1. Introduction
Intermetallic Ni
3Al-based alloys are a group of advanced materials which show potential outstanding physical and chemical properties, such as excellent oxidation and corrosion resistance, fairly high melting temperatures, relatively low densities and anomalous strengthening with increasing temperature. Extensive efforts have been made to develop Ni
3Al-based alloys into commercial use as high-temperature structural material in a bulk form, e.g., as-cast ingots, bars, plates, tubes,
etc. [
1,
2,
3,
4].
Thin foils made of Ni
3Al intermetallic alloy can be used in high-performance applications in the form of honeycomb structures, which have the advantage of being lightweight and showing high-stiffness and catalytic properties [
4,
5,
6,
7]. In previous work, a cold‑rolling based approach was set up which allows to obtain micro- or nano-structured Ni
3Al intermetallic foils as thin as 50 µm thickness [
8]. Another method to obtain similar thin foils-only with microcrystalline structure-was reported by Hirano
et al. [
9,
10], who rolled the directionally solidified ingots at room temperature. The mechanical properties of so-prepared Ni
3Al materials were studied in detail [
9,
10,
11]. The materials obtained with this method have good catalytic properties for alcohols and hydrocarbons decomposition [
5,
6,
12].
The present paper focuses on the catalytic decomposition of cyclohexane on Ni3Al intermetallic alloy thin foils. The performance of the catalyst, represented by conversion and its surface morphology changes, has been systematically investigated.
2. Results and Discussion
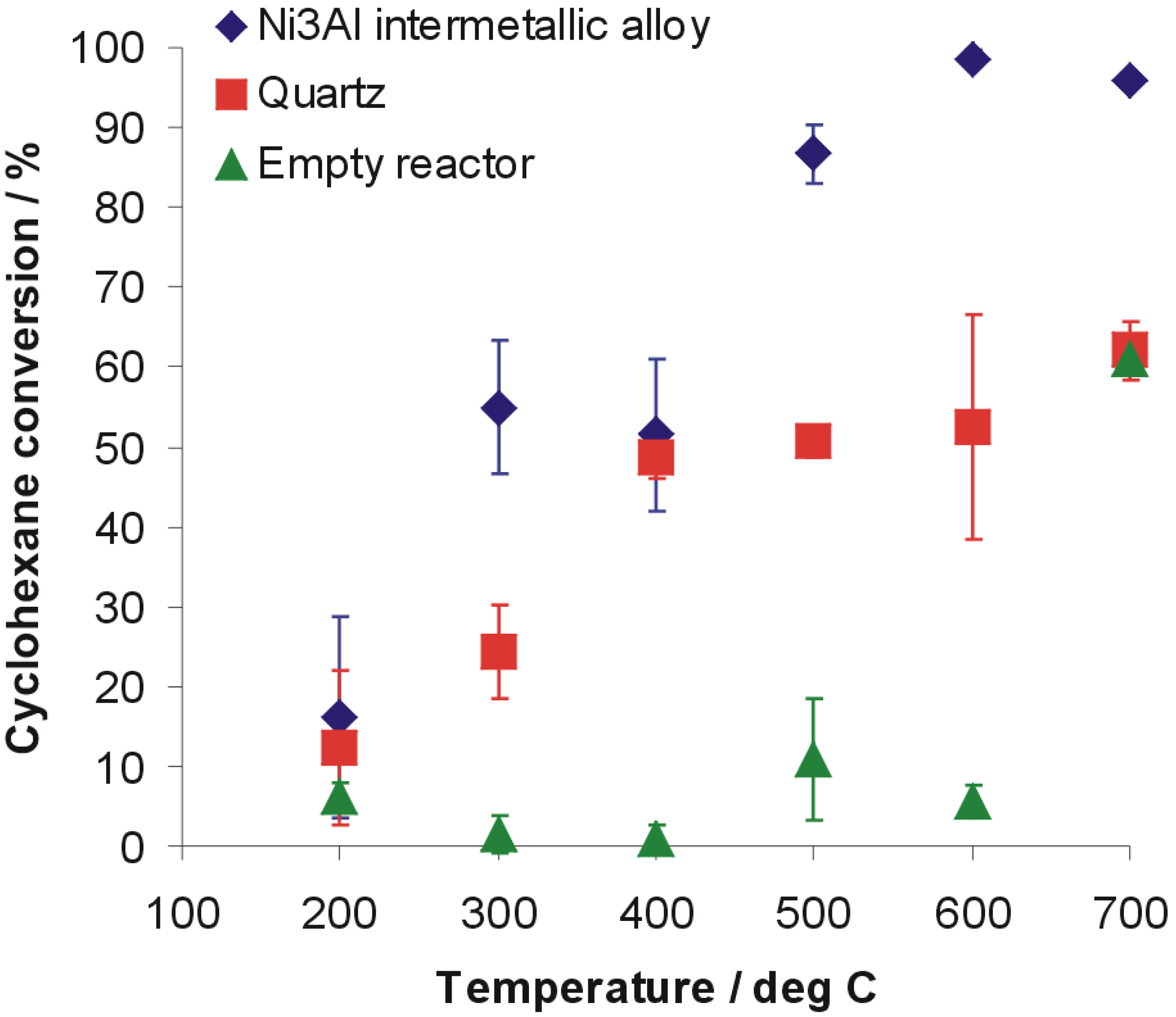
Figure 1 shows cyclohexane conversion
versus reaction temperature for Ni
3Al intermetallic alloy and quartz as a reference inert material and additionally by using empty reactor. The cyclohexane conversion on the pieces of Ni
