Characterization and Stability of Tanshinone IIA Solid Dispersions with Hydroxyapatite
Abstract
:1. Introduction
2. Experimental Section
2.1. Materials
2.2. Preparation of Solid Dispersions and Physical Mixtures
2.3. In vitro Dissolution Study
2.3.1. HPLC Analysis of TanIIA
2.3.2. In vitro Dissolution Studies
2.4. Differential Scanning Calorimetry (DSC)
2.5. Scanning Electron Microscopy (SEM)
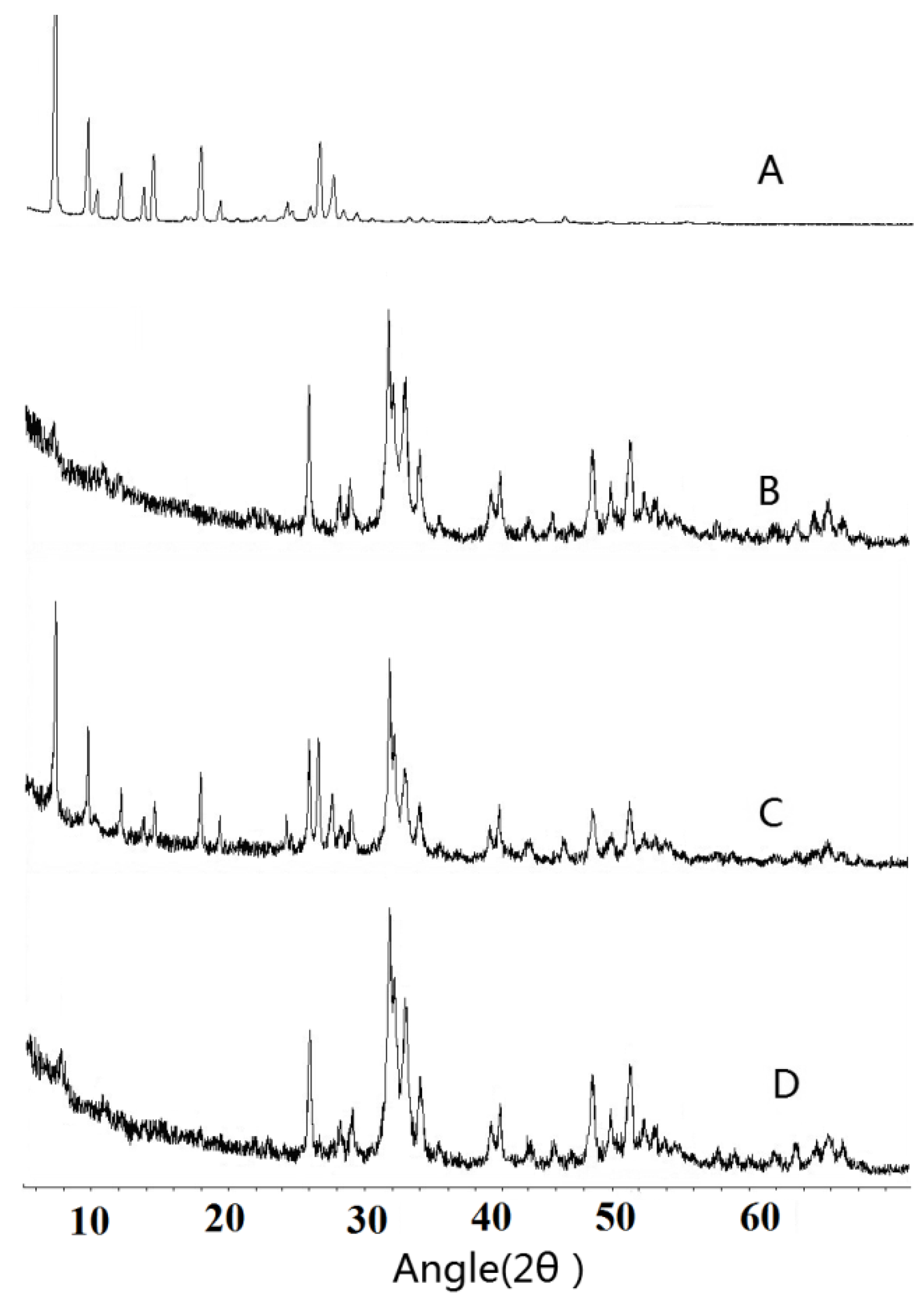
2.6. X-ray Powder Diffraction (XRPD)
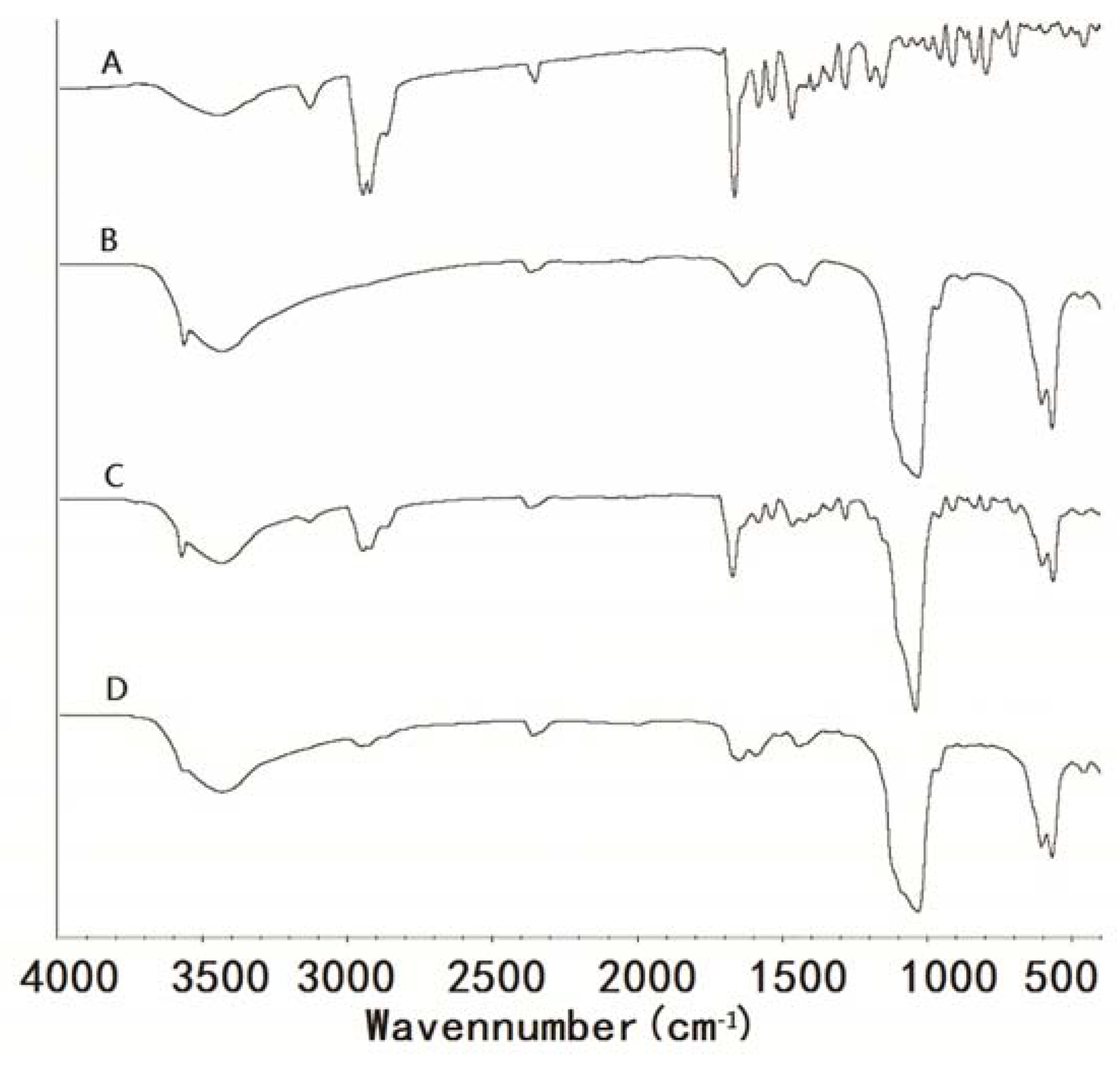
2.7. Fourier Transform Infrared Spectroscopy (FTIR)
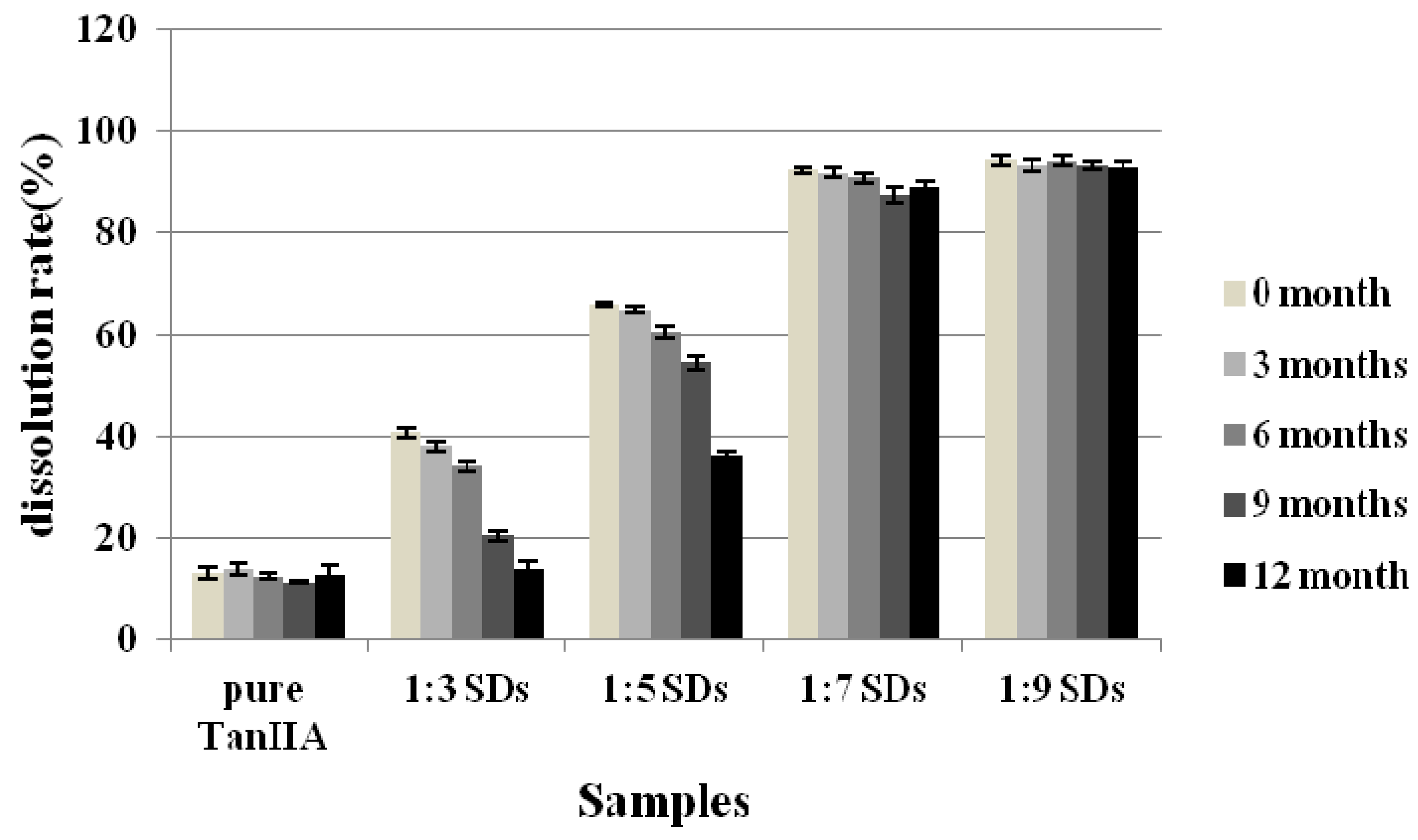
2.8. Stability Test
3. Results and Discussion
3.1. In vitro Dissolution Study

3.2. Scanning Electron Microscopy (SEM)

3.3. Differential Scanning Calorimetry (DSC)

3.4. X-ray Powder Diffraction (XRPD)
3.5. Fourier Transform Infrared (FTIR) Spectrophotometry
3.6. Stability Test
4. Conclusions
Acknowledgments
Conflict of Interest
References
- Wang, P.; Wu, X.; Bao, Y.; Fang, J.; Zhou, S.; Gao, J.; Pi, R.; Mou, Y.G.; Liu, P. Tanshinone IIA prevents cardiac remodeling through attenuating NAD (P)H oxidase-derived reactive oxygen species production in hypertensive rats. Pharmazie 2011, 66, 517–524. [Google Scholar] [PubMed]
- Wei, Y.; Li, P.; Wang, C.; Peng, Y.; Shu, L.; Jia, X.; Ma, W.; Wang, B. Metabolism of tanshinone IIA, cryptotanshinone and tanshinone I from Radix Salvia miltiorrhiza in zebrafish. Molecules 2012, 17, 8617–8632. [Google Scholar] [CrossRef] [PubMed]
- Gao, J.; Yang, G.; Pi, R.; Li, R.; Wang, P.; Zhang, H.; Le, K.; Chen, S.; Liu, P. Tanshinone IIA protects neonatal rat cardiomyocytes from adriamycin-induced apoptosis. Transl. Res. 2008, 151, 79–87. [Google Scholar] [CrossRef] [PubMed]
