 ± 0.011 | −0.016 ± 0.049 | −0.158 ± 0.034 | −0.137 ± 0.023 |
| NR + ZDA | ||||||
| 75 kGy | −0.285 ± 0.049 | 0.438 ± 0.142 | −0.466 ± 0.069 | −0.096 ± 0.015 | −0.292 ± 0.033 | −0.228 ± 0.014 |
| 150 kGy | −0.235 ± 0.047 | 0.277 ± 0.077 | −0.448 ± 0.031 | 0.055 ± 0.013 | −0.24 ± 0.029 | −0.213 ± 0.041 |
| 225 kGy | −0.204 ± 0.010 | 0.155 ± 0.072 | −0.435 ± 0.068 | −0.043 ± 0.049 | −0.188 ± 0.021 | −0.193 ± 0.021 |
| 300 kGy | −0.165 ± 0.057 | 0.114 ± 0.010 | −0.421 ± 0.039 | −0.047 ± 0.016 | −0.127 ± 0.039 | −0.109 ± 0.016 |
| NR + TAC | ||||||
| 75 kGy | −0.511 ± 0.014 | −0.605 ± 0.029 | −1.152 ± 0.310 | −0.592 ± 0.104 | −0.559 ± 0.054 | −0.432 ± 0.029 |
| 150 kGy | −0.441 ± 0.045 | −0.528 ± 0.069 | −1.016 ± 0.065 | −0.562 ± 0.052 | −0.52 ± 0.038 | −0.412 ± 0.041 |
| 225 kGy | −0.417 ± 0.011 | −0.454 ± 0.045 | −0.626 ± 0.034 | −0.256 ± 0.021 | −0.357 ± 0.032 | −0.304 ± 0.037 |
| 300 kGy | −0.352 ± 0.027 | −0.426 ± 0.041 | −0.605 ± 0.029 | −0.248 ± 0.025 | −0.336 ± 0.014 | −0.265 ± 0.025 |
| NR + TAIC | ||||||
| 75 kGy | −0.614 ± 0.037 | −1.002 ± 0.098 | −0.858 ± 0.012 | −0.541 ± 0.020 | −0.773 ± 0.059 | −0.745 ± 0.098 |
| 150 kGy | −0.485 ± 0.022 | −0.542 ± 0.025 | −0.822 ± 0.012 | −0.429 ± 0.041 | −0.498 ± 0.069 | −0.659 ± 0.101 |
| 225 kGy | −0.481 ± 0.103 | −0.486 ± 0.053 | −0.789 ± 0.012 | −0.408 ± 0.034 | −0.489 ± 0.021 | −0.65 ± 0.075 |
| 300 kGy | −0.477 ± 0.043 | −0.378 ± 0.093 | −0.647 ± 0.012 | −0.315 ± 0.036 | −0.497 ± 0.026 | −0.466 ± 0.102 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Craciun, G.; Manaila, E.; Stelescu, M.D. New Elastomeric Materials Based on Natural Rubber Obtained by Electron Beam Irradiation for Food and Pharmaceutical Use. Materials 2016, 9, 999. https://doi.org/10.3390/ma9120999
Craciun G, Manaila E, Stelescu MD. New Elastomeric Materials Based on Natural Rubber Obtained by Electron Beam Irradiation for Food and Pharmaceutical Use. Materials. 2016; 9(12):999. https://doi.org/10.3390/ma9120999
Chicago/Turabian StyleCraciun, Gabriela, Elena Manaila, and Maria Daniela Stelescu. 2016. "New Elastomeric Materials Based on Natural Rubber Obtained by Electron Beam Irradiation for Food and Pharmaceutical Use" Materials 9, no. 12: 999. https://doi.org/10.3390/ma9120999