3Al foil with increasing temperature is generally fast and starts already above 200 °C. Since 500 °C the conversion efficiency is 86.7% ± 3.6%, while the conversion efficiency of quartz is approximately 50.6% ± 1.9%. Analysis of cyclohexane presence after examination with empty reactor revealed a very low level of conversion (approximately 10% or below) up to a temperature of 600 °C. Increasing the temperature above that level leads to growing efficiency of cyclohexane decomposition in the empty reactor up to 61.1% ± 1.0%, a level comparable to the highest conversion obtained for decomposition reaction conducted on quartz.
A major advantage of the applied catalyst is its simple form, pieces of thin foil without any support, which makes this material cost‑effective despite the significant content of nickel. Additionally, no complex, expensive and sophisticated chemistry is applied to form the catalyst and this approach allows its formation in industrial quantities. Moreover, the catalyst performance is comparable to, or even better than, those obtained with sophisticated chemical methods. For example, with Ni
2P catalyst a conversion of cyclohexane close to 100% was achieved for similar temperatures, however in this case Al
2O
3 was applied as the support for the active material [
13]. Application of nanoporous materials as a support for the catalyst,
i.e., Au@TiO
2 also brings satisfactory catalytic results (approximately 10% of conversion at 150 °C). However, also in that case the catalyst manufacturing requires several steps [
14].
Figure 1.
Conversion of cyclohexane conducted on the pieces of Ni3Al thin foil, quartz and empty reactor as a function of the temperature.
Figure 1.
Conversion of cyclohexane conducted on the pieces of Ni3Al thin foil, quartz and empty reactor as a function of the temperature.
An additional advantage of using the Ni
3Al intermetallic alloy for hydrocarbons thermocatalytic decomposition is the formation of complex carbon nanostructures on the alloys surface (see
Figure 2). It has been reported that carbon nanostructures are outstanding for hydrocarbons,
i.e., cyclohexane decomposition [
15]. Typically, catalysts are being doped by the products of the reaction. In contrast, the products of the reaction—carbon nanostructures—also work as the catalyst with increased active surface area. Therefore, the catalytic performance of the Ni
3Al intermetallic alloy is being enhanced by the reaction products, which is another advantage of this material.