- Li, X.B.; Wang, W.; Zhou, G.J.; Li, Y.; Xie, X.M.; Zhou, T.S. Production of salvianolic acid B in roots of Salvia miltiorrhiza (Danshen) during the post-harvest drying process. Molecules 2012, 17, 2388–2407. [Google Scholar] [CrossRef] [PubMed]
- Qiao, Z.; Ma, J.; Liu, H. Evaluation of the antioxidant potential of Salvia miltiorrhiza ethanol extract in a rat model of ischemia-reperfusion injury. Molecules 2011, 16, 10002–10012. [Google Scholar] [CrossRef] [PubMed]
- Fu, J.; Huang, H.; Liu, J.; Pi, R.; Chen, J.; Liu, P. TSIIA protects cardiac myocytes against oxidative stress-triggered damage and apoptosis. Eur. J. Pharmacol. 2007, 568, 213–221. [Google Scholar] [CrossRef] [PubMed]
- Hao, H.; Wang, G.; Cui, N.; Li, J.; Xie, L.; Ding, Z. Pharmacokinetics, absorption and tissue distribution of tanshinone IIA solid dispersion. Planta Med. 2006, 72, 1311–1317. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.; Subedi, R.K.; Nepal, P.R.; Kim, Y.G.; Choi, H.K. Enhancement of solubility and dissolution rate of cryptotanshinone, tanshinone I and tanshinone IIA extracted from Salvia miltiorrhiza. Arch. Pharm. Res. 2012, 35, 1457–1464. [Google Scholar] [CrossRef] [PubMed]
- Chang, L.C.; Wu, C.L.; Liu, C.W.; Chuo, W.H.; Li, P.C.; Tsai, T.R. Preparation, characterization and cytotoxicity evaluation of tanshinone IIA nanoemulsions. J. Biomed. Nanotechnol. 2011, 7, 558–567. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.Y.; Lin, S.G.; Zhou, Z.W.; Chen, X.; Liang, J.; Liu, P.Q.; Duan, W.; Chowbay, B.; Wen, J.Y.; Li, C.G.; Zhou, S.F. Role of P-glycoprotein in the intestinal absorption of TSIIA, a major active ingredient in the root of Salvia miltiorrhiza. Curr. Drug Metab. 2007, 8, 325–340. [Google Scholar] [CrossRef] [PubMed]