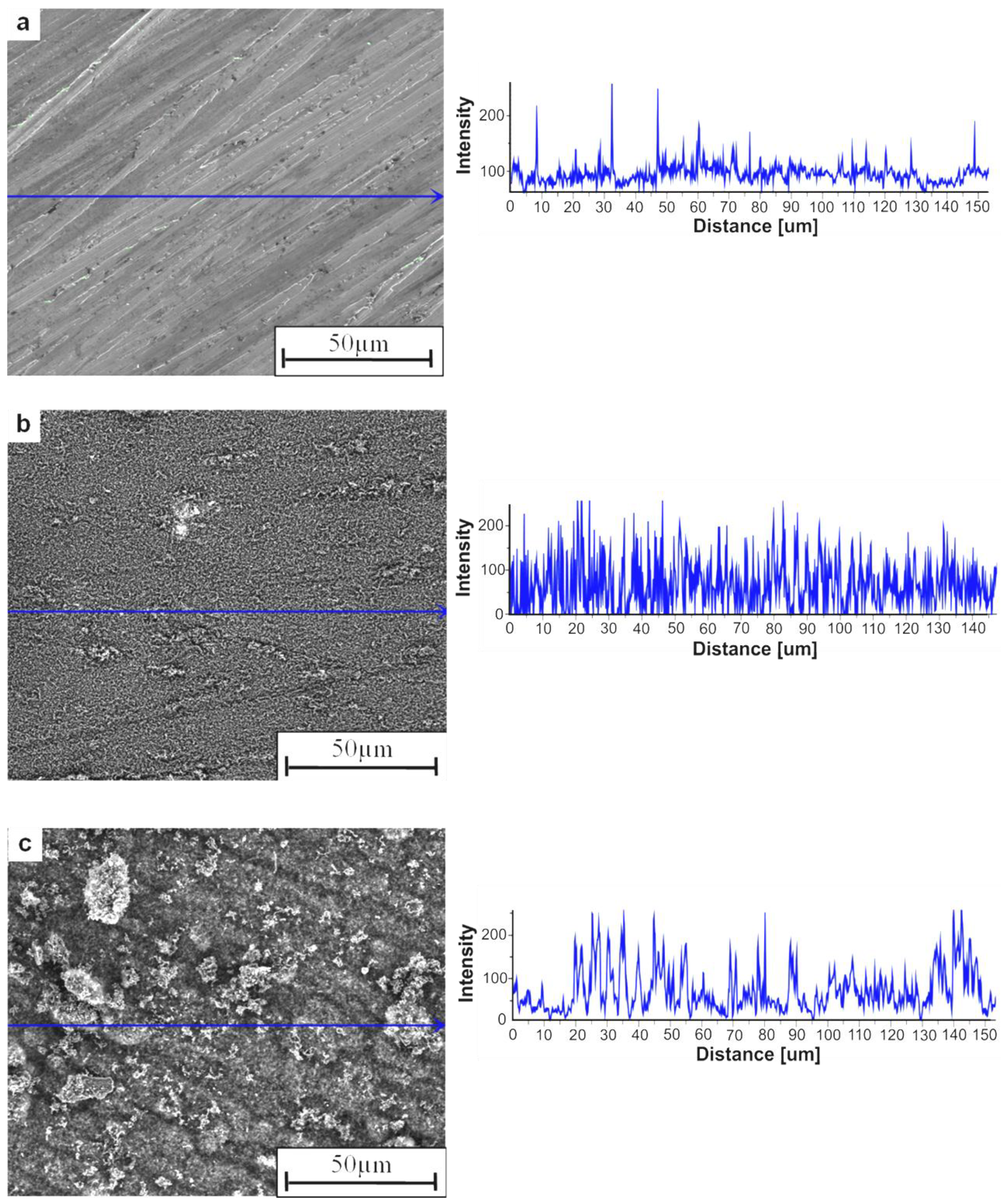
The pieces of Ni
3Al thin foil, which have clean and glossy surfaces before the experiment, are covered with a deposit that looks like carbon black after the catalytic examination (
Figure 2). The analysis of SEM morphology and intensity profiles of the Ni
3Al foils surface before and decomposition of cyclohexane at 500 °C, shows the morphology developed for the deposit (
Figure 2a,b). Further temperature increase up to 700 °C leads to the formation of clusters of carbon nanofibers (CNFs), which can be seen as a decrease in the intensity profiles (
Figure 2c,
Figure 3 and
Figure 4). Observation of the Ni
3Al foil by secondary electrons (SE) and backscattered electrons (BSE) signals in SEM, shows the presence of a complex structure of carbon nanofibers (CNFs) with various shapes and diameters, as well as metal-like contrast nanoparticles, deposited during cyclohexane decomposition (
Figure 2 and
Figure 3). The diffusion of carbon atoms through the catalyst centers leads to formation of tubular and platelet fibers on the foil surface. In the deposit, the tubular CNFs are dominant, with graphene layers rolled‑up in a cylindrical shape [
16] (with average diameter below 80 nm) and bearing the metal‑like particles at the tip (see
Figure 4c,d,f). In these CNFs, only a single face is available for precipitating carbon in the form of concentric graphite [
17].
Figure 2.
SEM morphology with intensity profiles of the surface of Ni3Al foil: (a) before experiments; and after decomposition of cyclohexane at: (b) 500 °C; (c) 700 °C.
Figure 2.