- Mao, S.J.; Hou, S.X.; Liang, Z.; Bi, Y.Q.; Wu, Y.; Li, H.; Jin, H. Ion-pair reversed-phase HPLC: Assay validation of sodium TSIIA sulfonate in mouse plasma. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2006, 831, 163–168. [Google Scholar] [CrossRef]
- Wang, L.; Jiang, X.; Xu, W.; Li, C. Complexation of tanshinone IIA with 2-hydroxypropyl-beta-cyclodextrin: Effect on aqueous solubility, dissolution rate, and intestinal absorption behavior in rats. Int. J. Pharm. 2007, 341, 58–67. [Google Scholar] [CrossRef] [PubMed]
- Beak, I.H.; Kim, M.S. Improved supersaturation and oral absorption of dutasteride by amorphous solid dispersions. Chem. Pharm. Bull. 2012, 60, 1468–1473. [Google Scholar] [CrossRef] [PubMed]
- Potluri, R.H.; Bandari, S.; Jukanti, R.; Veerareddy, P.R. Solubility enhancement and physicochemical characterization of carvedilol solid dispersion with Gelucire 50/13. Arch. Pharm. Res. 2011, 34, 51–57. [Google Scholar] [CrossRef] [PubMed]
- Planinšek, O.; Kovaèiè, B.; Vreèer, F. Carvedilol dissolution improvement by preparation of solid dispersions with porous silica. Int. J. Pharm. 2011, 406, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Venkatesh, G.M.; Barnett, M.E.; Owusu-Fordjour, C.; Galop, M. Detection of low levels of the amorphous phase in crystalline pharmaceutical materials by thermally stimulated current spectrometry. Pharm. Res. 2001, 18, 98–103. [Google Scholar] [CrossRef] [PubMed]
- Willart, J.F.; Descamps, M. Solid state amorphization of pharmaceuticals. Mol. Pharm. 2008, 5, 905–920. [Google Scholar] [CrossRef] [PubMed]
- Greco, S.; Authelin, J.R.; Leveder, C.; Segalini, A. A practical method to predict physical stability of amorphous solid dispersions. Pharm. Res. 2012, 29, 2792–2805. [Google Scholar] [CrossRef] [PubMed]
- Bhugra, C.; Rambhatla, S.; Bakri, A.; Duddu, S.P.; Miller, D.P.; Pikal, M.J.; Lechuga-Ballesteros, D. Prediction of the onset of crystallization of amorphous sucrose below the calorimetric glass transition temperature from correlations with mobility. J. Pharm. Sci. 2007, 96, 1258–1269. [Google Scholar] [CrossRef] [PubMed]
- Hong, J.; Shah, J.C.; Mcgonagle, M.D. Effect of cyclodextrin derivation and amorphous state of complex on accelerated degradation of ziprasidone. J. Pharm. Sci. 2011, 100, 2703–2716. [Google Scholar] [CrossRef] [PubMed]
- Kojima, T.; Higashi, K.; Suzuki, T.; Tomono, K.; Moribe, K.; Yamamoto, K. Stabilization of a supersaturated solution of mefenamic acid from a solid dispersion with EUDRAGIT® EPO. Pharm. Res. 2012, 29, 2777–2791. [Google Scholar] [CrossRef] [PubMed]
- Qian, F.; Wang, J.; Hartley, R.; Tao, J.; Haddadin, R.; Mathias, N.; Hussain, M. Solution behavior of PVP-VA and HPMC-AS-based amorphous solid dispersions and their bioavailability implications. Pharm. Res. 2012, 29, 2765–2776. [Google Scholar] [PubMed]
- Al-Obaidi, H.; Ke, P.; Brocchini, S.; Buckton, G. Characterization and stability of ternary solid dispersions with PVP and PHPMA. Int J. Pharm. 2011, 419, 20–27. [Google Scholar] [CrossRef] [PubMed]
- Al-Obaidi, H.; Brocchini, S.; Buckton, G. Anomalous properties of spray dried solid dispersions. J. Pharm Sci. 2009, 98, 4724–4737. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.X.; Yang, M.; van den Berg, F.; Pajander, J.; Rades, T.; Rantanen, J. Influence of solvent evaporation rate and formulation factors on solid dispersion physical stability. Eur. J. Pharm. Sci. 2011, 44, 610–620. [Google Scholar] [CrossRef] [PubMed]