SEM morphology with intensity profiles of the surface of Ni3Al foil: (a) before experiments; and after decomposition of cyclohexane at: (b) 500 °C; (c) 700 °C.
As the second component of the deposit, thicker platelet CNFs are observed, with an average diameter of approximately 500 nm [
18]. As reported by Rodriguez
et al. [
17] and Martin-Gullon
et al. [
18], the catalyst particles are located inside the fiber (see
Figure 4e), where carbon was formed by bidirectional mode. The platelet CNFs were formed simultaneously from opposite faces of the particle, which remains within the structure throughout the growth process [
17].
Figure 3.
SEM morphology of carbon nanofibres (CNFs) deposited on the surface of Ni3Al foil after decomposition of cyclohexane at 500 °C, obtained by using a backscattered electrons (BSE) detector.
Figure 3.
SEM morphology of carbon nanofibres (CNFs) deposited on the surface of Ni3Al foil after decomposition of cyclohexane at 500 °C, obtained by using a backscattered electrons (BSE) detector.
Figure 4.
SEM morphology of CNFs deposited on the surface of Ni3Al foil after decomposition of cyclohexane at 700 °C, obtained by using (a–c) SE and (d–f) BSE detectors.
Figure 4.
SEM morphology of CNFs deposited on the surface of Ni3Al foil after decomposition of cyclohexane at 700 °C, obtained by using (a–c) SE and (d–f) BSE detectors.
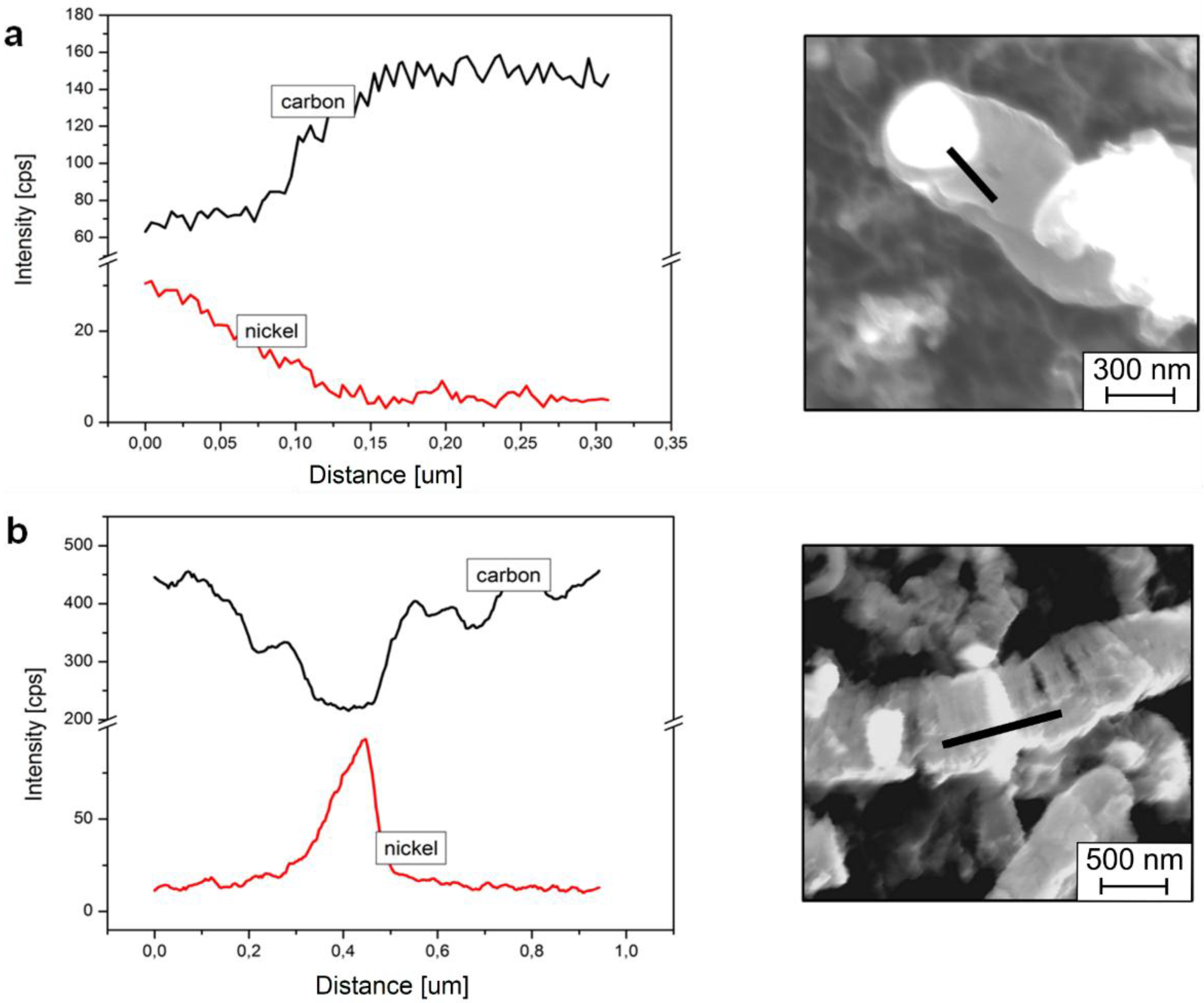
In the case of the above-mentioned small metal-like objects, the result of EDS has only qualitative significance, but it is clear that the interior of the tubular CNFs is also rich in nickel and depleted from carbon (see metal nanoparticle in
Figure 3b,
Figure 4c–f and
Figure 5b).
Figure 5.
The energy dispersive spectrometry (EDS) linear analysis along the nanofiber with nickel‑like nanoparticles in (a) tubular and (b) platelet CNF.
Figure 5.
The energy dispersive spectrometry (EDS) linear analysis along the nanofiber with nickel‑like nanoparticles in (a) tubular and (b) platelet CNF.
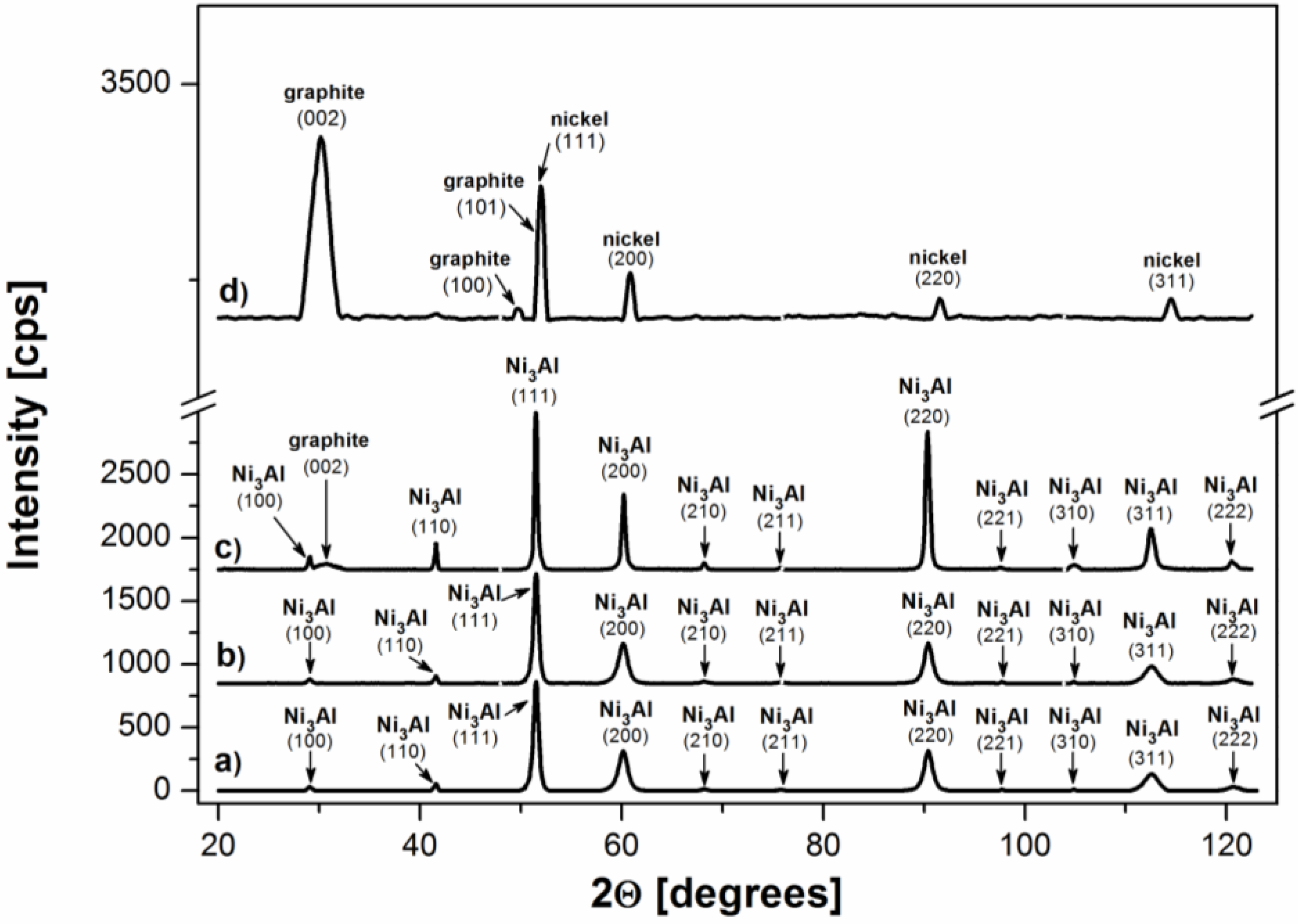
The XRD patterns of the examined foils surface after cyclohexane decomposition at 700 °C (
Figure 6a) show existence of Ni
3Al intermetallic compound with slightly visible peak of graphite only from (002) plane, but without any apparent nickel contribution. The XRD analysis of Ni
3Al foil after experiment at 500 °C shows only peaks characteristic for Ni
3Al phase. The lack of nickel lattice is connected with similar
fcc-based crystal structure to Ni
3Al-phase and strong coincidence in their lattice constants (3.524 Å and 3.567 Å, respectively).
The XRD patterns for deposit after cyclohexane decomposition at 700 °C scraped from the Ni
3Al foil (
Figure 6d) demonstrate that the catalytic reaction products contain only pure nickel and graphite. The small nickel particles, probably formed in accordance with the mechanism described in previous reports [
16,
17,
18], serve as an effective catalyst for cyclohexane decomposition, as well as CNFs, which are expected in common catalyst supports due to their large edges.
Figure 6.
XRD patterns of Ni3Al foil surface: (a) before experiments; after cyclohexane decomposition at: (b) 500 °C and (c) 700 °C as well as deposit scraped off of the surface of Ni3Al foils (d).
Figure 6.
XRD patterns of Ni3Al foil surface: (a) before experiments; after cyclohexane decomposition at: (b) 500 °C and (c) 700 °C as well as deposit scraped off of the surface of Ni3Al foils (d).
3. Materials and Methods
A Ni