- Bellucci, D.; Cannillo, V.; Sola, A. A New Highly Bioactive Composite for Scaffold Applications: A Feasibility Study. Materials 2011, 4, 339–354. [Google Scholar] [CrossRef]
- Jin, H.H.; Kim, D.H.; Kim, T.W.; Shin, K.K.; Jung, J.S.; Park, H.C.; Yoon, S.Y. In vivo evaluation of porous hydroxyapatite/chitosan-alginate composite scaffolds for bone tissue engineering. Int. J. Biol. Macromol. 2012, 51, 1079–1085. [Google Scholar] [CrossRef] [PubMed]
- Ishikawa, K. Bone Substitute Fabrication Based on Dissolution-Precipitation Reactions. Materials 2010, 3, 1138–1155. [Google Scholar] [CrossRef]
- Fu, S.; Ni, P.; Wang, B.; Chu, B.; Peng, J.; Zheng, L.; Zhao, X.; Luo, F.; Wei, Y.; Qian, Z. In vivo biocompatibility and osteogenesis of electrospun poly(ε-caprolactone)-poly(ethyleneglycol)-poly(ε-caprolactone)/nano-hydroxyapatite composite scaffold. Biomaterials 2012, 33, 8363–8371. [Google Scholar] [CrossRef] [PubMed]
- Venkatesan, J.; Kim, S.K. Effect of Temperature on Isolation and Characterization of Hydroxyapatite from Tuna (Thunnus obesus) Bone. Materials 2010, 3, 4761–4772. [Google Scholar] [CrossRef]
- Kong, L.; Gao, Y.; Cao, W.; Gong, Y.; Zhao, N.; Zhang, X. Preparation and characterization of nano-hydroxyapatite/chitosan composite scaffolds. J. Biomed. Mater. Res. A 2005, 75, 275–282. [Google Scholar] [CrossRef] [PubMed]
- Doostmohammadi, A.; Monshi, A.; Fathi, M.H.; Karbasi, S.; Braissant, O.; Daniels, A.U. Direct cytotoxicity evaluation of 63S bioactive glass and bone-derived hydroxyapatite particles using yeast model and human chondrocyte cells by microcalorimetry. J. Mater. Sci. Mater. Med. 2011, 22, 2293–2300. [Google Scholar] [CrossRef] [PubMed]