3Al intermetallic alloy with nominal composition of Ni–22.1Al–0.26Zr–0.1B (at%) was obtained by casting into shell-mold from pure elements melted in Ar atmosphere. The obtained ingots were cut into plates, cold-rolled up to 95% in few passes and recrystallized at 1100 °C for 1.5 h in argon atmosphere. As a result, 50 µm thick microcrystalline foil was obtained. More detailed procedures have been described previously [
8]. Before catalytic experiments, the Ni
3Al foil was mechanically polished and rinsed with acetone in an ultrasonic cleaner.
Catalytic decomposition of cyclohexane was carried out in a fixed bed quartz reactor preceded by preliminary quartz reactor where cyclohexane and air were being added and heated up. The investigations were conducted in a synthetic air atmosphere, with a composition of 21% O2, 10 ppm H2O or less in N2. The feed of the air and cyclohexane were set as 3 dm3/h by electronic flow controller and 3 mL/h of liquid hydrocarbon with infusion pump, respectively.
The catalytic activity of the Ni3Al intermetallic alloy was examined in the temperature range from 200 to 700 °C for 2 h. The gaseous products were analyzed online by the Clarus 500 gas-chromatograph equipped with Clarus 560S mass spectrometer (Perkin Elmer, Waltham, MA, USA). Small pieces of quartz were applied as the reference inert material. For all catalytic test the Ni3Al thin foil was cut into small pieces (approximately 2 × 3 mm2 each) and the total weight of quartz reactor feed was approximately 0.6 g per experiment. Four chromatograms were recorded for each reaction temperature.
The surface morphologies of representative samples were examined by SEM FEI Quanta 3d FEG (FEI, Hillsboro, OR, USA), with detectors of: SE and BSE, and with EDS equipment.
The phase structure of the surface products after catalysis was examined by XRD using a Seifert 3000 diffractometer (Seifert GmbH, Ahrensburg, Germany) with CoKα radiation at operating parameters of 30 mA, 50 kV and a step size of 0.02° per 3 s.