- Vohra, S.; Hennessy, K.M.; Sawyer, A.A.; Zhuo, Y.; Bellis, S.L. Comparison of mesenchymal stem cell and osteosarcoma cell adhesion to hydroxyapatite. J. Mater. Sci. Mater. Med. 2008, 19, 3567–3574. [Google Scholar] [CrossRef] [PubMed]
- Hennessy, K.M.; Clem, W.C.; Phipps, M.C.; Sawyer, A.A.; Shaikh, F.M.; Bellis, S.L. The effect of RGD peptides on osseointegration of hydroxyapatite biomaterials. Biomaterials 2008, 29, 3075–3083. [Google Scholar] [CrossRef] [PubMed]
- Wei, G.; Ma, P.X. Structure and properties of nano-hydroxyapatite/polymer composite scaffolds for bone tissue engineering. Biomaterials 2004, 25, 4749–4757. [Google Scholar] [CrossRef] [PubMed]
- Victor, S.P.; Sharma, C.P. Tryptophan complexed hydroxyapatite nanoparticles for immunoglobulin adsorption. J. Mater. Sci. Mater. Med. 2011, 22, 2219–2229. [Google Scholar] [CrossRef] [PubMed]
- Monkhouse, D.C.; Lach, J.L. Use of adsorbents in enhancement of drug dissolution II. J. Pharm. Sci. 1972, 61, 1435–1441. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Liu, X.; Gan, L.; Zhou, C.; Mo, J. Preparation and physicochemical characterizations of tanshinone IIA solid dispersion. Arch. Pharm. Res. 2011, 34, 949–959. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Liu, P.; Liu, J.P.; Zhang, W.L.; Yang, J.K.; Fan, Y.Q. Novel Tanshinone IIA ternary solid dispersion pellets prepared by a single-step technique: In vitro and in vivo evaluation. Eur. J. Pharm. Biopharm. 2012, 80, 426–432. [Google Scholar] [CrossRef] [PubMed]
- Shaltout, A.A.; Allam, M.A.; Moharram, M.A. FTIR spectroscopic, thermal and XRD characterization of hydroxyapatite from new natural sources. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2011, 83, 56–60. [Google Scholar] [CrossRef] [PubMed]
- Hwang, K.S.; Jeon, K.O.; Jeon, Y.S.; Kim, B.H. Hydroxyapatite forming ability of electrostatic spray pyrolysis derived calcium phosphate nano powder. J. Mater. Sci. Mater. Med. 2007, 18, 619–622. [Google Scholar] [CrossRef] [PubMed]
- Reves, B.T.; Jennings, J.A.; Bumgardner, J.D.; Haggard, W.O. Osteoinductivity Assessment of BMP-2 Loaded Composite Chitosan-Nano-Hydroxyapatite Scaffolds in a Rat Muscle Pouch. Materials 2011, 4, 1360–1374. [Google Scholar] [CrossRef]
- Kandori, K.; Oketani, M.; Wakamura, M. Effects of Ti(IV) substitution on protein adsorption behaviors of calcium hydroxyapatite particles. Colloids Surf. B Biointerfaces 2013, 101, 68–73. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, M.; Hu, J.; Gao, P.; Li, L.; Ali-Reynolds, A.; Chal, B.; Gupta, V.; Ma, C.; Mahajan, N.; Akrami, A.; Surapaneni, S. Enhanced bioavailability of a poorly soluble VR1 antagonist using an amorphous solid dispersion approach: A case study. Mol. Pharm. 2008, 5, 981–993. [Google Scholar] [CrossRef] [PubMed]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Wang, X.; Li, L.; Huo, W.; Hou, L.; Zhao, Z.; Li, W. Characterization and Stability of Tanshinone IIA Solid Dispersions with Hydroxyapatite. Materials 2013, 6, 805-816. https://doi.org/10.3390/ma6030805
Wang X, Li L, Huo W, Hou L, Zhao Z, Li W. Characterization and Stability of Tanshinone IIA Solid Dispersions with Hydroxyapatite. Materials. 2013; 6(3):805-816. https://doi.org/10.3390/ma6030805
Chicago/Turabian StyleWang, Xiaopan, Li Li, Wei Huo, Lulu Hou, Zhiying Zhao, and Weiguang Li. 2013. "Characterization and Stability of Tanshinone IIA Solid Dispersions with Hydroxyapatite" Materials 6, no. 3: 805-816. https://doi.org/10.3390/ma6030805



